Abstract
A 4–5-month-old intact male Indian flying fox (Pteropus giganteus) was presented to the Baton Rouge Zoo’s veterinary hospital with an acute onset of obtundation that was diagnosed with amebic encephalitis. Histologic examination revealed numerous amebic trophozoites within necrotic foci, affecting the occipital cerebrum and surrounding the mesencephalic aqueduct. The etiologic agent, Balamuthia mandrillaris, was determined by multiplex quantitative real-time polymerase chain reaction, immunohistochemistry, and indirect fluorescent antibody test. The current report documented a case of amebic encephalitis within the order Chiroptera.
A 4–5-month-old intact male captive-bred Indian flying fox (Pteropus giganteus) housed at the Baton Rouge Zoo was presented to the zoo’s veterinary hospital after being found on the ground of its enclosure. The bat was comatose at the time of presentation, was cold to the touch, and covered in its own urine and feces. Nonaffected cage-mates included 20 adults and 1 other captive-bred juvenile Indian flying fox. The bat enclosure measured 6 m × 6 m with a natural dirt floor and an aluminum roof. The walls were composed of cinder blocks extending from ground level to 1 m in height, with wire mesh (covered with tarps in the winter to maintain warmth) extending from this level to the ceiling. The ceiling contained a timed intermittent sprinkling system to keep the bats cool during warm weather months. The exhibit was not open to the public for walkthrough, but zookeepers entered the enclosure twice daily for routine animal husbandry. Food and water bowls were changed daily, located throughout the exhibit at a height no less than 1 m from the ground. No other animals within the enclosure displayed similar clinical signs. Despite supportive therapy, the bat died 7 hr after presentation and was submitted for autopsy to the Louisiana Animal Disease Diagnostic Laboratory (Baton Rouge, Louisiana).
An abbreviated autopsy was performed, and a transverse section of the mid-cerebellum and brainstem was submitted for rabies direct fluorescent antibody testing according to routine standards set forth by the Centers for Disease Control and Prevention (CDC; Atlanta, Georgia), with the remaining brain placed in 10% formalin. The autopsy was completed 24 hr later, following a negative test result for rabies. No significant gross findings were observed. However, transverse sections of fixed brain revealed multiple foci of malacia and reddening, including a 2-mm focus affecting the gray matter of the right occipital lobe adjacent to the longitudinal fissure (Fig. 1), and more prominent malacia affecting ~25% of the midbrain radiating from the mesencephalic aqueduct (Fig. 2). Histologically, these areas consisted of regionalized collapse and rarefaction of neuropil consistent with liquefactive necrosis, with numerous intralesional protozoal parasites. Organisms varied in size, measured 18–35 μm in diameter, had abundant granular eosinophilic cytoplasm with distinct small, dense, basophilic central to eccentrically placed nuclei with prominent karyosomes, and were consistent with ameba trophozoites (Fig. 3). A periodic acid–Schiff (PAS) stain highlighted the cell membrane and granular cytoplasm of the ameba (Fig. 4). Large numbers of neutrophils, hemorrhage, and occasional fibrinoid vascular necrosis were observed within areas of necrosis. A moderate number of lymphocytes, plasma cells, and lesser numbers of eosinophils and macrophages surrounded blood vessels and expanded the leptomeninges, primarily adjacent to areas affected by liquefactive necrosis. Additional tissues examined histologically included cervical and lumbar spinal cord, nasal passages, cribriform plate, eyes, lungs, heart, liver, spleen, tongue, stomach, small and large intestines, kidney, adrenal glands, testicles, skin from the patagia, thymus, and bone marrow. No amebas were observed in these organs supporting localized central nervous system (CNS) involvement. Other histologic changes included moderate lymphoid hyperplasia of splenic white pulp with low numbers of tingible-body macrophages, mild hydropic degeneration of centrilobular hepatocytes, incidental pneumoconiosis in the lung, and mild multifocal neutrophilic inflammation of the liver. Examination of the patagium identified overgrowth of large numbers of yeast-like organisms resembling Malassezia spp. in the surface keratin without evidence of scaling or inflammation. No histological evidence supportive of underlying immunosuppression was observed.
