Abstract
Blood triacylglycerol (TG) and lipoproteins are important variables for evaluating nutritional status of wildlife, but measurements are often expensive and difficult. Performance of a small, portable blood analyzer intended for human medical diagnostics was evaluated in measuring these variables in plasma and serum from free-ranging polar bears (Ursus maritimus), which are experiencing nutritional stress related to sea ice loss. The analyzer accurately tracked changes in concentration of total cholesterol (Ctotal), cholesterol associated with high-density lipoprotein (CHDL), and TG during a validation protocol of diluting samples and spiking them with exogenous cholesterol and glycerol. Values of Ctotal and TG agreed well with values obtained by other methods (ultracentrifugation followed by colorimetric assays); agreement was variable for values of cholesterol associated with specific lipoproteins. Similar to a study of captive polar bears, ultracentrifugation methods revealed greater TG in very low-density lipoproteins than in low-density lipoprotein, which is unusual and merits additional study.
Whether an animal is fasting or feeding is reflected by blood values of cholesterol associated with high-density lipoproteins (CHDL), low-density lipoproteins (CLDL), very low-density lipoproteins (CVLDL), and total cholesterol (Ctotal), as well as by values for triacylglycerols (TGHDL, TGLDL, TGVLDL, TGtotal). For example, Ctotal and TGtotal often rise during mobilization of endogenous fat while animals are fasting. 8 Thus, lipoprotein measurements provide important inferences regarding the nutritional state of free-ranging animals that may or may not be fasting. 11
Several methods are available for lipoprotein quantification, including colorimetric assays in lipid panels and ultracentrifugation techniques. 13 However, ultracentrifugation is expensive, labor-intensive, and not easily performed in research field camps; therefore, many researchers instead send samples to laboratories for colorimetric assays. 1 The objective of the current study was to validate assay applicability and performance for samples from wildlife species, particularly assays that have the potential to be performed in field camps, allowing immediate analysis and decreasing risks of sample deterioration. 5
Blood samples from polar bears (Ursus maritimus) were analyzed with a portable blood analyzer a designed for human diagnostic procedures. The analyzer is also available for veterinary diagnostics, b but the human version a includes software for lipoprotein analysis. Both instrumentsa,b are small (height 32 cm, width 15 cm, depth 20 cm, mass 5 kg) and can easily be used in field camps provided there is electricity (AC 100–240 volts and 50–60 Hz, DC 15 volts and 5.0 Amperes) and that ambient temperatures remain within 15°C and 32°C. Both instruments use disposable rotors c requiring 100 µl of sample. Thus, total equipment required for lipoprotein analysis includes the analyzer, a disposable rotors, c and a pipette. A printer is built into the analyzer, a and results are produced in approximately 12 min. Disposable lipid rotors c include assays for Ctotal, 9 CHDL, 19 and glycerol as a TG proxy.4,12 Other variables are calculated: CVLDL = TGtotal/5, and CLDL = Ctotal – CHDL – TGtotal/5. 7
The 3 assays have been certified by the Centers for Disease Control and Prevention (United States) for blood from fasted human beings and validated for point-of-care use with human patients. 19 The current study tested the assays on blood from free-ranging polar bears and compared the results to values obtained by ultracentrifugation followed by analysis using commercial kits. d Accurate lipoprotein measurements are particularly useful in polar bear research. Research suggests that sea ice loss is leading to extended fasting and potential population declines in these carnivores, 18 and lipoprotein measurements have been successfully used to understand polar bear nutritional state. 8 Additionally, polar bears are unique because they consume a high-fat diet (e.g., 80% seal blubber), 2 and a previous study of captive bears reported greater TGLDL than TGVLDL. 8 This is unusual because in human beings, LDLs are synthesized from TG-depleted VLDLs, implying that TGLDL is less than or equal to TGVLDL.
Portable analyzers enable the ideal scenario in which analyses are performed immediately after sample collection, avoiding deterioration risks of storage. 5 However, it is also important to validate assays if logistics prevent immediate analysis and samples must be stored. Validations were performed with samples collected during ongoing studies of polar bears in the Southern Beaufort and Chukchi seas, Alaska, USA (details on capture and sample collection described elsewhere 10 ). Blood was collected from the femoral vein or artery in ethylenediamine tetra-acetic acid (EDTA) and no-additive containers, e centrifuged at 2,000 RCF for 10 min, then plasma or serum was siphoned into cryovials and stored at −20°C or −40°C until analysis.
