Abstract
In vitro susceptibilities of 29 strains of Arcanobacterium pyogenes isolated from lung lesions of white-tailed deer (Odocoileus virginianus) with pneumonia were determined using the broth microdilution method to ascertain efficacious treatment options for pneumonic white-tailed deer. All 29 A. pyogenes strains tested were susceptible to ceftiofur, spectinomycin, tiamulin, and trimethoprim–sulfamethoxazole but were resistant to both danofloxacin and sulfadimethoxine. Likewise, all 29 isolates were either fully susceptible or intermediately susceptible to gentamicin (25 susceptible; 4 intermediate) and tulathromycin (25 susceptible; 4 intermediate). At least one isolate of A. pyogenes tested was resistant to ampicillin, chlortetracycline, clindamycin, enrofloxacin, florfenicol, oxytetracycline, penicillin, and tilmicosin suggesting their ineffectiveness in treating A. pyogenes–associated lung infections in white-tailed deer. Minimum inhibitory concentration (MIC) data for tylosin and neomycin could not be interpreted due to unavailability of Clinical and Laboratory Standards Institute (CLSI)-approved breakpoints for these 2 agents. In summary, based on MIC values, ceftiofur, spectinomycin, tiamulin, and trimethoprim–sulfamethoxazole are more efficacious than other antimicrobial agents for treating A. pyogenes–related pneumonia in white-tailed deer. However, ceftiofur may be preferred over the other 4 drugs as it is being widely used to treat respiratory disease in cattle and other animal species, as well as is available for single dose parenteral administration.
Keywords
Farming of white-tailed deer (Odocoileus virginianus) is a growing sector of agriculture in the United States and around the world. It is perhaps the fastest growing industry in rural areas in the United States. 1 Deer are raised commercially for the production of venison, harvest of antlers, urine collection, and for the population of hunting preserves. 1 According to the National Agricultural Statistical Service, in the year 2007, there were approximately 5,654 deer farms in the United States, a 15% increase from the previous census in 2002. 29 There is limited information available on the pathogens of farmed white-tailed deer, but a few reports on causes of mortality in captive deer have been published. The studies show that respiratory infections are one of the most common causes of morbidity and mortality in captive white-tailed deer herds.3,4,12 Although in most studies the etiologic agents were not identified, 3 studies in which bacteria were cultured from the tissues of dead white-tailed deer reported that Arcanobacterium pyogenes was the most common agent isolated from pneumonic lungs.11,12,17
Arcanobacterium pyogenes (formerly Actinomyces pyogenes and Corynebacterium pyogenes) is a Gram-positive, nonmotile, facultatively anaerobic, pleomorphic bacterium.23,26,27 It is a common inhabitant of the mucous membranes as well as an opportunistic pathogen of animals.23,26,27 In domesticated and free-ranging ruminants, A. pyogenes produces a wide range of suppurative disease conditions such as pneumonia, mastitis, abscessation, endometritis, pyometra, and umbilical infections, which involve visceral organs, skin, and joints. *
Despite its importance as a cause of fatal pneumonia, antimicrobial susceptibility of A. pyogenes isolated from the lungs of white-tailed deer has not previously been investigated. One study has looked into antimicrobial susceptibility patterns of A. pyogenes isolated from white-tailed deer with necrobacillosis; however, none of the A. pyogenes strains included in the study originated from the lungs. 5 There are currently no antibiotics labeled in the United States for use in cervids, and treatment options are therefore limited to off-label use of medications approved for cattle and other food animal species. The objective of the current study was to investigate the susceptibility of white-tailed deer lung isolates of A. pyogenes to antimicrobial agents that are used to treat pneumonia in ruminants. Such susceptibility data can be used to develop more targeted and efficacious treatments for pneumonic white-tailed deer.
