Abstract
A 4-y-old female and 3-y-old male rhesus macaque (Macaca mulatta), both housed in the same facility, died unexpectedly within 2 wk. Postmortem examination revealed severe gastric dilation in both macaques and gastric emphysema in the female macaque. Histologically, bacteria consistent with Sarcina sp. were present in both macaques within the lungs and lumen of the trachea, esophagus, and gastrointestinal (GI) tract without associated inflammation. Additionally, in the female macaque, the bacteria were found in the gastric mucosa and associated with emphysematous spaces in the gastric wall without associated inflammation. PCR and Sanger sequencing of amplicons were subsequently performed on GI contents and non-alimentary tissues from the 2 affected monkeys and on comparative samples from unaffected rhesus monkeys in the same facility and an adjacent primate facility. The cases were compared using the 2-tailed Fisher exact test (p-value at 95% confidence). PCR identified Sarcina in GI contents of both affected and unaffected monkeys (p = 0.6084) and in non-alimentary tissues of affected monkeys only (p = 0.0083). These results suggest that the presence of Sarcina sp. in non-alimentary tissues is associated with gastric distension, gas accumulation, and unexpected death in nonhuman primates.
Acute gastric dilation (AGD) associated with Sarcina bacteria is an emerging concern. Sarcina ventriculi is associated with gastrointestinal (GI) clinical signs in both humans and veterinary species, including delayed gastric emptying, gastric dilation, vomiting, and emphysematous gastritis.2,8,11,24,25 Additionally, in western chimpanzees (syn. West African chimpanzee; Pan troglodytes verus), Candidatus Sarcina troglodytae has been linked to a fatal disease known as epizootic neurologic and gastroenteric syndrome, which results in both GI and neurologic clinical signs, including ataxia, neuromuscular weakness, seizures, and rapid progression to death. 19 Sarcina species are gram-positive, anaerobic cocci in the family Clostridiaceae that are common in the environment, particularly soil and cereal grains, and can survive and grow in acidic environments, such as the stomach, with pH values as low as 1.1,8 They ferment carbohydrates and produce carbon dioxide, resulting in bloat and emphysema.2,8 S. ventriculi also produces ethanol, which may cause acetaldehyde accumulation, resulting in mucosal injury.1,6,8
Despite an association with severe clinical signs and postmortem findings in some instances, the pathogenicity of Sarcina has remained poorly defined, because, in many instances, it is not associated with histopathologic changes.2,11,15,16 The bacterium has been reported as an incidental finding in the GI tract of asymptomatic humans and macaques, suggesting that it may be a benign commensal in some cases.1,5,12,23 To complicate matters, the prevalence of Sarcina is difficult to assess because the bacteria are fastidious and difficult to culture, often requiring techniques such as PCR to confirm their presence.2,8,19 Identification is typically made when the organisms with distinct morphology are found with light microscopy—the microorganisms are cuboidal, 1.8–3 μm in diameter, and arranged in tetrad packets given their replication in at least 2 planes of growth. 1
We investigated Sarcina sp. as a possible cause of gastric dilation in 2 rhesus macaques that died unexpectedly at a primate facility. Identification of the bacteria and presumptive diagnosis of clinical sarcinosis were made using histologic examination. Our goals were to identify and confirm Sarcina in the affected monkeys using PCR, investigate the prevalence of Sarcina sp. in affected animals and rhesus monkey populations on a university campus, and rule out recently provided feed as a source of infection.
Materials and methods
Clinical cases
Two rhesus macaques (Macaca mulatta) from the same facility on a university campus (facility A) were found dead without premonitory clinical signs (monkey 1, a 4-y-old female; monkey 2, a 3-y-old male) in 2 wk in December 2021. The 2 monkeys were housed in the same room that was maintained on a 12/12 light/dark cycle. The monkeys were fed a nutritionally complete commercial primate biscuit diet (5037; LabDiet) twice daily; water was provided ad libitum. Additionally, standard food enrichment was provided as follows: a fruit or vegetable was provided 3 times a week; a flavored ice treat with a variety of embedded food items including grains, beans, seeds, oats, fruit, and/or vegetables was provided once a week; a rotating foraging device filled with cereal and popcorn, molasses and grits, or mashed potato and grains, beans, seeds, oats, fruit, and/or vegetables was provided once a week.
