Abstract
Nematode, cestode, protozoan, microsporidian, and pentastomid parasites affect domesticated and wild rabbits, hares, and jackrabbits of the genera Brachylagus, Lepus, Oryctolagus, Pentalagus, and Sylvilagus. Some endoparasite infections are of limited or no significance, whereas others have potentially profound consequences. Accurate identification of endoparasites of rabbits, hares, and jackrabbits is an important facet of the work of veterinary pathologists engaged in lagomorph pathology. Here I review endoparasites from the pathologist’s perspective, focusing on pathogenesis, lesions, and implications of infection. Stomach nematodes Graphidium strigosum and Obeliscoides cuniculi are infrequently pathogenic but may cause gastritis and gastric mucosal thickening. Nematodes Passalurus ambiguus, Protostrongylus spp., Trichostrongylus spp., and Trichuris spp. are rarely associated with disease. Adult Capillaria hepatica (syn. Calodium hepaticum) nematodes and non-embryonated eggs cause granulomatous hepatitis in wild Oryctolagus cuniculus and Lepus europaeus, resulting in multifocal, off-white, hepatic lesions, which may be misdiagnosed as hepatic eimeriosis. When the rabbit is an intermediate host for carnivore cestodes, the space-occupying effects of Cysticercus pisiformis and Coenurus serialis may have pathologic consequences. Eimeria stiedai is a major cause of white-spotted liver in O. cuniculus, particularly in juveniles. Enteric coccidiosis is a noteworthy cause of unthriftiness in young animals, and frequently manifests as diarrhea with grossly appreciable multifocal off-white intestinal lesions. O. cuniculus is the natural host for the zoonotic microsporidian Encephalitozoon cuniculi. Infection may be acute and focused mainly on the kidneys, or it may follow a chronic disease course, frequently with neurologic lesions. A latent carrier status may also develop.
In the order Lagomorpha, family Leporidae, domesticated and wild rabbits (genera Brachylagus, Oryctolagus, Pentalagus, and Sylvilagus) and hares and jackrabbits (genus Lepus) may be affected by nematodes, cestodes, protozoans, microsporidians, and pentastomids. In some cases, endoparasite infection has profound consequences for the host, whereas in other instances, endoparasite infection is of limited or no significance. Accurate diagnosis of endoparasite infections of rabbits, hares, and jackrabbits is thus an important facet of the work of veterinary pathologists engaged with lagomorph pathology.
In this review, I examine identification of endoparasites in lagomorphs from the perspective of the anatomic pathologist, focusing particularly on pathogenesis, lesions, diagnosis, and implications of infection for the infected animal, conspecifics, and other in-contact species. Conduction of fecal egg counts and fecal egg identification will not be discussed in detail as this constitutes part of the specialist work of veterinary parasitologists.
Hares and jackrabbits of the genus Lepus spp. will be referred to as hares. The discussion will focus on the European rabbit (Oryctolagus cuniculus) and hares, but reference will be made to the pygmy rabbit (Brachylagus), Amami rabbit (Pentalagus), and various other rabbits and cottontails (Sylvilagus spp.), for which there are relevant data.
