Abstract
A free-ranging, adult female two-toed sloth (Choloepus hoffmanni) was brought to a wildlife rescue center in Costa Rica with ocular and auricular myiasis and numerous skin lesions. After one month of unsuccessful systemic and topical antimicrobial treatment, the patient died. A postmortem examination was performed, and tissues were examined histologically, confirming disseminated amebic infection with intralesional trophozoites and cysts in the lungs, liver, eye, heart, spleen, and stomach. Immunohistochemistry identified the ameba as Acanthamoeba sp. A multiplex real-time PCR assay, 18S ribosomal DNA PCR, and sequencing performed on formalin-fixed, paraffin-embedded lung tissue confirmed the Acanthamoeba T17 genotype. The Acanthamoeba genus is in the group of free-living amebas that cause infection in humans and animals, and it is ubiquitous in the environment. Acanthamoeba T17 has been isolated from water and soil, but to our knowledge, this genotype has not been implicated in infections of animals previously and has not been reported from Costa Rica. Systemic Acanthamoeba infection has not been described in sloths previously. We provide a comprehensive literature review describing infections by free-living amebas of the genus Acanthamoeba spp., Balamuthia spp., and Naegleria spp. in domestic, zoo, and wild mammals.
Keywords
A free-ranging, adult female, two-toed sloth (Choloepus hoffmanni) was brought to a wildlife rescue center in Costa Rica with ocular and auricular myiasis and numerous skin lesions presumed to be caused by electric burns after climbing electric wires. The animal was weak and incoordinated. The skin lesions were distributed over the face, nose area, and tongue. After admission, the sloth was dewormed, and systemic and topical antimicrobial treatment was applied. Despite the partial healing of the skin lesions, weakness and incoordination persisted, and the patient died one month after admission to the rescue center.
