Abstract
Carbon nanotubes bound to tumor specific antibodies offer specific treatment for cancer cells without affecting surrounding tissue. The present study seeks to affirm the initial results of CNTs in cancer therapy by investigating the toxicological effect in mice injected with CNT-Ab followed by microwave hypothermia. We were particularly interested in evaluating the biodistribution, toxicity, and immune response that may be elicited from CNT-Ab exposure in mice. 4–5 week old mice (C57BL/6) were injected with various concentrations and combinations of multiwalled carbon nanotubes (MWCNT) conjugated with specific prostate-specific membrane antigen (PMSA) antibodies. After 1-week post-injection, mice were sacrificed followed by the collection of blood separated into serum, liver, kidney and other tissues for further analysis. Serum total protein concentration across the treatment groups was varied. No significant changes in albumin levels were detected when compared to the control group (No Treatment). Group YE (.125 mg/ml anti-PSMA-MWCNT + Microwave) was found to have consistently high blood serum analyte levels, indicating impaired liver and kidney function. Likewise, groups YB (Microwave only), YF [.5 mg/ml anti-PSMA-MWCNT (No Microwave)], and YG (.5 mg/ml plain MWCNT + Microwave) seemed to show indications of impaired liver function. Analysis of gene expression revealed a significant impact on the NF-KB inflammatory response pathway. NF-KB gene was up-regulated relative to controls in all treatment groups. These results seem to suggest marginal toxicity from the injection of CNT-Ab followed by microwave hyperthermia in mice subjects.
Keywords
Introduction
Cancer is a cluster of diseases that involves abnormal and uncontrollable cell growth which has the potential to spread to other parts of the body. It is a genetic disease caused by the alteration of genes that control cell growth and division. With more than 100 types of cancer, it is the second highest leading cause of death in the United States. 1 The complexities of cancer make the disease extremely difficult to treat once it has invaded more than one tissue type. Cancer treatments such as chemotherapy, radiation therapy, and immunotherapy have serious side effects and the potential for biological resistance. Existing efforts that target single cellular abnormalities have been proven to be effective despite many obstacles. 2 Further, hyperthermia irradiation has been combined with various nanoparticles for reducing cancer proliferation in cancer cell lines. 3,4 Recently, microwave ablation randomized clinical trials on BCLC-A hepatocellular carcinoma was carried-out. Results suggested that, early application of this method of treatment is effective. 5 The present study utilized microwave hyperthermia irradiation of CNT-conjugated antibody to target tissues.
Prostate cancer is one of the most lethal malignancies to men, second to lung cancer. Roughly 1 in 9 men will be diagnosed with prostate cancer and 1 out of 41 will die from it. Options for treatment are commonly surgical resection, while the remaining alternatives such as chemotherapy and radiation therapy have large potential for healthy cell death and undesired side effects. 6 Other established targeting therapies can have great shortcomings and low success rates.
Carbon nanotubes (CNTs) are nanostructured materials that are smaller than a single nanometer at one characteristic structural length. 7 These materials have been explored for various uses because of their strength, tenacity and impact resistance. 8 In this particular study, we are interested in their applications in medicine. Previous research has examined the binding kinetics of CNTs with biological materials such as hemeproteins. 9 Carbon nanotubes conjugated to antibodies (CNT-Abs) offer a specific binding to cancer cells without association to other normal cell types. The strategy proposed in this work makes use of the extraordinary absorptive properties of carbon nanotubes toward microwaves to promote the localized heating of both the nanotube and its associated tumor in vivo, thus diminishing or eliminating associated side effects of current cancer treatments that typically damage surrounding tissues. The function of the CNT-Abs at the cellular level provides specialized targeting of desired cell types.
Previous studies have shown that CNTs have the potential to trigger inflammation, cytotoxicity and oxidative stress. However, there is not yet a full understanding of the toxicity of CNTs. 10 Further studies have demonstrated that the degree of toxicity is dependent on the surface chemistry of the CNTs. Functionalized carbon-based nanomaterials are expected to present a greater dispersibility in an aqueous medium in comparison to non-functionalized carbon-based nanomaterials. The increased dispersibility can trigger an increased cell response. CNTs associated with any metal have been found to have a higher chance for negative reactions. 11 The direct interaction between carbon-based nanomaterials and the cell can enhance oxidative stress within the cell due to CNTs ability to stimulate the generation of reactive oxygen species (ROS), which can damage lipids, carbohydrates, proteins, and DNA. This oxidative stress has been implicated in the induction of inflammation in many studies examining CNTs both in vivo and in vitro. 12 Effectiveness of CNT treatment has also been found to be dependent on the tissue type. For instance, CNTs introduced to lung tissue had a significant negative impact on the elderly with respiratory problems. 13
The research reported in this manuscript seeks to show preliminary data about the effectiveness of CNT-Abs for targeted cancer treatment and to explore potential systemic and localized impact of CNT-Ab treatment followed by microwave hyperthermia. Hyperthermic ablation will cause the CNTs to heat up, thereby killing the surrounding tissues. We are particularly interested in evaluating the biodistribution, toxicity, and immune response that may be involved from the combination of injected MWCNTs and microwave irradiation in the mice C57BL/6 model. This study could provide new specific and localized treatment options for a variety of cancers without the invasiveness of surgery or the risk of tissue damage as in chemotherapy and radiation.
Materials and methods
PSMA-MWCNT preparation
MWCNTs (Cheap Tubes Inc, Grafton VT) were partially oxidized for 2 hours via UVO cleaner. This process also further sterilized the nanotubes. Then, the materials were sonicated after which sterile NHS, EDAC and 0.5 m MES buffer were added to the mixture and stirred on a hot plate for an hour. The mixture was then vacuum filtered and dried overnight in a 70°C vacuum. This step created the necessary functional groups to form peptide bonds with the antibodies. PSMA antibody was then added to the MWCNT mixture and stirred with a siliconized stir bar on a hot plate for an hour. After stirring, the mixture was centrifuged for 5 minutes at 5,000 rpm. The concentration was adjusted using 0.5 M MES buffer.
Fluorescent microscopy was used to verify antibody conjugation. Specifically, anti-PSMA AlexaFluor488® (BioLegend, San Diego CA) was employed in this mice study. The human prostate-specific membrane antigen (PSMA) is a murine IgG antibody. It emits green fluorescence when excited with light at 488 nm wavelength. PSMA has been found to be highly expressed in prostate tumors. This protein is a type II transmembrane protein. A detailed characterization of the CNT-Abs can be found in the literature. 14
Experimental design
Approximately 4–5 week old male mice were assigned to 8 different treatment groups, each group containing six mice. As specified in Table 1, treatment groups vary in CNT concentration, microwave exposure, and presence of prostate cancer specific antibodies (Prostate membrane specific antigen-PSMA). The mice were injected with multiwalled carbon nanotubes (MWCNT) conjugated with cancer specific antibodies (CNT-Abs/ PSMA-MWCNT) (Figure 1) subcutaneously into both right and left flank and microwaved at 150 watts for 5 seconds. After 1-week post-injection, blood samples were collected. Additionally, tissues, such as liver, kidney, and brain tissue, were obtained for future examination. Microwave irradiation power and time, and CNT concentration were previously optimized, revealing the highest tolerable dose for the mouse model under study. Mice were exposed to different microwave energy levels from 500 J to 6300 J and were monitored for 48 hours for tolerance. 5 s at 150 W was the highest dose tolerated and was therefore chosen for the experiments reported herein. Details about the irradiation optimization experiments have been published separately. 15
Summary of treatment groups. Mice were assigned into a total of eight control, anti-PSMA-MWCNT plus/minus microwave treatment groups.