Nematodes
Graphidium strigosum and Obeliscoides spp
Nematodes affecting the stomach of lagomorphs include Graphidium strigosum and Obeliscoides cuniculi. G. strigosum is particularly associated with infections in the European rabbit, 46 European brown hare (Lepus europaeus), 96 and Irish hare (Lepus timidus hibernicus; an endemic subspecies of the mountain hare). 6 O. cuniculi typically infects European rabbits, snowshoe hares (Lepus americanus), and eastern cottontail rabbits (Sylvilagus floridanus) in North America.90,91 Following the introduction of the eastern cottontail in Italy for hunting purposes, O. cuniculi has also been detected in wild European rabbits, 42 as well as in European brown hares in Ontario, Canada, 90 suggesting considerable overlap in host range. Obeliscoides pentalagi has been described in Amami rabbits (Pentalagus furnessi), 49 and recent descriptions of other disease processes in this species have also recorded the presence of gastric nematodes that were not further described. 74
Within European rabbits, the context within which the rabbits are kept strongly impacts the prevalence of stomach nematodes. A 1992–1996 survey of wild European rabbits in the United Kingdom found a 78% prevalence of G. strigosum, 2 which supports my own observations examining wild European rabbits from Cambridgeshire, United Kingdom, where I see a high prevalence of infection. By contrast, G. strigosum and O. cuniculi were detected at low levels in slaughtered European rabbits in Poland, with prevalences of 1.09% and 0.36%, respectively. Stomach nematodes are infrequently reported in European rabbits maintained as pets. 57
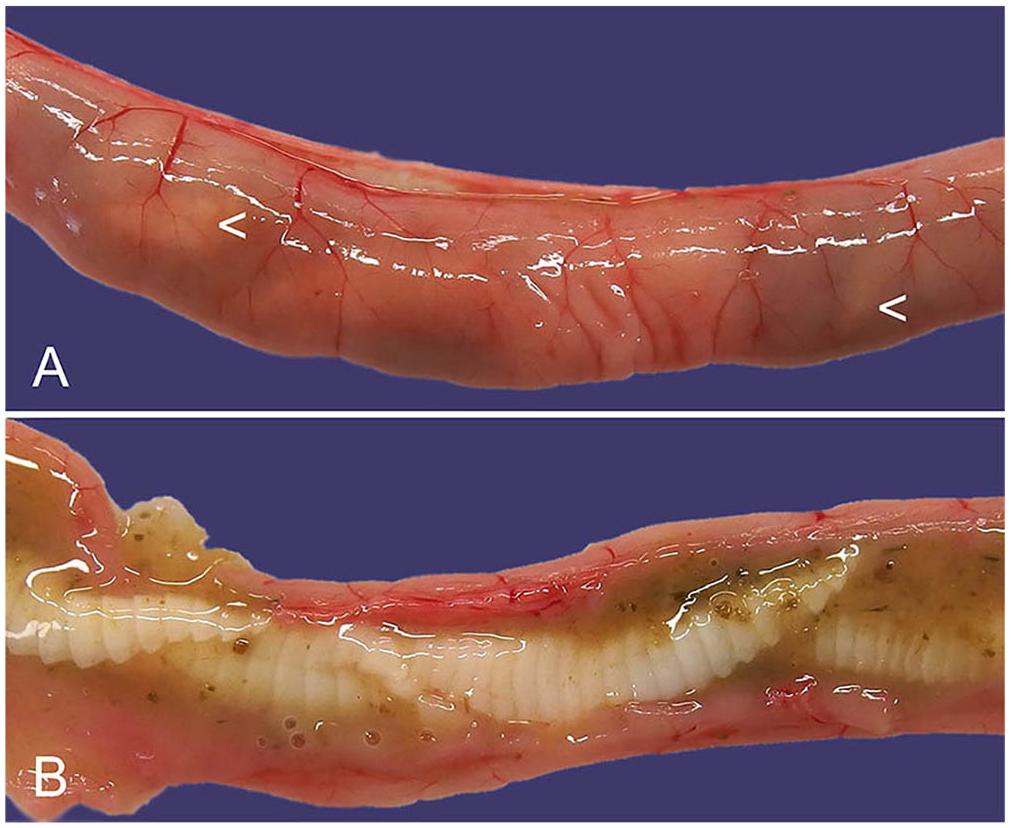
Both G. strigosum and O. cuniculi have a direct lifecycle, and third-stage larvae are infective. Infections are apparent macroscopically. In the case of G. strigosum, red nematodes are clearly visible in close association with the gastric mucosa (Fig. 1). 40 The adult males have a prominent caudal bursa that is visible macroscopically. 88 Microscopically, G. strigosum has a ridged cuticle, and the large intestine is lined by multinucleate cells. Brown chitinized spicules may be apparent in sections of adult males (Fig. 1). 51 Macroscopically, O. cuniculi is off-white, and the males also have a prominent bursa and chitinized spicules visible microscopically. 42 O. pentalagi is dark-red. 49
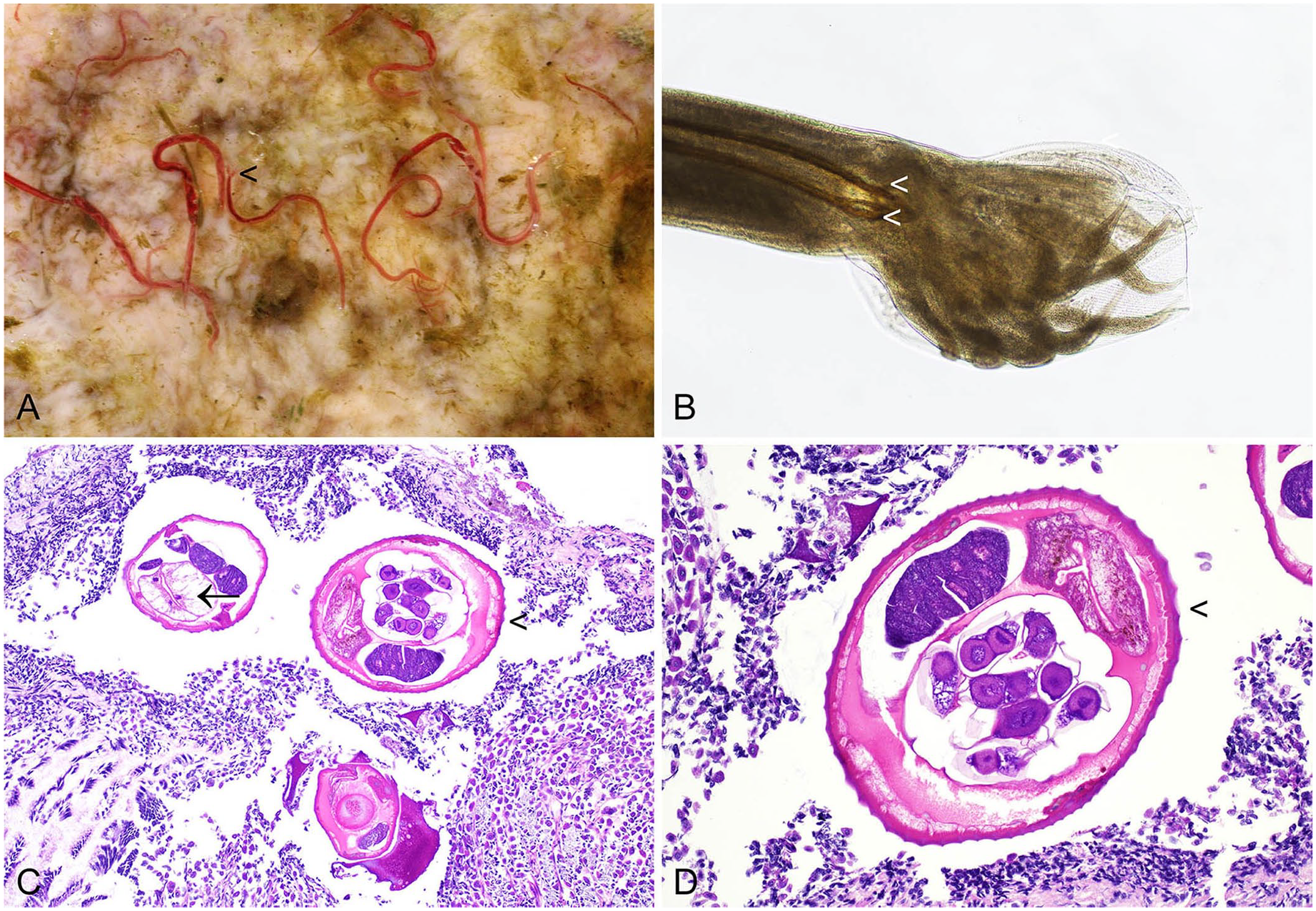
Graphidium strigosum in the stomach of a European rabbit.