Gross examination and histopathology
Routine postmortem examinations were performed on monkeys 1 and 2 within 24 h of death. Tissue samples were collected and fixed in 10% neutral-buffered formalin or collected fresh and stored at −20°C. Fresh-frozen alimentary tissues were stored with their contents. Formalin-fixed tissues were processed, paraffin-embedded, sectioned at 5 μm, and stained with H&E.
Bacterial culture of clinical cases
Gastric contents of monkey 1 underwent aerobic culture. Gastric contents of monkey 2 underwent aerobic and anaerobic culture.
Retrospective analysis
A retrospective search of rhesus monkey autopsies was performed on the database of primate facility A for 2001–2022 and an adjacent primate facility (facility B) for 1982–May 2023. The keywords used to search included the terms: Sarcina, Sarcina-like, gastritis, bloat.
Case selection for PCR testing
To confirm the identity of bacterial organisms that were histologically consistent with Sarcina sp. in monkeys 1 and 2, PCR was performed on frozen GI contents and tissue stored at −20°C.
To determine the prevalence of Sarcina in the rhesus monkey populations on campus, PCR was performed on frozen archived tissues from previously autopsied rhesus monkeys (Table 1) and rectal swabs from live healthy rhesus monkeys. Frozen archived samples included 30 tissues from 12 previous rhesus macaque postmortem examinations. The frozen archived samples were stored at −20°C and included 6 males and 6 females from facilities A and B. The monkeys were 3- to 6-y-old and were sampled between April 2013 and March 2022. Ten of the frozen archived tissue samples were from monkeys autopsied for reasons unrelated to GI disease. Two cases were from monkeys with gross evidence of GI disease from facility A. The GI cases included a 5-y-old female rhesus monkey (monkey 3) with colonic volvulus and colonic and cecal dilation, autopsied ~3 mo before the 2 clinical cases of AGD, and a 5-y-old male rhesus monkey (monkey 4) with historic typhlitis characterized by fibrous adhesions between the cecum and body wall. Rectal swabs from live healthy rhesus monkeys were obtained from 26 individuals in facility A between February 24, 2022, and February 25, 2022 (10 males and 16 females). After collection, the swabs were placed in dry 1.5-mL tubes, immediately frozen, and stored at −20°C.
Characterization of autopsied rhesus monkey cases and controls to assess the presence of Sarcina in tissues with PCR testing.
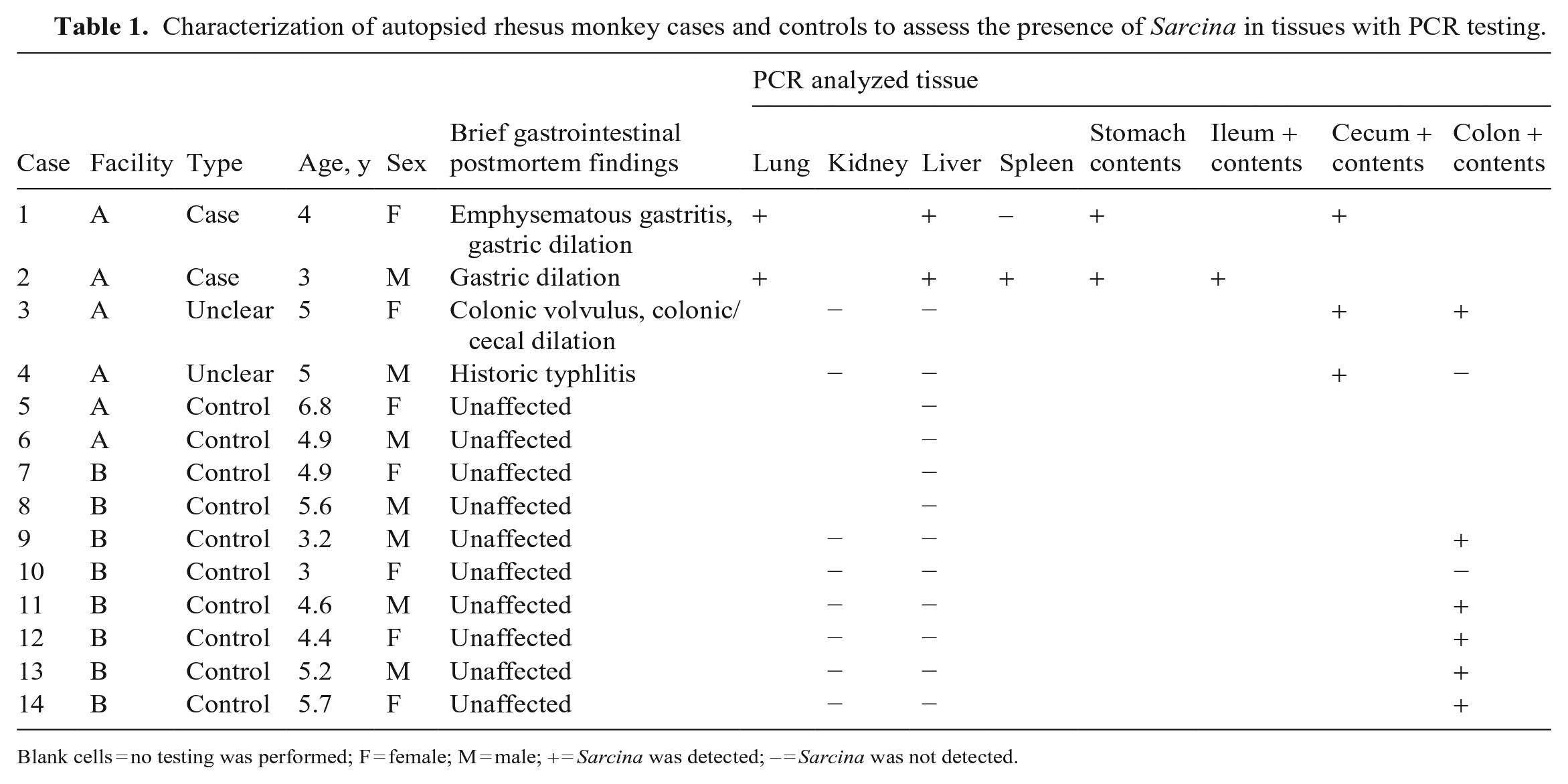