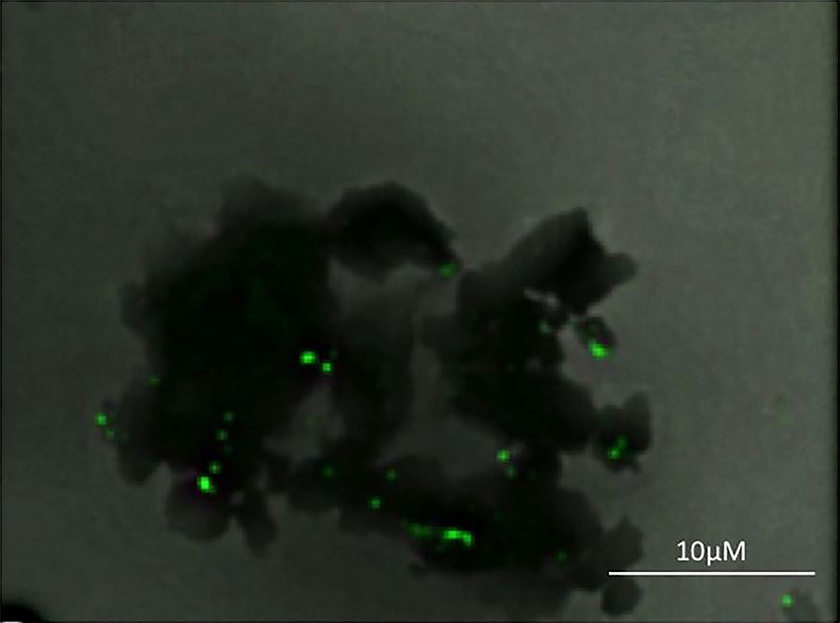
Functionalized multiwalled carbon nanotube used. Green color indicates the associated prostate cancer specific antibodies. The image corresponds to anti-PSMA conjugated CNTs imaged by a Zeiss Confocal Microscope model LSM 710 (Jena, Germany). The green color corresponds to the anti-PSMA AlexaFluor647 antibodies, which fluoresce green when excited at 647 nm.
Mice were housed at the Georgia Southern University Animal Facility (1176A Biological Sciences Fieldhouse). The animals were handled according to the principles outlined in the ILAR’s (Institute or Laboratory Animal Research) Guide for Care and Use of Laboratory Animals. Study protocols were reviewed and approved by Institutional Animal Care and Use Committee (IACUC Protocol Approval # I18024).
Serum samples were assayed to quantify systemic and localized impact of treatment. Further research onto the specific tissue types was conducted to analyze gene expression and the influence of treatment on immune and inflammatory responses.
Serum creatinine assay
The reagent was prepared to quantify creatinine using a 48 well plate. Reaction buffer was made up of a mixture of 1 mL creatinine sodium borate, 3 mL creatinine surfactant, and 2 mL creatinine NaOH. A standard was then prepared using a series of dilutions of the creatinine standard and HPLC-grade water. Samples ranging from A through H (Table 1) were analyzed using guidelines provided by the assay manufacturer [Cayman Chemical, Creatinine (serum) Colorimetric Assay, Ann Arbor Mi; Cat# 700460]. Approximately 15 mL of each sample was added to the respective wells along with 100 mL of creatinine reaction buffer and 100 mL of creatinine color reagent. The absorbance of the solutions was read at 495 nm, 1 and 7 minutes after incubation at room temperature. The creatinine concentrations were then determined via the standard curve.
Serum albumin assay
The level of albumin in serum was measured using a reagent kit provided by Pointe Scientific, Inc., (Canton, MI, Cat#: A7502-1L). Using guidelines provided by the kit manufacturer, albumin reagent was mixed with serum samples in their respective test tubes followed by incubation at room temperature. The UV spectrophotometer was reset, read, and recorded at 630 nm. Albumin concentrations were calculated using a standard curve generated by an albumin standard.
Serum liquid AST (aspartate aminotransferase) assay
The level of AST activity was also quantified in serum samples (Pointe Scientific, INC., Canton, MI, Cat#: A7561-450). Working reagents were pre-warmed followed by the addition of the respective serum samples using the assay protocol provided by the kit manufacturer. The absorbance was read and recorded in a UV spectrophotometer at 340 nm. Each sample was then returned to the incubator and the absorbance was read again after 1 minute and after 2 minutes. The average difference in absorbance per minute was calculated and multiplied by the factor 1768 to yield IU/L. Each sample was run three times, and each measurement was recorded in duplicates.
Tissue RNA extraction
Total RNA was extracted from liver, kidney, and brain tissues using Qiagen’s RNeasy mini RNA extraction kit. Approximately 20–30 mg of liver were weighed for samples A-H. Tissues were placed and homogenized in QIAzol Lysis reagent. Addition of gDNA eliminator solution and chloroform were performed followed by centrifugation at low temperature. Next, the RNA supernatant was removed, passed through an RNeasy spin column, and then the column was cleaned using RWT and RPE buffer. Finally, RNA samples were eluted from the spin columns using RNase free water. The quality and concentration of the total RNA were examined with a Nanodrop (Thermo Scientific Nanodrop, 2000/2000c Spectrophotometer, Cat#: ND-2000c) nucleic acid spectrophotometer. RNA gel electrophoresis was also performed to evaluate the quality of the RNA.
cDNA synthesis
Complementary DNA (cDNA) was synthesized from total RNA samples using the Bio-Rad’s iScript Reverse Transcription Supermix for RT-qPCR (Hercules, CA, USA) following manufacturer guidelines.
Gene expression
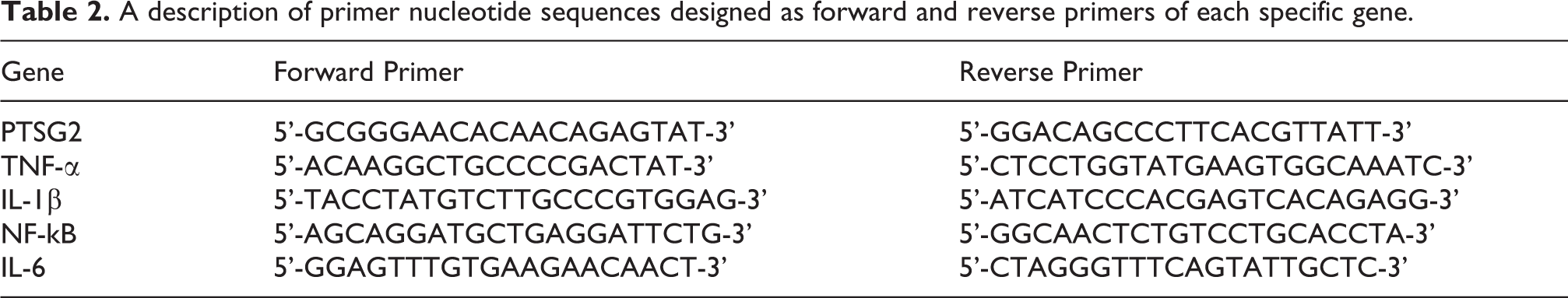
The expression of various genes related to inflammatory response in the treatment groups YA-YH was analyzed. Genes such as IL1B, IL6, PTSG2, TNF, and NFKB were analyzed using b-actin as a control gene. The reaction mixture comprised of forward and reverse primers specific to the gene of interest (Table 2), SsoFast EvaGreen Supermix (Bio-Rad, Hercules, CA, USA), nuclease free water, and cDNA. The reaction mixture was run on a Bio-Rad CFX96 Rt-PCR system to quantitatively measure the expression of mRNA transcripts.
