Abstract
The 1890s marked a significant milestone with the introduction of antibody-based agglutination and precipitation assays, revolutionizing the detection of bacterial pathogens in both animals and humans. This era also witnessed pivotal contributions to our understanding of humoral immunity, as researchers elucidated the structure and functions of antibody molecules, laying the groundwork for diagnostic applications. Among antibody isotypes, IgG is of paramount importance in diagnostic investigations given its definitive indication of infection or vaccination, coupled with its widespread presence and detectability across various specimen types, such as serum, colostrum, milk, oral fluids, urine, feces, and tissue exudate. Despite their resilience, immunoglobulins are susceptible to structural alterations induced by physicochemical and enzymatic processes, which can compromise the reliability of their detection. Here we review comprehensively the historical milestones, underlying mechanisms, and influencing factors (e.g., temperature, pH, storage) that shape the structural integrity and stability of IgG antibodies in aqueous solutions and various clinical specimens.
Historic discoveries in humoral immunity are closely intertwined with key advances in medicine and the prevention and control of infectious diseases in humans and animals. By the 1760s, a number of workers had observed that cowpox, a mild disease in humans caused by infection with vaccinia virus, conferred protection against smallpox. 7 “Vaccination” (Edward Jenner’s term) experiments in which people were intentionally exposed to vaccinia virus corroborated this idea. 61 Ultimately, the concept that an “antitoxin” produced by the body could counter a “toxin” produced by a pathogen was affirmed in the 1890s when Behring showed that “serum therapy” conferred resistance to diphtheria and tetanus.65,80 These protective substances were termed “antibody” by Paul Ehrlich in his publication, Collected Studies on Immunity. 33 Subsequent work with immune precipitates from horse and rabbit serum revealed 2 principal forms of antibody with distinct masses and sedimentation rates, namely, high (19S) and low (7S) molecular weights—known today as immunoglobulins M and G (IgM, IgG, respectively).34,53
The ability of antibodies to agglutinate bacteria was recognized in 1889, 64 and the concept was quickly used to develop assays for the detection of specific antibodies against a variety of bacterial pathogens of humans and animals (e.g., Salmonella spp., Brucella spp., Burkholderia mallei, Erysipelothrix rhusiopathiae). 85 This innovation was enthusiastically received, and by the early 1900s, highly portable agglutination test kits (“agglutometer”) were commercially available for typhoid in humans. 35
The development of antibody assays for viral pathogens was delayed until antimicrobials were available to control the growth of adventitious agents in cell cultures, but in 1941, the serendipitous observation that allantoic fluid from chick embryos infected with influenza A virus would agglutinate red blood cells led to the development of the hemagglutination and hemagglutination-inhibition tests.55,56
Subsequently, different detection technologies based on the use of antibodies were described (e.g., the use of fluorescein-labeled antibodies and fluorescence microscopy were used to visualize pneumococcal antigens in tissue sections). 24 The pace of technological innovation was accelerated in the post–World War II era. As an example, radioimmunoassay (RIA) was described in 1956 and later applied to a variety of targets (e.g., hormones, vitamins, enzymes).10,145 Derived from the principles of RIA, the ELISA was described in 1971,37,134,144 and quickly adapted for the detection of antibodies against a variety of pathogens. 113 Shortly thereafter, the discovery that monoclonal antibodies (mAbs) could be produced by fusing cultured myeloma cells and spleen cells from an immunized mouse 68 created new opportunities in both therapeutics and testing, with the lateral flow immunoassay format being perhaps its most widely recognized testing application. Other touchstones in our brief timeline include the production of recombinant protein and antibody engineering through phage display, 119 and the development of alternative immunoassay platforms such as AlphaLISA, 128 and fluorescent microsphere–based immunoassay. 9 In general, despite the development of various molecular technologies, the specific detection of antibodies remains an important tool to confirm and monitor effective exposure to a pathogen or antigen via infection or vaccination.
To our short chronology of antibody testing should be added one of the major contributions to disease control and eradication: that individuals infected with wild-type viruses could potentially be differentiated from vaccinated individuals by differences in their antibody responses. 131 For example, the DIVA (differentiating infected from vaccinated individuals) vaccination and testing strategy was successfully used in pseudorabies (Aujeszky’s disease virus, ADV) eradication based on a fundamental understanding of the functions of the key ADV glycoproteins. The same strategy is used during infectious bovine rhinotracheitis control. This test was a prerequisite for developing efficacious ADV marker vaccines and companion detection tests that made its eradication possible.122,132,133
Although IgG antibodies are a crucial class of therapeutics in human medicine, particularly in oncology, chronic inflammation, cardiovascular, transplantation, and infectious diseases, their application in veterinary medicine is relatively rare. Consequently, we do not cover their use in the veterinary field in our review.
For serologic testing to fulfill its full capability, the structure of the antibodies should be maintained close to native conditions. Refrigerated transport is the most common, if not the only, recommended practice for transporting specimens to veterinary diagnostic laboratories. However, several other physical and chemical effects discussed here can also alter the characteristics of antibodies and thus should be considered. Here we review the general structure of antibodies, specifically IgG, processes that may affect antibody structure, and some effects that should also be considered in specimen handling for maintaining antibody stability in clinical specimens. We performed our bibliography search with the main keywords “antibody structure”, “antibody stability”, “antibody denaturation”, and “antibody storage condition”, and incorporated the addition of specific physicochemical effects and specimen storage conditions including “aggregation”, “deamidation”, “oxidation’, “temperature”, “pH”, “freeze-thaw”, and “buffer composition” in Google and PubMed for publications before July 2022.