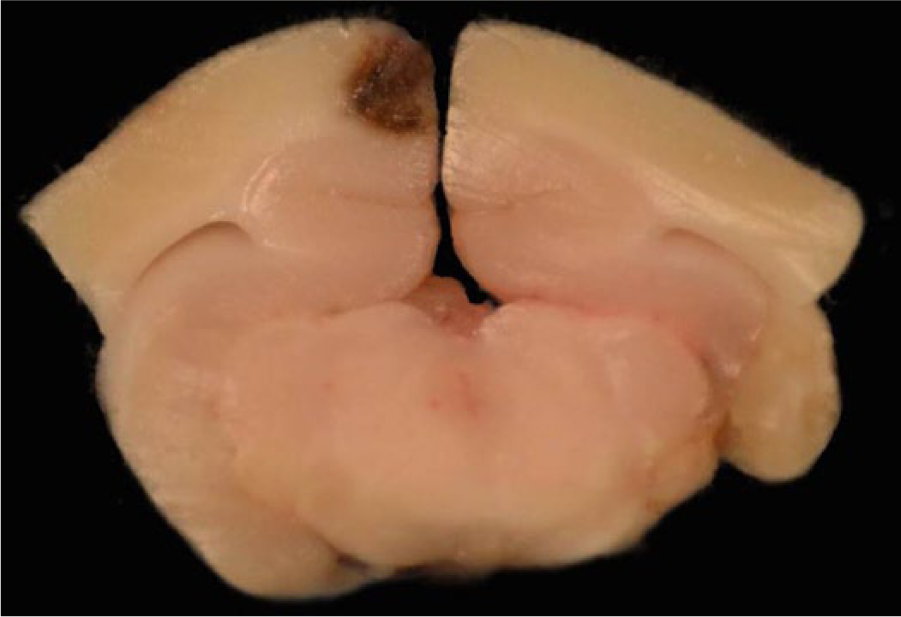
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. The right cerebral occipital lobe contains a focal area of malacia and reddening within the superficial gray matter along the longitudinal fissure.
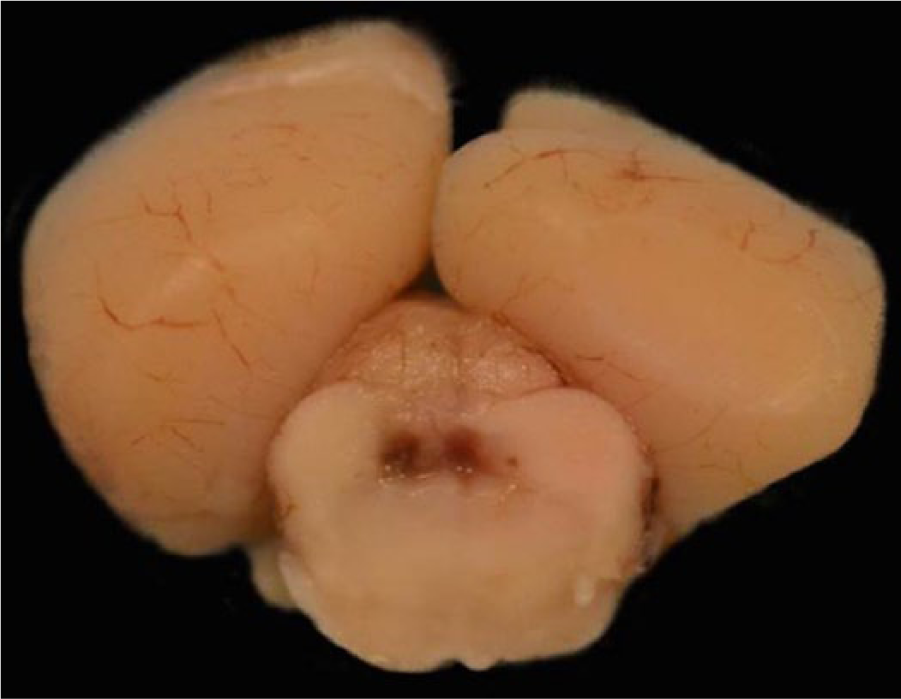
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. Similar changes surround the mesencephalic aqueduct within the midbrain, extending locally into adjacent neuropil.