A description of primer nucleotide sequences designed as forward and reverse primers of each specific gene.
Data analysis
The statistical analysis of data was performed using analysis of variance (ANOVA). Albumin concentration, creatinine concentration, total protein concentration, and AST activity were evaluated for significant changes comparing the treated groups to the control group. Significant differences between the treatment groups were denoted as either *p < 0.05, or **p < 0.01, and the data was presented as mean ± standard error.
Results
Quantification of albumin, AST, creatinine, and total protein levels in serum
An evaluation of the serum albumin concentration of the control and treatment groups was performed, allowing a comparison of the different treatment groups and their systemic effects. Figure 2 illustrates the mean serum albumin in g/dL. Group YA (no treatment) is the control group. Compared to the control group, no significant change was noted in the treated groups.
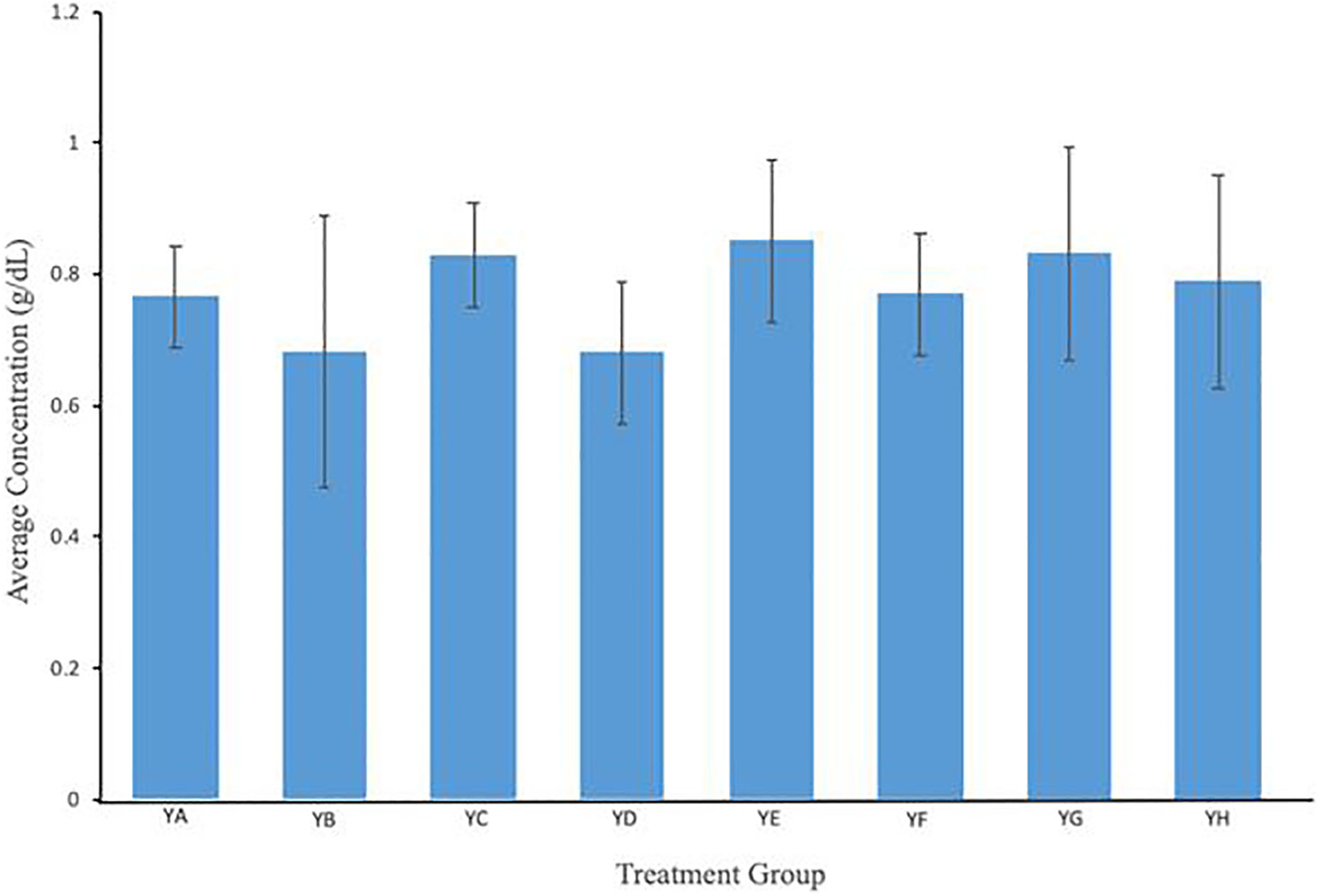
Mean serum albumin levels in mice treated with various combinations of CNT-Ab and microwave radiation. Albumin concentration was not significantly altered in treated groups in comparison to control mice. Treatment groups included: YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave) and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave).
In Figure 3, the serum creatinine concentrations of all experimental groups is shown. Creatinine concentrations of groups YB (Microwave only), YD (.125 mg/mL plain MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT + No Microwave), YG (.5 mg/mL plain MWCNT + Microwave) and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave) were significantly reduced when compared to the control group (YA). Likewise, YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave) & YE (.125 mg/mL anti-PSMA-MWCNT + Microwave) treatment groups did not show any significant differences when compared to the control group.
