Abstract
A 7-mo-old farmed white-tailed deer fawn (Odocoileus virginianus) died after several weeks of progressive deterioration associated with endoparasitism and respiratory signs. A field autopsy was performed, and lung tissue was submitted for histologic examination. The findings were consistent with necrosuppurative bronchointerstitial pneumonia with intranuclear viral inclusions. Immunofluorescence using fluorescently labeled polyclonal antibodies to bovine adenovirus 3 and 5 was positive. To rule out cross-reactivity with other adenoviruses, formalin-fixed, paraffin-embedded tissue sections were submitted for genome sequence analysis, which revealed a 99.6% match to Deer mastadenovirus B (formerly Odocoileus adenovirus 2, OdAdV2). To our knowledge, natural clinical disease associated with OdAdV2 has not been reported previously.
Adenoviruses are nonencapsulated, icosahedral, double-stranded DNA viruses of 70–90 nm diameter 7 that typically cause host–specific, subclinical infection that can lead to mild respiratory or enteric disease. Clinical disease is most commonly seen in immunocompromised animals, and adenoviral infection can predispose animals to bacterial pneumonia. The family Adenoviridae includes the genera Atadenovirus and Mastadenovirus, and adenoviruses that affect porcine, canine, equine, ovine, bovine, and cervid species.7,8
Two cervid adenoviruses have been detected over the past 25 y. Most reported adenoviral cases in deer have been caused by Deer atadenovirus A (hereafter, OdAdV1; formerly deer adenovirus 1, Odocoileus adenovirus 1). 9 This virus is the cause of adenoviral hemorrhagic disease (ADH), which has been reported in many domestic and wild cervids.2,12,14,16,17 ADH is characterized by severe vasculitis in the lungs, intestine, and other organs. More recently, Deer mastadenovirus B (hereafter, OdAdV2; formerly, cervid adenovirus 1, deer adenovirus 2, Odocoileus adenovirus 2) was discovered incidentally in nasal secretions and buffy coat samples of farmed white-tailed deer (WTD) fawns. 11 Inoculation of this virus into WTD fawns resulted in mild respiratory disease, pyrexia, and decreased thymic weight. 11 In that study, the closest genetic match to OdAdV2 was bovine adenovirus 3 (BAdV3; Bovine mastadenovirus B) with 76% sequence identity. Here we describe a case of natural OdAdV2 pneumonia in a farmed WTD fawn.
A 7-mo-old farmed WTD fawn died after several weeks of progressive deterioration associated with chronic diarrhea and nonspecific respiratory signs. The fawn was historically underweight, pot-bellied, and had been treated for chronic gastrointestinal parasitism with moxidectin, albendazole, and ponazuril. A field autopsy was performed, and lung tissue was submitted to the Texas A&M Veterinary Medical Diagnostic Laboratory (College Station, TX, USA) for histologic examination. No gross description was provided by the referring veterinarian. Fixed lung samples were processed routinely, embedded in paraffin wax, sectioned at 5 µm, and stained with H&E.
Microscopically, lung lesions were consistent with bronchointerstitial pneumonia. Multifocally, bronchi and bronchioles were dilated and filled with sloughed epithelial cells, necrotic cellular debris, and numerous neutrophils (Fig. 1). Within the alveoli were dense infiltrates of neutrophils, fewer macrophages, and accumulations of fibrin, serum protein, mucin, and necrotic cellular debris (Fig. 2). The bronchial and bronchiolar epithelium was hyperplastic with areas of necrosis, and many epithelial cells had 8–12-µm intranuclear inclusion bodies (INIBs). Most of the INIBs were basophilic and centered within the nucleus; others were more eosinophilic with margination of nuclear chromatin (Fig. 3). Alveolar septa were hyperemic, expanded by edema, and contained mildly increased numbers of neutrophils and lymphocytes. Interlobular septa and perivascular spaces were also expanded by edema. Bacteria were not visible on H&E or Gram stains.