Antibody structure and function
Before the discovery of antibody molecular structure, the separation of proteins into multiple fractions, such as α-, β-, and γ- (which correspond to IgG in the modern name) globulins, from serum had preliminarily indicated the multiple isotypes according to their mobility during electrophoresis. 13 Subsequently, based on the distinct masses and sedimentation rates obtained by ultracentrifugation, precipitates from serum were classified based on their high (19S) or low (7S) molecular weights,34,53 and allowed further antibody purification. 53 This technique, together with antibody immunoreactivity characteristics, led to the discovery of more antibody isotypes. 13
Five antibody isotypes (IgA, IgE, IgG, IgM, IgD) are commonly found in mammals, 90 with additional antibody isotypes present in non-mammalian species (i.e., IgY in birds, IgX in amphibians). 130 In mammals, the proportion of each isotype varies across species, specimen types, and specific pathogens to which the animals were exposed. 90 We focus here on IgG because of its main role in veterinary diagnostic medicine, 83 largely because IgG is the most abundant immunoglobulin in serum (e.g., ~80–90% of the total serum antibody in swine). 26 Detectable levels of IgG can also be found in nearly every compartment and clinical specimen, including serum or plasma, colostrum, oral fluids, urine, feces, and meat or tissue exudate16,30,36,45,78,89,97,100 due to the widespread distribution of neonatal antibody Fc receptors (FcRs) in epithelial tissues,109,143 which actively facilitate this transportation across tissue barriers by selective binding to IgG, regardless of animal age. 147 Moreover, neonatal FcRs also possibly explain the higher and longer-lasting levels of IgG 90 compared to other antibody isotypes, such as IgA and IgM. 109
The structure of antibodies was hypothesized in 1940 by Pauling, 95 and the discovery of the molecular structure was later demonstrated in 1959, 1 leading to the current Y-shaped structure of IgG antibody.31,99 Workers in the 1960s established methods to fragment IgG, described the amino acid composition of antibody structures, and explored their functions.32,92 Finally, in 1977, an understanding of the 3-dimensional structure of the intact glycoproteic structure of the IgG molecule was achieved through X-ray crystallography.93,116
The mammalian IgG antibody is a Y-shaped glycoprotein 93 with the end of each arm of the “Y” (fragment antigen-binding region, or Fab, region) containing a hypervariable region (HV) that defines the specificity of the antibody’s antigen-binding properties (Fig. 1). 93 The base of the “Y” (fragment crystallizable, or Fc, region) is involved in the activation of complement, as well as interactions with specific FcRs associated with phagocytosis, cell-mediated cytotoxicity, transplacental transport of antibody, and antibody catabolism.16,105,136 The Fab and Fc portions of the antibody structure are connected by a hinge that allows the molecule to twist and flex during interactions with antigens, FcRs, and complement. 105 The hinge region varies in length among isotypes, as observed in the human antibody, with the increase in length corresponding to an increasing number of amino acids and disulfide bridges, 93 and also a greater susceptibility to degradation. 142
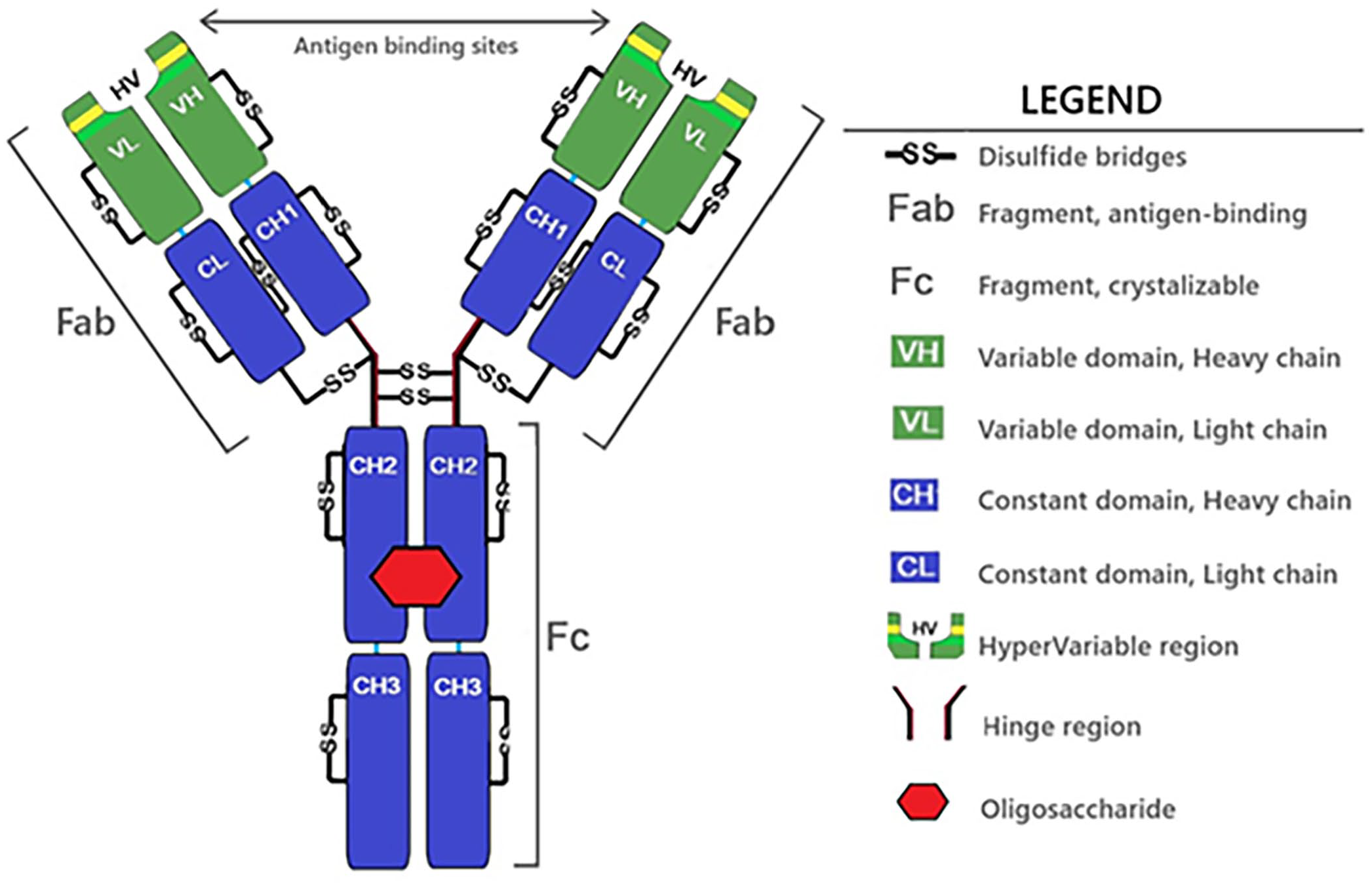
Primary or monomeric antibody (IgG) structure. VH+CH1+CH2+CH3 domains form the heavy chain, and VL+CL domains form the light chain. All domains include intra-chain disulfide bridges. Inter-chain disulfide bridges connect the light with the heavy chains. The Fabs and Fc functional portions assemble the IgG structure through the hinge region and disulfide bridges.