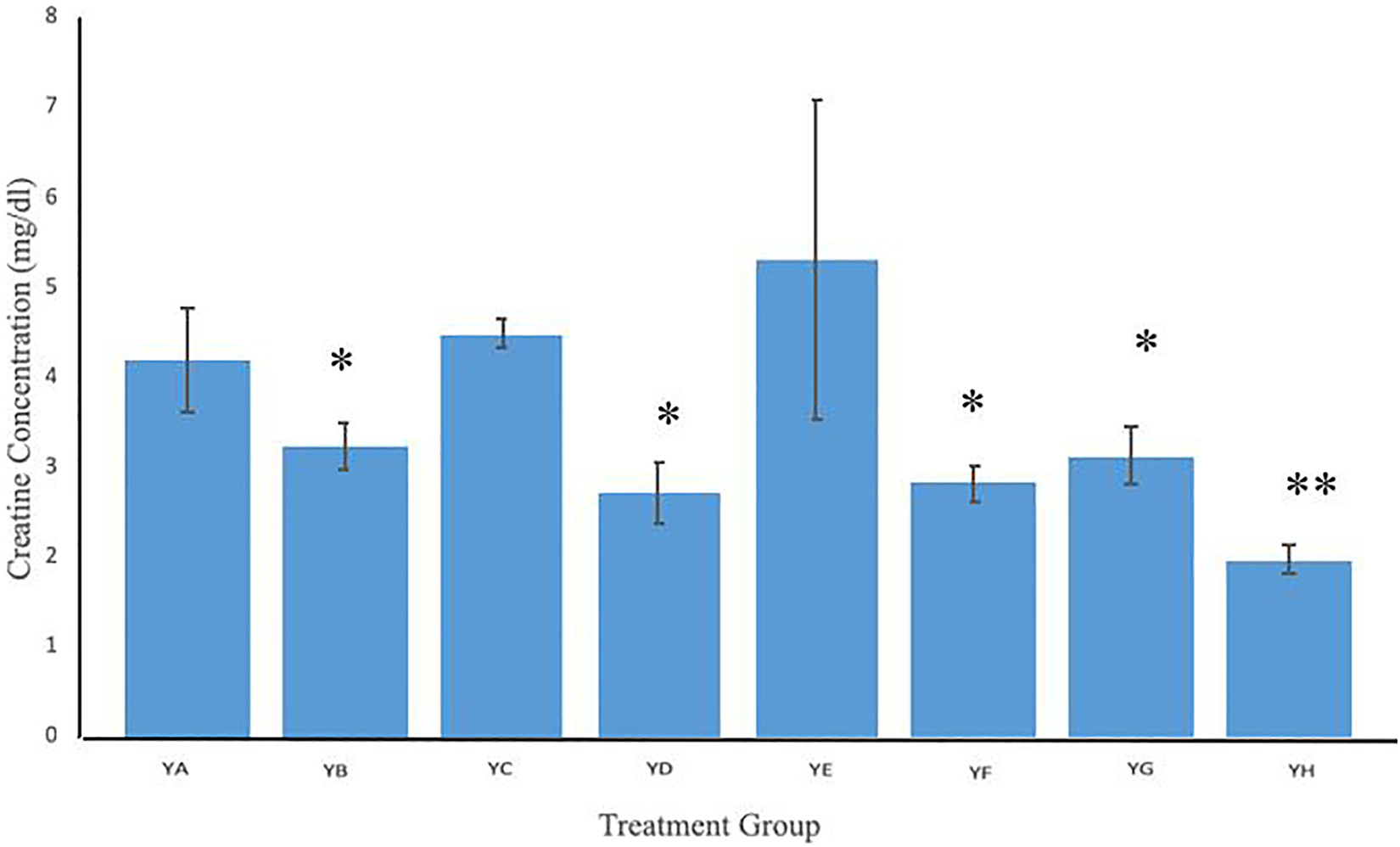
Mean creatinine in the serum of mice exposed to various concentrations of anti-PSMA-MWCNT in the presence of microwave irradiation. Significantly (*P < 0.05; **P < 0.01) lower levels of creatinine were found in YB (Microwave only), YD (.125 mg/mL plain MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave) groups. Creatinine levels in YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave) and YE (.125 mg/mL anti-PSMA-MWCNT + Microwave) groups were not altered significantly.
Evaluation of AST was performed across treatment and control groups. Figure 4 illustrates AST activity levels in mice for groups YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave) for serum AST. Compared to YA (no treatment), groups YB (Microwave only), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave) and YG (.5 mg/mL plain MWCNT + Microwave) showed significantly increased AST activity in blood serum. On the contrary, YD (.125 mg/mL plain MWCNT + Microwave) and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave) mice appeared to show significantly reduced levels of serum AST activity. No change was observed in the YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave) treated mice.
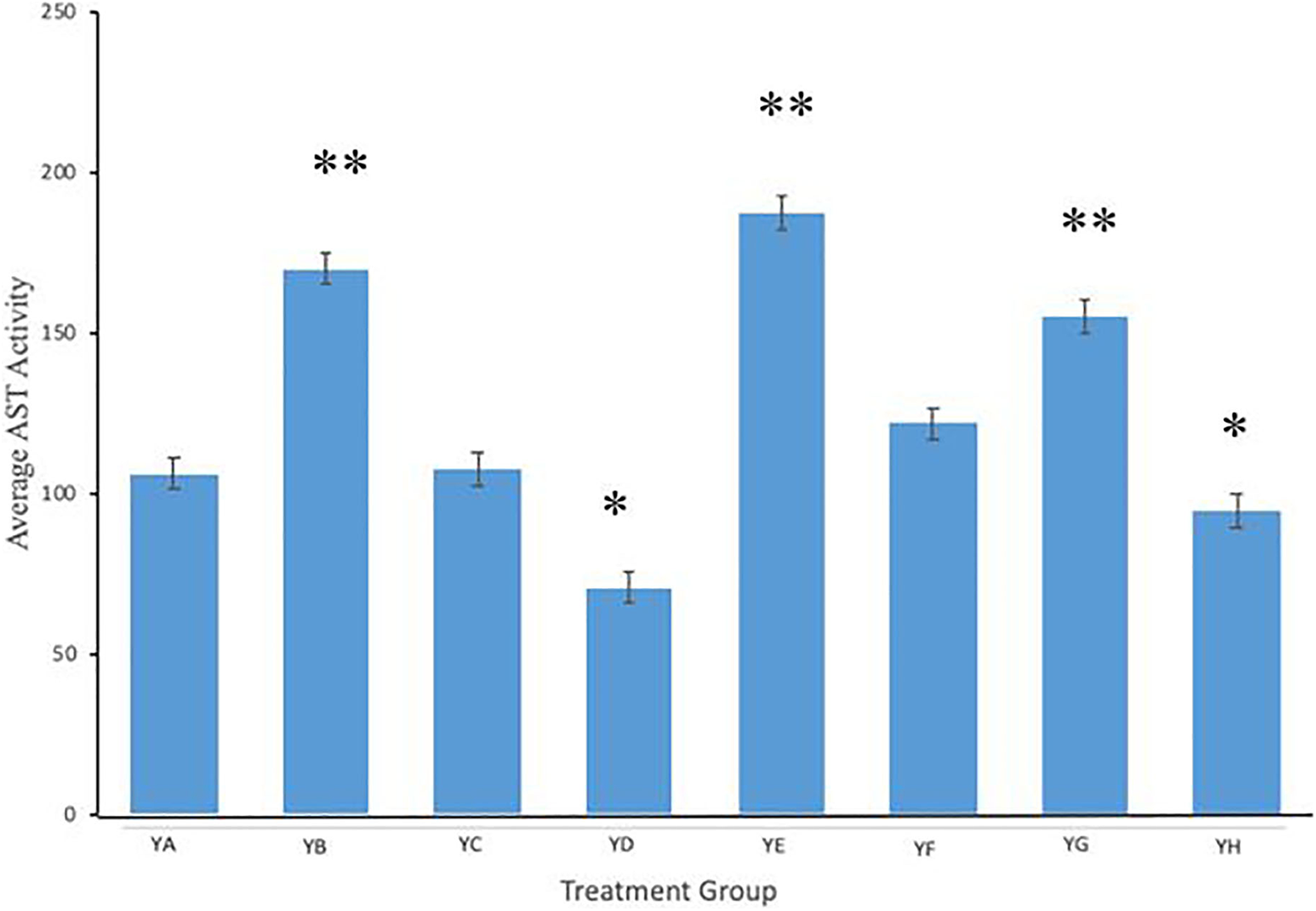
The activity of AST (IU/L) in serum of mice intramuscularly injected with various amounts of anti-PSMA-MWCNT plus/minus microwave irradiation. Compared to the control group, significantly elevated AST (*P < 0.05; **P < 0.01) was seen in YB, YE and YG. On the contrary, serum AST in YD and YH mice was significantly reduced. Treatment groups included YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave).