Blank cells = no testing was performed; F = female; M = male; + = Sarcina was detected; – = Sarcina was not detected.
To assess feed contamination as a possible source of Sarcina infection, PCR was performed on two 11.4-kg (25-lb) bags of LabDiet from the same shipment as the feed provided to monkeys 1 and 2 the day before death. The bags were stored at ambient temperature after opening, and 12 samples from throughout each bag were removed for analysis.
Sarcina-specific PCR assay and sequencing
DNA was extracted from frozen non-alimentary tissue samples using a commercial DNA extraction kit intended for tissues (DNeasy blood and tissue kit; Qiagen) and from frozen GI contents, rectal swabs, and monkey chow using a commercial DNA extraction kit intended for samples with high levels of PCR inhibitors (DNeasy PowerLyzer PowerSoil kit; Qiagen) according to the manufacturer’s instructions. Isolated DNA was used as template for Sarcina PCR as described previously. 19 Briefly, 25-µL reactions containing 12.5 µL of PCR master mix (HotStar 2× PCR master mix; Qiagen), 0.25 µL each of 10 µM forward and reverse primers (F: 5′-TGAAAGGCATCTTTTAACAATCAAAG-3′; R: 5′-TACCGTCATTATCGTCCCTAAA-3′), 10 µL of molecular-grade water, and 2 µL of template DNA were cycled under the following conditions: 95°C for 15 min (once), 94°C for 30 s, 48°C for 30 s, 72°C for 30 s (29 cycles), 72°C for 10 min (once), and 4°C hold. PCR products (289 bp expected length) were electrophoresed on 1.5% agarose gel with ethidium bromide and DNA length standards, visualized under UV light, and photographed. Amplicons were then excised and purified using a commercial kit (Zymoclean gel DNA recovery kit; Zymo Research) and Sanger sequenced at the University of Wisconsin–Madison Biotechnology Center (Madison, WI, USA).
Statistical analysis
Odds ratios and 95% CIs were calculated to assess the strength of association between the presence of Sarcina in samples and clinical signs and outcomes, with Fisher exact tests used to calculate associated p-values.
Results
Gross and histopathologic findings of clinical cases
Gross examination of monkeys 1 and 2 showed severe gastric dilation, pulmonary congestion and atelectasis, pleural effusion, intraluminal digesta within the trachea suggestive of aspiration, focally extensive tan hepatic discoloration suggestive of blanching, and disseminated petechiae and ecchymoses. Additionally, monkey 1 had gastric emphysema (Fig. 1). Monkey 1 had a slightly thin body condition, and monkey 2 had a moderate body condition.
