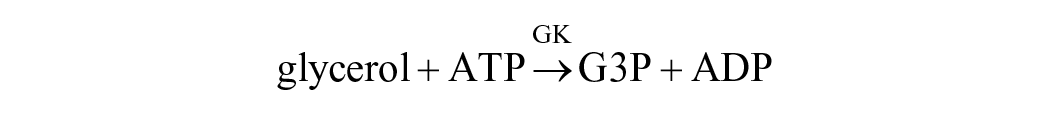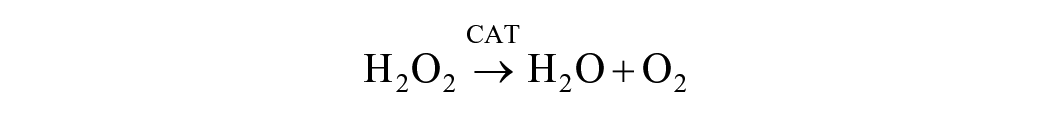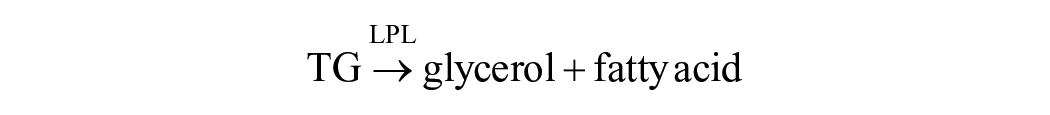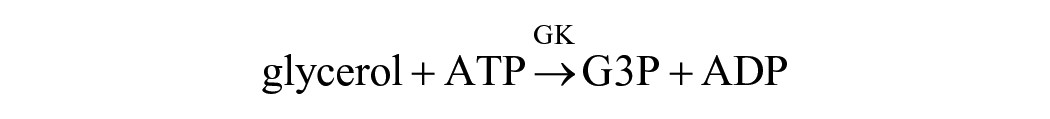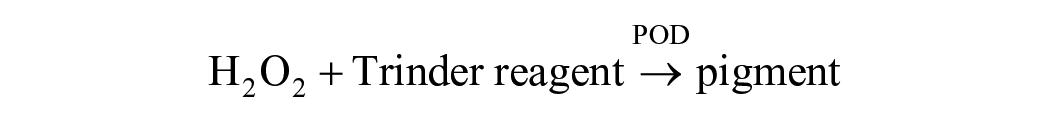Abstract
Two methods of measuring triacylglycerol (TG) in the blood are used in clinical laboratories. The glycerol-blanked TG analysis method is used primarily in Japan; the total glyceride measurement method is used in most countries, including the United States. The latter method includes free glycerol in the blood, which is known to be slightly higher in humans. However, the extent to which the 2 methods differ in companion animals is unknown. We used cryopreserved blood from dogs and cats that visited a secondary veterinary hospital to investigate the relationship between the data obtained using the 2 TG analysis methods. The median ratio (5th and 95th percentiles) of total glyceride measurement:glycerol-blanked TG measurement ratios was 1.24 (1.08 and 1.77) for dogs and 2.00 (1.44 and 3.66) for cats, with the total glycerol method clearly having higher values. The Passing–Bablok regression equation comparing the total glyceride method (y) and glycerol-blanked method (x) was y = 1.049x + 0.119 for dogs and y = 1.476x + 0.177 for cats. The estimated free glycerol value calculated from the difference between the 2 TG measurements was strongly correlated with the measured free glycerol value (dog, r = 0.7905; cat, r = 0.8708), indicating that free glycerol in the blood was the cause of the TG measurement discrepancy. Therefore, the TG concentrations obtained from total glyceride assays in dogs and cats may contain non-negligible levels of free glycerol.