Total protein concentration is reported in Figure 5. Serum total protein concentration values mirror that of the albumin concentration. Although the treated groups were either slightly lower or higher, these values were within the margin of error. None of the treatment groups showed any significantly elevated or diminished total protein levels when compared to the control mice.
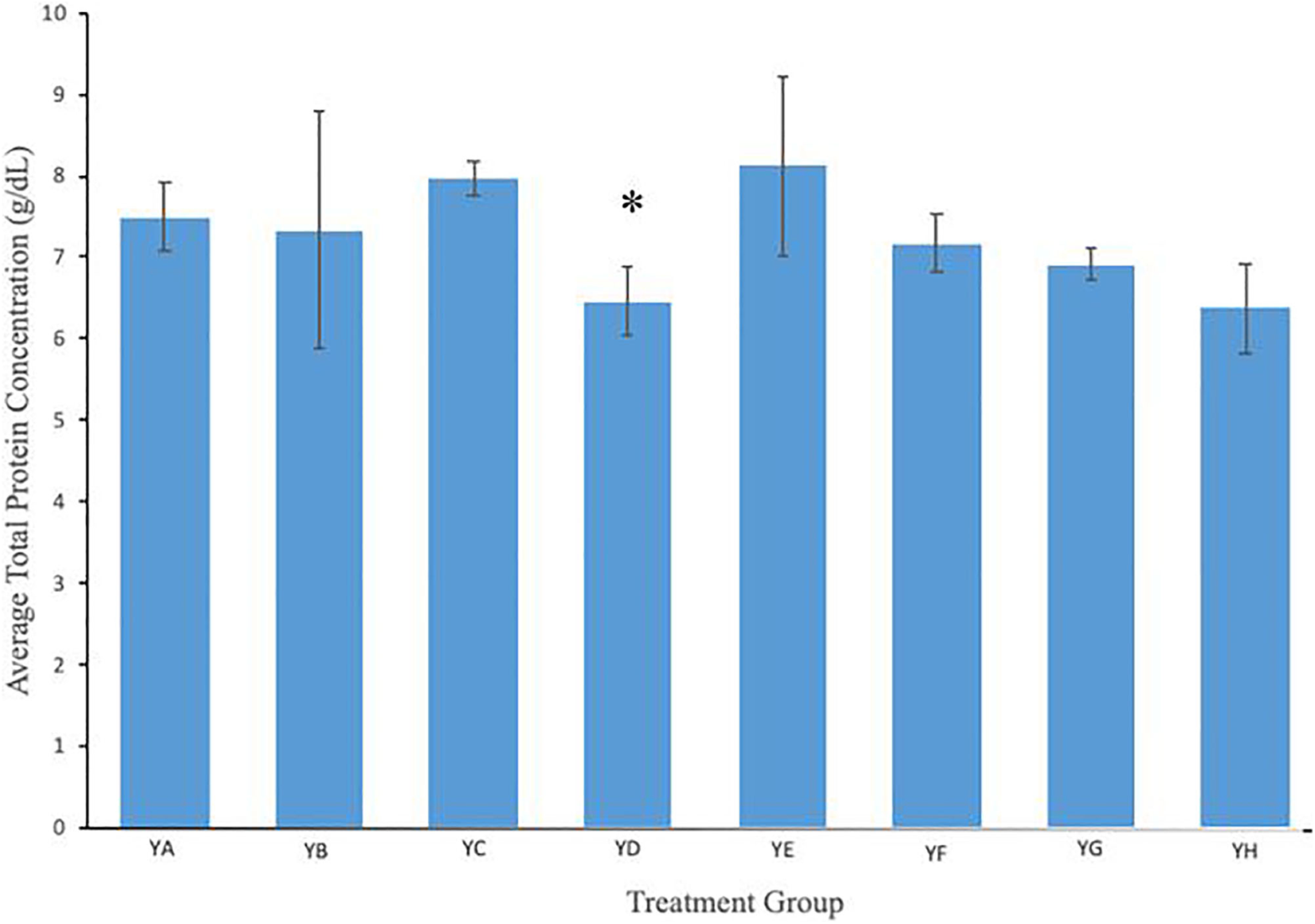
Serum total protein levels in mice exposed to different amounts of anti-PSMA-MWCNT plus/minus microwave irradiation. The level of total protein in the serum was relatively constant for all groups. In comparison to the control group, no significant change in the amount of total protein was noted in any of the groups except for the YD mice. Treatment groups included YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave). Total protein in the YD (.125 mg/mL plain MWCNT + Microwave) treatment group was slightly reduced (*P < 0.05).
Expression of inflammatory genes in the brain, liver, and kidney tissues
Brain, hepatic, and renal relative expression of select inflammatory genes, IL1B, IL6, NF-KB, PTSG2, and TNF, were measured in each experimental group. A brief description of each of these genes is provided below. 16,17
IL1B is a member of the interleukin 1 cytokine family. The cytokine is produced by activated macrophages which is proteolytically processed to its active form by caspase. This cytokine is an important mediator of the inflammatory response, and is involved in a variety of cellular activities, including cell proliferation, differentiation, and apoptosis. 16
IL6 gene encodes a cytokine that functions in inflammation and the maturation of B cells. The protein is primarily produced at sites of acute and chronic inflammation, where it is secreted into the serum and induces a transcriptional inflammatory response through interleukin 6 receptor, alpha. 17
NFKB is a transcription regulator that is activated by various intra- and extra-cellular stimuli such as cytokines, oxidant-free radicals, and bacterial or viral products. Activated NFKB stimulates the expression of genes involved in a wide variety of biological functions. Inappropriate activation of NFKB has been associated with a number of inflammatory diseases. 18
The PTSG2 gene encodes the inducible isozyme PTSG2. It is regulated by specific stimulatory events and is responsible for the prostanoid biosynthesis involved in inflammation and mitogenesis. 19
TNF-alpha gene encodes a multifunctional proinflammatory cytokine that belongs to the tumor necrosis factor (TNF) superfamily. This cytokine is mainly secreted by macrophages. This cytokine is involved in the regulation of a wide spectrum of biological processes including cell proliferation, differentiation, and apoptosis. This cytokine has been implicated in a variety of diseases, including autoimmune diseases, insulin resistance, and cancer. 20
Gene expression of select inflammatory genes in the liver tissues of treated mice are displayed in Figure 6. Genes IL1B and PTSG2 were not expressed for any of the treatment groups. All treatment groups had a down-regulation in expression of IL6 and TNF compared to the control. Groups YD (.125 mg/mL plain MWCNT + Microwave) and YE (.125 mg/mL anti-PSMA-MWCNT + Microwave) had an up-regulation for NFKB, while YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT No Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), and YG (.5 mg/mL plain MWCNT + Microwave) were all down-regulated when compared to the control.
