Abstract
We performed a retrospective study of all case submissions for the rabies virus (RABV) direct fluorescent antibody test (DFAT) requested of the Tifton Veterinary Diagnostic and Investigational Laboratory (Tifton, GA, USA) between July 2010 and June 2021. Submitted were 792 samples from 23 animal species from 89 counties in Georgia, and 4 neighboring counties in Florida, 1 in South Carolina, and 1 in Alabama. In 13 (1.6%) cases, the DFAT result was inconclusive; 779 (98.4%) cases had a conclusive (positive or negative) test result. Of these 779 cases, 79 (10.1%) tested positive across 10 species. The remaining 700 (89.9%) cases were negative. The main reason for submission for RABV testing was human exposure to a potentially rabid animal in 414 (52.3%) cases. Among the 79 positive cases, 74 (93.7%) involved wildlife; raccoons (51 cases; 68.9%) were the primary host confirmed with RABV infection, followed by skunk and fox (8 cases each; 10.8%), bobcat (5 cases; 6.8%), and bats (2 cases; 2.7%). Only 5 domestic animals (6.3% of the positive cases) tested positive during our study period; one from each of the bovine, canine, caprine, equine, and feline species. Hence, the sylvatic cycle plays the predominant role in circulating RABV infection in our study area.
Rabies is a fatal zoonotic viral disease that continues to threaten both human and animal health worldwide, despite the availability of vaccines that can prevent rabies.6,15 It is estimated that globally rabies is responsible for > 60,000 human deaths annually. 15 In addition, ~ 15 million people take rabies post-exposure prophylaxis (PEP) each year. 15 The cost for rabies PEP averaged $3,800 per person in 2017–2018 in the United States (range: $1,200–$6,500), which results in an average annual estimated PEP cost of $209 million (range: $66–358 million). 12 This estimate does not include costs for hospital treatment or wound care. 12
Rabies virus (RABV; Rhabdoviridae, Lyssavirus rabies) is a negative-sense, single-stranded RNA virus. 15 Infection typically begins in muscle as a result of a bite from a rabid animal, from whence the virus travels across neuromuscular junctions to peripheral nerves and uses retrograde axonal movement to arrive at the CNS. 3 RABV then reverses its movement and migrates out of the CNS in an anterograde pathway towards the periphery. Centrifugal dissemination, mainly to the salivary glands, where the virus can simply be passed on in the saliva through a bite, is required to resume the cycle. 3 Non-bite exposure consists of inhalation of aerosolized RABV particles, organ and corneal transplants, and contamination of open wounds, abrasions, and mucous membranes with infectious material, such as brain tissue or saliva from a rabid animal; however, very few cases of the disease have been reported due to these routes of exposure. 15 The incubation time for RABV infection in humans and animals can last 20–90 d. 15 Rabies is almost invariably a deadly disease, and once CNS signs develop, mortality reaches 100%. 15 PEP can be effective in preventing clinical signs and disease if administered immediately after exposure. Given that the exposure event should be easy to identify, and the incubation period is long, PEP immunization can induce a protective immune response.6,15
There are dozens of RABV strains, each of which is associated closely with a host mammalian species in either the orders Chiroptera (bats) or Carnivora (dogs, raccoons, foxes, skunks). 3 In Georgia and nearby states, the predominant terrestrial RABV variant is the eastern raccoon variant. 7 Bat RABV variants are widespread in the United States, including our study area. 16 RABV has 2 epidemiologic cycles that are interconnected, the urban and sylvatic cycles. The urban cycle has pet dogs and cats as vectors or reservoirs, and the sylvatic cycle has wild mammals, such as fox, raccoon, jackal, wolf, badger, mongoose, and bats, as vectors or reservoirs. 15 The urban cycle is maintained mostly in community and stray dogs; transmission to pet dogs generates an additional load to humans at risk of rabies worldwide. 15 In the United States, rabies is enzootic in no fewer than 20 species of bats and terrestrial hosts, which include raccoons, skunks, foxes, and, solely in Puerto Rico, the Indian mongoose. 8 Due to extensive vaccination and control programs, the United States declared canine rabies–free status in 2007. 2 However, wildlife variant RABVs in specific reservoirs still have remarkable public and animal health, as well as economic, consequences throughout the United States. 2 Since the 1990s, oral vaccination has been the leading strategy for wildlife rabies control in the United States. 2