Structurally, the IgG antibody monomer is composed of 4 chains: 2 identical light chains, each comprised of 2 compact globular subdivisions or domains (VL and CL), and 2 identical heavy chains with 4 domains (VH and CH 1–3). 15 Intra-chain and inter-chain disulfide bridges hold the molecule together and provide stability. 142 Intra-chain disulfide bridges link 2 amino acids within the same domain, and inter-chain disulfide bridges link 2 amino acids on different heavy or light chains. 76 An oligosaccharide located between the heavy chains functions in modulating antibody interaction with complement molecules and FcRs, and also reduces the susceptibility of the antibody to degradation by proteases.5,87,102,142
Despite the similar molecular weights among IgG subclasses, the general IgG structure may differ from the length and flexibility of the hinge region to the number of disulfide bond bridges. These differences can affect the arrangement of the Fab arms within the Fc region, and thus cause the differences in effector functions among IgG subclasses. 136 However, beyond the general IgG structure, the diversity of the structure among some mammalian species should also be noted. Camelids (camels, llamas, alpacas, guanacos, etc.) are one exception, with the major type of antibodies differing from the general model by containing heavy-chain–only antibodies without the light chain and CH1 domain. 47 The difference in antibody structure raised curiosity about the mechanism and the function of a “smaller” variable region 41 ; the characteristics from the structure also enabled the single-domain fragments of camelid antibodies for pharmaceutical applications.49,130 In cows, antibodies also differ from the general mammalian IgG structure; the extra-long third complementarity-determining regions in the heavy chain were originally observed in IgM114 and later determined to be a trend existing across isotypes. 140 The “stalk and knob” analogy for the structure 141 may explain the antigen-binding affinity of the antibody toward the binding site surrounded by the complex glycan architecture 121 ; the exact reason behind the evolution of this structure remains unknown.
Physicochemical processes that affect IgG stability
Antibody stability is the tendency of the molecule to maintain its structural integrity and functionality over time. 59 IgG tends to be more stable than other proteins because of its globular structure, with hydrogen bonds in the peptide backbone structure and side-chain interactions in a 3-dimensional structure, making its conformation rigid. 77 Awareness of these physicochemical processes is important because these changes can affect their stability and functionality (i.e., the specificity and strength of antigen binding).
Specific limitations in the following discussion should be recognized: 1) most of the information is based on studies of human antibodies, particularly of the IgG class, 2) the mechanisms involved are not always fully understood, and 3) the high degree of homology in the mammalian IgG primary structure suggests a similar pattern of susceptibility among antibodies within the isotype, but minor variations in structure or composition likewise infer some variability in stability.16,38,136
Denaturation
At its simplest, 4 levels of protein structure are recognized: primary (amino acid sequences), secondary (alpha helices and beta sheet formation), tertiary (3-dimensional folded structure), and quaternary (assemblies of >1 amino acid chain). Denaturation of proteins is thus defined as changes in conformation (unfolding) that affect structure and/or functionality. 77 Changes in the tertiary or quaternary structures may be reversible (refolding); changes in the primary or secondary structures are irreversible. Antibodies fulfill their biological activity only when they are correctly folded; unfolding leads to conformational instability, permanent denaturation, and changes in binding activity, including inter-antibody interactions.77,108