At postmortem examination, the cranial lobe of the left lung had a 3.5-cm diameter, raised, pale-tan nodular area (Fig. 1A). On cut surface, the nodule had pliable, soft, light-red to pink areas alternating with firm, pale-tan to white areas (Fig. 1B). The right lung had numerous 0.2–0.5-cm subpleural blebs. The remaining organs were grossly unremarkable.
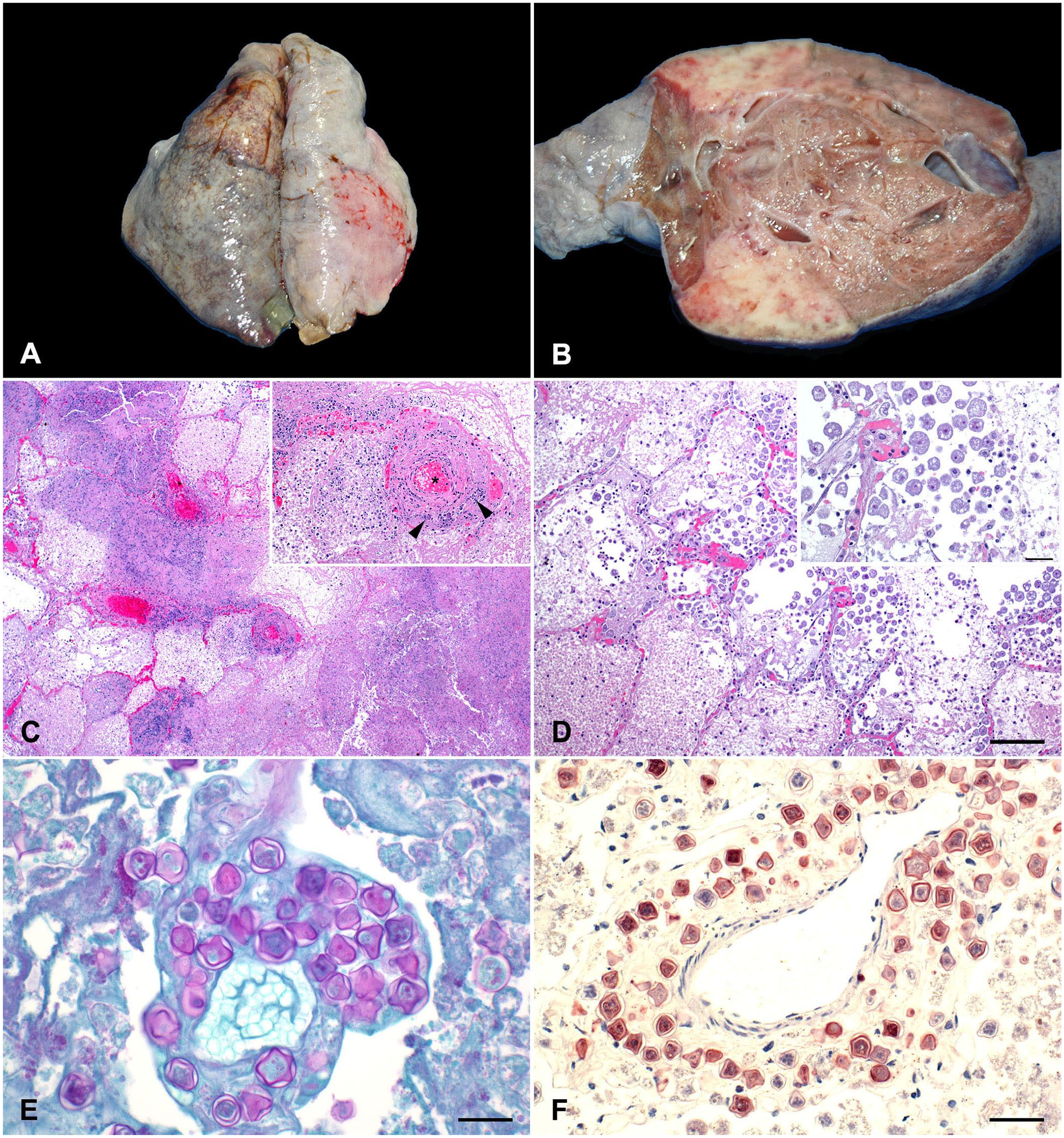
Systemic infection caused by Acanthamoeba genotype T17 in a free-ranging, adult two-toed sloth.
Samples of lungs, liver, heart, spleen, eye, tongue, and intestine were collected and fixed by immersion in 10% neutral-buffered formalin, pH 7.2, and processed routinely to produce 4-µm thick, H&E-stained slides. Selected sections were stained with periodic acid–Schiff (PAS) stain.
Histologically, alveoli were obscured and effaced by fibrin, and eosinophilic and karyorrhectic necrotic debris, intermixed with non-degenerate and degenerate neutrophils in extensive, multifocal-to-coalescing areas of coagulative necrosis of the pulmonary parenchyma (Fig. 1C). Pulmonary vessels within the areas of necrosis contained fibrin thrombi and had hyalinized vascular walls, heavily infiltrated by fragmented granulocyte nuclei (Fig. 1C). Myriad extracellular, round, 20–35-µm, amebic trophozoites with abundant amphophilic, lacy-to-vacuolated cytoplasm, and a single, round, centric-to-eccentric karyosome containing a single nucleolus congregated at the boundary of the necrotic and viable tissue, and were surrounded by neutrophilic infiltrate (Fig. 1D). Occasionally, PAS-positive, double-walled encysted trophozoites with undulating membranes were observed surrounded by degenerate neutrophils, histiocytes, lymphocytes, and plasma cells (Fig. 1E). Extracellular trophozoites surrounded by areas of necrosis were also observed in liver, eye, heart, spleen, and stomach.
Selected sections of the lung were processed by immunohistochemistry (IHC) for Naegleri fowleri, Acanthamoeba sp., and Balamuthia mandrillaris using ameba-specific polyclonal antisera and pepsin for antigen retrieval, as described previously.16,19 Encysted amebic trophozoites were positively stained with Acanthamoeba IHC stain (Fig. 1F). IHC for N. fowleri and B. mandrillaris was negative.