Several tests are available for the detection of RABV.11,17 Primary detection tests include the direct fluorescent antibody test (DFAT), the direct rapid immunohistochemistry test (dRIT), and the pan-lyssavirus reverse-transcription PCR (RT-PCR) assay. The DFAT, dRIT, and RT-PCR assay provide an accurate result in 98–100% of cases for all lyssavirus strains if appropriate conjugates or primers/probe are used. 17 The DFAT, which is recommended by both the WHO and WOAH, is the most widely used test for RABV detection. The DFAT is highly sensitive and specific (96–99%) and is considered the gold standard.11,17 The dRIT can be used as an alternative to DFAT in routine RABV testing as it has similar sensitivity and specificity, and the dRIT can be used in laboratories that do not have access to a fluorescence microscope. 17 Virus isolation can be used as a confirmatory test if the DFAT, dRIT, other antigen detection tests, or an RT-PCR assay, give inconclusive results. 17 Wherever possible, virus isolation in cell culture should replace the mouse inoculation test. Serologic tests such as virus neutralization (VN) and ELISA are suitable for monitoring the antibody response of vaccinated animals for rabies control. For measuring antibody responses to vaccination prior to international animal movement or trade, only VN results (fluorescent antibody VN test, and rapid fluorescent focus inhibition test) are acceptable. 17 Serologic tests are not allowed as primary detection tests for RABV. 17
The Tifton Veterinary Diagnostic and Investigation Laboratory (TVDIL), College of Veterinary Medicine, University of Georgia (UGA; Tifton, GA, USA) routinely receives requests for RABV testing in submissions of various species of wildlife and domestic animals from South Georgia and nearby counties. We investigated which species are submitted most commonly for RABV testing, which species are commonly positive, how many cases are autopsy submissions, the counties from which the animals originated, and the most common reasons that RABV testing is sought.
We searched all case submissions to the TVDIL for rabies testing between July 2010 and June 2021. Research ethics approval was not required for our study because it was a retrospective study using information from dead animals. The TVDIL received 792 RABV test request samples during the study period (Fig. 1). Submissions included whole body, head, or brain. Submissions to the TVDIL are from local veterinarians and directly from owners who do not have a veterinarian; wild animals for RABV testing; or farm animals for autopsy. In 208 cases, the animal was submitted for autopsy at the TVDIL. In the cases in which an autopsy was not performed, the laboratory was responsible for either removing the head or removing the brain and shipping it to one of the regional health public laboratories or another diagnostic laboratory for testing. Submissions for RABV testing can also be shipped directly to a regional health public laboratory by referring veterinarians. However, the public health laboratories only accept submissions of the head or the brain. Therefore, many clinicians prefer to submit the entire carcass to the TVDIL to have the head or brain removed instead of performing this task in their clinics.
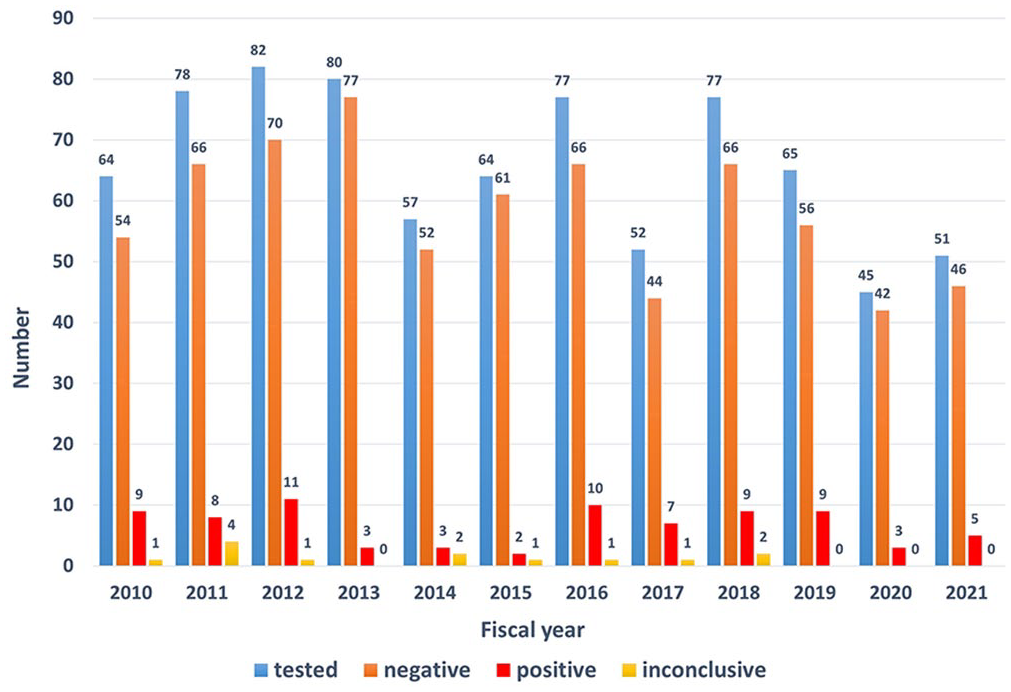
Results of a retrospective rabies surveillance study of 792 samples across 23 animal species submitted for a direct fluorescent antibody test per fiscal year (2010–2021) in the southern United States.