Aggregation
First described in 1954, aggregation or “oligomerization” is protein self-association.59,77 Proteins may unfold and aggregate by physical association with one another and without any changes in primary structure (physical aggregation) or by the formation of new covalent bond(s) (chemical aggregation).67,110 Physical aggregation occurs after the tertiary structure unfolds sufficiently to expose hydrophobic zones and change the antibody polarity, thereby triggering aggregation. 40 Chemical aggregation results from the formation of disulfide bonds among antibodies or antibody fragments. 40
Isomerization
Isomerization is the transformation of one molecule into another because of a rearrangement of atoms, resulting in different molecular structures despite identical atomic composition, 48 including the transfer of the peptide backbone from the α-carbon of an aspartate or asparagine to the β-carbon.44,139 In particular, aspartate is an amino acid component of antibodies that is highly susceptible to isomerization when it is followed by small amino acids (e.g., serine, alanine, cysteine, glycine) and/or when it is in a slightly acidic environment.17,88,139 The occurrence of amino acid isomerization in the Fab HV region increases its flexibility and thereby unpredictably affects binding affinity through modification of the peptide structure. 17 However, compared to the homogeneity of mAbs, the effect of isomerization of aspartate residues on natural polyclonal antibodies in vivo could be minimal. 17 Overall, the effect of isomerization on the antibody structure, stability, and specific conditions and mechanisms under which aspartate isomerization occurs remain unclear.88,123
Deamidation
Deamidation is a spontaneous reaction in which the side-chain amide linkage in either an asparagine or glutamine residue is hydrolyzed to form a free carboxylic acid, thereby affecting the primary structure of the protein.21,59,107 Deamidation has been studied, often in conjunction with isomerization, since the 1980s as a means of altering protein structure and function. 44 Because asparagine and glutamine are primarily located in the light chain of the IgG Fab and HV regions, deamidation directly affects antibody binding affinity (Fig. 2). 123 Deamidation of IgG can occur under temperature and/or pH stress.21,46 Susceptible regions of the IgG molecular have been identified, but the mechanisms of deamidation are poorly understood. 123
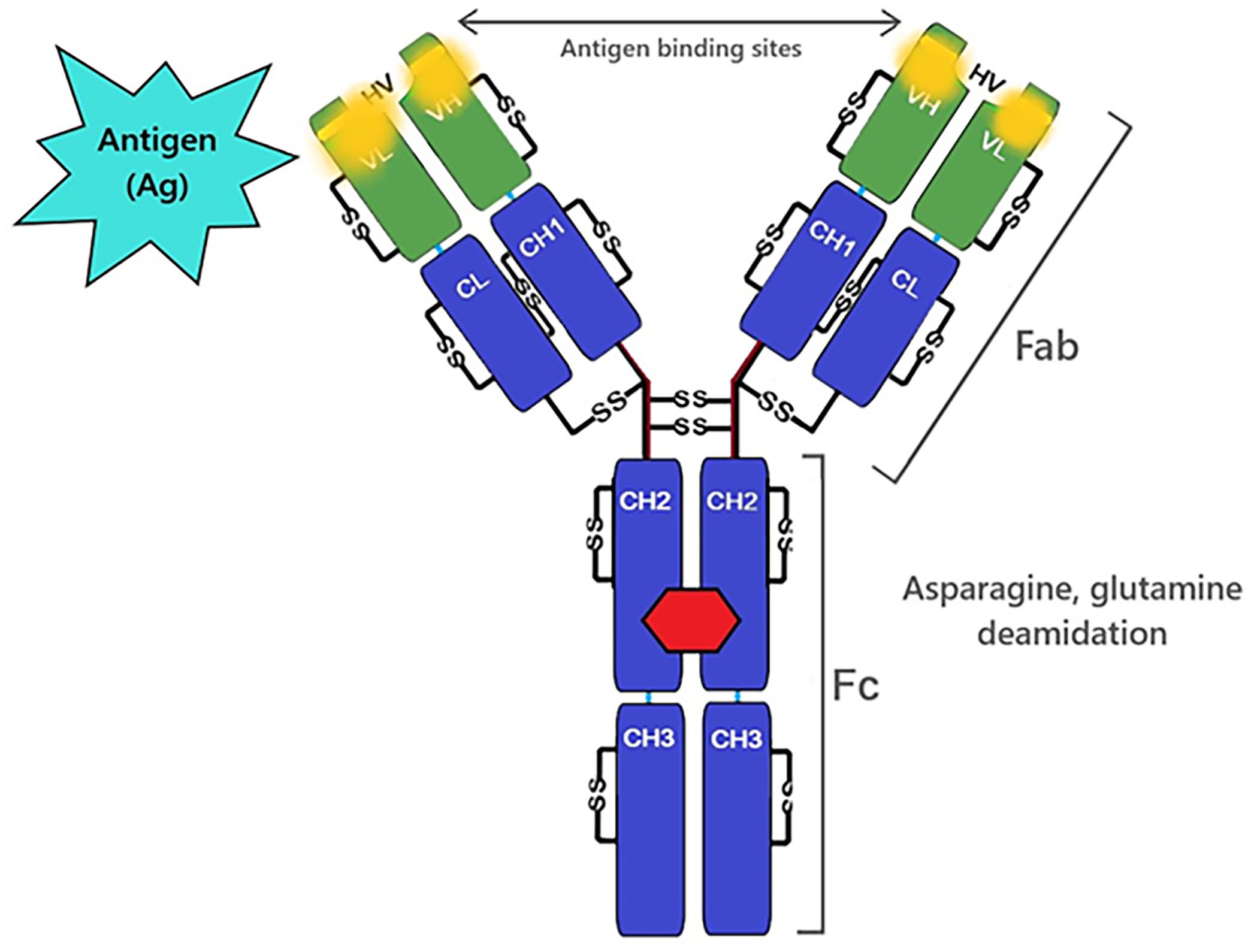
Deamidation of asparagine or glutamine (amino acids involved in antigen-antibody interactions) may affect antigen binding.
Polymerization
Polymerization is the combination of 2 or more small and similar molecules (monomers) to form a new macromolecule (polymer).52,146 Antibody polymerization is the result of the formation of new covalent bonds between 2 or more similar protein structures (protein cross-linking), with IgM and IgA being examples of naturally occurring polymers.20,52 Polymerization can be produced by chemicals that generate covalent bonds between lysine amino acids in the heavy chains of the IgG structure. For example, glutaraldehyde generates larger insoluble IgG polymers under pH 7 or smaller soluble IgG polymers under pH 9. 146 In contrast with other antibody physicochemical processes, polymerization is generally not temperature dependent. 146
Oxidation