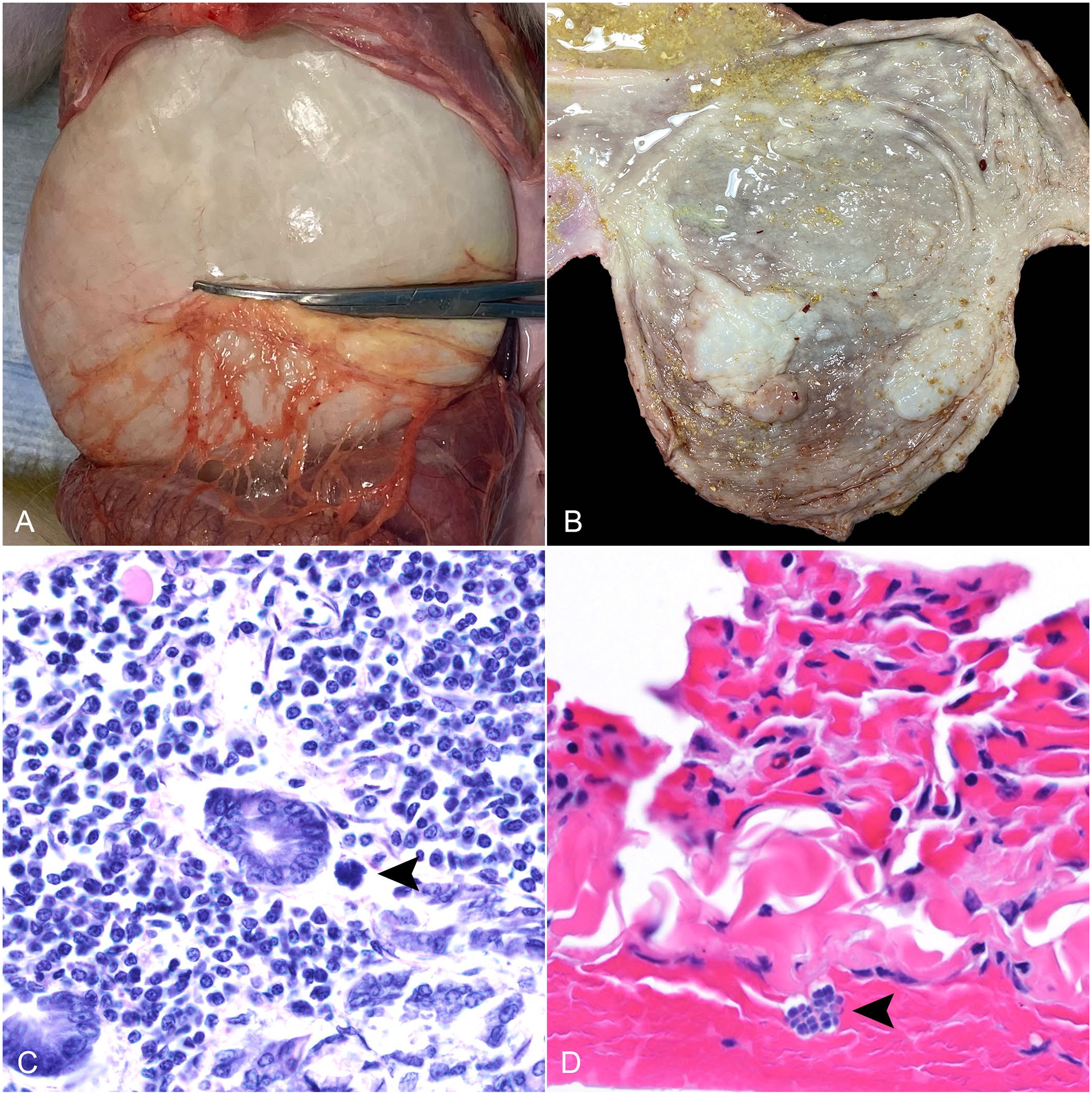
Gross and histologic findings associated with Sarcina sp. in a rhesus monkey.