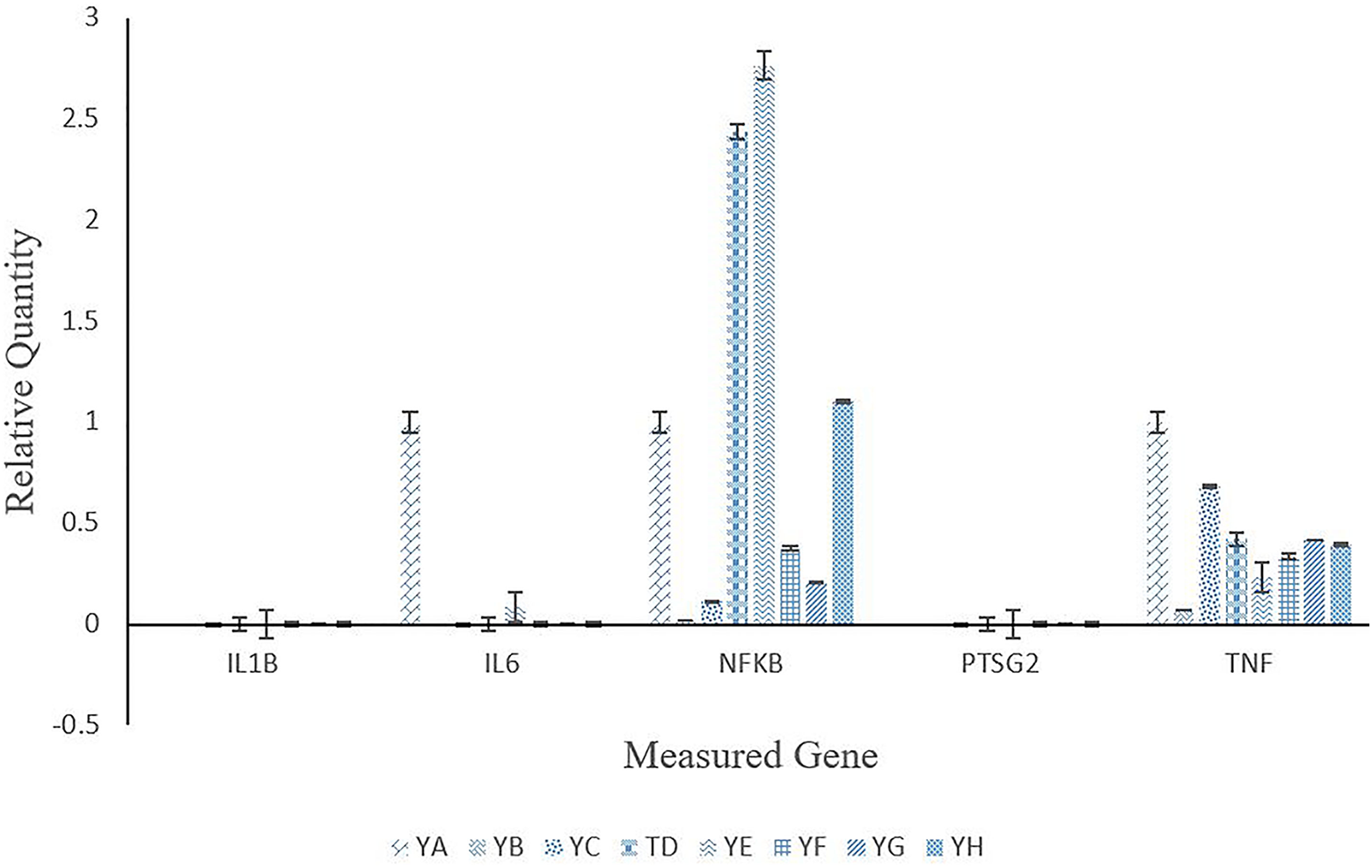
Hepatic relative gene expression mice treated to varying levels of anti-PSMA-MWCNT plus/minus microwave irradiation. Treatment groups included YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave) and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave).
Gene expression in brain tissue is presented in Figure 7 for all treatment groups. Across treatment groups, gene IL1B was not expressed, while genes IL6, PTSG2, and TNF were down-regulated when compared to the control. For the gene NFKB, all treatment groups were up-regulated in comparison to the control group.
Relative gene expression in the brain of mice that underwent exposure to different amounts of anti-PSMA-MWCNT plus/minus microwave hyperthermia. Treatment groups included YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave).
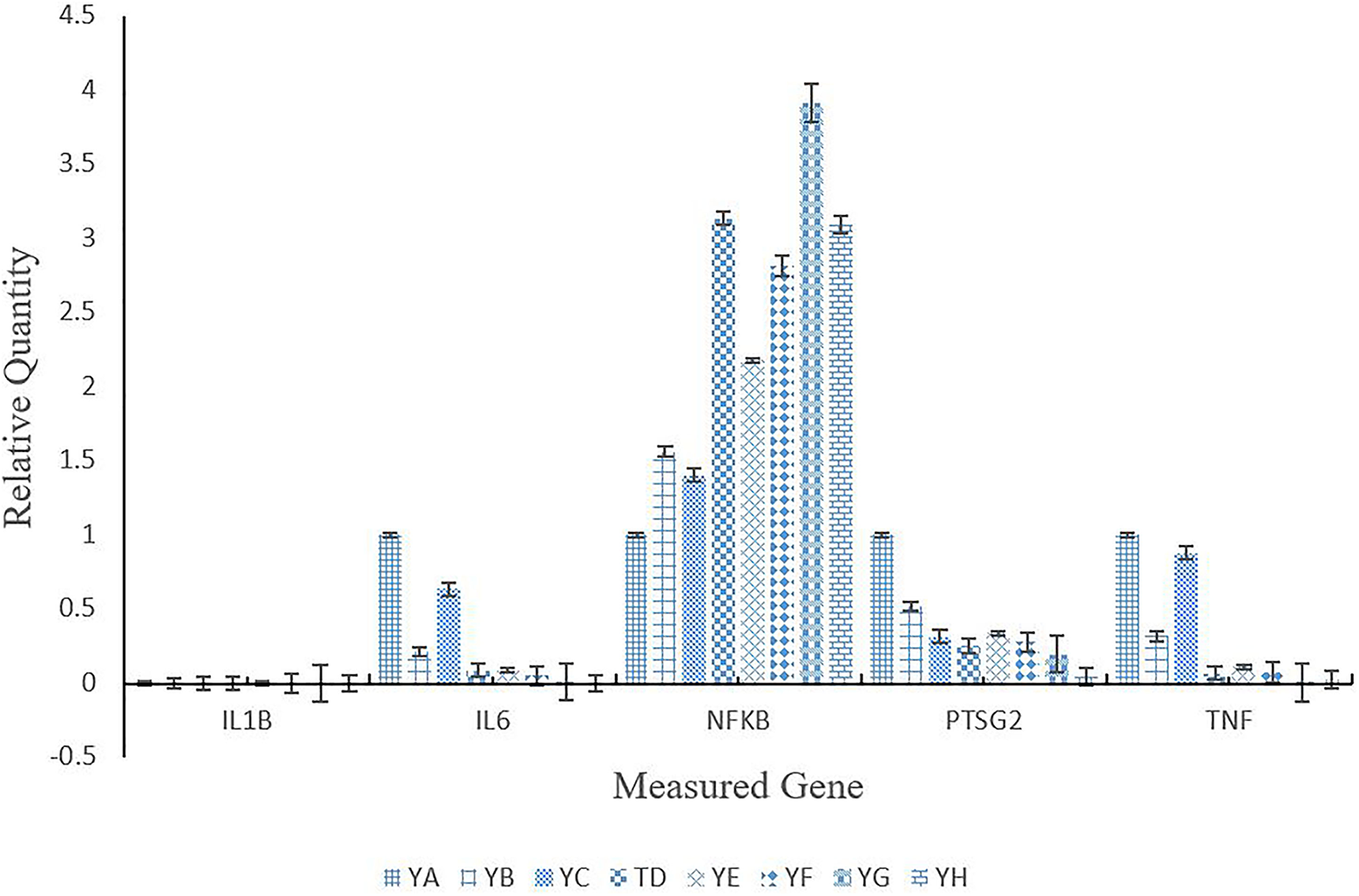
The expression of select inflammatory genes in kidney tissue is shown in Figure 8 for all treatment groups. IL1B was not expressed for treated or control groups, and gene TNF was down-regulated across all treatment groups. Genes IL6 and PTSG2 were down-regulated across treatment groups with the exception of group YB (Microwave only) which was slightly up-regulated. For the gene NFKB, all treatment groups were up-regulated in comparison to the control group.