Animal clinical testing, particularly blood testing, makes extensive use of standardized analyzers and clinical reagents designed for human medical use. One example is triacylglycerol (TG) measurement reagents, which are used routinely in the veterinary field. TG is an energy source composed of a glycerol backbone with 3 fatty acids attached. Although monoglycerides and diglycerides are present in small amounts in the blood, TG accounts for nearly all neutral fat in humans.9,15 Fatty acids are β-oxidized in mitochondria and used to generate ATP, and the glycerol backbone is also used to generate ATP in the glycolytic system.9,15 Blood TG can be derived from dietary sources or synthesized in the body; in healthy humans and other animals, blood TG levels increase after eating.9,12,20 Clinically, TG is used as an indicator of nutritional status and to investigate the pathophysiology of hyperlipidemia, which occurs in humans,13,18 dogs,3,19 and cats. 11
Among the TG-measuring reagents used in general clinical testing, including the reagents compared in our study, lipoprotein lipase cleaves fatty acids from TG, and the concentration of the remaining glycerol skeleton is measured rather than TG itself. Although international standardization in the field of human clinical testing is progressing, TG measurements have not been standardized. Assay methods are classified into 2 categories. One method, primarily used in Japan, employs wet reagents based on the glycerol-blanking TG assay method recommended by the Japanese Society of Clinical Chemistry (JSCC). 2 This routine testing method eliminates free glycerol from the blood sample as a first reaction and allows lipoprotein lipase to act on TG as a second reaction, as follows:
Free glycerol scavenging reaction:
TG analytical reaction:
ADP = adenosine diphosphate; ATP = adenosine triphosphate; CAT = catalase; G3P = glycerol 3-phosphate; GK = glycerol kinase; GPO = glycerol 3-phosphate oxidase; LPL = lipoprotein lipase; POD = peroxidase.
Hence, the concentrations of TG, monoacylglycerol, and diacylglycerol-derived glycerol are determined. However, most countries have adopted the reference method of the U.S. Centers for Disease Control and Prevention, 4 which determines total glycerol in the blood, including free glycerol as well as triglyceride-derived glycerol. In this method, no free glycerol scavenging reaction occurs. Total glycerol may also be measured using a dry reagent analysis method, which is the method that is used most extensively in veterinary medicine.5,7
In humans, free glycerol accounts for ~5% of total glycerol; 6 therefore, although some researchers believe that no clinically significant effects would occur without using the glycerol-blanked TG measurement method,8,10 others contend that the total glycerol measurement method overestimates TG and is clinically problematic. 14 The debate persists over which method is more appropriate. Therefore, clinically measured TG may differ in test item composition, depending on the TG measurement method used. This variability applies to both human and animal clinical tests that use human reagents. However, there is limited discussion on the distinctions among the various TG measurement methods in the clinical examination of companion animals, and the extent of divergence of these methods remains unclear.
We investigated the relationship between the glycerol-blanked TG and total glycerol measurement methods in dogs and cats. We hypothesized that the difference between the methods corresponds to the free glycerol concentration in blood. To test this hypothesis, we measured the free glycerol concentration. We also compared our results with human blood tested under the same conditions.
Materials and methods
Ethics approval for research involving animals and humans
We used residual specimens from humans, dogs, and cats after completion of clinical examinations. Blood was not drawn from the animals in our investigation; therefore, the study was not subject to the ethical considerations of the Okayama University of Science Ethics Committee on Clinical Investigation (examination 2020-0007). Adult human specimens were anonymized. The study design was approved by the Ethical Committee for Medical and Health Research Involving Human Subjects of the Okayama University of Science (approval 2–11). In addition, blood donor consent was waived by the internal ethics committee of the university.
Sample collection
Human blood samples were used after a full anonymization procedure. Individual dog and cat information and owner information were also used after being anonymized. Residual blood samples were collected from 135 dogs and 103 cats and stored for 6 mo at the Biomedical Science Examination and Research Center, Okayama University of Science (Imabari, Ehime, Japan). Residual blood samples were also collected from 96 adult humans who underwent health checkups at Ehime Rinken (Matsuyama, Ehime, Japan). For both humans and animals, blood samples were collected under fasting conditions. When comparing 2 analytical methods, it is necessary to analyze samples covering a wide range of concentrations, from low to high, and collected from subjects with diverse backgrounds. Therefore, we applied no health selection criteria when collecting blood samples from animals or humans, and no exclusion was made based on the presence of hemolysis, hyperbilirubinemia, or chylous blood.