In the present study, the susceptibility profiles of 29 strains of A. pyogenes isolated from the lungs of captive white-tailed deer (n = 29) with gross pathologic lesions of pneumonia to 18 antimicrobials were examined. The cases were derived from diagnostic service submissions to the Animal Diagnostic Laboratory at The Pennsylvania State University (University Park, Pennsylvania) for a period of 6 months from July 2009 to January 2010. Deer originated from 26 different farms representing 3 U.S. states (Pennsylvania: 23 farms; Ohio: 2 farms; Missouri: 1 farm). Of the 29 deer, 17 were males (<1 year, n = 3; 1–5 years, n = 12; >5 years, n = 1; age unspecified, n = 1), and 10 were females (<1 year, n = 1; 1–5 years, n = 6; >5 years, n = 2; age unspecified, n = 1 ), whereas the gender of 2 were unspecified (<1 year, n = 1; 1–5 years, n = 1). Bacterial isolates were confirmed as A. pyogenes by colony morphology on blood agar, Gram-staining, conventional biochemical analysis, and 16S ribosomal RNA gene sequencing. Antimicrobial drugs were selected based on the availability and label claims for treating pneumonia in food-producing animals.
Antimicrobial susceptibility testing was performed with an automated system a using the bovine/porcine (with tulathromycin) minimum inhibitory concentration (MIC) format (BOPO6F) in accordance with the manufacturer’s instructions and Clinical and Laboratory Standards Institute (CLSI) guidelines. 30 The antibacterial agents included in the selected plate format were ampicillin, ceftiofur, chlortetracycline, clindamycin, danofloxacin, enrofloxacin, florfenicol, gentamicin, neomycin, oxytetracycline, penicillin, spectinomycin, sulfadimethoxine, tiamulin, tilmicosin, trimethoprim– sulfamethoxazole, and tulathromycin. Although neomycin is not indicated for treating animals, it was included for quality control purposes. Arcanobacterium pyogenes isolates were grown on 5% sheep blood agar b and incubated overnight at 35–37°C. Several isolated colonies were suspended in trypticase soy broth or demineralized water, and the suspension was adjusted to approximate a 0.5 McFarland turbidity standard. A 10-µl volume of the adjusted suspension was then transferred into 10 ml of Mueller–Hinton broth with TES biological buffer (N-Tris[hydroxymethyl]methyl-2-aminoethanesulfonic acid) and lysed horse blood a to give a final inoculum density of approximately 5 × 105 colony-forming units/ml in the wells of the broth microdilution trays. It was necessary to supplement the broth with 10% fetal calf serum, c a modification to the CLSI methodology required due to the fastidious nature of A. pyogenes. The inoculums (50 µl) were dispensed into each well of the 96-well plates. a The plates were sealed and incubated at 35–37°C for 18–24 hr. The MIC value was defined as the lowest antimicrobial concentration that resulted in no growth of the isolate.
The MIC results were interpreted according to the breakpoints of the CLSI guidelines. 30 Since CLSI-approved MIC breakpoints are not available for A. pyogenes from any animal species, the CLSI-approved MIC breakpoints for other veterinary pathogens were used for interpreting MIC values (Table 1). In detail, the CLSI-approved MIC breakpoints for bovine respiratory disease pathogens were used whenever they were available. In the instances where MIC breakpoints for bovine respiratory disease pathogens were not available, CLSI-approved MIC breakpoints for other pathogens were used in the following order based on their availability: 1) swine respiratory pathogens, 2) undefined veterinary pathogens, and 3) Staphylococcus species. Quality control testing was performed weekly by using Staphylococcus aureus ATCC 29213, d Enterococcus faecalis ATCC 29212, d Escherichia coli ATCC 25922, d and Pseudomonas aeruginosa ATCC 27853 d bacterial strains.
Concentrations and breakpoints of antibacterial agents used to test antimicrobial susceptibility of 29 Arcanobacterium pyogenes strains isolated from white-tailed deer (Odocoileus virginianus) with pneumonia.