Four validation experiments (A–D) were conducted as follows: A) plasma was diluted, then exogenous CHDL and TG were added, and percent recovery measured with lipid rotors c after both manipulations; B) results were compared from lipid rotors when analyzing serum versus EDTA plasma samples; C) results were compared from lipid rotors and kits d for Ctotal and TGtotal; and D) ultracentrifugation was used to separate HDL, LDL, and VLDL, then cholesterol and TG were quantified for each class.
In experiment A, preliminary trials with 2 bears indicated plasma contained CHDL levels above the detection limit of the blood analyzer a (100 mg/dl). Bear 1 required a 20:1 dilution (volume of plasma-to-sterile saline), and bear 2 required a 3:4 dilution, to reduce CHDL to a measurable concentration. Additional dilutions were then performed (bear 1—5:2, 4:3, 3:4, 2:5; bear 2—1:2, 1:3, 1:4, 1:7). For each dilution, CHDL and Ctotal were measured on 3 separate rotors; mean values were calculated, then divided by predicted values (based on the first dilution), and the results were converted to percent recovery (%). To each dilution, 4 μl of exogenous CHDL (606 mg/dl) f was added, and new expected concentrations were calculated. Measurements for CHDL and Ctotal were repeated and the percent recovery calculated.
Triacylglycerol was measured on bear 3 without dilution, as the concentration was within detection limits. The sample was then diluted (7:2, 9:7, 1:2, and 1:7) and percent recovery calculated for each dilution. Next, 5 μl of exogenous glycerol used as a TG standard (1,000 mg/dl) g was added to each dilution, TG measurements were repeated, and the percent recovery was calculated.
For consistency with previous studies of lipoproteins in bears, 6 the plasma was analyzed. However, the anticoagulant EDTA may negatively bias some colorimetric measurements of CHDL 20 although measurements of Ctotal seem unaffected. 9 In experiment B, lipid rotor c assays were performed on both serum and EDTA plasma for 10 bears, and results were compared using linear regression.
In experiment C, Ctotal and TGtotal were measured in plasma from 8 bears, using kits d and lipid rotors. c Results were compared with linear regression. In experiment D, ultracentrifugation was used to create a gradient based on density, separating HDL, LDL, and VLDL. For 8 plasma samples, 21 fractions of lipoproteins 6 were identified: VLDL (1–3), LDL (3–11), and HDL (11–20). Some VLDL remained on the meniscus surface in the separation column, forming fractions 20 and 21. In each fraction, cholesterol and TG were measured using the same kits as in experiment C. The area under the curve was then calculated for each fraction and multiplied by Ctotal and TGtotal to derive cholesterol and TG for each lipoprotein.
In experiment A, lipid rotors c accurately measured diluted and spiked samples of plasma. After dilution and spiking, mean recovery rates (± standard deviation) for CHDL and Ctotal were 91% (±3%) to 105% (±3%). Recovery rates for TG after dilution and spiking were 100% (±2%) to 106% (±7%).
In experiment B, EDTA plasma samples yielded data similar to serum samples. Of the 10 bears for which both sample types were analyzed, 1 sample of EDTA plasma had extremely low values of Ctotal (38 mg/dl), CHDL (22 mg/dl), and TGtotal (43 mg/dl), whereas the serum appeared normal (353 mg/dl, 187 mg/dl, and 255 mg/dl, respectively); this sample was excluded from further analyses. For TGtotal, EDTA plasma yielded results nearly identical to serum (in linear regression, 95% confidence intervals for slope included 1, and for intercept included 0). In contrast, slopes differed from 1 and intercepts differed from 0 for Ctotal and CHDL, indicating these variables were measured differently (Fig. 1).