Oxidation is a chemical process in which a molecule loses electrons. Storage conditions such as temperature and light exposure can be factors causing stress that leads to the degradation of molecules through oxidation. 70 In antibody molecules, oxidation modifies interactions between amino acids and may disrupt peptide bonds, thereby affecting the primary and even high-order structure levels of the protein. 126 The amino acid methionine is distributed throughout the IgG molecule, 88 and it is highly susceptible to oxidation (Fig. 3). 70 Specifically, the oxidation of methionine in the Fc region affects the antibody’s capacity to interact with FcRs and complement.11,22,76 Other amino acids are susceptible to oxidation (e.g., tryptophan, phenylalanine, tyrosine, cysteine, histidine), but their location in the interior of the molecule restricts their exposure to oxidizers.22,42,59
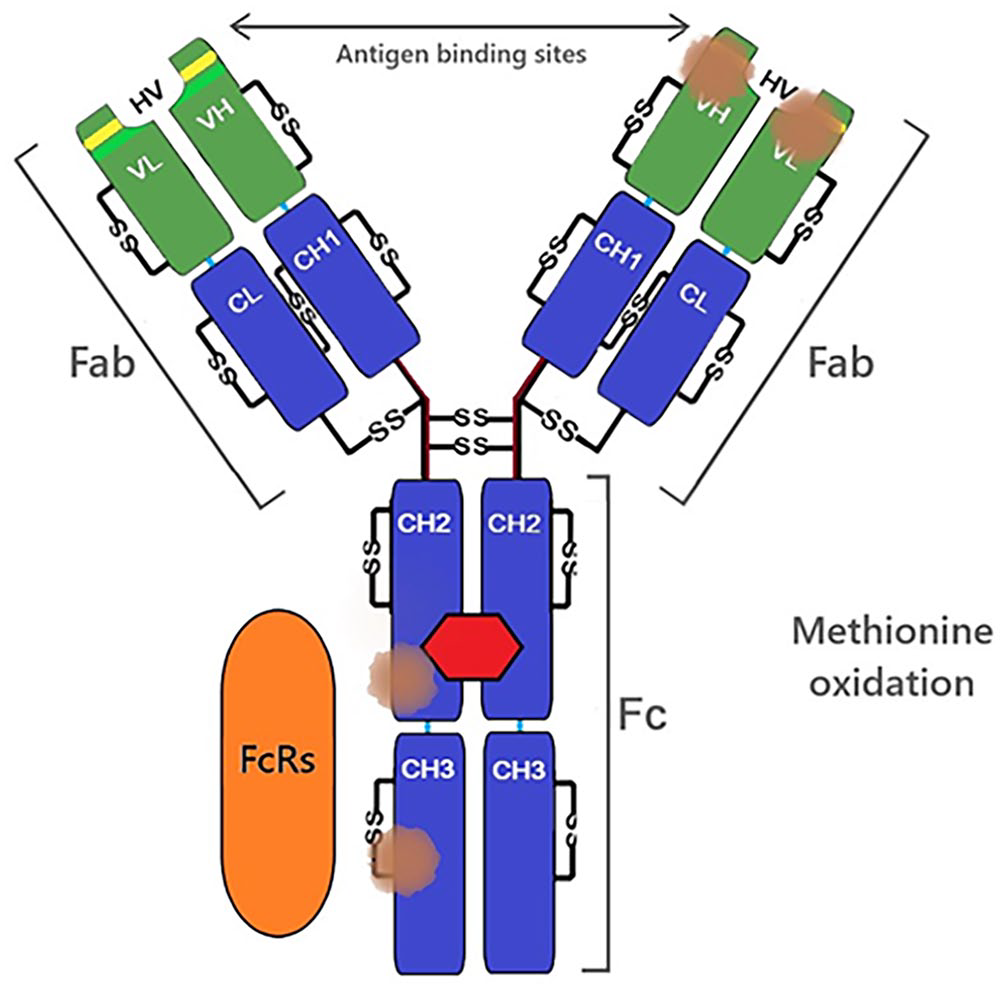
Methionine oxidation in heavy-chain regions may affect FcRs-IgG interactions. Methionine oxidation in the hypervariable region may affect antigen-IgG interactions.
Fragmentation via hydrolysis
Antibody fragmentation is a change in the structure of the protein backbone due to pH or enzymatic catalysis. 137 We review here non-enzymatic mechanisms that lead to antibody fragmentation; we discuss enzymatic processes involved in antibody fragmentation in the following section.
Hydrolysis is a non-enzymatic mechanism responsible for most IgG fragmentations, 43 and it is regularly observed at peptide bonds of the hinge region and is influenced by its flexibility. 25 Hydrolysis refers basically to the replacement of a molecule’s atoms by atoms of water, forming an intermediate that leads to the cleavage of peptide bonds, therefore, fragmenting the primary structure of the antibody. 137 Some amino acid peptide bonds are more susceptible to fragmentation by hydrolysis than others (i.e., peptide bonds next to aspartate, lysine, cysteine, and phenylalanine). 25 This process is time and pH-dependent, with a low reaction at neutral pH and a higher reaction in both acidic and alkaline conditions.25,43 In living organisms, the hydrolysis process is usually speeded by enzymes, 8 as further described in the following section.
Enzymatic processes that affect antibody
The enzymatic degradation of hemoglobin by papain and pepsin was originally reported in the 1930s.3,4 Using the same approach, the reduction in antigen-antibody interactions after enzymatic antibody fragmentation was reported in the 1950s.91,98 Papain and pepsin are nonspecific proteases capable of fragmenting all antibody isotypes under physiologic conditions (pH 7, 37°C; Fig. 4). Papain has higher cleavage activity on the amino acids phenylalanine, lysine, and glycine, 8 whereas pepsin cleaves peptide linkages formed by amino groups of aromatic and acidic amino acids, such as phenylalanine, tryptophan, and tyrosine (Fig. 4). 39 Papain preferentially cleaves IgG at the hinge region, dividing the antibody into the Fc portion and Fab portions (Fig. 4A). 8 Pepsin cleaves the IgG molecule below the hinge region, dividing the antibody into a linked Fab portion and an often-fragmented Fc portion (Fig. 4B).8,75 Enzymatic fragmentation at the hinge region is also used by bacteria to evade phagocytosis (e.g., the family of immunoglobulin-degrading enzymes [IdeS] produced by Streptococcus pyogenes, 138 and from Streptococcus suis).114,120 Some IdeS enzymes have highly specific proteolytic activity (e.g., the S. suis IgdE enzyme specifically cleaves the porcine IgG hinge region, 120 and the S. suis IdeSsuis enzyme exclusively cleaves porcine IgM). 114 IgG Fab and Fc fragments retain specific binding functions, but the separate components are unable to trigger a complete immunologic response (agglutination, precipitation, opsonization, etc.) 112 and do not perform adequately in detection assays.
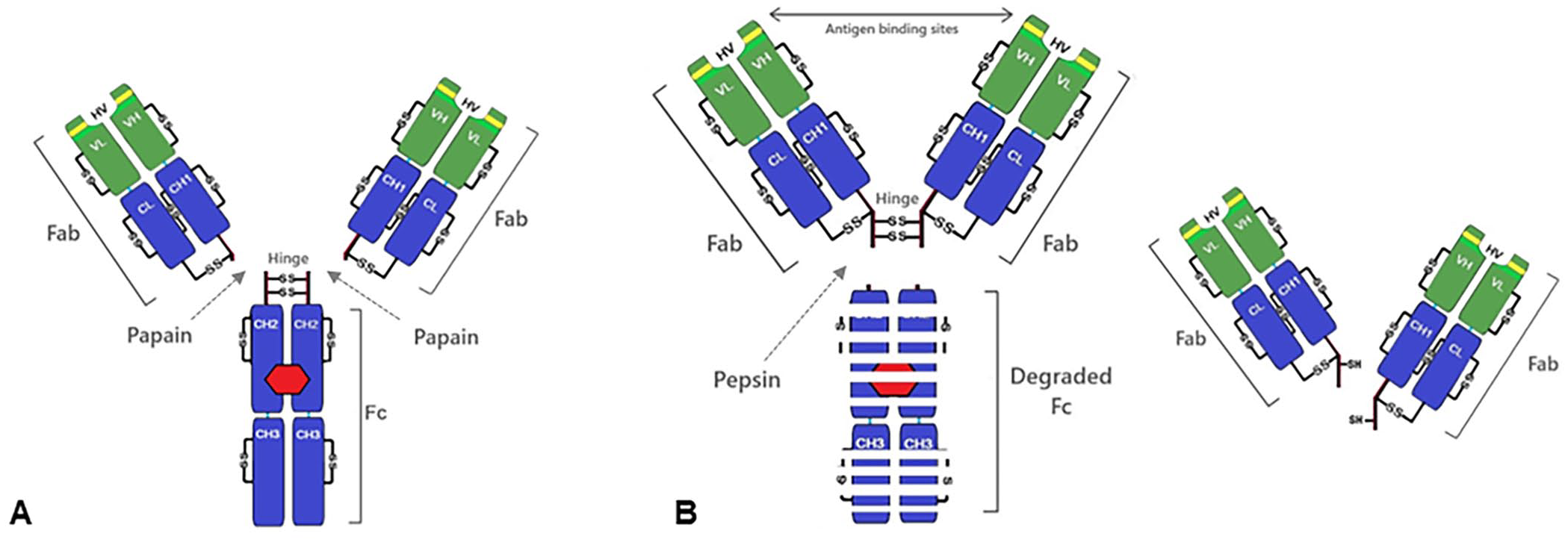
Antibody fragmentation by enzymes.