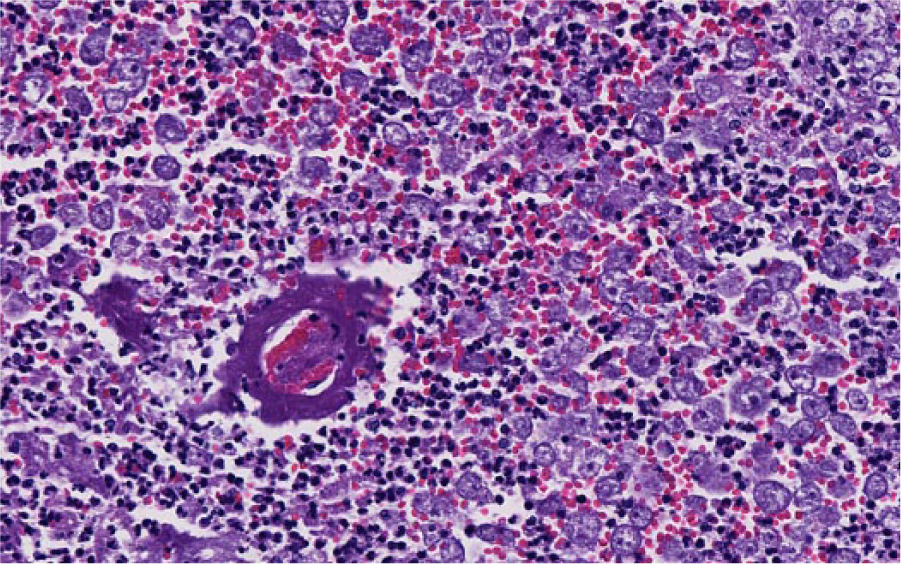
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. Representative of periventricular malacia observed in Figure 2 characterized by liquefactive necrosis with numerous variably sized ameba trophozoites, hemorrhage, neutrophils, and a prominent hyalinized vessel (fibrinoid necrosis). Hematoxylin and eosin.
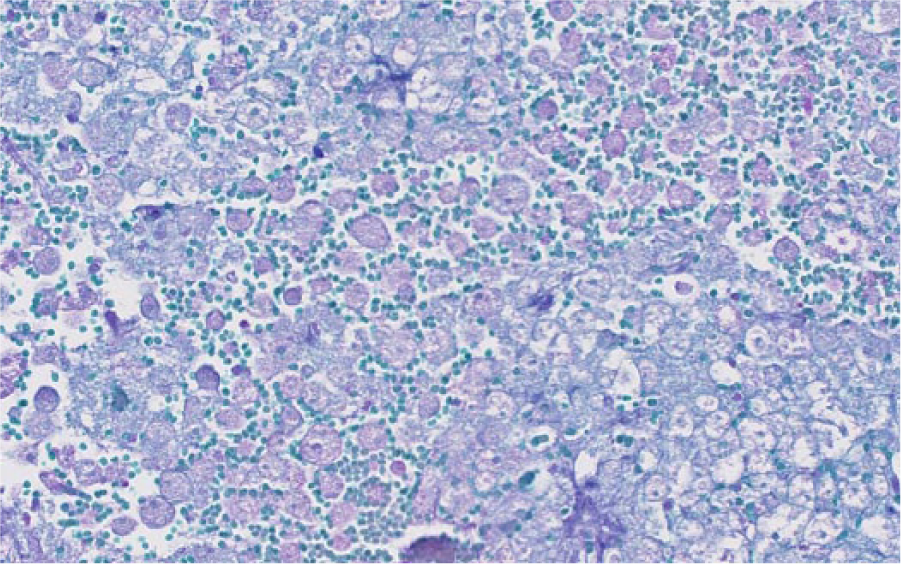
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. Glycoproteins within protozoal cell membranes and cytoplasm highlight purple with periodic acid–Schiff stain.
Frozen brain submitted to the CDC Ameba lab for multiplex quantitative real-time polymerase chain reaction (qPCR) was strongly positive with Balamuthia mandrillaris–specific primers, with a quantification cycle value of 25.88. 18 Pooled heart, liver, lung, kidney, and spleen were negative for the presence of B. mandrillaris DNA. Additionally, fixed brain submitted to the CDC Ameba lab for an indirect fluorescent antibody test (IFAT) specific for B. mandrillaris, was strongly positive (Fig. 5), while negative on nasal passages (decalcified), lung, liver, and kidneys. Fixed brain was submitted to the University of California–Davis Veterinary Medical Teaching Hospital Histology Laboratory (Davis, California) for Balamuthia spp.–specific immunohistochemistry (IHC), and was strongly positive (Fig. 6). Four environmental samples representative of the bat enclosure substrate were assessed by the CDC using their multiplex qPCR techniques; all samples were negative. Testing of water and food sources within the enclosure was deemed unnecessary as these were changed out on a daily basis. Appropriate positive and negative controls were used in all diagnostic tests performed in this case.