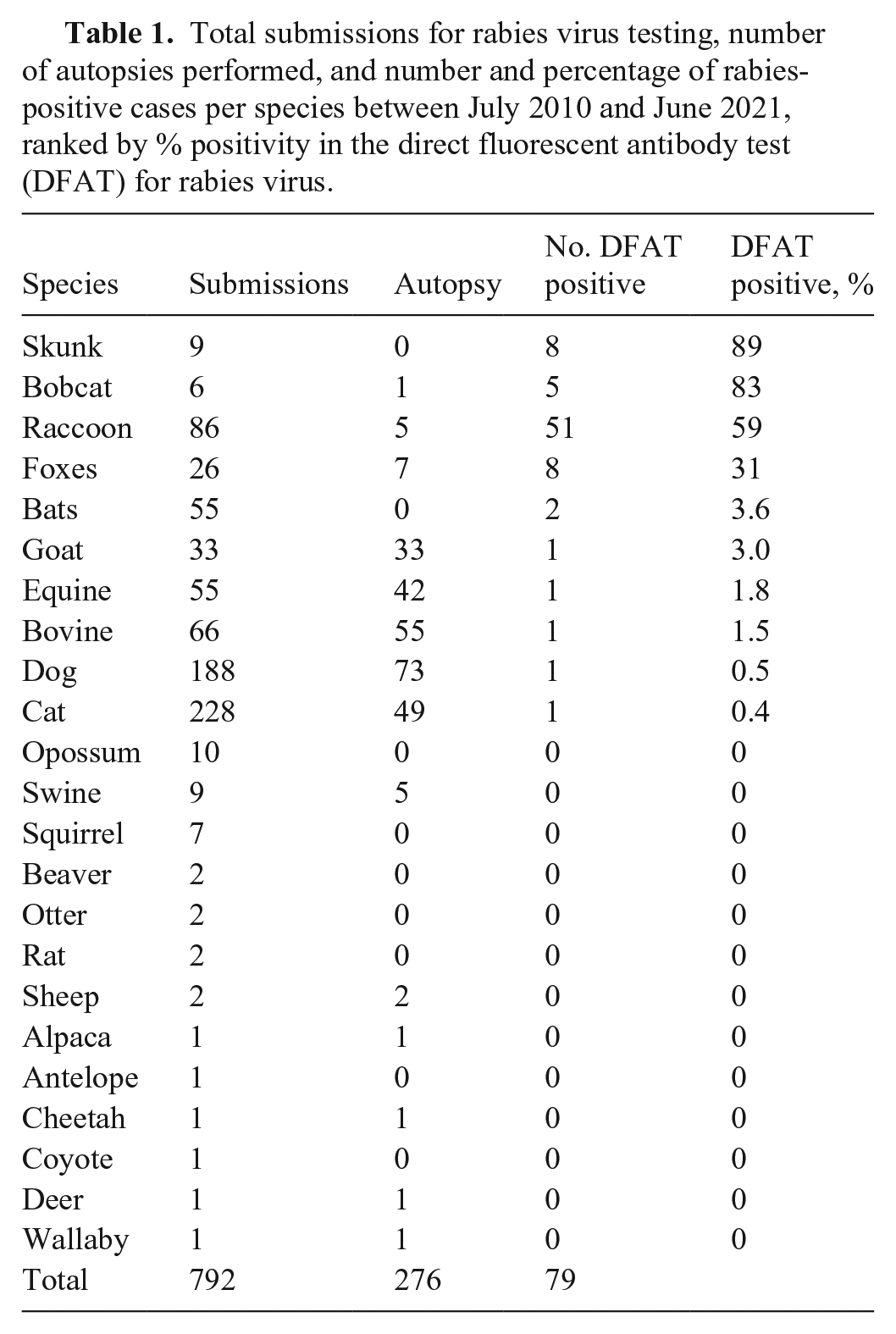
RABV status was confirmed in brain samples using the DFAT; reliable results on fresh specimens can be obtained in < 2 h. Test sensitivity depends on the specimen, the degree of autolysis, and the sample type. 17 RABV DFAT is not available at the TVDIL. Samples were forwarded from the TVDIL for a DFAT at 1 of 4 laboratories: UGA–Athens Veterinary Diagnostic Laboratory (39 cases; AVDL, Athens, GA, USA), and Georgia Public Health Laboratories in the cities of Albany (291 cases), Decatur (121 cases), and Waycross (341 cases). There are no fees for rabies testing at the 3 Georgia Public Health Laboratories; a fee is charged for testing at the UGA–AVDL. Out-of-state submissions are not tested at the Georgia Public Health Laboratories. The animal submissions for rabies testing were from 23 animal species (Table 1). In 13 (1.6%) cases, the DFAT result was inconclusive (sample was unsuitable or unsatisfactory for testing); 779 (98.4%) cases had a conclusive (positive or negative) test result; 700 (89.9%) cases were negative. Of the 779 DFAT conclusive test result cases, 79 (10.1%) tested positive across 10 animal species (Table 1).
Total submissions for rabies virus testing, number of autopsies performed, and number and percentage of rabies-positive cases per species between July 2010 and June 2021, ranked by % positivity in the direct fluorescent antibody test (DFAT) for rabies virus.