Lithium heparin was used as an anticoagulant in dog and cat blood samples. No anticoagulant was used for human blood samples. Blood samples were centrifuged (1,500 × g, 10 min) to separate serum or plasma, which was then frozen at −80°C until further analyzed. The time elapsed from thawing to analysis was <3 h. This was the second thawing for human samples; for cat and dog samples, this was the first thawing.
Analysis of blood TG values
The glycerol-blanked TG analysis was conducted using commercial reagents for automated biochemical analyzers (Cholestest TG, JSCC transferable method; Sekisui Medical). Similarly, the total glyceride assay was performed using commercial reagents (Cholestest N TG; Sekisui Medical). Calibration was performed using commercial equipment (Cholestest N calibrator; Sekisui Medical). Commercial analytical QC serum samples were used (QAP-Trol 1X and 2X; Sysmex). In the glycerol-blanked TG analysis, the validity of the calibration was confirmed by ensuring that it fell within the manufacturer’s specified range for QAT-Trols. However, for the total glycerol method, given the unavailability of QC materials assigned using this method, the validity of the calibration was confirmed using the TG calibrator. To maintain analytical accuracy in both methods, QAP-Trols were utilized to confirm that the CV was <5% in parallel accuracy and intra-laboratory reproducibility
Blood TG (the glycerol-blanked TG analysis and total glyceride measurement) and free glycerol were analyzed with an automatic biochemistry analyzer (type 3100; Hitachi High-Tech), according to the manufacturer’s instructions. Analytical conditions, such as the mixing ratio of specimens and reagents and measurement time, were set to the recommended values of the reagent manufacturer. The measurement range for the 2 methods was 0.03–22.6 mmol/L (3–2,000 mg/dL).
Free glycerol in blood was measured using a liquid glycerol standard (Enzytec; R-Biopharm). Calibrators and QC materials were obtained from R-Biopharm. The measurement range of this analysis was 0.11–2.71 mmol/L (1–25 mg/dL).
Statistical analysis
The TG values detected using the glycerol-blanked TG (x-axis) and total glycerol (y-axis) measurement methods were plotted in a scatter diagram. The correlation between the measured TG values of both methods was determined using Pearson correlation coefficients using SPSS Statistics 19 for Windows (IBM). Furthermore, a validation support application (v.6.3, JSCC, https://jscc-jp.gr.jp/file/senmon/Validation-Support-V63.xlsm) was used to perform a Passing–Bablok regression analysis to confirm the relationship between the 2 measurement methods. Statistical significance was set at p ≤ 0.05. To evaluate the degree of agreement and type of error between TG measurements for the 2 assays, Bland–Altman plots were generated, depicting the mean of the glycerol-blanked TG and total glycerol measurements on the x-axis and the difference between the 2 measurements on the y-axis. Normality of the data was confirmed using QQ plots and the Shapiro–Wilk test. If normality was confirmed, the agreement limits for both of the TG analyses were calculated as x̄ ± 1.96SD, with the upper and lower ranges displayed on the graph.