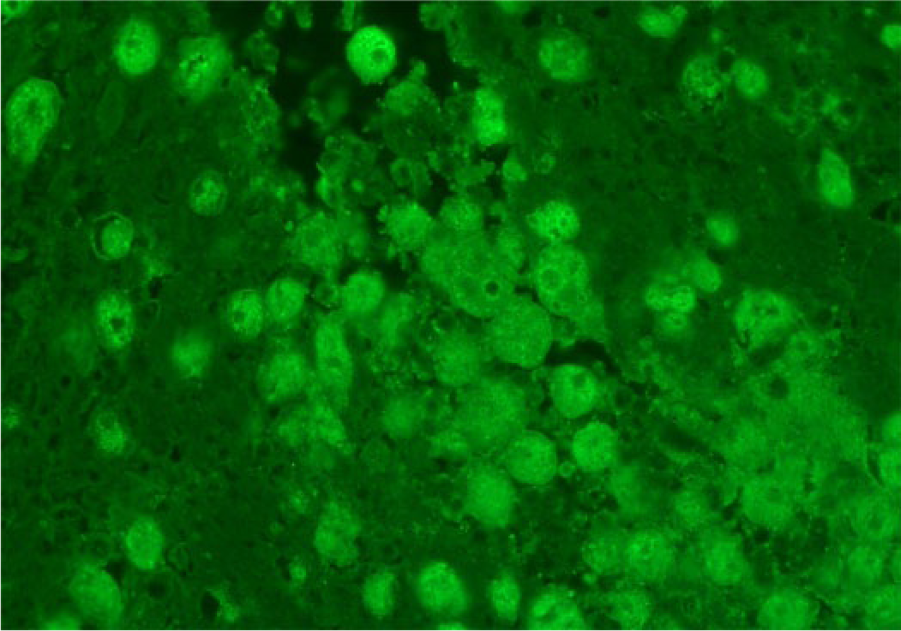
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. Indirect fluorescent antibody test highlights antigen specific for Balamuthia mandrillaris, with strong bright green fluorescence (polyclonal rabbit anti-Balamuthia serum R-146).
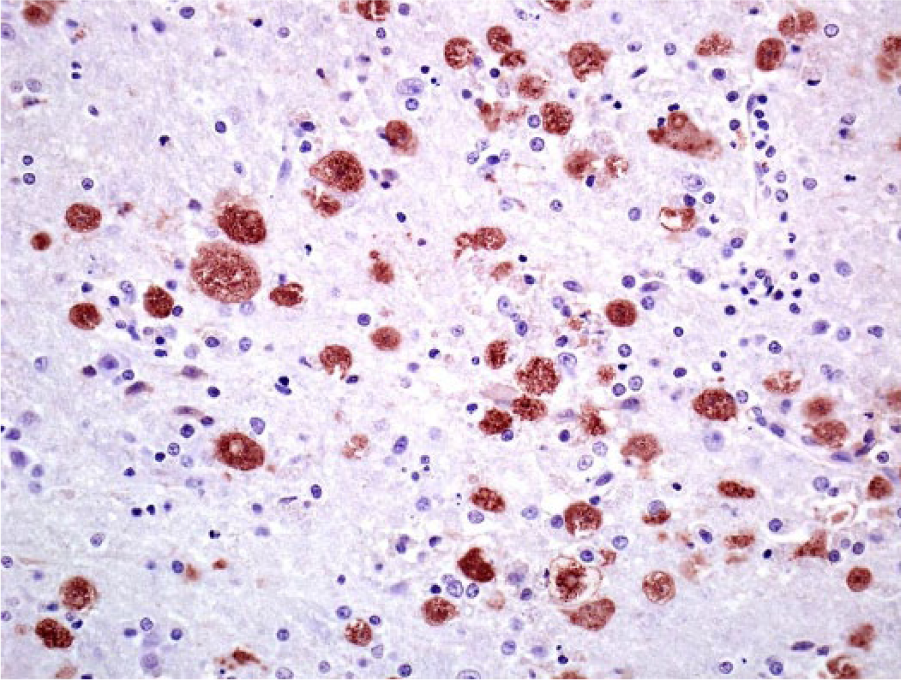
Indian flying fox (Pteropus giganteus); right occipital cerebrum and midbrain. Immunohistochemistry highlights antigen specific for Balamuthia spp. with strong red chromogenic labeling (polyclonal rabbit anti-Balamuthia serum).