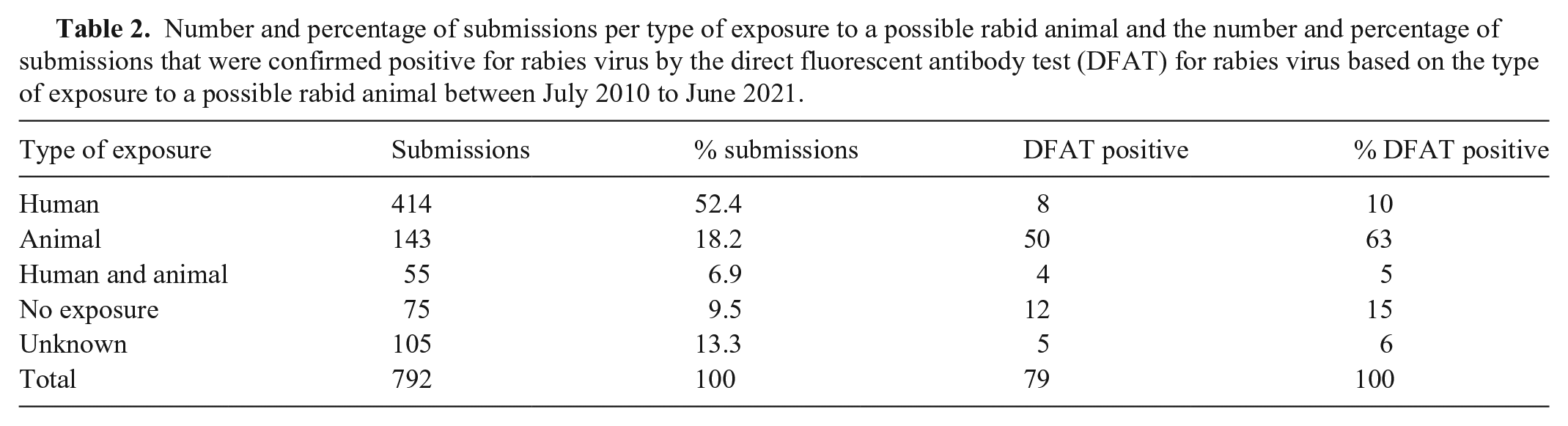
Submissions originated from 89 counties in Georgia, and 4 neighboring counties in Florida, 1 in South Carolina, and 1 in Alabama. Submissions from other states are by either a veterinarian practicing in both states or because of a veterinarian’s personal preference to submit to the TVDIL. The reason for submission for RABV testing was human exposure in 414 (52.3%) cases, animal exposure in 143 (18.1%) cases, and both human and animal exposure in 55 (6.9%) cases (Table 2). No exposure was recorded in 75 (9.5%) cases. The history of exposure was unknown in 105 (13.3%) cases, of which 73 were autopsy cases in which the reason for testing was prompted by neurologic clinical signs, histopathologic findings, or submitting veterinarian request. An autopsy was performed in 276 (34.8%) cases, and included autopsies performed by TVDIL pathologists (208 cases) or by the submitting veterinarian (68 cases), in which either the head or brain was received for testing (Table 1). Autopsy was performed in only 3 (3.8%) of the positive cases. In all autopsy cases, the brain was examined histologically for the detection of Negri bodies (Fig. 2) and mononuclear ence-phalitis.