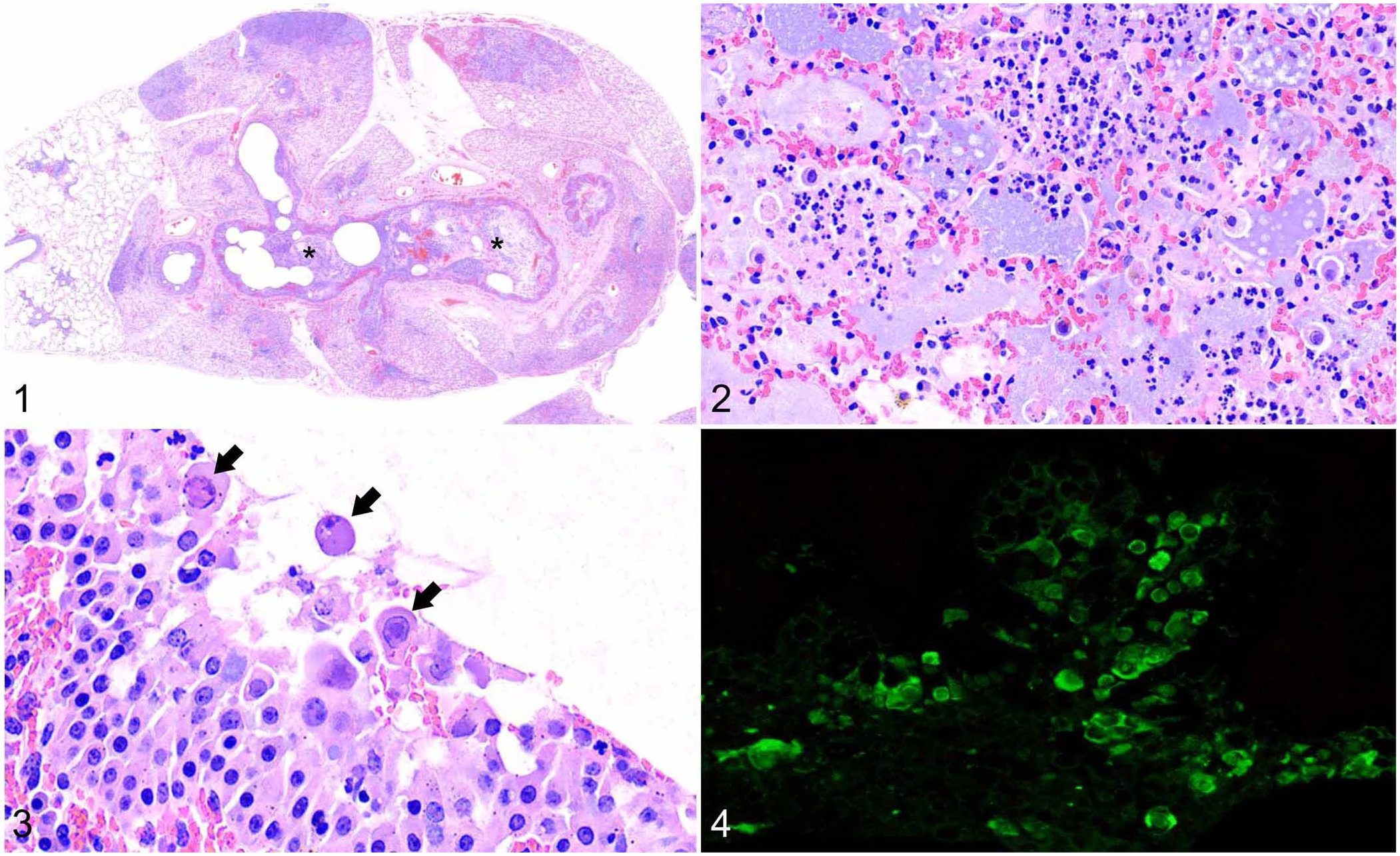
Deer mastadenovirus B (OdAdV2)-associated bronchointerstitial pneumonia in a white-tailed deer (Odocoileus virginianus) fawn. H&E.
For immunofluorescence analysis, formalin-fixed, 5-μm sections of lung were mounted on positively charged slides, allowed to dry overnight, and then deparaffinized and rehydrated. Antigen unmasking was conducted (proteinase K solution, 0.2 mg/mL; Sigma-Aldrich) for 1 min at room temperature, then slides were washed in PBS 3 times for 5 min each wash. Following the final wash, 100 µL of 5× fluorescein isothiocyanate–labeled direct conjugates to BAdV3 (VMRD) were added to tissue sections and incubated for 5 h at 37°C in a humidified chamber. Following incubation, slides were washed 3 times in PBS for 5–10 min each wash, mounting medium (10% glycerol in PBS) was added, and slides were coverslipped and viewed with a fluorescence microscope. The INIBs observed in bronchial and bronchiolar epithelial cells were labeled for BAdV3 (Fig. 4).
We extracted DNA from formalin-fixed, paraffin-embedded (FFPE) pneumonic lung tissue. A library was constructed using 600 ng of genomic DNA (Hyper PCR-free library prep kit, Kapa Biosystems; SciClone NGS instrument, Perkin Elmer). Size selection was accomplished through a paramagnetic bead cleanup after DNA fragmentation with a target insert size of 350 bp. The concentration of the final library was accurately determined through qPCR (Kapa Biosystems), and the library was diluted to 2.5 µM and sequenced (NovaSeq6000; Illumina) generating 2 × 150 paired-end reads.
The sequence reads were originally aligned to the bovine adenovirus reference genome using a server running software v.07.021.609.3.9.3 (v.3.9.3 Dragen; Illumina). Because mapping rates were lower than expected and there were regions of low coverage to that reference genome, we suspected that another virus would likely serve as a better reference. Sequences from the genomic alignments were extracted and a search for better matches was performed using BLASTN (https://blast.ncbi.nlm.nih.gov/Blast.cgi). Most of the top alignments were to OdAdV2 isolate 1319 genomic sequences. The genome analysis was repeated using the OdAdV2 isolate 1319 (NC_034834.1) reference genome, and variants were called to identify the percent identity between the isolate and OdAdV2. The OdAdV2 genome is 33,945 bases in length. The nucleotide sequence of the nucleic acid extract was 99.6% identical to the OdAdV2 isolate 1319 reference (128 single nucleotide changes) with an additional 20 small indels 1–4 nucleotides in length.