Histologically, there was mild-to-moderate autolysis throughout most of the GI tract, with the exception of multifocal marked autolysis in the duodenum, jejunum, and ileocecocolic junction of monkey 1 and cecum of monkey 2. Both monkeys had tetrad packets of cuboidal 1.8–3-μm bacteria within the lumen of the GI tract, esophagus, trachea, and airways. Monkey 1 also had gas-filled emphysematous pockets in the gastric submucosa and less commonly in the gastric mucosa and muscularis with small numbers of bacteria around and rarely within emphysematous pockets, as well as free within the gastric lamina propria (Fig. 1). Additionally, in monkey 1, bacteria were present on the pleural surface of the lungs and surface of the tongue.
Bacterial isolation
Aerobic culture of gastric contents of monkey 1 yielded heavy growth of mixed bacterial microbiota including alpha-hemolytic streptococci and commensal gram-negative rods. Aerobic culture of gastric contents of monkey 2 yielded heavy growth of mixed microbiota including alpha-hemolytic streptococci and coliforms. Anaerobic culture of gastric contents of monkey 2 yielded heavy growth of mixed microbiota including Ligilactobacillus murinus and probable normal microbiota. No Clostridium spp. were isolated.
Retrospective analysis
Between 2001 and 2022, 84 rhesus monkey autopsies were recovered from the database at facility A. Of these autopsies, 3 of 84 rhesus monkeys had histologic evidence of Sarcina. Two of the 3 cases are monkeys 1 and 2 of our study. The third case occurred in a reportedly healthy 1-y-old male rhesus monkey with gastric dilation; Sarcina-like bacteria were found within the lumen of the trachea. An additional case of gastric dilation of unknown etiology was found in an acutely dead 3-y-old female rhesus macaque.
Of the 8,656 rhesus monkey autopsies recovered from the database at facility B between 1982–May 2023, 1 rhesus monkey had histologic evidence of Sarcina. The case occurred in a 17-y-old female simian retrovirus–positive (SRV+) rhesus monkey with AGD and perforation. Tetramers and octamers of Sarcina-like organisms were evident histologically in the tissues at the rupture site only. PCR testing could not be performed on tissues from the suspected sarcinosis cases.
PCR assay and sequencing
All GI content samples from 4 of 4 cases were positive for Sarcina DNA, as were 5 of 6 non-alimentary tissues from these same animals (Tables 1, 2). By contrast, no (0 of 12) non-alimentary tissue samples from previously autopsied controls were positive, although 5 of 6 GI content samples from these same controls were positive for Sarcina DNA (Tables 1, 2). Most rectal swabs from healthy live individuals were positive for Sarcina DNA (24 of 26, 92%; Table 2). No monkey diet samples were positive for Sarcina DNA (0 of 24; Table 2).
Results of PCR testing for Sarcina sp. in 4 cases, 38 control rhesus monkeys, and feed.

Statistical analysis
PCR positivity for Sarcina in non-alimentary tissue samples was significantly associated with case status (odds ratio = 91.7; 95% CI: 3.2–2,620; Fisher exact p = 0.0083, 2-tailed). However, PCR positivity for Sarcina in GI content samples was not significantly associated with case status (odds ratio = 2.5; 95% CI: 0.1–76.1; Fisher exact p = 0.608, 2-tailed).
Discussion
AGD is a sporadic, often fatal condition in captive and semi-captive nonhuman primates.3,4,7,9,10,12 –14,17 –22 A definitive etiology and pathogenesis of AGD has yet to be determined; however, a number of predisposing factors are thought to contribute to its development in nonhuman primates, including intragastric fermentation associated with Clostridium perfringens, recent anesthesia, overfeeding, altered feeding schedules, excessive water consumption, and stress.3,4,7,9,10,12 –14,17,20 –22 There has been an increase in reports of Sarcina-associated AGD and emphysematous gastritis (EG) in the human and veterinary literature, suggesting that Sarcina-associated AGD and EG is an emerging concern.2,8,11,19,23 –25
Our 2 cases of AGD, one of which also had gastric emphysema, had abundant bacteria morphologically consistent with Sarcina sp. in the absence of microbiologic evidence of C. perfringens. Although there was no evidence of associated acute inflammation, the presence of Sarcina in the airways was indicative of aspiration and suggests, at minimum, that a dysbiosis of Sarcina contributed to clinical disease. In our retrospective search of rhesus monkey autopsies at facilities A and B, we found organisms with morphology similar to Sarcina in 4 of 8,740 autopsies. These autopsies included monkeys 1 and 2, and 2 additional monkeys with AGD. This finding demonstrates that organisms with Sarcina-like morphology are not found routinely in autopsy cases at these facilities, and is supportive of clinical sarcinosis in monkeys 1 and 2.