Number and percentage of submissions per type of exposure to a possible rabid animal and the number and percentage of submissions that were confirmed positive for rabies virus by the direct fluorescent antibody test (DFAT) for rabies virus based on the type of exposure to a possible rabid animal between July 2010 to June 2021.
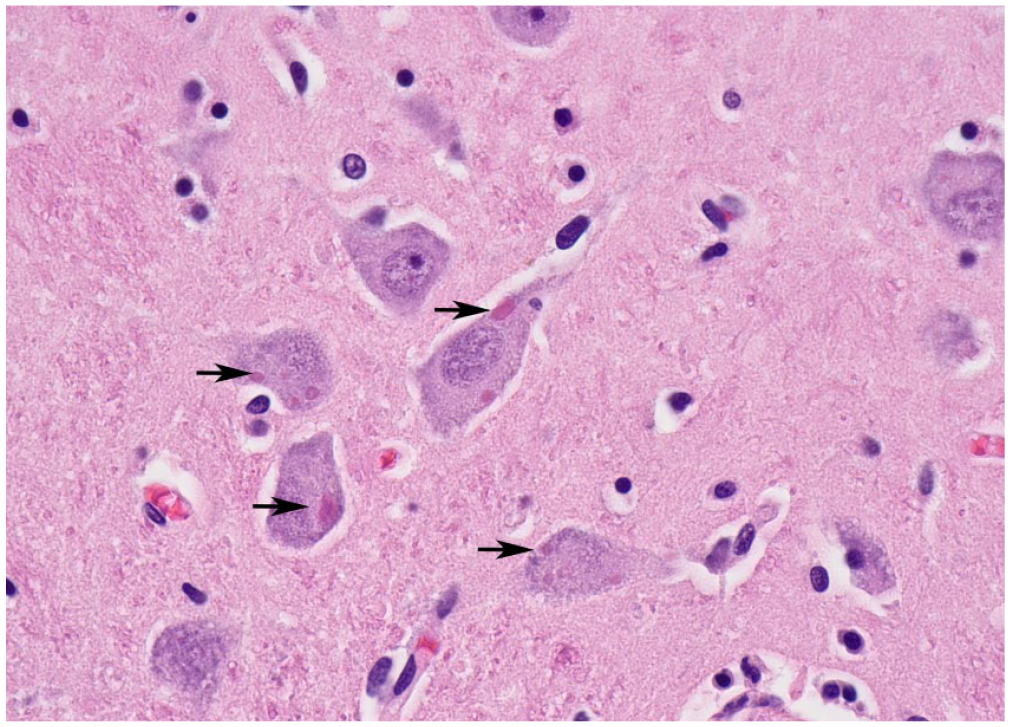
Brain section from the rabid dog, with eosinophilic intracytoplasmic viral inclusion bodies (Negri bodies) in neurons (arrows). H&E.
The lowest percentage of positive cases was observed in June (3.9%), followed by January (4.8%), August (7.0%), July (9.7%), and March (9.9%), with the remaining months having 10% or more positive cases each, with the highest percentage seen in May (13.8%).