Formalin-fixed, paraffin-embedded sections of the lung were processed for molecular testing. A multiplex real-time PCR (rtPCR) assay for simultaneous detection of 3 common pathogenic free-living amebas (Acanthamoeba spp., B. mandrillaris, N. fowleri) previously described in the literature 55 was only positive for Acanthamoeba sp. The available nucleic acids subsequently underwent subgenic 18S ribosomal DNA PCR and sequencing for genus and genotype identification using the JDP1-2 primers described previously. 62 The 503-bp PCR product was purified from the gel, sequenced, and identified using BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The sequence matched with 98.0% and 97.8% identity to 2 Acanthamoeba T17 genotypes (GenBank GU808279.1, GU808278.1, respectively) isolated from freshwater ponds in Thailand. 49 Our ancillary molecular testing combined with gross and histologic findings confirmed systemic infection caused by Acanthamoeba genotype T17 (deposited in GenBank as PP400729).
A comprehensive search of Google, PubMed, CABI Direct, Web of Science, and Scopus, using search terms “Acanthamoeba T17”, “systemic amoebiasis”, “free-living amoeba”, “two-toed sloth”, “wildlife”, “animal” and “Choloepus hoffmanni”, did not yield any reports of systemic amebiasis caused by Acanthamoeba T17 in domestic or wild animals. Similar criteria using the search terms “free-living amoeba”, “systemic amoebiasis”, “infection”, “domestic animal”, “wild animal”, and “wildlife” was used to create review Tables 1–5.
Selected cases of naturally occurred free-living amebic infection in dogs.

CAV = canine adenovirus; CDV = canine distemper virus; CM = castrated male; F = female; FLA = free-living ameba; IHC = immunohistochemistry; IFA = indirect immunofluorescence assay; KCS = keratoconjunctivitis sicca; M = male; NS = not specified; SF = spayed female.
Diagnosis was made based on the histologic morphology of the amebas.
PCR was performed on paraffin-embedded samples from the cornea and was unsuccessful due to insufficient nucleic acids, degradation of DNA after fixation time, and quality of the fixative or inadequate primers.
Selected cases of naturally occurring free-living amebic infection in cats.

CM = castrated male; DSH = domestic shorthair; FLA = free-living ameba; NS = not specified.
Selected cases of naturally occurring free-living amebic infection in ruminants.

CM = castrated male; F = female; FLA = free-living amoeba; IHC = immunohistochemistry; IFA = indirect immunofluorescence assay; M = male; NS = not specified.
Selected cases of naturally occurred free-living amebic infection in horses.

CM = castrated male; F = female; FLA = free-living ameba; IHC = immunohistochemistry; IFA = indirect immunofluorescence assay; M = male; NS = not specified.
Selected cases of naturally occurring free-living amebic infection in zoo and wild animals.

CM = castrated male; F = female; FLA = free-living ameba; IHC = immunohistochemistry; IFA = indirect immunofluorescence assay; M = male; SIV = simian immunodeficiency virus.
Acanthamoeba T17 genotype was first isolated from environmental water samples in Thailand. 49 Additionally, T17 isolates were retrieved from soil and environmental water in Brazil and recreational hot springs in Malaysia.43,45,53 However, the sequences of the Malaysia isolate were later reanalyzed and reclassified as T18. 14 T17 belongs to group 1 of Acanthamoeba; species and genotypes in group 1 are mostly environmental and considered less pathogenic than those in groups 2 and 3. 14 Environmental genotypes, including T7, T9, T17, and T18, are thermotolerant and capable of inducing cytopathic effects in cell cultures at 40–42°C, 14 which suggests that they are capable of causing disease in vertebrates. Experimental studies using the T17 environmental genotypes isolated from Brazil and Thailand revealed that the agent grew at 37°C and 42°C (Brazilian isolates); however, no growth was reported in the Thailand isolates at the same temperatures.42,49 An experimental study demonstrated that the tolerance of genotypes T4, T5, T11, and T12 to 39°C is not correlated with pathogenicity. 34 However, this does not have a direct relevance in our case given that the body temperature range recorded in free-ranging two-toed sloths in Costa Rica is 33.2–35.7°C. 50 The lack of molecular testing in many clinical cases is a possible cause of the lack of published reports of disease caused by this genotype. 14
It is unclear whether the sloth in our case became infected before or after the clinical intervention at the wildlife rescue center. The definitive source of infection remains obscure. However, we speculate that the animal acquired the organism from environmental sources (water or soil) before admission to the center and the stress induced by a traumatic event (possible electrocution) and veterinary intervention exacerbated the disease. Acanthamoeba of the T17 genotype has not been reported previously in Costa Rica; only a few reports about Acanthamoeba T4 and T5 isolated from environmental sources, such as water, shower, and dental equipment units, have been published.12,58–60 Acanthamoeba genotype T17 infection should be considered as a differential diagnosis in cases of amebiasis in animals.