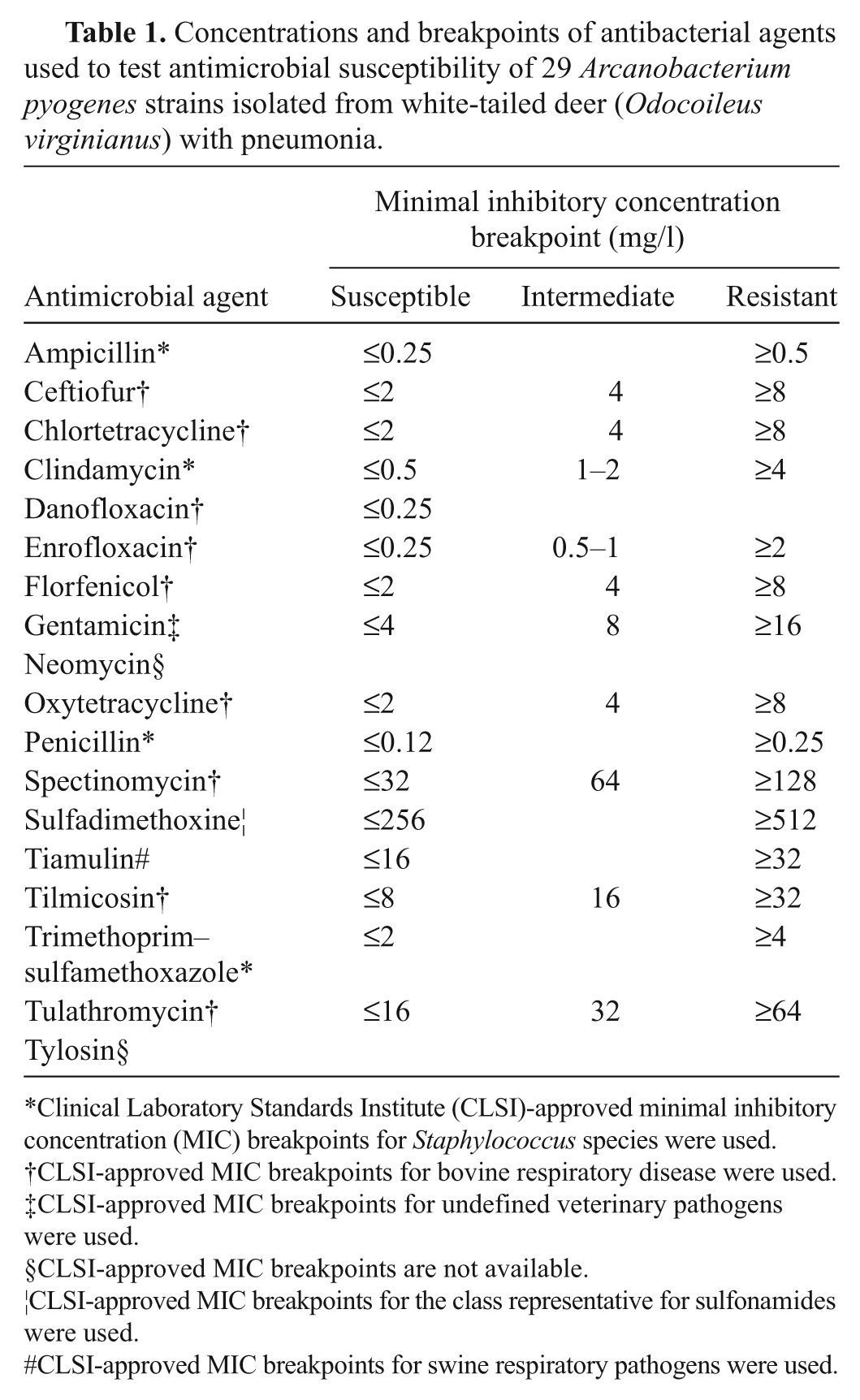
Clinical Laboratory Standards Institute (CLSI)-approved minimal inhibitory concentration (MIC) breakpoints for Staphylococcus species were used.
CLSI-approved MIC breakpoints for bovine respiratory disease were used.
CLSI-approved MIC breakpoints for undefined veterinary pathogens were used.
CLSI-approved MIC breakpoints are not available.
CLSI-approved MIC breakpoints for the class representative for sulfonamides were used.
CLSI-approved MIC breakpoints for swine respiratory pathogens were used.
