Abstract
Here we describe a case of fatal amebic gastritis associated with Naegleria australiensis infection in an 11-mo-old Linnaeus’s two-toed sloth (Choloepus didactylus). The sloth had a history of weight loss and intermittent diarrhea for 18 d, and subsequently died despite empirical treatment. Postmortem findings included emaciation, gastric dilation with fluid content, and fibrinonecrotic gastritis with intralesional amebic trophozoites and cysts in the glandular region of the fundus. Transmission electron microscopy ruled out Amoebozoa of the family Entamoebidae based on the presence of mitochondria in the amoeboid organisms. PCR for pan–free-living amebae followed by next-generation sequencing of the PCR product revealed 99% identity with Naegleria australiensis. Gastric amebiasis has been reported sporadically in macropods and in leaf-eating monkeys with a sacculated stomach. To our knowledge, gastric amebiasis has not been reported previously in a sloth, which also has a sacculated and multi-chambered stomach.
In contrast to parasitic amebae, free-living amebae (FLA) are ubiquitous and live freely in the environment without requiring a host to complete their life cycle. Although most FLAs do not cause disease in humans and animals, a few FLAs are known to be pathogenic, and these include Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. 17 These pathogenic FLAs are poorly adapted to the host, therefore causing fatal infections following their acquisition from the environment. 17 The genus Naegleria contains 47 species, and the formation of a flagellate form is characteristic of this genus. 6 Naegleria spp. have 3 stages in their life cycle: a dormant cyst, an actively multiplying and feeding trophozoite, and a swimming flagellate stage. 14 Among the 47 Naegleria species, only N. fowleri causes natural disease in humans and animals. This disease, called primary amebic meningoencephalitis (PAM), is an acute necrotizing meningoencephalitis that has a 95% mortality rate. 6 The source of infection is usually contaminated warm water, and healthy young individuals are often affected.14,17 Encephalitis caused by N. fowleri has also been reported in cattle, sheep, and a tapir. 1 N. australiensis and N. italica can cause meningoencephalitis in experimentally infected mice;5,7 however, natural diseases caused by these species have not been reported. 6 We report here a case of gastric amebiasis in a Linnaeus’s two-toed sloth (syn. southern two-toed sloth; Choloepus didactylus) associated with N. australiensis infection.
An 11-mo-old, female Linnaeus’s two-toed sloth, imported from South America to a wildlife preserve in South Louisiana, USA, at 3 mo of age, developed intermittent diarrhea and marked weight loss (~25% of body weight) over 18 d. The sloth was housed in an exhibit that contained 4 additional sloths of the same species but in separate cages (Suppl. Table 1). No parasites, including, but not limited to, Giardia sp. trophozoites or cysts, were seen on repeated fecal flotation tests; fecal cultures for Salmonella sp. and Campylobacter sp. were negative. The sloth was treated empirically with fluids, simethicone, psyllium (Metamucil; Procter & Gamble), di-tri-octahedral smectite (Bio-Sponge: Platinum Performance), and rectal transfaunation containing diluted normal adult sloth feces. Despite treatment, the sloth died 18 d after the onset of clinical signs. The sloth was submitted for postmortem examination to the Louisiana Animal Disease Diagnostic Laboratory (LADDL), Louisiana State University (Baton Rouge, LA, USA).
On postmortem examination, the sloth was emaciated with mild hydrothorax and moderate ascites. The sacculated and multi-chambered stomach (Fig. 1) was markedly distended with a large amount of green opaque fluid mixed with fibrous plant ingesta. The mucosa of the glandular region at the fundus was multifocally reddened, roughened, and occasionally covered by fibrinonecrotic membranes (Figs. 1, 2). The mucosa of the prepyloric glandular region was diffusely red but smooth. The non-glandular regions of the stomach were unremarkable. The small intestine contained thick, gray, fluid digesta, and the colon contained soft, brown, non-formed feces. Samples of stomach, pancreas, lung, liver, kidney, thyroid and adrenal glands, urinary bladder, heart, skeletal muscle, large intestine, and brain were collected in 10% neutral-buffered formalin, processed routinely, sectioned at 4 μm, and stained with H&E.