At the DNA sequence level, our nucleic acid extract had 3 nucleotide variants within the polymerase gene with respect to the OdAdV2 reference. The nucleotide sequence of the DNA-dependent polymerase gene from the genomic alignments was extracted using IGV (https://software.broadinstitute.org/software/igv/) and translated into a protein sequence. The protein sequence was aligned to the OdAdV2 DNA polymerase using BLASTP (https://blast.ncbi.nlm.nih.gov/Blast.cgi) to identify differences in the protein sequences. Of the 3 nucleotide variants, 1 is a synonymous substitution. The other 2 variants are adjacent and result in a single amino acid change (from leucine to arginine) in the translated protein sequence.
Respiratory disease is highly prevalent in deer, yet comparatively few reports have delineated the common etiologies involved and their interplay. Similar to production calves, farm-raised fawns are particularly susceptible to environmental stress–induced bacterial and viral pneumonias leading to high death losses. A study reported that half of all mortalities in 160 captive WTD occurred in animals < 1-y-old, with 42% of those deaths associated with respiratory disease. 1 Although that study determined that bacterial etiologies such as Fusobacterium spp., Mannheimia haemolytica, Trueperella pyogenes, and Escherichia coli comprised most of these pneumonias, viral pneumonias caused by parainfluenza, 4 coronavirus, 4 astrovirus, 13 and adenoviruses have been reported.
Reported deer adenoviruses include OdAdV116,17 and OdAdV2. 11 OdAdV1 infection, responsible for ADH in many deer species, is characterized by widespread vasculitis and endothelial INIBs,6,9,12,14–17 neither of which were noted in our case. We retrieved no cases of natural disease associated with OdAdV2 in a search of Google, CAB Direct, PubMed, Scopus, and Web of Science, suggesting that no descriptions of this condition have been reported in WTD. In the one report of experimental infection of WTD with OdAdV2, the gross lesions consisted of multifocal pulmonary congestion and edema. 11 Similar gross lesions have been described in calves infected with BAdV3. 10 Histologic lesions of experimental OdAdV2 infection in deer included mild neutrophilic bronchiolitis, thymic hemorrhage, lymphoid depletion, and hyperplasia of bronchiolar-associated lymphoid tissues (BALT) with no mention of INIBs. 11 Unfortunately, only lung tissue was available for histologic evaluation in our case; hence, we cannot confirm if thymic hemorrhage and lymphoid depletion were present. The microscopic lesions observed in our case most closely resemble those observed in BAdV3-infected cattle,3,10 namely necrosuppurative bronchointerstitial pneumonia with prominent INIBs. The lesions also resemble those described in a case of adenoviral pneumonia in a reindeer caused by a novel virus that is closely related to both OdAdV2 and BAdV3. 5
Genetic analysis based on the hexon gene indicates that OdAdV2 has 76% homology to BAdV3, 11 which explains its cross-reactivity with anti-BAdV3 antibodies. Although this cross-reactivity suggests that interspecies transmission is plausible, there is no evidence that OdAdV2 causes disease in non-cervids, to date. Although OdAdV2 has been propagated in bovine-derived cells, it reached significantly higher titers in cervid-derived cells, and antibody titer levels in dairy cattle co-housed with exposed WTD fawns were higher for BAdV3 than OdAdV2. 11
The clinician described our case as a chronic “poor-doer” since birth. Adenoviruses typically cause significant disease only in young or immunocompromised animals. 8 It is possible that the fawn experienced failure of passive transfer, and this could have predisposed it to chronic endoparasitism and eventual adenoviral pneumonia. The clinician reported that although most fawns at this farm develop respiratory signs, few individuals die (Hoskins EC, pers. comm., 2023 Jan 16). Additional study is needed to determine if OdAdV2 is circulating within the herd and if other respiratory mortality cases are associated with this virus. OdAdV2 pneumonia may be a rare event. A retrospective serologic survey 11 showed that the virus had been circulating in a herd for ≥ 14 y with no report of clinical disease, and viral inoculation of immunocompetent fawns resulted in only mild disease.
The possible contribution of bacterial infection to the pneumonia in our case cannot be excluded, but bacteria were not observed with either H&E or Gram stains. Bacterial culture was not performed because only formalin-fixed lung was submitted.
OdAdV2 should be considered as a potential cause of pneumonia in WTD, especially in younger immunocompromised animals. The virus is closely related to BAdV3 and may cross-react with BAdV3 antibodies.
Footnotes
Acknowledgements
We thank Drs. Aline Rodrigues Hoffmann, Mayane Faccin, and Caitlin Older for assistance with photographs.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