Considerations for clinical specimens
In this section, we discuss the physicochemical and enzymatic processes described above in the context of specimen handling and the diagnostic functionality of IgG.
Temperature
The temperature-by-time–dependent denaturation of IgG is one of the most important considerations in the handling and storage of diagnostic samples (Table 1).2,40,142 However, a simple formula describing this relationship has not been described, perhaps because the reports in the peer-reviewed literature differ considerably in 1) antibody evaluated, 2) specimen or matrix, 3) temperature and time evaluated, 4) assay(s) used to measure the effect, and 5) strength of the experimental design.
Temperature-time–dependent processes that may affect IgG stability.

Increased temperatures over time cause the unfolding of the secondary and tertiary structures of IgG, which, together with aggregation, lead to protein denaturation. 135 Earlier studies reported that 20 min at 70°C was sufficient to completely denature IgG in PBS at pH 8.1. 135 Heat-induced changes in protein structure can lead to antibody aggregation. As an example, IgG was reported to aggregate partly in PBS (pH 7.2) after 15 min at 77°C, 50 or the reduced concentration of monomeric IgG into an aggregated state in a formulation (pH 7.5) after 72 h at 60°C, or 4 wk at 40°C. 40
In addition to aggregation, thermal stress can facilitate other physicochemical processes that affect IgG functionality. 59 For instance, the oxidation of IgG in a buffered solution containing sodium acetate, sodium chloride, and polysorbate (pH 5.0), after 2 wk of exposure at 5°C, resulted in specific oxidation of 10% of the methionine in the Fc region, 17% at 30°C, and 52% at 40°C. 70 Deamidation of IgG can also be triggered by prolonged exposure (2–4 wk) at 25°C, or in a shorter time at an elevated temperature of 40°C (pH 6.0), which can result in deamidation in the heavy or light chain and the subsequent decrease of IgG binding activity. 46
The intermediate storage of antibody solutions at high temperatures can lead to instability because of denaturation of the protein structure and further aggregation. Likewise, thermal stress can also be associated with low temperatures (i.e., crystallization of certain solvents), and changes in the pH due to solvent freezing may affect protein stability. 72 Although IgG is highly resistant to repeated “standard” freeze-thaw cycles, snap freezing should be avoided because abrupt changes in temperature affect antibody integrity and increase aggregation. 111 A good practice of splitting a sample into multiple aliquots is recommended to avoid multiple freeze-thaw cycles. For short-term storage and transportation, specimens containing antibodies should be maintained preferably at a moderate-to-low temperature (~4°C) rather than frozen if subject to multiple freeze-thaw cycles. 62 As for long-term storage via frozen solution under −20 or −80°C or lyophilization, multiple factors (e.g., material of the storage container, aliquot volume [one-time use], type of cryo-protectant, concentration of the antibody in the stock solution), should be considered for long-term stability. 62
Several considerations are involved when assessing how freeze-thawing affects the stability of immunoglobulins as many stress factors included within the process may cause the proteins to aggregate, including but not limited to the change of temperature, crystallization of the solvent and/or the excipient, pH change during freezing, and the buffer choice.12,50 For example, the addition of excipients may affect the turbidity of the solution undergoing freeze-thawing, but whether this has an effect on the instability of antibodies remains under investigation.57,60 In these studies, IgG1 maintained its protein conformation better around a neutral pH (6–8) compared to an acidic pH (3). 57 Additional studies showed that the speed of freezing the antibody formulation may affect antibody integrity 111 ; however, various studies indicated that multiple freeze-thaw cycles had no significant effect on antibody aggregation.50,57,111 Overall, in contrast to the effect of thermal stress on antibodies, it can be concluded that the freeze-thaw cycle that specimens commonly undergo in the laboratory setting has little effect on IgG stability as demonstrated by reports showing that different serologic tests against various pathogens remained effective after serum samples underwent multiple freeze-thaw cycles.18,96,104,115,125
In addition to temperature and time effects, a further consideration for the stability of IgG in specimens is bacterial contamination. Unpublished pilot experiments showed that ELISA-detectable IgG declined after 2 d in swine oral fluids held at 30°C, but was extended to 12 d in samples held at 10°C, 14 d at 4°C, and months or more in samples stored at −20°C or −80°C (Table 2). Lower temperatures were associated both with increased IgG stability and with less bacterial proliferation in the specimen (Table 2).54,101 A similar effect was observed in samples treated with antimicrobial and/or antiproteolytic agents. Thus, ELISA-detectable antibody was stable for up to one month at room temperature in human saliva collected with a commercial device containing antimicrobial and antiproteolytic compounds. 124 Likewise, similar trends were observed in swine oral fluids treated with a bacteriostat (0.01% chlorhexidine digluconate). 101
Oral fluid IgG stability (ELISA-detectable IgG).