Free-living amebas: biology and detection
Free-living amebas (FLAs) are protozoan species that may infect humans and other animals.63,67 Most FLAs are opportunistic pathogens, with the most commonly incriminated species belonging to the genera Acanthamoeba, Balamuthia, Naegleria, and Sappinia.63,67 These microorganisms can be found in the environment, including dust, soil, air, and water from swimming pools, irrigation canals, hot springs, sea, and sewers. 67 Acanthamoeba is the most common FLA found in the environment, vegetables, and stool samples from several animal species.63,67
Methods used to detect FLAs in animal tissues include histology, IHC, immunofluorescence, molecular methods, such as multiplex rtPCR, and culture. 55 Subtle differences between the most frequently identified infective genera of FLAs can be noticed in histology. For example, Balamuthia trophozoites are more pleomorphic, have several nucleoli, and their cysts have 3 layers. In contrast, Acanthamoeba trophozoites are less pleomorphic, and contain one nucleolus; cysts are 2-walled.48,67 Naegleria cysts are not found in tissues.48,67 A fourth free-living amoeba, Sappinia sp., has binucleate cysts with trophozoites found in tissues.24,67
In humans, Acanthamoeba is recognized as the causative agent of Acanthamoeba keratitis and Acanthamoeba granulomatous encephalitis. 64 Systemic amebic infection has a high mortality rate in humans due to the lack of effective specific antiamebic compounds. However, the use of several drugs including ketoconazole, fluconazole, sulfadiazine, pentamidine isethionate, amphotericin B, azithromycin, itraconazole, or rifampicin has been reported to be successful in a limited number of cases. 64
Acanthamoeba has a cosmopolitan distribution comprising numerous habitats in soil and water from tropical to Arctic regions of the world. 63 Acanthamoeba is thermotolerant, capable of surviving at temperatures above 37°C and in a wide range of salinity, osmolarity, and pH conditions.63,67 This is probably why Acanthamoeba organisms have been found in tissue cultures, body fluids of mammals, and distilled water.63,67 The life cycle of this agent comprises 2 stages. The first stage is the invasive and parasitic trophozoite (8–40 µm) with a single nucleus and fine adherent acanthopodia, allowing cellular movements and predation of other microorganisms. 67 In adverse conditions that jeopardize nutrient acquisition, a second stage is present, the resilient uninucleate, double-walled dormant cyst (8–28 µm) containing an endo- and ectocyst. 67
The >24 species of Acanthamoeba described are classified morphologically into 3 groups. 39 Group 1 has large cysts (>18 µm) with stellate endocysts and smooth ectocysts; group 2 has smaller cysts (<18 µm) with ovoid-to-stellate endocysts and wavy endocysts; and group 3 has 19-µm cysts with ovoid endocysts and smooth-or-wavy endocysts. 63 Most isolated pathogenic species belong to group 2. 63 However, by using PCR and sequencing of the 18S rRNA gene, Acanthamoeba has been classified into 23 genotypes (T1–23).39,54,62 The most prevalent genotype is T4. 22
The diagnosis of Acanthamoeba infection can be achieved by demonstration of the amebas within the infected tissue by histologic examination and IHC. Determination of the species and/or genotype, however, requires molecular testing. 55
Other species of FLAs implicated in human and animal disease are N. fowleri and B. mandrillaris. N. fowleri is the etiologic agent of primary amebic meningoencephalitis (PAM) and is found in soil and water.63,67 The only Naegleria species that has been isolated from human cases is N. fowleri.63,67 The life cycle includes the amebic and cystic forms.63,67 B. mandrillaris is another FLA implicated in cases of Balamuthia amebic encephalitis (BAE) and, like other FLAs, is present in soil and water.63,67 Its life cycle includes large trophozoites (larger than Acanthamoeba and Naegleria, >60 µm) and 3-layered cysts.63,67 When first isolated, Balamuthia was described as a leptomyxid ameba due to its similar morphologic features; however, further genotyping analysis revealed that it was phylogenetically different from the leptomyxids. 63 Sappinia diploidea was identified only once, in a single non-fatal encephalitis case in a man, with only trophozoite forms. 24 This ameba has been isolated from environmental sources and feces from humans, elk, bison, and cattle.63,67 Willaertia magna is a thermophilic FLA similar to Naegleria, with 4 flagella and no reported infections in human medicine; it is non-pathogenic in mouse experimental models and has only been reported in ulcerative gastritis in a dog.63,65 W. magna has been isolated from bovine feces and thermal waters. 63