Among the 79 positive cases, 74 (94%) involved wildlife, with raccoons (51 cases; 69%) representing the primary host confirmed with rabies, followed by skunk and fox (8 cases each; 118%), bobcat (5 cases; 7%), and bats (2 cases; 3%). Only 5 domestic animals tested positive during the study period, representing only 6% of the positive cases, one each from bovine, canine, caprine, equine, and feline species.
In 51 (65%) of the RABV-positive cases, clinical history and/or clinical signs were provided with the submission. The only positive cow vocalized excessively, was aggressive, and died during an attempt to restrain. A rabid skunk was reported to be killed on the same property 6 wk before the clinical signs were observed in the cow. The only positive dog submitted for autopsy was sick for 4 d, was lethargic and drooling, followed by aggressive behavior towards other dogs in the household, open-mouth breathing, abnormal behavior, and cage aggression. The dog was found comatose in its cage, rapidly progressing to cardiopulmonary arrest and death. This was an outdoor dog with no known exposure and a history of rabies vaccination 4 y before developing the disease. The type of vaccine or how many doses were administered was not provided with the history submission. The only positive cat was an 8-mo-old cat submitted due to human exposure because a person was bitten and scratched by the rabid cat.
In the 2 positive bats, both were submitted due to animal exposure. In one case, an unvaccinated kitten caught the bat, and in the other case, the bat possibly had bitten a dog. In 5 of the 8 positive foxes, a history was provided. In one of the cases, a fox bit a dog. In the other case, both a cat and a dog were exposed. Three of the submissions were due to human exposure. In 2 cases, a history of superficial bites was provided without details. In the third case, an unprovoked rabid fox attacked and bit a person on the right foot.
A history was provided in only 5 of 8 positive skunk cases. In all 5 cases, history implied that a dog was exposed. A history was provided in 4 of 5 positive bobcat cases. In 2 cases, submission was due to human exposure and in 1 of these cases, an unprovoked bobcat attacked and jumped on a person who was bitten in the left forearm. In one of the submissions, both a dog and a person were bitten by a bobcat in the backyard. In the last case, a goat killed a bobcat.
History was provided for 34 of the 51 positive raccoons in our study. In 24 cases, submission was due to dog exposure. In 19 cases, the rabid raccoon fought with a dog or multiple dogs, and either the dog(s) killed the raccoon, or the dog(s) owners killed it. In 3 cases, submission was due to domestic cat exposure; in all 3 cases, a rabid raccoon attacked the cat. In 2 other cases, a horse and a goat were exposed to a rabid raccoon. In 2 cases, the raccoon either had neurologic signs or was walking around the property. In 1 of the cases, a 2-mo-old orphaned raccoon of a litter of 5 animals bit the person that tried to catch it. The only positive raccoon that an autopsy was performed on belonged to a wildlife sanctuary. This raccoon behaved aggressively when ~ 1.5-mo-old and died suddenly at 3.5-mo-old; it was seen fighting with another raccoon in the same cage.
Human exposure in 12 of the positive cases was documented in 4 foxes, 3 raccoons, 2 bobcats, and 1 each of horse, cat, and dog. In 9 cases, history was provided. Bite wound was documented for the cat, 3 foxes, 2 bobcats, and 1 raccoon. In one of the histories provided, the person was not sure if bitten by her pet cat or the rabid raccoon. Several people were exposed to the positive dog, but the nature of exposure was not documented. The main source of exposure for domestic animals was raccoons in 34 of the positive cases in which animal exposure was documented alone or in conjunction with human exposure, followed by skunk (8 cases), foxes (4 cases), and bobcats (4 cases).