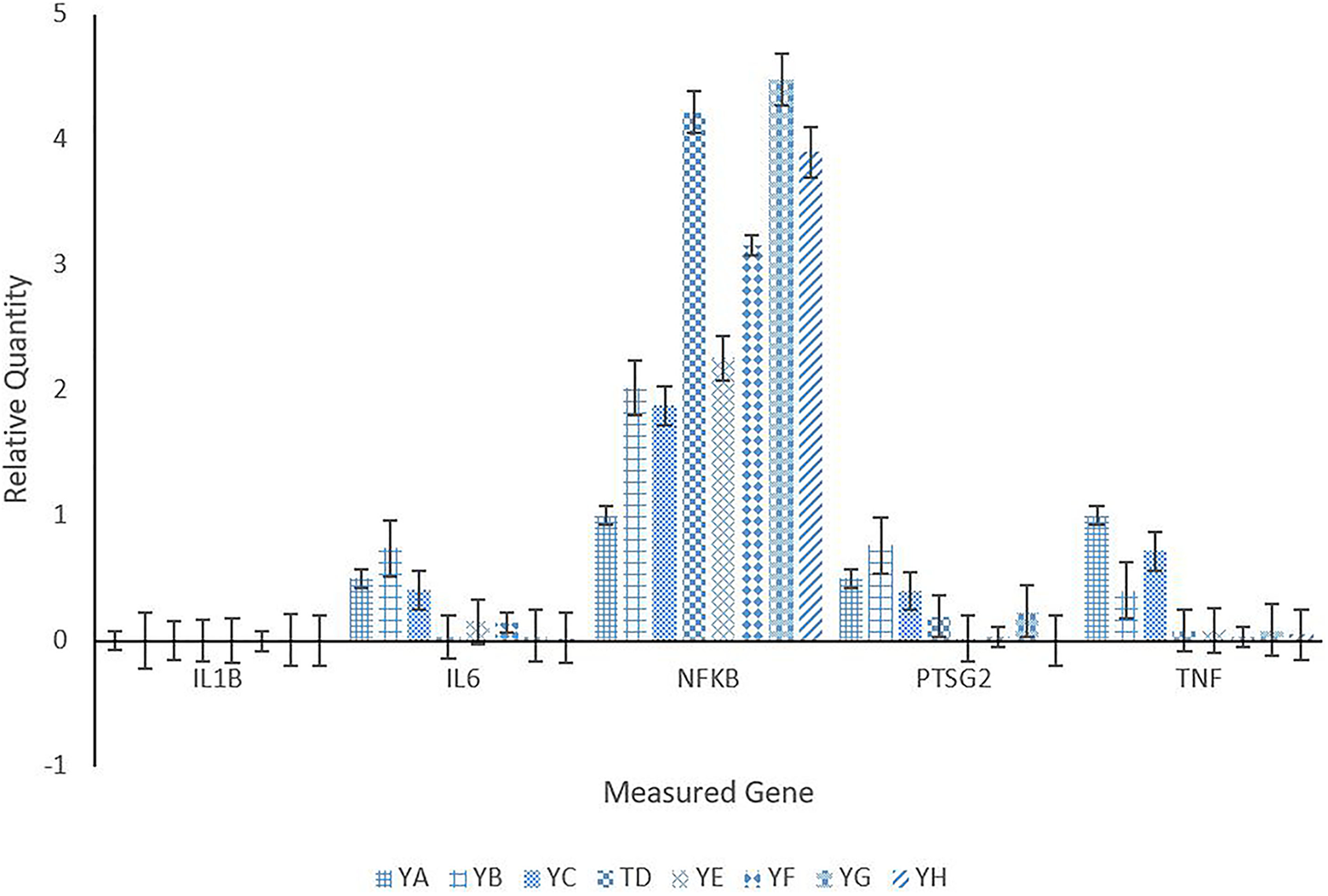
Renal relative gene expression of mice injected with varying concentrations of anti-PSMA-MWCNT plus/minus microwave irradiation. Treatment groups included YA (no treatment), YB (Microwave only), YC (.125 mg/mL anti-PSMA-MWCNT + No Microwave), YD (.125 mg/mL plain MWCNT + Microwave), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), YG (.5 mg/mL plain MWCNT + Microwave), and YH (.5 mg/mL anti-PSMA-MWCNT + Microwave).
Results portray the effect of treatment on important immune responses and whether or not a response is induced. Figures 6, 7, and 8 illustrate relative expression in liver, brain, and kidney tissues, respectively. IL1B was not expressed in any tissues across treatment groups. Genes Il6, PTSG2, and TNF were all under-expressed across treatment groups and tissue types. Gene NFKB is of greatest interest due to the significant overexpression that most treatment groups portrayed across tissue types.
Discussion
Clinical tests of the serum were conducted to examine the levels of creatinine, albumin, aspartate aminotransferase (AST) and total protein within mice exposed to CNTs conjugated with antibodies. These assays provide insight into systemic impacts of the treatment, including hepatotoxicity and nephrotoxicity, while investigation of gene expression provides insight into specific and localized effects on the kidney, brain, and liver tissues. These assays enable us to examine the biodistribution of injected CNT-Ab followed by hyperthermia. Understanding the distribution of CNTs throughout the body and modes of clearance is an important factor when exploring its potential use as a therapy.