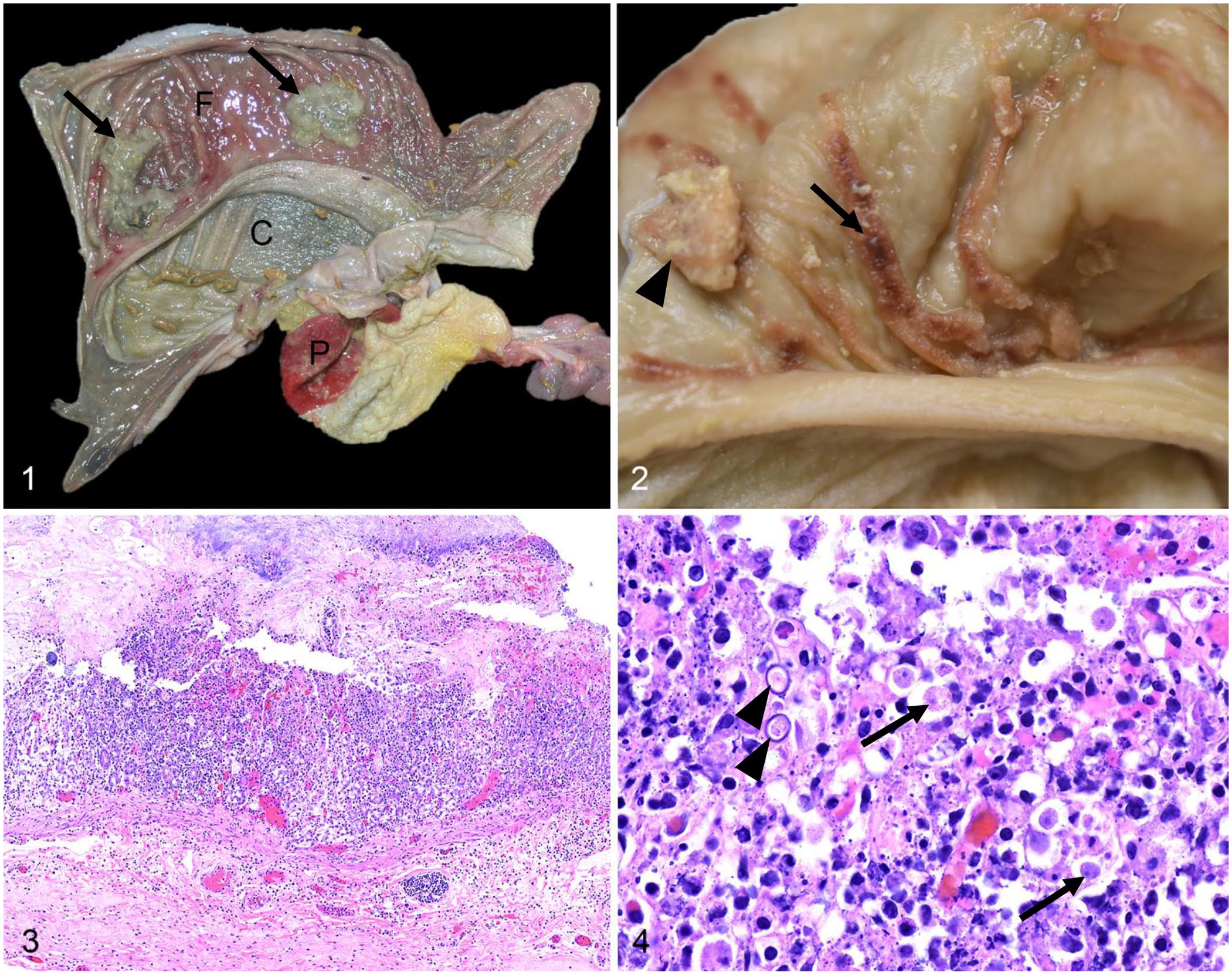
Amebic gastritis in a Linnaeus’s two-toed sloth.