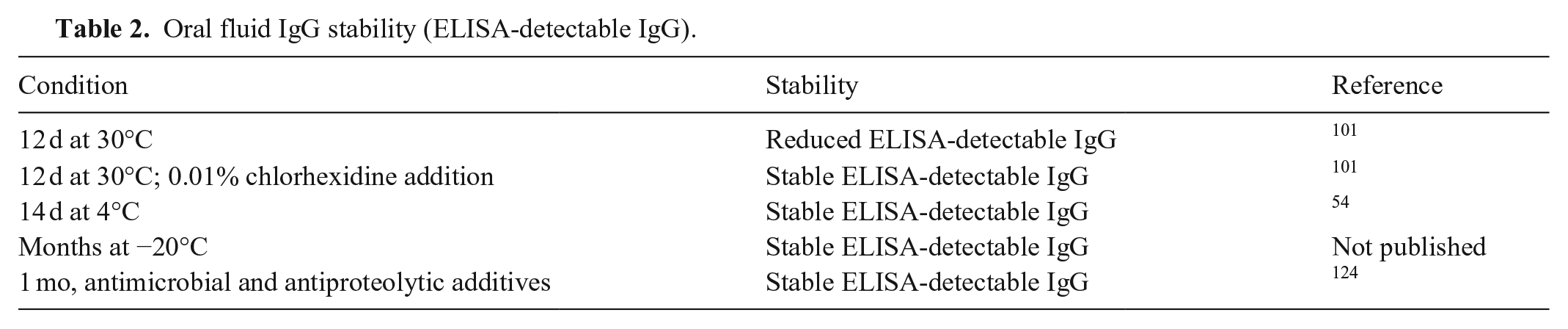
Hydrogen ion concentration (pH)
Changes in the solution pH can affect antibody stability by altering the distribution of charges on the protein surface, and even causing structural changes and denaturation. 135 These structural changes affect the biological functionality of antibodies by reducing their binding specificity and by triggering aggregation.
The storage of antibodies at extreme pH is not recommended due to the fact that IgG is naturally suspended in biological aqueous solutions with stable and physiologic pH conditions (pH ~7), but pH stability varies among antibody subclasses. Reports have indicated that at room temperature (20–25°C), changing the pH from 7 to 3, and from 7 to 10 by the addition of NaOH or HCl, and returning the pH to 7 after 30 min, markedly affects (35–76%) the binding specificity of human IgM, IgG1, and IgG2a,b subclasses through changes in the amino acid sequence of the antigen-binding region. 129 Changes in the pH can also cause antibody deamidation under alkaline conditions (pH 8–9) at room temperature (25°C). Specifically, increasing asparagine deamidation has been reported in both IgG heavy- and light-chain HV regions after 3 and 7 d of incubation, reducing binding activity by 10% and 20–25%, respectively. 46 Functionally, deamidation in the light chain in one of the complementarity-determining regions is unlikely to fully eliminate the entire antibody-binding ability, but the degree to which the specimen function will be affected typically depends on how long the specimens stay under the condition change and to what extent the pH change is happening. 46
Furthermore, structural differences in the inter-chain disulfide bridges and HV region amino acids or their Fc region can also make the difference in the susceptibility of antibody subclasses to pH changes.58,71,129 Differences in aggregation were also demonstrated using solutions of different IgG subclasses held to 40°C for 1 mo under different pH conditions compared with fresh-prepared homologs, and it was concluded that pH-dependency varied depending on the variable region and differences in the proximity of the variable region to CH2 regions. 58
Even though extreme pH might not be a process occurring purposefully during the storage of clinical specimens, it should still be considered because pH change or other considerations play an indirect role in other conditions (e.g., pH change during freezing or the optimal working pH with the addition of other excipients62,73,79), and pH can trigger factors affecting the stability of the antibody, such as fragmentation by hydrolysis.25,43
Ionic strength (salts)
The term “salts” defines any chemical compound consisting of an assembly of cations, or positively charged ions (e.g., chloride, acetate, citrate, carbonate, phosphate, sulfate), with anions, or negatively charged ions (e.g., hydrogen, potassium, calcium, sodium). 82 The addition of salts into protein and antibody aqueous solutions causes changes in protein hydrophobicity, surface charge distribution, or even changes in the structure, which might induce a shift in solubility and stability, resulting in aggregation and/or precipitation of the protein contained in the salt-added solution.6,73,84
The IgGs are amphipathic molecules (i.e., they exhibit a hydrophilic surface but also hydrophobic properties in their internal structure). 40 The effect of salts on the hydrophobicity and, therefore, on the stability of the antibodies, varies depending on factors such as buffer type, 63 the type of salt (chaotropic or kosmotropic, according to the Hofmeister series), 81 the protein-ion interaction, and the concentration of the salt in the buffer.6,118,127 Protein-protein interactions are also affected by changes in the ionic strength; an increase of ions may increase (salting-in) or decrease (salting-out) protein solubility, thus influencing aggregation.27,59,66,106
From a testing perspective, the selection of a specific buffer, along with other factors described here, can affect the properties and stability of antibodies. The choice of sodium phosphate or other buffers with the addition of salts can reduce the pH of IgG solutions via salt crystallization and, together with the following freeze-thaw conditions, leads to antibody aggregation and precipitation. 50 Antibody aggregation induced via supplementation of different salts was also observed at different pHs, 69 or via lyophilization. 111 In any case, the addition of salts during the preparation of buffer solutions for specimens containing antibodies should be customized and fine-optimized according to specific buffer composition and pH.6,59
Freeze-dry (lyophilization) and air-dry
The aqueous state of antibody solutions often facilitates degradation processes and thus antibodies and many other proteins are often manufactured by a lyophilization process or stored in a dry state.74,111 Clinical specimens are not routinely lyophilized; however, when long-term freezing or cryopreservation by liquid nitrogen is not possible, lyophilization is a good alternative to preserve specimens and conjugate antibodies, which would be suitable for antibody testing and other assays. 28
Lyophilization removes the water components from a solution or specimen through freeze-drying in a vacuum atmosphere. The lyophilized specimen (powder) is stored in sealed vacuum ampoules or vials that can later be reconstituted by the addition of sterile water or an alternative solvent. 28 With similar purposes to lyophilization, the air-dry process uses hot air to dry the particles and transforms the antibody solutions into solids. 103 In both cases, the changes in temperature and pressures that occur during these processes affect the antibody structure and thus their biological activity. The aggregation often reported after reconstitution of antibody may be due to the increased protein-protein interaction in the absence of solvent, 86 or structure misfolding occurring during the freezing process.23,74
Stabilizing excipients (“stabilizers”) are pharmaceutical ingredients that are used in product formulations. 51 In antibody solutions, stabilizers usually consist of sugars and salts that are extremely important for antibody stability during lyophilization and air-drying, and for long-term storage and preservation.23,111 Excipients for antibody stabilization may include different bulking agents (substances maintaining crystalline structure during freezing, e.g., mannitol) or lyoprotectants (substances remaining amorphous for antibody stabilization, e.g., trehalose).60,73,117 In addition, the use of surfactants should also be considered during excipient formulations.60,73 Besides excipients, the stability of antibodies undergoing lyophilization can be affected by the speed of freezing, 111 the moisture of the formulation, 14 the formulation pH and selected buffer,94,142 and the final concentration of the antibody in the formulation.111,142