In our study, 98.4% of cases had a conclusive (positive or negative) test result over a period of 10 y, which is similar to national records; in 2020 and 2021, 97.3% and 97.5% of the U.S. submissions had a conclusive result, either positive or negative, respectively.8,9 Although only 1 cat and 1 dog tested positive for rabies during our study period, cats (228) and dogs (188) were the species submitted most commonly for testing during our study period. Although now a disease mainly in wildlife, there is still a fear of transmission of rabies from companion animals in the United States. This was also evident in a study in Tennessee 5 in which most dogs were tested because of having bitten a person rather than any clinical suspicion of rabies; based on the discrepancy between rabies epidemiology and animal submissions for testing, testing requests were likely not indicative of the rabies risk in most circumstances. 5
We found variation in the percentage of positive cases across months and variation in the number of cases submitted for rabies across the years (Fig. 1). This could be explained by the diverse ecology that affects the transmission dynamics of RABV among its bat and carnivore reservoirs. 3 Contributions to RABV ecology and maintenance of the disease include annual and multiannual cycles among host reservoirs, spillover from one host species to other species, population densities of hosts, demographic structure, spatial structure (rivers, mountains, and other landscape barriers), vaccination campaigns, dependence on human-provided resources for host species, natural host dispersal, and intentional or unintentional human interference with host dispersal.3,7 These factors also likely contribute to seasonal and annual variations observed in positive cases of the disease across a specific geographic area and could in part explain our findings. In the United States, the number of animal samples submitted for rabies testing in the calendar years 2020 and 2021 were significantly lower than the pre–COVID-19 years.8,9 This decline in testing was attributed to the influence of the COVID-19 pandemic on human–animal interactions and overworked public health systems. 9
We found that the main reason to seek rabies testing at the TVDIL was human exposure to a potentially rabid animal (Table 2). Although the main reason for testing was human exposure, > 50% of the positive submissions were observed in cases in which the reason for testing was domestic animal exposure to a suspected rabid animal. Wildlife exposure was the main source of human and animal exposure to RABV. Among the 79 positive cases in our study, 74 (93.7%) involved wildlife, which is comparable to the 91.5% reported in U.S. wildlife during 2021. 8 Nationally, bats and raccoons were 33.9% and 28.1%, respectively, of the rabid wildlife reported in 2021, followed by skunks (18.9%) and foxes (8.6%). 8 One reason that raccoons appear overrepresented, and bats appear to be underrepresented in our study, may be related to the geographic region because in our area the raccoon rabies variant is predominant.7,8,16 Another reason may be that animal exposure was mainly observed in domestic dogs that encountered a rabid raccoon or a skunk. Information on the fate of dogs exposed to a rabid raccoon or skunk was unavailable. RABV variants maintained in mesocarnivores typically circulate in separated geographic regions of the United States. 8 In the geographic area of our study, when the variant is reported, rabid raccoons are usually found to be infected with the eastern raccoon variant.7 –9 Raccoons are likely the main reservoir as well as the main species to transmit rabies to pet dogs and cats in our region.
Human exposure in our study was documented as associated with the following positive animals: foxes, raccoons, bobcats, a horse, a cat, and a dog. There were no documented human deaths due to rabies in Georgia or neighboring states from which a few samples were submitted (FL, SC, AL), 8 suggesting that none of these people succumbed to RABV infection. Bats are considered a primary source of human rabies in the United States. 8 Bats were the second most tested wildlife species in our study (55 cases); however, only 2 cases (4%) tested positive for RABV. In both cases, tests were sought due to animal exposure, not human exposure. In 2021, there were 5 confirmed human deaths in the United States due to rabies, 4 occurred after direct contact with a bat and 1 occurred after a bite from a dog abroad. 9 There were no reported human deaths attributed to rabies in 2019 and 2020.8,9 The most frequent reason that patients did not receive PEP was the lack of knowledge that bats can transmit rabies. 9 RABV isolates from bats are more virulent when inserted superficially into the epidermis because the bat RABV replicates more quickly in non-neuronal cells and at lower temperatures than does the dog RABV. 15 Percutaneous infection likely happens in the course of unnoticed skin touch and shallow bites and scratches. 15 Most human rabies cases in the United States are caused by viral variants from insectivorous bats. 1 Rabies as a potential cause of mass mortality events in bats has been also reported in the United States. 1