On microscopic examination, the glandular region at the fundus had severe multifocal fibrinonecrotic gastritis. The mucosa was necrotic, hemorrhagic, and infiltrated by moderate numbers of plasma cells, lymphocytes, fewer neutrophils, and macrophages (Fig. 3). Large numbers of round, 8–10-μm trophozoites and fewer 12–14 × 8–9-μm oval cysts infiltrated the necrotic mucosa and occasionally the muscularis mucosae and the superficial submucosa (Fig. 4); a few trophozoites were in the cytoplasm of macrophages. The trophozoites had pale basophilic granular cytoplasm and a round, dark, basophilic, 1–2-μm nucleus. The cysts had a 1-μm thick, basophilic cyst wall and contained an internal structure similar to the trophozoites described above. The necrotic mucosa was covered by a fibrinonecrotic membrane mixed with numerous mixed bacteria, and a few trophozoites and cysts. The mucosa of the prepyloric glandular region was congested. The adjacent prepyloric non-glandular mucosa had occasional erosions and micropustules. Other histologic changes included lymphoid hyperplasia of the gastric lymph node, mild lymphoid depletion of Peyer patches and splenic white pulp, depletion of pancreatic zymogen granules, and serous atrophy of peripancreatic fat. No other significant lesions were noted in other tissues examined.
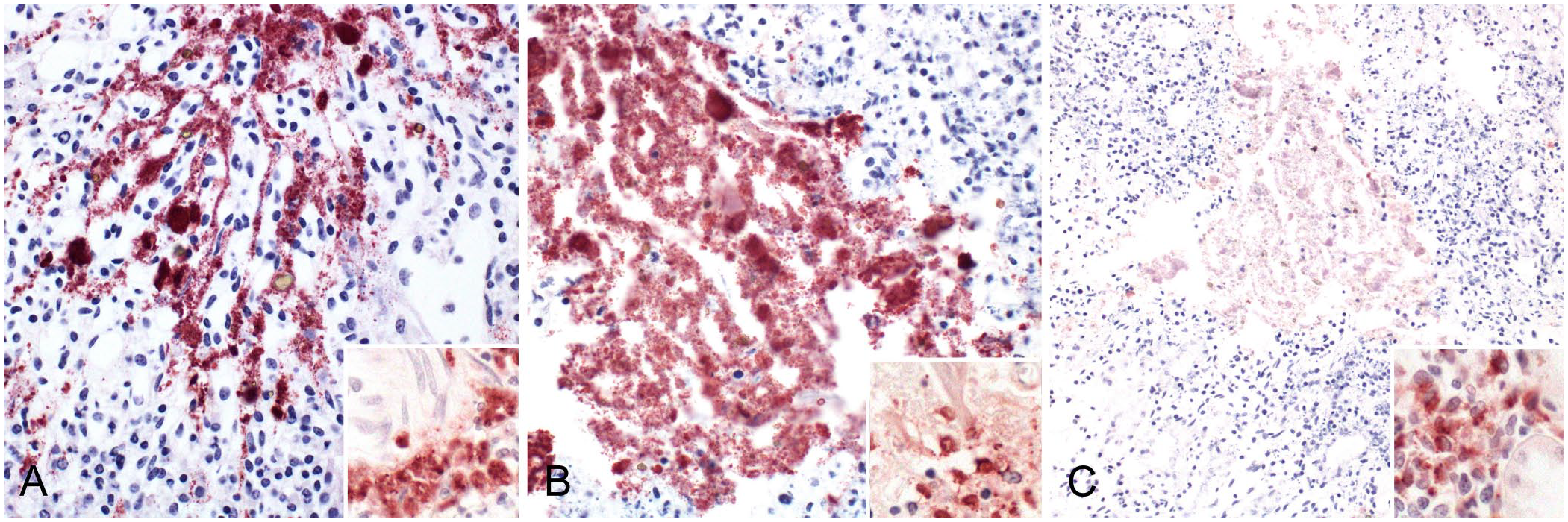
PCR for Entamoeba sp. was performed on formalin-fixed, paraffin-embedded (FFPE) stomach sections at the Zoological Medicine and Wildlife Disease Laboratory, University of Florida (Gainesville, FL, USA) and on the frozen stomach sample at the Free-Living and Intestinal Amebas (FLIA) Laboratory, Centers for Disease Control and Prevention (CDC; Atlanta, GA, USA); however, both were negative. Transmission electron microscopy (TEM) was performed on formalin-fixed stomach tissue. Formalin-fixed, 1–2 mm3 gastric specimens were post-fixed in Karnovsky solution and in 1% osmium tetroxide 0.1 M cacodylate buffer, processed and embedded in resin, sectioned, and stained with toluidine blue. TEM revealed intralesional round trophozoites and cystic forms of a eukaryotic ameboid microorganism, free and within macrophages. Trophozoites measured 9.93 ± 1.25 μm (x̄ ± SD) by 8.96 ± 1.4, containing a nucleus, endoplasm, ectoplasm, mitochondria with branching tubular cristae, endoplasmic reticulum, vacuoles, and autolysosomes. The cystic form had a wall of ~0.16–0.77 μm and organelle composition similar to trophozoites. These features were most consistent with a free-living Amoebozoa, and the presence of mitochondria ruled out Amoebozoa of the family Entamoebidae (Figs. 5–7), consistent with the PCR results that were negative for Entamoeba sp. Immunohistochemistry (IHC) for Acanthamoeba sp., Balamuthia sp., and Naegleria sp. was performed using pepsin-mediated antigen retrieval, and rabbit anti-Acanthamoeba, -Balamuthia, or -Naegleria polyclonal antibodies (provided by CDC). IHC revealed positive immunolabeling for Acanthamoeba sp. and Naegleria sp., and no immunolabeling for Balamuthia sp. (Fig. 8).