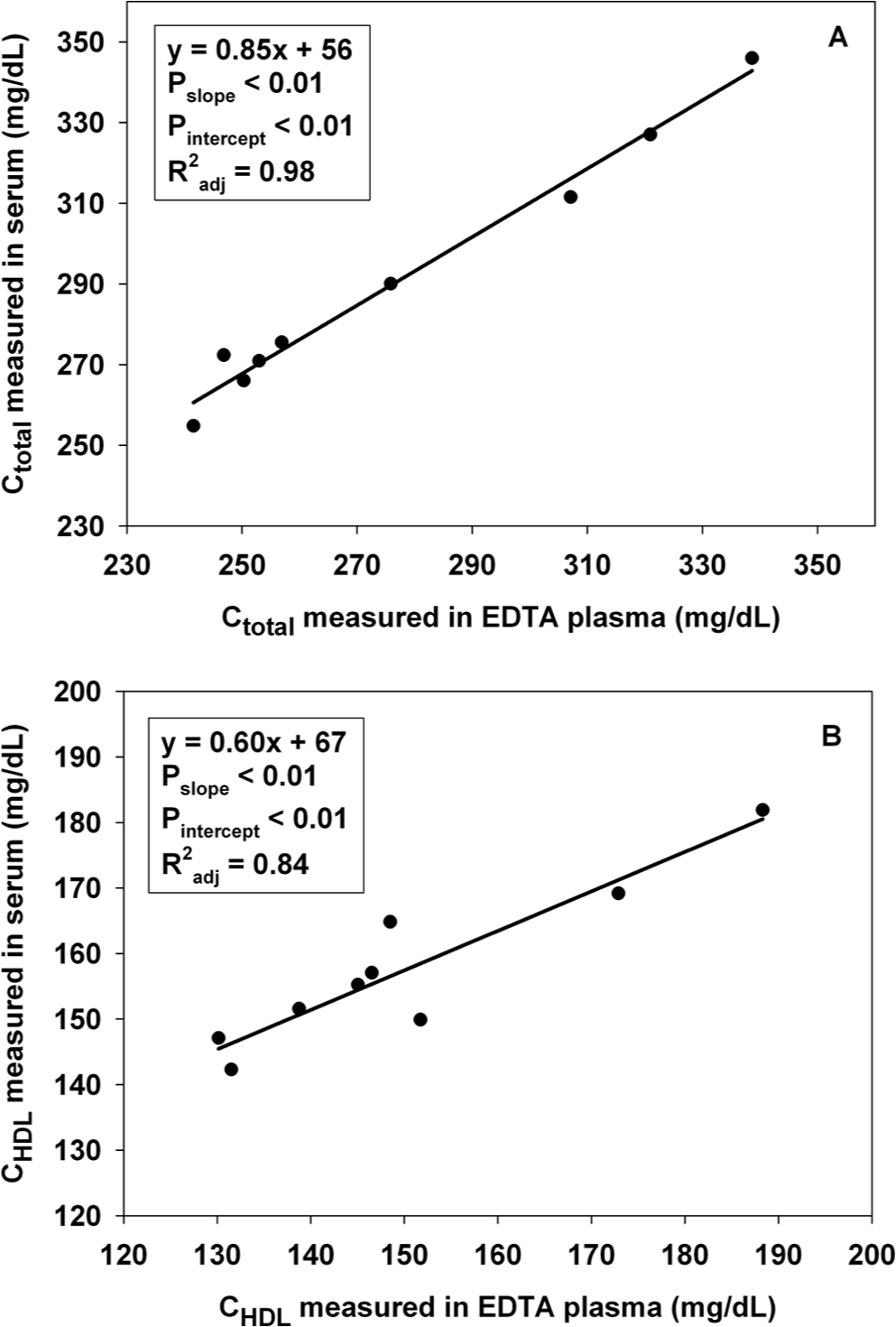
Relation between measurements performed on ethylenediamine tetra-acetic acid (EDTA) plasma samples and serum samples from free-ranging polar bears (Ursus maritimus), of (
In experiment C, measurements of Ctotal and TGtotal were similar between lipid rotors c and kits d (Fig. 2). In experiment D, values of CHDL, CLDL, and CVLDL were inconsistent between the lipid rotor and the ultracentrifugation separation followed by measurements with the same kits used in experiment C (Fig. 3). The following TG content (mean ± SD; mg/dl) was determined: TGHDL (52 ± 13), TGLDL (221 ± 78), and TGVLDL (23 ± 11).