To date, 5 genera of free-living ameba are known to cause infection of the CNS in humans including multiple Acanthamoeba spp., B. mandrillaris, Naegleria fowleri, Paravahlkampfia francinae n. sp. CDCV595, and Sappinia diploidea; all but S. diploidea have been reported in veterinary medicine as causes of disease.23,24 Free-living pathogenic ameba possess a minimum of 2 life stages, including a trophozoite stage, which feed on a combination of other protozoa, bacteria, and environmental debris, and a dormant cyst stage that reverts to a trophozoite stage on favorable environmental conditions. Naegleria fowleri contains an additional flagellate stage, and cysts are absent within affected tissue sections. 24 In addition to encephalitis, Acanthamoeba spp. and B. mandrillaris may also manifest as a multiorgan disease, affecting the skin and lungs. 12 Acanthamoeba spp. can cause keratitis, most frequently in people who use contact lenses, specifically if this practice includes use of contaminated water sources. 12
Water-borne transmission is common in human cases of amebiasis, with risk factors including exposure to contaminated water sources for recreation and drinking, although soil exposure from agricultural work, gardening, desert motorcycling, mountain biking, or playing in soil have also been other reported sources. 11 Interestingly, the flying fox enclosure that housed the bat of this report used a sprinkler system to keep the bats cool on hot days, which had been in use the weeks prior to case presentation. This practice was discontinued on diagnosis of amebic encephalitis, as the sprinkler system usage was considered a potential risk factor, inhibiting our ability to test water samples as a source of B. mandrillaris. As all other tested sources within the enclosure were negative for free-living ameba, and the Indian flying foxes interact minimally with the enclosure’s ground substrate, contaminated water from the sprinkler system was considered the most likely, though still speculative, source of infection in this case.
While some environmental and host-determined risk factors are shared among species of free-living ameba, the clinical course of disease is highly variable across species. Amebic encephalitis from N. fowleri typically manifests with an acute onset as early as 24 hr postexposure and affects young children and adults exposed to contaminated water sources referred to as primary amebic meningoencephalitis (PAM). 24 Similar cases of acute onset have also been described in multiple animal species infected by B. mandrillaris. In the 3 canine case reports published, the first of the infected dogs had been diagnosed with lymphoma and inflammatory bowel disease, while the second had a history of long-term steroid administration, and the third and most recently reported case was seemingly immunocompetent; however, all these animals shared a recent history of exposure to stagnant water and lacked apparent skin lesions.5–8 In humans, disease caused by Acanthamoeba spp. and B. mandrillaris traditionally has slower clinical onset and is referred to as granulomatous amebic encephalitis (GAE), with the majority of cases occurring in immunosuppressed patients. 11 Balamuthia mandrillaris can also cause disease in immunocompetent children and older individuals. 11 The bat of our report was considered otherwise healthy prior to the development of acute neurologic signs, with no histologic findings to support extracranial pathology or immunodeficiency. Additionally, qPCR and IFAT for B. mandrillaris were negative on all tissues sampled apart from the brain, leaving the exact portal of CNS entry undetermined.
Neurological balamuthiosis was first reported by the San Diego Zoo in 1986 in a mandrill and has subsequently been documented in gorillas, baboons, gibbons, monkeys, horses, sheep, and dogs.5–7,10,19 Amebic encephalitis has been invariably fatal in animals with rare documentation of survival in humans. 9 As antemortem diagnosis is rarely obtained in veterinary medicine, treatment is generally empirical, consisting of a combination of antifungals and antibiotics. Autopsy represents the most common diagnostic modality, likely because of the acute presentation in most cases and lack of clinical awareness of this uncommon pathogen. Amebic encephalitis is a relatively rare condition in humans and animals, and the vast majority of cases have been diagnosed in highly developed countries. This observation likely reflects the greater density of medical professionals and greater number of postmortem examinations performed in such countries, rather than a geographic niche, and raises concern for global underreporting of the disease. While nonviral infectious agents were included as initial possible differential diagnoses in this case, the diagnostic approach was shaped by public health considerations. Rabies should always be the first differential diagnosis ruled out in any acute neurologic case involving bats because of its profound public health significance. Additional infectious CNS diseases observed in megabats include additional non–rabies lyssaviruses, Angiostrongylus cantonensis verminous encephalitis, tick paralysis, toxoplasmosis, listeriosis, and bacterial meningitis.1,3,8,20 Although megabats are reservoir hosts for Hendra virus and Nipah virus, natural neurological disease associated with Hendra virus has been limited to humans and horses, while clinical disease associated with Nipah virus has been limited to pigs and humans. 2