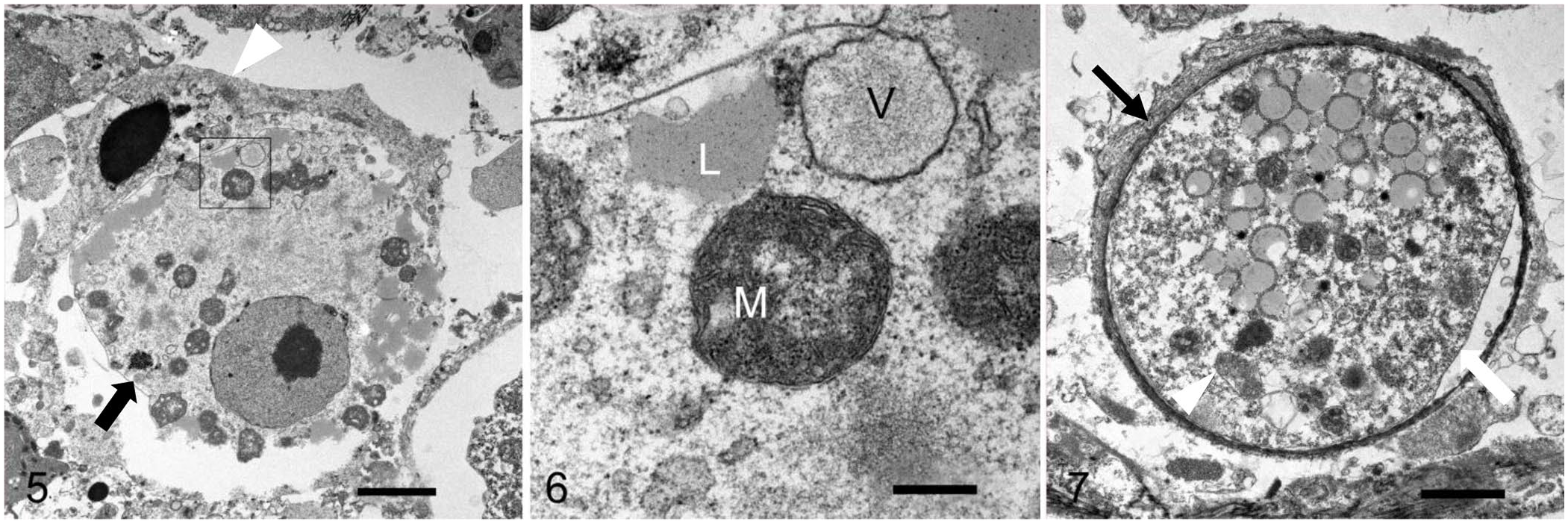
Amebic gastritis in a Linnaeus’s two-toed sloth. Transmission electron microscopy.
Amebic gastritis in a Linnaeus’s two-toed sloth.