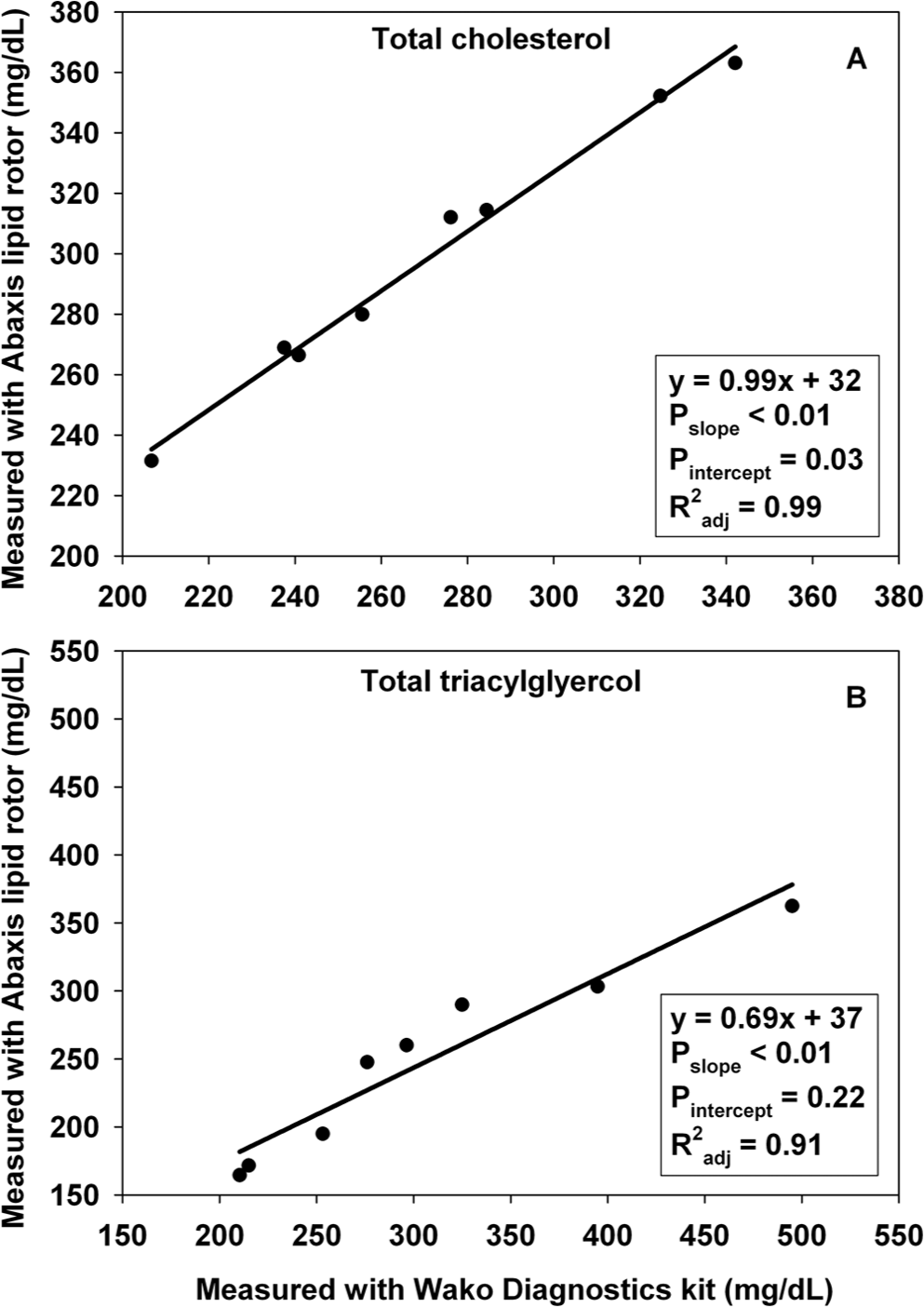
Relation between measurements from commercial kits and lipid rotors for (
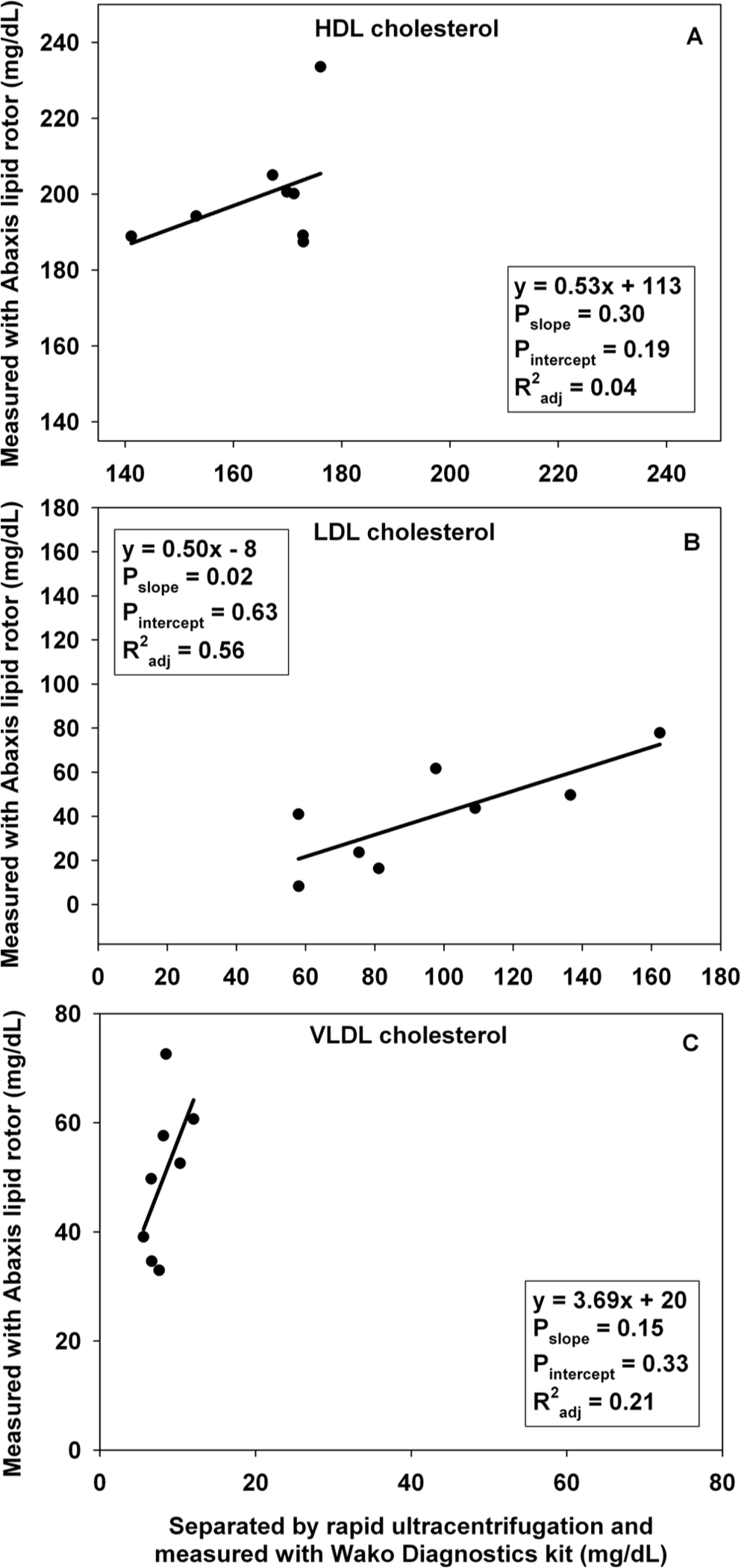
Relation between measurements from lipoprotein separation by ultracentrifugation and analysis with a commercial kit and lipid rotors, for ethylenediamine tetra-acetic acid plasma samples from free-ranging polar bears (Ursus maritimus). (
Lipid rotors c accurately measured diluted samples and samples with added HDL and glycerol, indicating there were no interfering substances in polar bear plasma. The EDTA plasma samples yielded lower Ctotal and CHDL than serum samples. This may be because EDTA causes osmotic shifting of water out of red blood cells, 20 influencing the concentration of Ctotal and CHDL in plasma (although TGtotal was not reduced). Alternatively, it is possible that EDTA interferes with the chemical reactions within the rotor. Regardless, significant slope parameters and high R2 values suggest a consistent and precise relationship, allowing for reliable correction of data derived from plasma samples. One bear (of 10) exhibited extremely low values of all variables in EDTA plasma, but normal values in serum; it is unclear whether this is a risk when analyzing EDTA plasma or a chance occurrence. It is recommended that 1 sample type be used when comparing individuals or sampling events and that the results be compared to the published range of the species.
