Abstract
Serum and urine samples from 30 cows (15 pregnant and 15 nonpregnant) from each of 10 Georgia dairy herds (total cows = 300) were examined by microscopic agglutination testing (MAT) and direct fluorescent antibody testing (FAT), respectively. Seven of the 10 herds had at least 1 cow with a positive FAT, and all of the herds had at least 1 cow with a reciprocal MAT titer ≥100 for 1 or more serovars. Serological testing was not helpful in identifying the infecting serovar for cows with a positive FAT result. The MAT titers for all 7 of the serovars evaluated were significantly correlated with one another, with 17 (81%) of the 21 Spearman rank correlation coefficients ≥0.4 in magnitude. Twenty (56%) of 36 FAT-positive cows did not have a titer that was highest for any particular serovar. Four of the 7 herds that reported using a Leptospira borgpetersenii serovar Hardjo-bovis vaccine had one or more FAT-positive cows compared with 3 out of 3 herds that reported they were not using this type of vaccine, although this difference was not statistically significant.
Introduction
Early embryonic death and infertility can lead to significant economic losses for cattle producers. Many infectious agents including Leptospira are associated with infertility, early embryonic death, and abortions. 1 The role of Leptospira borgpetersenii serovar Hardjo in infertility is a debated issue. According to review articles, overall herd prevalence of Leptospira infection in U.S. dairies and beef cow operations is estimated to be 35–50%. 1–3 Leptospira serovars Hardjo, Pomona, and Grippotyphosa are implicated in bovine abortions. 3,7 It has been recommended that the diagnosis of Leptospira infection in an individual cow can be made by combining serological testing with detection of leptospires in the urine. 12 In the current study, serological titers were examined and compared against 7 Leptospira serovars detected by microscopic agglutination test (MAT) and the presence of Leptospira in urine samples detected by direct fluorescent antibody testing (FAT) in pregnant and nonpregnant dairy cows.
Materials and methods
The study was performed in compliance with institutional guidelines for research on animals, and owner consent and a waiver of liability was obtained from each producer before testing. Ten dairy farms in Georgia were selected for the current study, and within each herd, a convenience sample of 15 pregnant and 15 nonpregnant lactating cows that were at least 200 days in lactation was selected. Urine from each cow was collected for direct FAT for Leptospira, and blood was collected for MAT.
Mid-stream urine (from the second or third voiding) was collected from each animal in a 50-ml tube after intramuscular administration of furosemide at a dose rate of 1 mg/kg. The urine samples were processed for direct FAT within 24 hr of collection using previously published protocols with modifications. 8,9,11 Briefly, 3 ml of urine was centrifuged at 10,000 × g, and the pellet was washed in 1.5 ml of sterile phosphate buffered saline (PBS) 2 times. Smears were prepared on circles of microscopic slides using 15 μl of the pellet. Slides were fixed in chilled acetone for 15 min and were treated with fluorescein isothiocyanate–conjugated anti-Leptospira polyclonal antibody a for 1.5 hr at 37°C. The slides were washed 3 times in PBS and air dried; then a mounting fluid and cover slip were applied. The slides were examined under the 40x objective of a fluorescent microscope. The samples with typical morphology of Leptospira and positive fluorescence were recorded as positive. Microscopic agglutination testing was performed on serum against 7 Leptospira serovars (Autumnalis, Bratislava, Canicola, Grippotyphosa, Hardjo, Icterohemorrhagiae, and Pomona) following Veterinary Diagnostic and Investigational Laboratory (VDIL; Tifton, GA) standard operating protocol for MAT testing. All samples were tested at a dilution of 1:50, and all positive samples were tested at 2-fold dilutions ranging from 1:100 to 1:6,400. Positive control serums were run with each testing for quality control purposes.
Summary of direct fluorescent antibody test (FAT) and microscopic agglutination test (MAT) results for Leptospira testing performed on 30 cows in each of 10 Georgia dairy herds.