A pan PCR for FLA was performed at LADDL on nucleic acids extracted from fresh-frozen gastric specimens using a previously reported primer pair targeting the 18S rRNA gene to detect pan-FLA (5′-CGCGGTAATTCCAGCTCCAATAGC-3′ [forward] and 5′-CAGGTTAAGGTCTCGTTCGTTAAC-3′ [reverse]); 22 PCR was conducted (OneStep Ahead RT-PCR kit; Qiagen) with the following cycling conditions: a reverse-transcription step (15 min at 45°C) followed by 95°C for 5 min, 40 cycles of 95°C for 15 s, 55°C for 15 s, and 68°C for 2 min, and a final extension step at 68°C. Genomic DNA for Acanthamoeba sp. (NR-46511), N. fowleri (NR-50144), B. mandrillaris (NR-50145), Entamoeba histolytica (NR-175), and E. invadens (NR-15226) were used as controls (BEI Resources). Pan-FLA PCR yielded a PCR product of 900–1,000 bp. Next-generation sequencing of the PCR product (GenBank SUB14214271 Seq6334_18S PP316205) and bioinformatic analysis revealed 99.5% identity to the Naegleria australiensis 18S rRNA gene. Therefore, the amebic organism associated with fatal gastritis in this sloth was identified as N. australiensis.
Gastric amebiasis has been reported sporadically in a few species that have a sacculated stomach with less acidic gastric pH. These included macropods (red kangaroos, and a dama wallaby) and leaf-eating monkeys (a silvered leaf monkey and black-and-white colobus monkeys).12,13,16,19 Compared to monogastric species, such as dogs and humans (in which the gastric pH is 1.5–2.0), the gastric pH in these monkeys and macropods that have a sacculated stomach is higher, in the range of 5.0–6.7 in leaf-eating monkeys and 4.6–8.0 in macropods. 19 It is speculated that this higher pH could facilitate amebic infection. Sloths also have a sacculated stomach with 3 large chambers that mix and ferment vegetation (Fig. 1). The pH of the sacculated portion of the stomach has been reported to be 5.2–6.7 in Hoffmann’s two-toed sloths (Choloepus hoffmanni) and ~6.0 in three-toed sloths (Bradypus tridactylus) compared to the prepyloric compartment in which the pH is 1.6–4.8 (Hoffmann’s two-toed sloths) and 2.0 (three-toed sloths).8,15 To our knowledge, the gastric pH of the Linnaeus’s two-toed sloth has not been reported; however, it is presumed to be higher (i.e., less acidic) in the cardiac and fundic regions as in other sloth species.
Pathogenic gastrointestinal amebiasis is usually caused by Entamoeba sp., a parasitic ameba that colonizes the intestinal tract of various animal species. E. histolytica causes colitis and hepatic abscesses in humans, and E. nuttalli causes a similar disease in non-human primates. 11 E. invadens can cause colitis in snakes, and E. ranarum can cause colitis with or without hepatic involvement in amphibians. 18
Pathogenic gastrointestinal amebiasis caused by free-living amebae is rare. To our knowledge, only 3 reports describe gastric amebiasis associated with FLA infection in: 1) a giant anteater (Myrmecophaga tridactyla) in which both Entamoeba spp. and Acanthamoeba spp. trophozoites were identified in ulcerative gastritis with perforation, 4 2) a woman in which Acanthamoeba sp. trophozoites and cysts were identified in a peptic ulcer that led to perforation, 21 and 3) a dog that had gastric ulcers with perforation and amebic trophozoites of Willaertia sp. 20 The prevalence of FLAs in the mammalian gastrointestinal tract has been poorly investigated. It is yet unclear whether FLAs are bystander organisms of the gastrointestinal tract or actually colonizers and, therefore, serve as primary or opportunistic pathogens of the gastrointestinal tract. 2 In the 3 aforementioned reports, the amebic infection was considered secondary or opportunistic but a significant contributing factor to the gastric lesions. In our case, no comorbidities or lesions that could have predisposed to the amebic infection were identified. Although it is difficult to determine if N. australiensis acted as a primary pathogen or an opportunistic infection secondary to multifactorial conditions such as stress and dysbiosis, we hypothesize that N. australiensis played a significant role in the development of the gastric lesions. We retrieved no cases of natural diseases associated with N. australiensis in a search of Google, PubMed, and Scopus, using search terms “N. australiensis” and “N. australiensis infection”, suggesting that this condition has not been reported in sloths or other species.