Measurements of Ctotal and TGtotal from lipid rotors c and kits d accurately tracked each other. It is not clear why CHDL measurements differed between lipid rotors and ultracentrifugation. The lipid rotor precipitates non-HDL lipoproteins and quantifies the remaining cholesterol that must be associated with HDL. However, the non-HDL precipitation may be inaccurate because it depends on recognition of apolipoprotein-B, which can vary in structure among species 14 and may differ between human beings and polar bears. The ultracentrifugation method separates lipoproteins by density and then quantifies cholesterol in each layer. In dogs, HDL are divided into 2 subtypes, 1 of which overlaps with LDL in density. 15 It is not known if polar bears possess HDL subtypes similar to dogs; if so, this may make it difficult to separate HDL and LDL by density. Neither method should be affected by chylomicrons, which likely precipitate with non-HDL in lipid rotors, and which have a much lower density than HDL and thus, in ultracentrifugation, should not appear in the HDL layer. Causes of the discrepancy between methods are unclear and merit further investigation.
Dissimilarities in CHDL between lipid rotors c and ultracentrifugation must have contributed to inconsistencies in CLDL because the blood analyzer a software uses CHDL to calculate CLDL. Intriguingly, CVLDL calculated from the rotor measurement of TGtotal was approximately 6× higher than CVLDL measured after ultracentrifugation. This pattern, in combination with the unusual distribution of TG among VLDL and LDL (discussed in the following), suggests that lipoproteins may be different in polar bears than in human beings.
Ultracentrifugation methods revealed that, similar to polar bears in captivity, 8 free-ranging polar bears consistently have greater TGLDL than TGVLDL. In contrast, TGLDL is less than or equal to TGVLDL in black bears, 6 human beings, 3 rats, 17 and ground squirrels. 16 Dogs with greater TGLDL than TGVLDL have been reported in studies where, similar to the current study, the VLDL fractions may have contained chylomicrons.17,21 In dogs, however, TGLDL was about 2× TGVLDL whereas in the present study, TGLDL was approximately 10× TGVLDL. It is speculated that if HDL and LDL do not separate completely because of an overlap in density, TGLDL may include TGHDL, elevating the former measurement. This topic requires further study.
In summary, lipid rotors c yielded accurate, precise measurements of Ctotal and TGtotal, but more variable measurements of cholesterol associated with specific lipoproteins, in blood from free-ranging polar bears. These results, in combination with ease of operation and field portability, indicate that the portable blood analyzer a evaluated in the current study is a useful tool for research on polar bears, and potentially other wildlife species.
Footnotes
a.
Abaxis Piccolo, Abaxis Inc., Union City, CA.
b.
Abaxis VS2 Vetscan, Abaxis Inc., Union City, CA.
c.
Lipid panel 400-7144, Abaxis Inc., Union City, CA.
d.
Cholesterol E 439-17501 and L-type TG M 461-08992; Wako Diagnostics, Richmond, VA.
e.
Vacutainer, BD, Franklin Lakes, NJ.
f.
Sigma L8039, Sigma-Aldrich, St. Louis, MO.
g.
10010509, Cayman Chemicals, Ann Arbor, MI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Principal funding was provided by National Science Foundation (OPP 0732713). Additional support was provided by US Geological Survey, Climate and Land Use Change Research and Development Program; U.S. Environmental Protection Agency’s Science to Achieve Results Fellowship Program (917373); Wyoming NASA Space Grant Consortium (NNG05G165H); and the University of Wyoming. Funding support does not imply approval or endorsement by any of these parties.