Results
Distribution of TG concentrations in the glycerol-blanked TG analysis and total glyceride measurement method
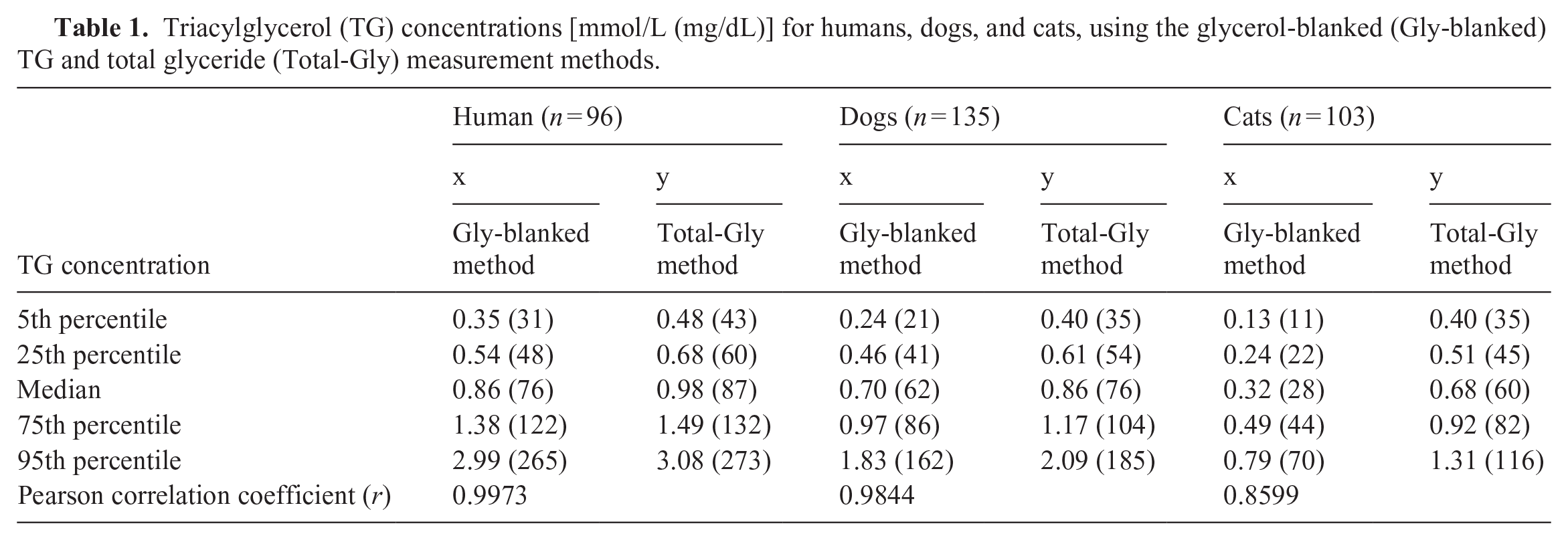
Across all samples from all species, the TG measurement values obtained using the total glyceride measurement method exceeded those obtained using the glycerol-blanked TG analysis (Table 1). The median (5th and 95th percentiles) ratio of total glyceride measurement:glycerol-blanked TG measurements was 1.11 (1.02 and 1.42) in humans, 1.24 (1.08 and 1.77) in dogs, and 2.00 (1.46 and 3.74) in cats (Fig. 1).
Triacylglycerol (TG) concentrations [mmol/L (mg/dL)] for humans, dogs, and cats, using the glycerol-blanked (Gly-blanked) TG and total glyceride (Total-Gly) measurement methods.

The ratio of total glyceride measurement method to the glycerol-blanked triacylglycerol measurement method in humans, dogs, and cats.
There was a positive linear correlation between the 2 TG measurement methods in humans (Fig. 2A), dogs (Fig. 3A), and cats (Fig. 4A); however, a stronger linear distribution was observed in humans and dogs than in cats. The Pearson correlation coefficient was 0.9973 for humans, 0.9844 for dogs, and 0.8599 for cats, with cats scoring slightly lower than the others (Table 1). The Passing–Bablok regression equation was y = 0.988x + 0.113 for humans, y = 1.049x + 0.119 for dogs, and y = 1.476x + 0.177 for cats.