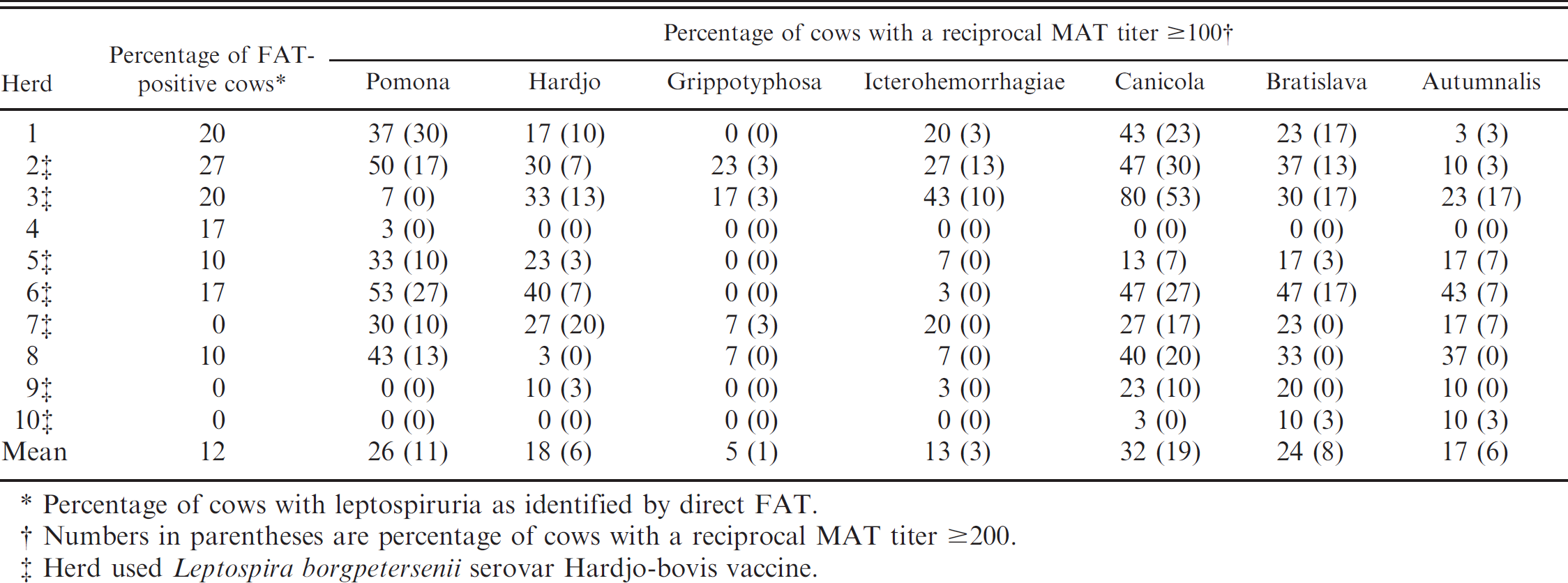
Percentage of cows with leptospiruria as identified by direct FAT.
Numbers in parentheses are percentage of cows with a reciprocal MAT titer ≥200.
Herd used Leptospira borgpetersenii serovar Hardjo-bovis vaccine.
Statistical analyses
Within herds that had 1 or more FAT-positive cows, the proportions of pregnant and nonpregnant cows with positive FAT results were compared using a Mantel–Haenszel chi-square test (stratified by herd), and the maximum log2 transformed MAT titers were compared between direct FAT-positive and -negative cows by using a linear mixed model with herd included as a random effect. The pairwise correlations of MAT titers between different serovars were evaluated using the Spearman rank correlation coefficient. Analyses were performed using commercially available software. b All testing was performed assuming a 2-sided alternative hypothesis, and P-values <0.05 were considered statistically significant.
Results
Seven of the 10 herds had at least 1 cow with leptospiruria, and all herds had at least 1 cow with a reciprocal MAT titer ≥100 for 1 or more serovars (Table 1). Four (57%) of 7 herds that reported using a L. borgpetersenii serovar Hardjo-bovis vaccine had 1 or more direct FAT-positive cows compared with 3 (100%) out of 3 herds that reported they were not using this type of vaccine, although this difference in proportions was not statistically significant (Fisher's exact test, P = 0.475).
Thirty-six cows from 7 herds had leptospires identified in their urine, and the distribution of maximum MAT titers for these cows is shown in Figure 1. Twenty-two (61%) of the direct FAT-positive cows had a maximum titer of ≤100, and only 1 cow had a maximum titer of 800. Notably, 9 (25%) direct FAT-positive cows did not demonstrate agglutination on the MAT test for any serovar (i.e., titers for all serovars in these cows were <50). Twenty (56%) of the 36 direct FAT-positive cows did not have a titer that was highest for any particular serovar. Of the remaining 16 cows, 11 had their highest titer to Leptospira interrogans serovar Canicola and 5 had their highest titer to L. interrogans serovar Pomona. No cows had their highest titer to L. interrogans serovar Hardjo or any of the other serovars (Autumnalis, Bratislava, Grippotyphosa, or Icterohemorrhagiae).
Microscopic agglutination testing titers for all 7 of the serovars evaluated were significantly correlated with one another (P < 0.001). Of the 21 pairwise comparisons, 17 (81%) of the Spearman rank correlation coefficients were ≥0.4 in magnitude, 11 (52%) were ≥0.5, and 6 (29%) were ≥0.6 (Table 2), indicating a high degree of correlation between serological titers of the Leptospira serovars tested.