Sugars as stabilizers
Sugar-based stabilizers containing combinations of sucrose, trehalose, mannitol, or sorbitol have been demonstrated to act as antibody stabilizers during freezing of liquid formulations and various drying processes.23,73 This stabilization effect may occur because at low temperatures the “sugars” change into a glassy structure when the water in the solution turns into ice and the non-crystallizing components (i.e., antibodies) remain with restricted molecular mobility in this glassy matrix after drying. 29 Although the specific mechanism through which sugars preserve antibody structure during the freezing process remains unclear, some hypotheses suggest that antibodies are stabilized by connecting the proteins with hydrogen bonds. 19
The effect and importance of stabilizers on the stability of IgG after air-drying (100°C) had been demonstrated by comparing the recovered monomer and preserved protein secondary structure between IgG solutions with no stabilizers versus solutions based on the combination of different sugar stabilizers including trehalose, beta cyclodextrin, and hydroxypropyl beta cyclodextrin. 103 Furthermore, the biological function of a mAb using the same stabilizer formulation had a higher binding affinity in an ELISA when stabilizers were used. 103 For lyophilization, the use of trehalose or sucrose at appropriate concentration, or their combination with mannitol, provided higher antibody stability and lower aggregation rates after lyophilization and storage. 23
The appropriate composition of sugar stabilizers and their characteristics of crystallizing or non-crystallizing in the solution, the pH of the solution, and the concentrations of both buffers and antibody are fundamental variables that impact the extent to which sugars maintain the native state of the antibodies and the biological activity of the lyophilized product after reconstitution.19,23,29,94,103 The use of stabilizers must be a customized process in which antibody stability will depend not only on the stabilizer solution but also the correct crystallizing temperatures selected during lyophilization or air-drying.
Conclusions, limitations, and scope for future research
We have compiled information from various studies about different factors and conditions affecting the structural integrity and stability of antibodies, with particular emphasis on clinical samples used routinely for veterinary testing. The stability of antibodies in the testing laboratory setting reflects the intrinsic physicochemical processes that affect antibody structural integrity and the characteristics of the specimen. As for any other analyte, the stability of antibodies in a specimen is a function of property variation over time upon specific storage conditions. Stability studies have shown differences, or even inconsistent results, due to the lack of standardized experimental designs and heterogeneity of the chosen specifications. Previous guidelines for validation of sample collection did not include a specific evaluation of stability. Veterinary diagnostic laboratories should define stability limits, treatment, and recommendations for storage conditions, which could apply to different specimens (Table 3), and use them as a cause for rejection before processing the sample, in spite of no apparent visible deterioration.
General recommendations to preserve specimens containing antibodies.

The specific mechanisms are discussed within the article.
We must acknowledge certain limitations found during our bibliographic search. Most publications were based on humanized mAbs; veterinary clinical specimens across species contain polyclonal antibodies instead. Polyclonal antibodies are generally more stable than mAbs as they are less susceptible to a broader range of pH and ionic strength. 75 Structural differences in mammalian IgG subclasses can manifest themselves as variation in susceptibility to physicochemical and external factors that affect antibody stability and diagnostic functionality; however, there are exceptions (e.g., camelids having a type of IgG with heavy chain only 49 ). Mammals share a common core structure in their IgG, and thus general conclusions may be broadly applicable to laboratory testing; differences among antibody subclasses and isotypes should also be considered. 58 Importantly, most antibody degradation research is based on structural characteristics after physical or chemical processing or on the thermodynamics of the antibody structure. However, whether their binding affinity is severely compromised should be further investigated. Several publications have suggested that storage conditions may cause instability of the antibody structure yet did not lead to a complete loss of binding affinity.46,103
Noteworthy, many of the physical and/or chemical processes and testing considerations discussed here should not be treated as independent phenomena or mechanisms towards altering the composition, structure, and stability of antibodies. For example, freezing does not induce stress just by decreasing the temperature. The freezing process also alters the state of the solution, which includes a change in the pH, which in turn can be affected by antibody and buffer concentration, excipients, salts, and sugar-stabilizers. 73 Additional factors (e.g., exposure to light or mechanical stress [shaking, stirring]) may be hazardous for antibodies in different specimens; however, they are less likely to cause instability of antibody-containing specimens under typical laboratory scenarios.67,77,101
Most peer-reviewed research was focused on the stability of IgG in serum samples, which has historically been the most frequently used specimen in clinical laboratories. However, new specimen types are increasingly used in the veterinary testing setting (e.g., mammary secretions, fecal specimens, oral fluids). These specimens have different characteristics and may require additional manipulation outlines (e.g., the addition of a bacteriostat improved the stability of antibody in swine oral fluid over time). 101 The correct identification and understanding of the fundamental processes that affect IgG functionality and stability in different matrices results in the correct specimen manipulation and thus testing accuracy.
The stability of antibodies throughout the testing setting reflects the intrinsic physicochemical processes that affect the structural integrity of the antibody and the characteristics of the specimen. Our document can serve as a guide to veterinary diagnostic laboratories, helping them evaluate the characteristics of their sample collection, processing, and short-term and long-term storage processes towards maintaining antibody stability.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