Table 2 shows the frequency of occurrence of MICs of the antimicrobial agents and overall antimicrobial resistance or susceptibility profiles of A. pyogenes strains. All 29 A. pyogenes strains included in the current study were sensitive to ceftiofur, spectinomycin, tiamulin, and trimethoprim–sulfamethoxazole with MIC values of ≤0.25 mg/l, ≤1 mg/l, ≤16 mg/l, and ≤2 mg/l, respectively. Also, all the isolates were either fully or intermediately susceptible to gentamycin and tulathromycin. In detail, for gentamicin, 25 (86.2%) isolates were fully susceptible (MIC ≤4 mg/l), and 4 (13.7%) were intermediately susceptible (MIC = 8 mg/l). For tulathromycin, 25 (86.2%) isolates were fully susceptible with an MIC of ≤16 mg/l, and 4 (13.7%) isolates were intermediately susceptible with an MIC of 32 mg/l. In contrast, all the strains tested had an MIC of >256 mg/l to sulfadimethoxine and ≥0.5 mg/l to danofloxacin, and therefore were resistant to both agents. The effectiveness of tylosin in treating pneumonia associated with A. pyogenes could not be ascertained, as no veterinary species-specific or human-specific interpretive criterion was available. At least one strain of A. pyogenes was resistant to ampicillin, clindamycin, chlortetracycline, enrofloxacin, florfenicol, oxytetracycline, penicillin, and tilmicosin. In detail, for ampicillin, 24 (82.8%) isolates were fully susceptible (MIC ≤0.25 mg/l), and 5 (17.2%) were resistant (MIC ≥0.5). For clindamycin, 20 (69%) isolates were fully susceptible (MIC ≤0.25 mg/l), 4 (13.7%) were intermediately susceptible (MIC = 1–2 mg/l), and 5 (17.2%) were resistant (MIC ≥4). For chlortetracycline, 11 (37.9%) isolates were fully susceptible (MIC ≤2 mg/l), 4 (13.7%) were intermediately susceptible (MIC = 4 mg/l), and 14 (48.3%) were resistant (MIC ≥8). For enrofloxacin, 27 (93.1%) isolates were intermediately susceptible (MIC = 0.5–1 mg/l), and 2 (6.9%) were resistant (MIC ≥2). For florfenicol, 25 (86.2%) isolates were fully susceptible (MIC ≤1 mg/l), 3 (10.3%) were intermediately susceptible (MIC = 4 mg/l), and 1 (3.4%) isolate was resistant (MIC ≥8). For oxytetracycline, 11 (37.9%) isolates were fully susceptible (MIC ≤2 mg/l), 5 (17.2%) were intermediately susceptible (MIC = 4 mg/l), and 13 (44.8%) were resistant (MIC ≥8 mg/l). For penicillin, only 24 (82.8%) isolates were susceptible with MIC values of ≤0.12 mg/l. Finally, for tilmicosin, 19 (65.5%) isolates were fully susceptible (MIC ≤8 mg/l), 3 (10.3%) were intermediately susceptible (MIC = 16 mg/l), and 7 (24.1%) were resistant (MIC ≥32 mg/l).
Minimal inhibitory concentration (MIC) of the 18 antimicrobial agents for 29 Arcanobacterium pyogenes strains from white-tailed deer (Odocoileus virginianus) with pneumonia.