The florid amebic infiltrate present in this case made for less of a diagnostic challenge, but in more subtle cases ameba may be mistaken for gitter cells histologically, and are readily differentiated with IFAT and IHC techniques. The histologic features of Acanthamoeba spp., B. mandrillaris, and N. fowleri are very similar, but B. mandrillaris may be differentiated with routine hematoxylin and eosin staining (HE) by the occasional presence of binucleation. 21 The multiplex qPCR is based on 3 primer sets targeting 18S ribosomal RNA gene sequences of Acanthamoeba spp., B. mandrillaris, and N. fowleri. This modality is extremely sensitive, capable of identifying the presence of a single organism. 18 In addition to fresh frozen tissues, multiplex qPCR can be used for assessing environmental samples, aiding in epidemiological investigations. Acanthamoeba spp. are ubiquitous within the environment and are easily isolated and cultured in vitro from soil and water sources, while environmental isolation of B. mandrillaris is extremely rare, with detection of nucleic acids by PCR being the most common form of detection in water and soil samples. 11 Formalin-fixed tissues are not optimal for qPCR because of the nucleic acid and protein crosslinking, whereas IHC and IFAT are highly effective in formalin-fixed tissues. 22 Because of the unique biological and ecological niches occupied by each ameba species, identification may help guide preventative measures to mitigate future disease.
While the lesions of amebic encephalitis share commonalities across species, the portals of entry into the CNS vary. Naegleria fowleri is unique in that it directly infiltrates the cranial cavity by migrating across the olfactory epithelium and cribriform plate after entering the nasal passages via exposure to contaminated water, while Acanthamoeba spp. and B. mandrillaris may gain entry via hematogenous dissemination, inhalation, or breaks in the skin. 24 The blood–brain barrier is capable of preventing access to the majority of exogenous pathogens and is naturally absent in some areas of the ventricular system, including sections along the third and fourth ventricles. 16 This may explain the prominent periventricular involvement observed in this and some human case studies. 17 Aside from exploiting this natural anatomical phenomenon, neuropathogenic protozoa utilize multiple mechanisms to breach the blood–brain barrier and invade the CNS. These include contact-dependent factors involving transcellular and paracellular endothelial migration, and contact-independent factors including extracellular toxins. 4 Galactose-binding protein has been shown to be necessary for adhesion of B. mandrillaris to human brain microvascular endothelial capillaries (BMEC). 14 Administration of purified galactose-binding protein prior to inoculation of BMEC acts as a competitive agonist and inhibits B. mandrillaris cytotoxicity, suggesting primary attachment plays a role in endothelial cell death. 14 The major protease produced by B. mandrillaris is abolished when pretreated with a metalloprotease inhibitor (10-phenanthroline), supporting its role as a metalloprotease. 15 Interestingly, inoculation of BMEC with B. mandrillaris proteases disrupts monolayers without affecting BMEC cellular viability. These findings suggest metalloproteases may be important virulence factors of free-living ameba that aid in CNS invasion by targeting cellular adhesions and basement membrane constituents, 2 important components of the blood–brain barrier. 13
The current case documents the occurrence of amebic encephalitis from B. mandrillaris in the order Chiroptera, as determined by multiplex qPCR, IFAT, and IHC. These findings highlight the importance of routine autopsy of animals, which allows for greater understanding of the host range and environmental risk factors for rare pathogens such as B. mandrillaris.
Footnotes
Acknowledgements
We thank Dr. Gary Balsamo, the Louisiana State Public Health Veterinarian, who was instrumental in connecting us with the proper authorities at the CDC. Additional thanks goes to Dr. Francisco Uzal and CAHFS San Bernardino–UC Davis Histology Laboratory for providing the IHC images.
Authors’ note
The findings and conclusions in this report are those of the authors and do not necessarily represent those of the CDC.
Authors’ contributions
NA Crossland contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. I Ali contributed to analysis and interpretation of data; critically revised the manuscript; and gave final approval. C Higbie contributed to acquisition of data; drafted the manuscript; and critically revised the manuscript. J Jackson contributed to analysis and interpretation of data, and critically revised the manuscript. G Pirie contributed to acquisition of data, and critically revised the manuscript. R Bauer contributed to analysis and interpretation of data; critically revised the manuscript; and gave final approval. NA Crossland and R Bauer agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