Within herds that had 1 or more direct FAT-positive cows, there was no significant difference in the proportions of pregnant and nonpregnant cows with a positive direct FAT result (15% and 19%, respectively; Mantel–Haenszel chi-square, P = 0.467), and no significant difference in the geometric mean maximum MAT titers of direct FAT-positive and -negative cows (47.4 and 48.6, respectively; linear mixed model with herd as a random effect, P = 0.677).
Discussion
Although it has been suggested that examining the serological profile may help to identify the infecting serovar for leptospiruric cows, for 20 (56%) of the direct FAT-positive cows in the current study, there was no single serovar that yielded a uniquely high titer by MAT testing. There was also no significant difference in the maximum MAT titers of direct FAT-positive and -negative cattle. These results suggest that the usefulness of current methodology for serological testing for determining the infecting serovar in cattle should be revisited.
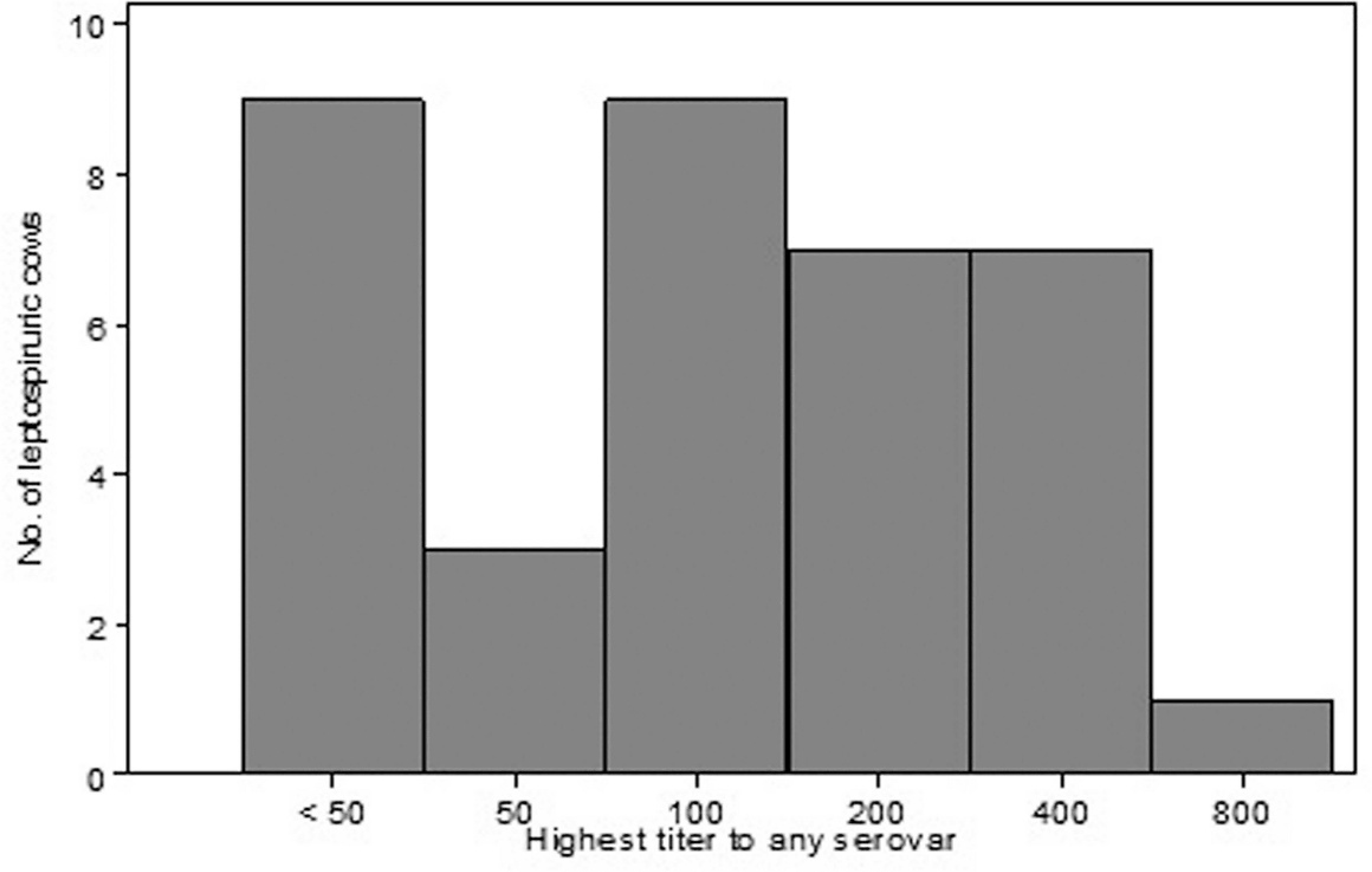
Distribution of maximum microscopic agglutination testing titers for 36 cows with leptospiruria as identified by fluorescent antibody testing. Titers were determined for 7 serovars: Autumnalis, Bratislava, Canicola, Grippotyphosa, Hardjo, Icterohemorrhagiae, and Pomona.