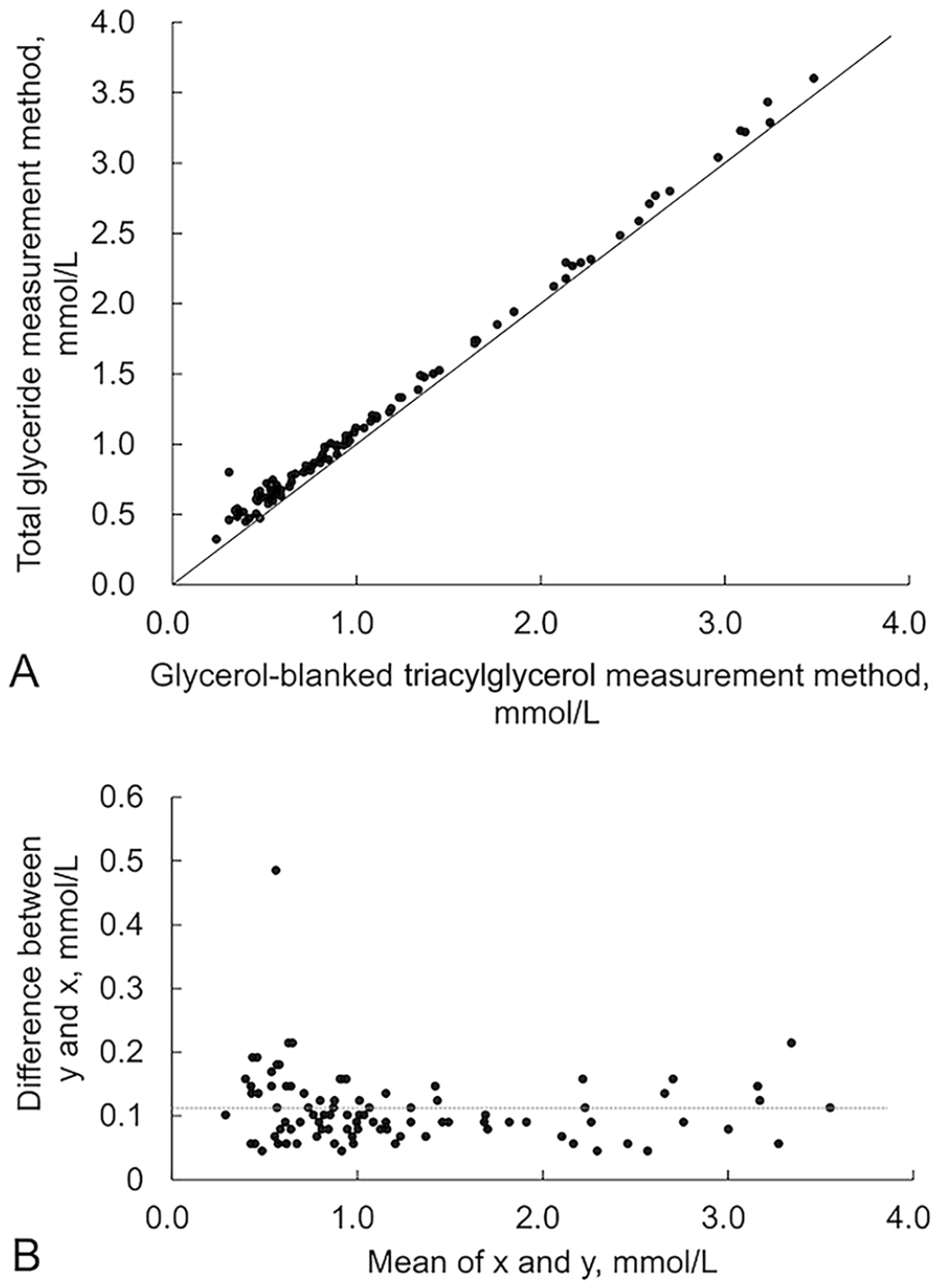

The Bland–Altman plot had a bias toward positive values on the y-axis (total glyceride measurement method – glycerol-blanked TG method) for humans, dogs, and cats, indicating a consistent systematic error between measurement values (Figs. 2–4). Specifically, the values obtained from the total glyceride measurement method were consistently higher than those from the glycerol-blanked TG analysis. In cats, the difference on the y-axis between the 2 methods increased as the mean value on the x-axis increased, indicating a proportional systematic error between the 2 methods. As the differences between the 2 methods did not have a normal distribution, the limit of agreement is not shown.