Creatinine is a waste product remaining from the metabolism of creatine phosphate in the muscles. Creatinine assays reveal important information about kidney function and filtration rate as well. High levels of serum creatinine can thus indicate improper kidney function, decreased glomerular filtration rate, and can also be indicative of intense exercise, dehydration, and high protein diets. 21 Mean serum creatinine levels of the treatment groups were significantly lower in comparison to the control mice with the exception of treatment group YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), which exhibited a much larger creatinine concentration. This indicates potential kidney impairment for the YE treatment group. Creatinine is formed largely in the muscles by the removal of water from creatine phosphate. While high levels of creatinine are not life threatening, it may be indicative of other underlying health issues such as chronic kidney disease. A study by Yadav and coworker (2014) showed that serum creatinine levels are directly related to the severity of kidney disease and thus a great biomarker for kidney impairment. 22
Albumin is the most abundant globular blood transport protein. Albumin binds substances including water, calcium, sodium, potassium, fatty acids, hormones, etc. In addition to being a nutrient carrier, it also serves to regulate the oncotic pressure of blood. Low levels of serum albumin can indicate liver damage, kidney disease, malabsorption, malnutrition, and malignancies, while high levels of albumin typically indicate dehydration. 23 There were no notable deviations in albumin levels across treatment groups compared to the controls.
Aspartate aminotransferase (AST) is an important enzyme in amino acid metabolism that is found predominantly in the liver, but also in lower amounts in the heart, brain, muscle, and kidney tissues. AST levels serve as indicators of liver function and inflammation. Healthy individuals have naturally low AST levels, however when tissue that contains AST is damaged, the substance is released into the blood leading to increased levels. 24 An increase in AST activity was found in groups YB (microwave only), YE (.125 mg/mL anti-PSMA-MWCNT + Microwave), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), and YG (.5 mg/mL plain MWCNT + Microwave). These results suggest that CNT-Ab exposure might be indicative of potential damage to liver function from these treatment groups. A study by Hennes and colleagues on pediatric patients with blunt abdominal trauma was able to identify hepatic injury using AST as a biomarker with 100% sensitivity and 93% specificity. They found that AST levels had a significant correlation with positive results on CT scans used to diagnose patients. Elevated AST levels do not have a direct impact on any system, but are a reliable biomarker for liver damage. 25
Proteins are important building blocks for organs, muscles, hormones, and enzymes, and are therefore essential to the overall health of an organism. Fluctuations in the number of proteins in blood serum can result in fatigue, weight loss, and inflammatory disease, and can be indicative of underlying kidney or liver diseases. High protein levels can indicate inflammation, cancer, dehydration, or liver/kidney disease, while low protein levels can indicate malnutrition, malabsorption, congestive heart failure, and liver/kidney disease. 26 Levels of total protein in blood serum among treatment and control mice was unchanged.
The evaluation of kidney and liver function is of great importance when determining toxicity of a certain substance or treatment. Both organs play key roles in removing toxins from the body and maintaining a systemic equilibrium of vital nutrients needed for functioning. 27 Thus, determining the potential for hepatotoxicity and nephrotoxicity are imperative in determining the human health risk. The liver functions to filter the blood coming from the digestive tract before dissemination to the rest of the body among other metabolism and storage functions. The role of the kidney is to remove any waste from the blood to be passed in the urine and to maintain hydration and electrolyte levels. 28 Treatment group YE (.125 mg/mL anti-PSMA-MWCNT + Microwave treatment) was found to have consistently high creatinine, AST, and total protein levels, all suggesting toxicity in the liver and kidneys of exposed mice. No other treatment groups appeared to have shown levels of these biomarkers that may be indicative of toxicity. However, groups YB (microwave only), YF (.5 mg/mL anti-PSMA-MWCNT No Microwave), and YG (.5 mg/mL plain MWCNT + Microwave) had higher AST levels which may suggest impaired liver function. It is unclear why treatment group YE elicited such a response in the mice, yet groups YF and YG only showed signs of liver damage even though a greater amount of MWCNT’s were used for treatment.
An investigation of gene expression can reveal which genes are turned on or off in a specific cell type, and can be used to quantify the gene’s relative expression. The genes studied are described in Table 2. These genes play important roles in the immune and inflammatory response. Measurement of these genes in brain, liver, and kidney tissues may provide information about whether a gene is overexpressed or under-expressed in treated groups.
Gene IL1B was not expressed for the treatment or control groups in any of the tissues. Genes IL6, PTSG2, and TNF were down-regulated for all treatment groups in the brain, liver, and kidney tissues. The gene of greatest interest was NFKB, which was highly expressed in all tissues for each treatment group. The magnitude of expression compared to the control suggests a significant up-regulation in the expression of this inflammatory response gene. NFKB is a transcription regulator that is activated by various cellular stimuli, such as cytokines, oxidant-free radicals, and bacterial or viral products. Activated NFKB stimulates the expression of genes involved in a wide variety of biological functions, including response to infection. Inappropriate activation of NFKB has been associated with a number of inflammatory diseases. 29 Up-regulation of this gene indicates a localized effect on these tissues that could be associated to a number of cytotoxic factors, including exposure to CNT-Ab plus microwave irradiation.
We believe microwave hyperthermia in combination with nanoparticles hold promise for the treatment of various cancers, including prostate cancer. There is quite some evidence to suggest that nanoparticles accumulate in tumors. In this study, CNTs in conjugation with antibodies have been explored for treatment of malignant tumors. However, one of the key challenges of using CNTs with microwave relates to the non-uniform heating of cells which necessitates a lengthy irradiation time even with the acceleration of hyperthermia facilitated by CNTs. However, this challenge can be overcome with the relatively low levels of microwave energy causing CNTs to reach temperatures capable of causing cell ablation.
Conclusion
In summary, the groups that received the combined CNT-Ab dose were found to have consistently higher serum creatinine, AST, and total protein levels that may be indicative of potential liver and kidney impairment. No other treatment groups were determined to have impaired kidney function; but groups YB (microwave only), YF (0.5 mg/mL anti-PSMA-MWCNT and no microwave), and YG (0.5 mg/mL anti-PSMA-MWCNT plus microwave) had higher AST levels which may suggest impaired liver function. Investigation of gene expression revealed an overexpression of the NFKB gene, which may impact the inflammatory response pathway. These results suggest that exposure to treatment may induce hepatotoxicity and nephrotoxicity. One week post treatment, higher levels of CNT-Ab exposure elicits the most hepatic and renal toxicological response. It does not appear the brain was affected by either CNT-Ab exposure alone or in combination with microwave hyperthermia. In the future, we will examine tissues for histopathological modifications. Other potential future studies might include intra-tumor injection in a human xenograft prostate cancer model and application of a localized microwave source with a power that is fully optimized.
Footnotes
Abbreviations
PSMA—prostate-specific membrane antigen; Ab—anti-PSMA antibodies; MWCNT—Multiwalled carbon nanotubes; RT-qPCR—real time quantitative polymerase chain reaction; AST—Aspartate aminotransferase; IL1B—Interleukin 1 beta; IL6—Interleukin 6; Nuclear Factor kappa-light-chain-enhancer of activated B cells; PTSG2—Prostaglandin-endoperoxide synthase 2; TNF—Tumor necrosis factor
Acknowledgments
We want to acknowledge the assistance of Mr Craig Banks, the Animal Facility Manager of Georgia Southern University. Moses Y Kusi assisted with some of the animal care procedures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Cancer Society under Grant #IRG-14-193-01 and the College Office of Undergraduate Research, Georgia Southern University.