Leptospira borgpetersenii serovar Hardjo types A and B are presumed to be the predominant serovars circulating in the North American cattle population, and previous studies have shown that experimental infection with L. borgpetersenii serovar Hardjo results in a detectable serological response, 10–12 but none of the cows in the current study had a higher titer to Hardjo than to any other serovar. This finding may have been because cattle failed to mount a strong serological response against the host-adapted serovar, or because the serovar used for testing did not cross-react with the infecting strain. Leptospira interrogans serovar Hardjo-prajitno is not typically found outside the United Kingdom, but it is the genotype used for MAT testing in the United States. A previous comparative serological study 4 also found that it was more sensitive for the detection of Hardjo antibodies than were the L. borgpetersenii Hardjo genotypes.
Spearman rank correlations between microscopic agglutination testing titers of different Leptospira serovars in serum collected from 300 Georgia dairy cows. *
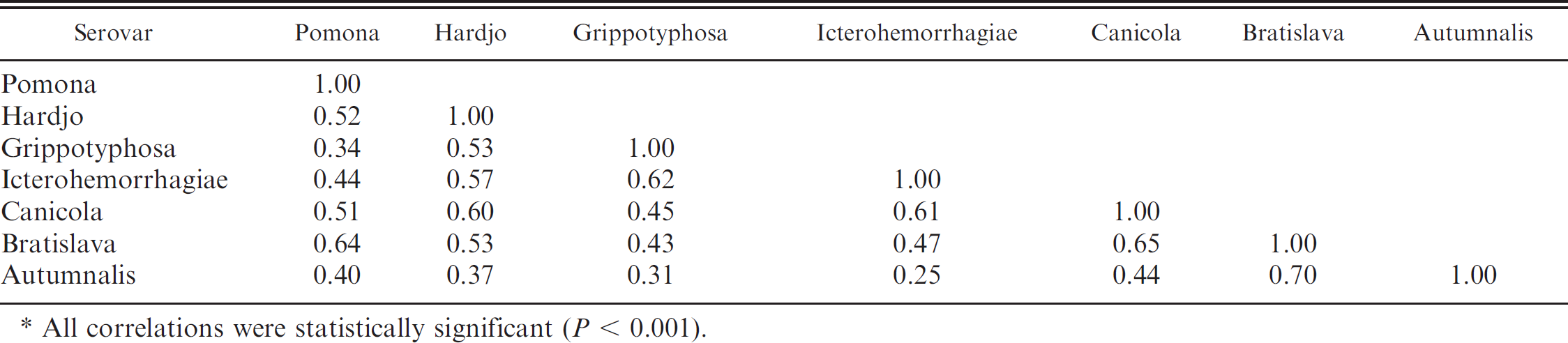
All correlations were statistically significant (P < 0.001).
The 3 farms that were FAT-negative for Leptospira reported that they administered intramuscular injections of oxytetracycline at the end of lactation in addition to vaccine containing L. borgpetersenii serovar Hardjo in an attempt to aid control of leptospirosis as recommended by the vaccine manufacturer. From the current study, it is not clear whether the antibiotic treatment, vaccination, or the combined effect of both resulted in a negative herd status for these 3 farms. The effect of vaccination alone on control of the disease could not be determined from the present study. In another study, 5 it was concluded that administration of monovalent leptospira vaccine in conjunction with systemic administration of oxytetracycline did not have any significant effect on reproductive performance of the cows. As Leptospira are prevalent and can cause economic losses to the cattle industry, 11 and considering the potential zoonotic impact 6 of these pathogens, further research is needed to isolate and characterize the Leptospira strains circulating in the cattle population and to develop better diagnostic methods for detection of infection.
Acknowledgements
The authors would like to thank the Milk Check-off established by Southeast Milk Inc., for funding this project, and the staff of the serology and bacteriology sections of the Veterinary Diagnostic and Investigational Laboratory, UGA/CVM, Tifton, GA, for technical support.
Footnotes
a.
National Veterinary Services Laboratory, Ames, IA.
b.
Stata 10.0, StataCorp LP, College Station, TX.