Free glycerol concentration in blood
The median (5th and 95th percentiles) measured free glycerol values for each species were: 0.22 (0.11–0.33) mmol/L [2 (1–3) mg/dL] for humans, 0.22 (0.0–0.43) mmol/L [2 (0–4) mg/dL] for dogs, and 0.43 (0.22–0.76) mmol/L [4 (2–7) mg/dL] for cats. Some dog samples fell below the lower limit of quantification of 0.11 mmol/L (1 mg/dL) for free glycerol.
The calculated free glycerol obtained by subtracting the glycerol-blanked TG analysis method from the total glyceride assay method was: 0.10 (0.05–0.19) mmol/L [1 (0–2) mg/dL] for humans; 0.16 (0.09–0.34) mmol/L [1 (1–3) mg/dL] for dogs; and 0.34 (0.17–0.68) mmol/L [3 (2–6) mg/dL] for cats. Measured free glycerol values and calculated values (Fig. 5) were strongly and positively correlated for humans (r = 0.7669), dogs (r = 0.7905), and cats (r = 0.8708).

Scatter plots of the measured free glycerol and calculated free glycerol values in
Discussion
We found that the values obtained using the total glyceride measurement method were higher than those from the glycerol-blanked TG assay across most human, canine, and feline samples. Furthermore, regression analysis of the measured values from the total glyceride (y) and glycerol-blanked TG (x) measurement methods revealed that the slope of the regression equation was closest to 1 in humans (0.988), followed by dogs (1.049) and cats (1.476). This indicates that the values from the 2 measurement methods for dogs and cats deviated more from each other than they did for humans. In particular, the discrepancy between the 2 measurement values was greater in cats. The differences were more pronounced in cats than in humans; the estimated free glycerol concentration calculated from the difference between the 2 TG assays was strongly correlated with the measured free glycerol in blood. This suggests that the difference between the 2 TG assays may be attributed to the inclusion of free glycerol in the total glycerol measurement method. Given that in cats, the difference between total glycerol and glycerol-blanked TG measurements is greater than that in humans and dogs, cats should not be treated identically.
Free glycerol concentrations in blood increase when TG is hydrolyzed by lipases during fasting. Hormone-sensitive lipase acts on TG degradation in adipose tissue, and its action is inhibited by insulin; therefore, the free glycerol concentration in blood is known to decrease with feeding.1,9 In our study, the blood samples from humans, dogs, and cats were collected during fasting. Therefore, it is presumed that the free glycerol concentration in blood was higher than that after eating, potentially representing another reason for the disparity between the results of the 2 TG assays.
The total glycerol measurement method has been found to be inadequate for TG assessment in laboratory animals, such as mice, Beagle dogs, and miniature swine, because of the influence of increasing free glycerol concentration during fasting.16,17 In these reports, there was a 1.5-fold difference between the TG values obtained using the 2 methods in Beagle dogs. We found that the median ratio of total glycerol:glycerol-blanked TG measurements in dogs was 1.24, which is similar to the values reported previously. 17
One limitation of our study is that the specimens were cryopreserved; therefore, the percentage of free glycerol may have been slightly higher than that in fresh blood. Furthermore, only one analysis reagent was used for the TG assay, and the reactivities of products from other companies may differ slightly.
Footnotes
Acknowledgements
We thank Chie Tanaka of the Biomedical Science Examination and Research Center, Okayama University of Science, for her assistance with the biochemical analysis of TG.
Declaration of conflicting interests
Akihisa Hata was awarded a contract research budget from Yamada Bee Company, for a study unrelated to the current research theme. The author declares no conflicts of interest regarding our study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.