Our study also provides insight into the prevalence and pathogenicity of Sarcina spp. in rhesus monkeys. Sarcina spp. were present in the GI contents of all monkeys, including those clinically affected and unaffected, and in rectal swabs from healthy monkeys from 2 different facilities. However, Sarcina spp. were present in non-alimentary tissues only in affected monkeys, indicated by both morphology and PCR. Notably, this pattern is identical to the report of a similar clinical syndrome in chimpanzees. 19 This finding suggests that Sarcina may be a normal inhabitant of the alimentary tract and that systemic bacterial dissemination may contribute to disease progression in individuals that normally carry Sarcina. Alternatively, Sarcina may be a complex of species or strains that are morphologically similar but cause different lesions, as has been suggested in the case of chimpanzees. 19 We note that the PCR and sequencing approach that we used does not distinguish among known Sarcina species and, to our knowledge, no such assay exists.
There are several limitations to our investigation. For example, we performed anaerobic culture of gastric contents only on monkey 2. Despite the absence of anaerobic culture on monkey 1, based on the presence of Sarcina in tissue sections and relative absence of incidental Sarcina from histologic study of other monkeys in the colony, we believe that the lesions are best attributed to Sarcina.
Additionally, in our cases, no inciting cause of bacterial overgrowth and dissemination was identified. In our study, the affected monkeys were fed twice a day. After the second case of fatal AGD was identified, the facility staff fed the remaining monkeys 3×/d out of caution, beginning on December 23, 2021. The monkeys in the affected room were fed 3×/d for ~6 mo and returned to 2×/day feeding on June 9, 2022. No additional cases of AGD or sarcinosis were identified. The efficacy of the change in feeding schedule was difficult to assess, and the high prevalence of asymptomatic Sarcina in the monkey colony made other interventions difficult. Monkey chow samples from the facility where monkeys 1 and 2 were housed were PCR negative, suggesting that the diet was not the source of the ubiquitous GI colonization in these monkeys. Although we did not identify a definitive source of colonization in our study, future studies aimed at better understanding sources of GI colonization in captive nonhuman primates may include comparative sampling of maternal and neonatal intestinal flora to determine whether maternal intestinal flora may serve as an early source of colonization. 27
PCR could not be performed on tissues from the 2 additional cases of suspected sarcinosis that we found, nor on the additional case of AGD of unknown etiology that was recovered from the databases at facilities A and B. We, therefore, recommend that future studies should evaluate the presence or absence of Sarcina in tissues using PCR in cases of AGD. Additionally, our database search was limited to rhesus macaques. Future retrospective studies with large nonhuman primate databases should be expanded to include additional nonhuman primate species, such as cynomolgus monkeys (Macaca fascicularis).
Death from AGD is thought to result from circulatory failure resulting from increased abdominal pressure compressing the caudal vena cava; pulmonary restriction caused by increased abdominal pressure limiting diaphragmatic movement; and/or severe cardiac failure as a result of vagal reflex. 26 Monkeys 1 and 2 had evidence of compression of both the caudal vena cava and diaphragm, suggesting that circulatory failure and/or pulmonary restriction may have contributed to death.
Intragastric colonization with subsequent fermentation, gas production, and dissemination to non-alimentary tissues by Sarcina sp. was the presumptive cause of fatal AGD in monkeys 1 and 2. Sarcina should be considered as a differential etiology for nonhuman primates with AGD and gastric emphysema. The distinct morphology of this organism allows presumptive identification when seen histologically. Because Sarcina spp. are quite difficult to culture, PCR and subsequent amplicon sequencing is the preferred method for confirmation of a presumptive morphologic diagnosis.2,8,19
Footnotes
Acknowledgements
We thank Dr. Andres Mejia from the University of Wisconsin–Research Animal Resources and Compliance, and Ivy Herkert and Faye Hartman from the University of Wisconsin–Madison Veterinary Medical Teaching Hospital Clinical Pathology Laboratory, for their investigative assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our investigation was supported by departmental funds for the Research Animal Resources and Compliance unit and Harlow Primate Laboratory, and U.S. Fish and Wildlife Service, Great Ape Conservation Fund–Africa, award F22AP01521-00.