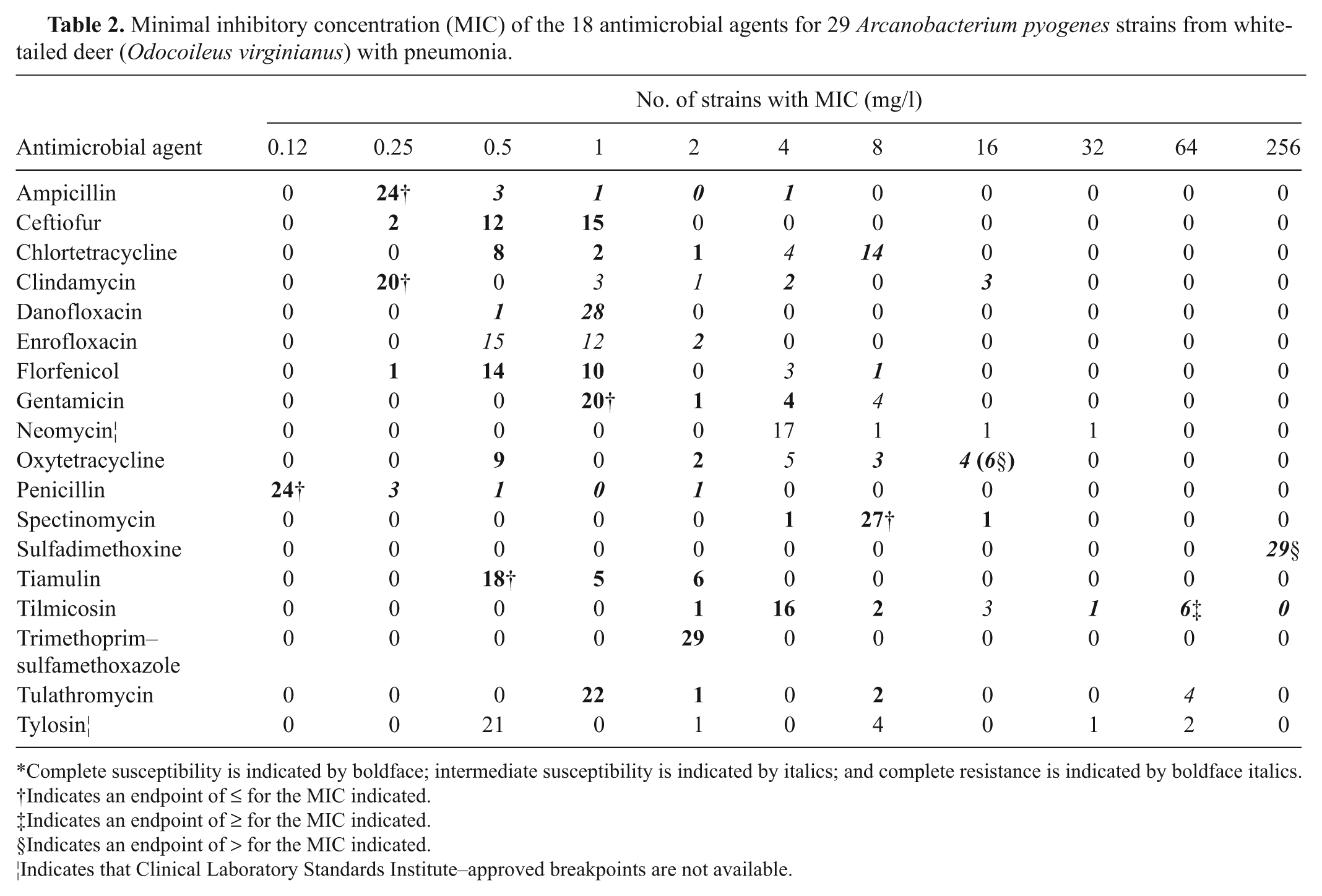
Complete susceptibility is indicated by boldface; intermediate susceptibility is indicated by italics; and complete resistance is indicated by boldface italics.
Indicates an endpoint of ≤ for the MIC indicated.
Indicates an endpoint of ≥ for the MIC indicated.
Indicates an endpoint of > for the MIC indicated.
Indicates that Clinical Laboratory Standards Institute–approved breakpoints are not available.