During our study period, only 1 of 188 (0.53%) dog submissions was positive for rabies. This was an autopsy case performed by a TVDIL pathologist. The dog was reported as having immunization for rabies 4 y prior. There was no follow-up information from the Georgia Public Health laboratory in which the sample was tested to know which strain of RABV was present in the rabid dog. Rabies is considered uncommon in previously vaccinated dogs but can occur even in dogs with current vaccination status. 10 Therefore, veterinary practitioners and pathologists should always consider rabies as a differential diagnosis for any dog or cat with clinical signs consistent with rabies, independent of vaccination history. 10 The canine RABV variant (CRVV) has been eradicated from the United States since 2007. 2 Maintaining this eradicated status requires viral characterization of every rabid dog. This has been challenging; the average viral characterization rate during 2015–2020 was only 55% 8 ; improvements have been made and the characterization rate increased to 86% of cases in 2021. 8 From 1938 to 2018, 588 cases of human rabies were reported in the United States. 12 The eradication of CRVV in the United States has resulted in a 10-fold reduction in human rabies fatalities described from 1938 through 2018. 12 Throughout 1960–2018, among 125 described human rabies fatalities, 89 were U.S.-acquired, including 6 organ transplantation cases. Among all U.S.-acquired cases, 62 (70%) were caused by bat RABV variants. Since 1960, 36 (28%) U.S. citizens have succumbed to rabies infection transmitted from dogs when traveling overseas. 12 Dog-related human rabies fatalities are endemic in most developing countries, primarily in Africa and Asia. 4 In the United States, the Centers for Disease Control and Prevention (CDC) controls the importation of dogs to avoid the re-establishment of CRVV. 13 Worldwide, CRVV is responsible for 98% of human fatalities due to rabies. 13 However, a trend of falsified, inaccurate, or incomplete rabies vaccination certificates was seen during 2019 and 2020, indicating a trend toward the attempted importation of unsatisfactorily vaccinated dogs into the United States, which could reintroduce CRVV. 13
All TVDIL personnel handling rabies-suspect samples are required to have pre-exposure prophylaxis (PrEP) vaccination. All vaccinated employees are also required to have a biennial rabies titer check. In addition, other measures are implemented, such as the use of proper PPE when handling samples and holding other tests on fresh specimens until DFAT results are known. In 2022, the Advisory Committee on Immunization Practices made several updates to the rabies PrEP recommendations, including the use of 2-dose intramuscular rabies vaccination and the minimum acceptable rabies antibody titer to be 0.5 IU/mL. 14 During January 2000–December 2020, 52 human rabies cases were identified in the United States, 38 of which were infections obtained within the United States; none was an individual who had received PrEP. 14
Our study indicates that non-domestic animals (primarily raccoons) were the main species affected and may play a significant role in circulating RABV infection in our study area. Several limitations may have influenced the findings of our study. One is that we only evaluated a 10-y period, which may have impacted the evaluation of any trends in epidemic cycles and transmission dynamics. In addition, given that ours was a retrospective study, the information on clinical history, clinical findings, and nature of exposure, was limited to the information recovered from the available files; in many cases, this information was not provided at the time of the submission. Therefore, this information may not be representative of the population affected. The geographic area studied is biased to the area that is within the reach of the TVDIL and may not be an accurate representation of the actual positive cases within this geographic area because affected animals may not have been submitted for testing due to the physical distance or have been submitted to other diagnostic laboratories. Finally, our study is restricted to only submissions that came through the TVDIL and does not incorporate submissions from the same region during this period that were submitted directly to the regional public health laboratories equipped for rabies testing.
Some of the strengths and implications of our study need further consideration. Only 10% of the animals tested were positive for rabies during the study period, suggesting that most rabies-suspect cases are negative for rabies in our area. Most submissions to the diagnostic laboratory that are positive involve wildlife species, mainly raccoons, therefore lab employees should be aware and take extra caution when handling these specimens as they likely are positive. Although most submissions are due to human exposure to a potentially rabid animal, the likelihood of a positive case in this circumstance is considered low. Our study demonstrates that a broad range of domestic and non-domestic animals were tested for rabies during the study period and that the TVDIL, as the only veterinary diagnostic laboratory in South Georgia, plays a pivotal role in providing local rabies surveillance and support for owners as an intermediary between the owner and the laboratories equipped for rabies testing.
Footnotes
Acknowledgements
We thank the current and past staff of the necropsy and receiving sections at the Tifton Diagnostic Veterinary and Investigational Laboratory for their work on all rabies submissions to the laboratory used for our study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Katie A. Dawson and Emily L. Atkinson received internship stipends from the USDA-NIFA-Higher Education Challenge Grant and from the USDA-NIFA-Non-Land Grant Colleges of Agriculture Grant between 2022 and 2023. Publication costs for this manuscript will be covered by the Tifton Veterinary Diagnostic and Investigational Laboratory.