Sloths, as arboreal folivores, have a unique physiology adapted to their calorie-poor diet. They have a multi-chambered stomach and a slow digestive rate facilitating fermentation of feed and nutrient absorption. Their low body temperature and metabolic rate aid their survival on such a diet but limits their ability to thermoregulate resulting in fluctuating core body temperature; therefore, ambient temperature affects the fermentation rate of gut digesta and food intake. 3 Furthermore, sloths’ gut microbiota is influenced by the type of diet and differs from other herbivores including arboreal folivores. 9 These factors may influence gastric homeostasis predisposing to gastrointestinal diseases; however, further studies to correlate their gastrointestinal physiology and occurrence of gastrointestinal disease are needed.
The source of infection in our case remains undetermined. N. australiensis is widespread and has been detected in Europe, North America, Asia, and Oceania. 6 Naegleria spp. are typically detected in water sources such as irrigation canals, rivers, lakes, geothermal water sources, swimming pools, cooling water for industrial use, as well as in the soil. 14 Even though not tested, food, water, or soil in the exhibit where this animal was located could have served as potential sources of N. australiensis. Whether this disease affected other sloths in the exhibition is unclear. Although another 18-mo-old sloth in the same enclosure concurrently had similar clinical signs, that sloth recovered with empirical treatment and the cause was never identified. Due to the thermotolerance of N. fowleri and the seasonality of PAM outbreaks associated with this organism, there is a significant concern that the disease prevalence may increase with global warming. 14 N. australiensis is also thermotolerant and can grow at temperatures up to 42°C. 7 Whether this microorganism has the potential to become an emerging pathogen in connection to global warming remains to be determined.
Lastly, our case underscores the importance of a multimodal diagnostic approach in identifying poorly characterized microorganisms. Initially, we suspected the amebic organism to be Entamoeba sp. given that this genus is commonly associated with gastrointestinal amebiasis. However, TEM revealed the presence of mitochondria, which are not present in Entamoeba sp., leading us to identify the microorganism as a free-living ameba. Immunolabeling with anti-Acanthamoeba sp. polyclonal antibody to characterize the intralesional amebae yielded positive immunolabeling. However, PCR to amplify Acanthamoeba sp. using multiple sets of both reported 10 and in-house designed primers targeting the 18S rRNA gene on different regions of fresh-frozen gastric specimens and FFPE gastric specimens was unsuccessful (data not shown). Based on the polyclonal nature of the antibodies used, cross-reactivity with this species of Naegleria is possible. Additionally, the polyclonal anti-Naegleria antibody, likely raised against N. fowleri, immunolabeled N. australiensis. This is in contrast to previous reports stating that N. australiensis has different serologic and biochemical characteristics compared to N. fowleri, and antiserum raised against N. fowleri does not label N. australiensis.5,7 This discrepancy may be due to the polyclonal nature of the antibody. This highlights the value of electron microscopy and the importance of correlating immunohistochemical staining with molecular testing when identifying less-studied microorganisms.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241268315 – Supplemental material for Fatal gastric amebiasis in a Linnaeus’s two-toed sloth associated with Naegleria australiensis infection
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241268315 for Fatal gastric amebiasis in a Linnaeus’s two-toed sloth associated with Naegleria australiensis infection by Jeongha Lee, Meena Braden, Anibal Guillermo Armien Medianero, Francisco A. Uzal, Ganwu Li, Daniel B. Paulsen and Mariano Carossino in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the Histology & Immunohistochemistry and Microbiology sections at the Louisiana Animal Disease Diagnostic Laboratory, Christina Heard at CAHFS for the outstanding TEM preparations, Dr. Michael Garner (Northwest ZooPath, Monroe, WA) for early consultation, Dr. Ibne Karim M. Ali (FLIA Laboratory, Centers for Disease Control and Prevention) for consultation and for performing PCR for Entamoeba sp., and the Zoological Medicine and Wildlife Disease Laboratory, University of Florida for performing PCR for Entamoeba sp. The following reagents were obtained through BEI Resources, NIAID, NIH: genomic DNA from Acanthamoeba sp., strain CDC:12741:1, NR-46511; genomic DNA from Naegleria fowleri, strain CDC:V414, NR-50144; genomic DNA from Balamuthia mandrillaris, strain CDC:V188, NR-50145; Entamoeba histolytica, HK-9, NR-175; and Entamoeba invadens, strain IP-1, NR-15226.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our work was supported by funds provided by the Louisiana Animal Disease Diagnostic Laboratory, Louisiana State University School of Veterinary Medicine (PG009095) and start-up funds provided by LSU Vet Med to Dr. Mariano Carossino (PG009641).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