Although there have been several studies on the antimicrobial susceptibilities of A. pyogenes causing disease in cattle, goats, sheep, and swine, only 1 study has looked into the susceptibilities of A. pyogenes isolated from white-tailed deer.5,6,10,14,31 Nevertheless, the study evaluated A. pyogenes isolated from various lesions of necrobacillosis but did not involve a single strain of A. pyogenes isolated from deer lung. 5
All A. pyogenes isolates tested were highly susceptible to ceftiofur, spectinomycin, tiamulin, and trimethoprim–sulfamethoxazole, suggesting their potential use in treating A. pyogenes–associated pneumonia. Ceftiofur, a third generation cephalosporin, belongs to the beta-lactam group of antibiotics,18,30 is effective against both Gram-positive and -negative bacteria, and is administered subcutaneously (cattle) or intramuscularly (pigs) as a single-dose treatment.18,30 Ceftiofur is specifically indicated against both bovine respiratory pathogens (Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni) and swine respiratory pathogens (Actinobacillus pleuropneumoniae, P. multocida, Salmonella choleraesuis, and Streptococcus suis). The drug has a 13-day pre-slaughter withdrawal period in cattle and 14-day withdrawal period in pigs when following label use as a single treatment. Ceftiofur is efficacious against anaerobic bacteria as well, which may be useful in treating infections due to A. pyogenes, because most A. pyogenes infections including pneumonia in white-tailed deer are co-infected with anaerobic bacteria such as Fusobacterium species. 12 Based on the MIC values obtained in the present study, spectinomycin is another drug of choice that can be effectively used to treat A. pyogenes–associated pneumonia in white-tailed deer. The drug is an aminocyclitol, and has been used in cattle to treat respiratory diseases including those associated with Mycoplasma.19,30 In cattle, spectinomycin is administered subcutaneously once daily for 3–5 days. Tiamulin, another drug of choice based on MIC values, is a semisynthetic derivative of pleuromutilin, and is extensively used in veterinary medicine. Although tiamulin is effective against Gram-positive and -negative bacteria including anaerobes, it is available only for oral medication, which may preclude its use in ruminants. 30 The MIC of orally administered drugs may not reliably predict in vivo efficacy even in monogastric species such as poultry and swine in which they are commonly used. Trimethoprim–sulfamethoxazole, a combination of trimethoprim and a sulfonamide, has a broad spectrum of activity due to synergism.20,30 However, in the United States, trimethoprim–sulfamethoxazole is not indicated for food-producing animals. The other antibacterial agents that produced acceptable MIC values (bacteria were either fully susceptible or intermediately susceptible with no resistant strains) are gentamicin and tulathromycin. Of these 2 antibacterial drugs, tulathromycin, which is a bactericidal macrolide, is preferred over gentamycin. Similar to ceftiofur, tulathromycin has been widely used in cattle to treat bovine respiratory disease complex where it is administered as a single subcutaneous injection, and in pigs to treat swine respiratory disease complex where it is administered as a single intramuscular injection.21,30 Out of 29 A. pyogenes isolates tested, 25 were fully susceptible to tulathromycin, whereas the other 4 were intermediately susceptible. It is a widely accepted norm that A. pyogenes is susceptible to penicillins. 30 However, out of 29 A. pyogenes strains tested in the current study, 5 strains were resistant to ampicillin and penicillin, demonstrating the importance of antimicrobial susceptibility testing before attempting to treat A. pyogenes–associated infections in deer.
The results of the present study demonstrated that A. pyogenes was resistant to some of the antimicrobial drugs that are commonly used to treat respiratory infections in food-producing animals, in particular, cattle and pigs. A notable discrepancy was that 5 (17.2%) isolates of A. pyogenes tested in the current study were resistant to penicillin and ampicillin, whereas A. pyogenes from bovine and porcine species, as well as A. pyogenes from non-lung lesions of white-tailed deer with necrobacillosis tested in previous studies, were susceptible to penicillin.5,6,31 Also, 1 study observed a difference between susceptible patterns of A. pyogenes isolated from bovine udder and bovine uterus to oxytetracycline and trimethoprim–sulfadiazine. 6 Most of the isolates that originated from the uterus were resistant to oxytetracycline and trimethoprim–sulfadiazine, whereas udder isolates were sensitive to both drugs. 6 Another study found that bovine and porcine isolates of A. pyogenes behaved differently to certain antibacterial drugs where more isolates of porcine origin were resistant to oxytetracycline, erythromycin, tilmicosin, and lincomycin as compared to those isolated from cattle. 31 Together, these studies suggest that antimicrobial susceptibility patterns of A. pyogenes may vary depending on the host species and the anatomical site from which they were isolated. To ascertain whether this difference is in fact due to species or niche specificity of certain A. pyogenes strains, molecular characterization of A. pyogenes isolated from various host species and anatomical sites is warranted.
Footnotes
a.
Sensititre®, Trek Diagnostic Systems, Cleveland, OH.
b.
Remel Inc., Lenexa, KS.
c.
Sigma-Aldrich, St. Louis, MO.
d.
American Type Culture Collection, Manassas, VA.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