FLA infections in animals
Naturally occurring infections caused by FLAs in domestic, zoo, and wild mammals have been extensively described in the veterinary literature since 1956 4 (Tables 1–5). In companion animals, most of the cases have been reported in dogs, with a characteristic multisystemic distribution of infection and an almost invariably fatal outcome (Table 1).3–5,17,18,20,66,68 Less frequently, fatal localized infections have been described in the CNS causing meningoencephalitis in dogs.9,13,32,51,57 Examples of non-lethal localized infections reported in canine patients include cutaneous and prostatic infections.11,41 Two cases of amebic keratoconjunctivitis were described in dogs, with similar clinical and pathologic features to those described in humans. Although these 2 cases had a favorable outcome after treatment, PCR identification of the organisms was unsuccessful, and the diagnosis was based only on histologic features and therefore remained presumptive. 6 Occasionally, Acanthamoeba is found in the cornea of dogs, causing keratitis, or subclinically.35,69 FLA infections reported in dogs include those caused by B. mandrillaris, Acanthamoeba castellanii, A. culbertsoni, A. healyi T12, and Acanthamoeba genotypes T1, T3, T4, T5, and T16.5,11,13,18,32,36,51,66,68
In cats, systemic infection by FLAs is uncommon (Table 2), with a single report of Acanthamoeba T4 infection causing chronic sclerokeratitis in a cat. 40 Studies of corneal scrapes or swabs from stray cats have demonstrated that these animals can harbor Acanthamoeba genotypes T2 and T4 subclinically.1,33,46
N. fowleri is the most common FLA causing infection in ruminants; most of the cases were associated with encephalitis in bovids (Table 3).8,16,21,30,31,47,52,70 Encephalitis by Acanthamoeba sp. was described in a cow. 26 In horses, Acanthamoeba sp. has caused disseminated infection (Table 4)27,37,38; localized infections were produced within the placenta by A. hatchetti and in the lungs by Acanthamoeba sp.7,56 In pigs, naturally occurring infection by FLAs has not been reported commonly, but Yucatan minipigs have been used as animal models for research on Acanthamoeba keratitis in humans.2,29
Several case reports have described clinical and fatal infections by FLA in zoo and wild animals in captivity; most of them were caused by B. mandrillaris (Table 5). This microorganism is a well-recognized cause of meningoencephalitis, with or without systemic dissemination, in gorillas and other non-human primates.10,15,25,28,44,61 Disseminated infection in a Siberian tiger was caused by B. mandrillaris. 48 N. fowleri causes sporadic infection in animals belonging to the order Perissodactyla, with case reports of meningoencephalitis and meningoencephalomyelitis in a tapir and a south-central black rhinoceros, respectively.42,72 Meningoencephalitis caused by Acanthamoeba sp. and Acanthamoeba genotype T4 was described in non-human primates.23,71
FLA infections occur sporadically in domestic, zoo, and wild animals with a reported fatal outcome. In companion animals, Acanthamoeba sp. infections are more commonly reported to cause multisystemic infections; in ruminants, N. fowleri is an agent with tropism for the CNS. Zoo and wild animal cases are scant, with an overrepresentation of non-human primates affected by B. mandrillaris. Environmental genotypes of Acanthamoeba can cause disseminated infections in people.
Footnotes
Acknowledgements
We thank the Histology Laboratory staff of the California Animal Health and Food Safety Laboratory System, University of California–Davis, San Bernardino Branch for technical assistance and slide preparation for the immunohistochemistry procedures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
We did not receive any specific grant from public, commercial, or not-for-profit funding agencies.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the CDC.