Gastric nematodes are infrequently pathogenic in rabbits and hares, but O. cuniculi may cause gastritis, gastric mucosal thickening, petechiation, and small white mucosal foci. In experimental infections, histologically there may be edema and infiltrates of lymphocytes and eosinophils. 115 G. strigosum infection in European brown hares is suggested to be more pathogenic than in O. cuniculus, with high burdens potentially resulting in anemia. 33 However, this suggestion is somewhat controversial, with another author noting lower G. strigosum burdens in hares compared to wild European rabbits sampled from the same locality, and no apparent gastric lesions in the infected hares; the difference in infection level may have been due to relative resistance of hares to the infective third-stage larvae of G. strigosum, or different grazing behaviors of hares compared to rabbits may have led to differing levels of exposure. 14
Trichostrongylus spp.
Intestinal strongyles include Trichostrongylus calcaratus and T. retortaeformis, and, in both cases, adults are found in the small intestine. T. affinis also affects lagomorphs, and adult nematodes are found in the cecum and large intestine. T. calcaratus and T. affinis have been detected in eastern cottontail rabbits, 39 and T. retortaeformis is associated with infections in European rabbits and hares.2,6,14,30 T. calcaratus and T. affinis have been identified in eastern cottontail rabbits in Italy and represent nematode species exotic to the Italian ecosystem.53,135 Conversely, Italian eastern cottontails may also be infected with T. retortaeformis, which naturally occurs in European lagomorphs. 53
Grossly, Trichostrongylus spp. are small and difficult to identify. Histologic features are those of the family Trichostrongylidae to which Trichostrongylus spp. belong. Trichostrongylus spp. receive little attention as a source of significant disease in lagomorphs, although in one study, control European rabbit does had 20% more offspring survive to the point of weaning than did does experimentally infected with T. retortaeformis. 34 Myxoma viral infection may alter the susceptibility of European rabbits to T. retortaeformis infection. Rabbits that were infected with myxoma virus had higher nematode burdens and maintained high levels of infection for longer, the latter suggestive of a disrupted ability to clear the nematode infection.15,22
Passalurus ambiguus
Passalurus ambiguus is the rabbit pinworm, an oxyurid nematode that is present in the cecum and large intestine. P. ambiguus has a direct lifecycle, in which eggs are deposited around the anus and egg ingestion occurs during cecotrophy.
In Finland, P. ambiguus eggs were found in 3% of fecal samples from pet rabbits, 86 and the same proportion of samples were affected in a German study of rabbit fecal specimens. 107 Prevalence in wild European rabbits is likely to be considerably higher.2,14 P. ambiguus has also been identified in eastern cottontails,39,140 European brown hares, 123 and the Italian hare (syn. Corsican hare; L. corsicanus), 30 suggesting a wide host range.
Adult nematodes may be detected in the cecum and large intestine, or in the cecotropes or feces. Nematodes are off-white, thread-like, and 5–10 mm long. 57 Sub-grossly, the females have pointed tails (Fig. 2A). P. ambiguus may also be detected as an incidental finding in histologic sections from the cecum or large intestine. Characteristic histologic features include a muscular esophagus, platymyarian musculature, and double lateral alae (Fig. 2B). P. ambiguus infection is typically considered nonpathogenic, but it is possible for heavy infestations in young rabbits to manifest as perianal pruritus, and the presence of large numbers of nematodes may contribute to the enteritis complex. 57
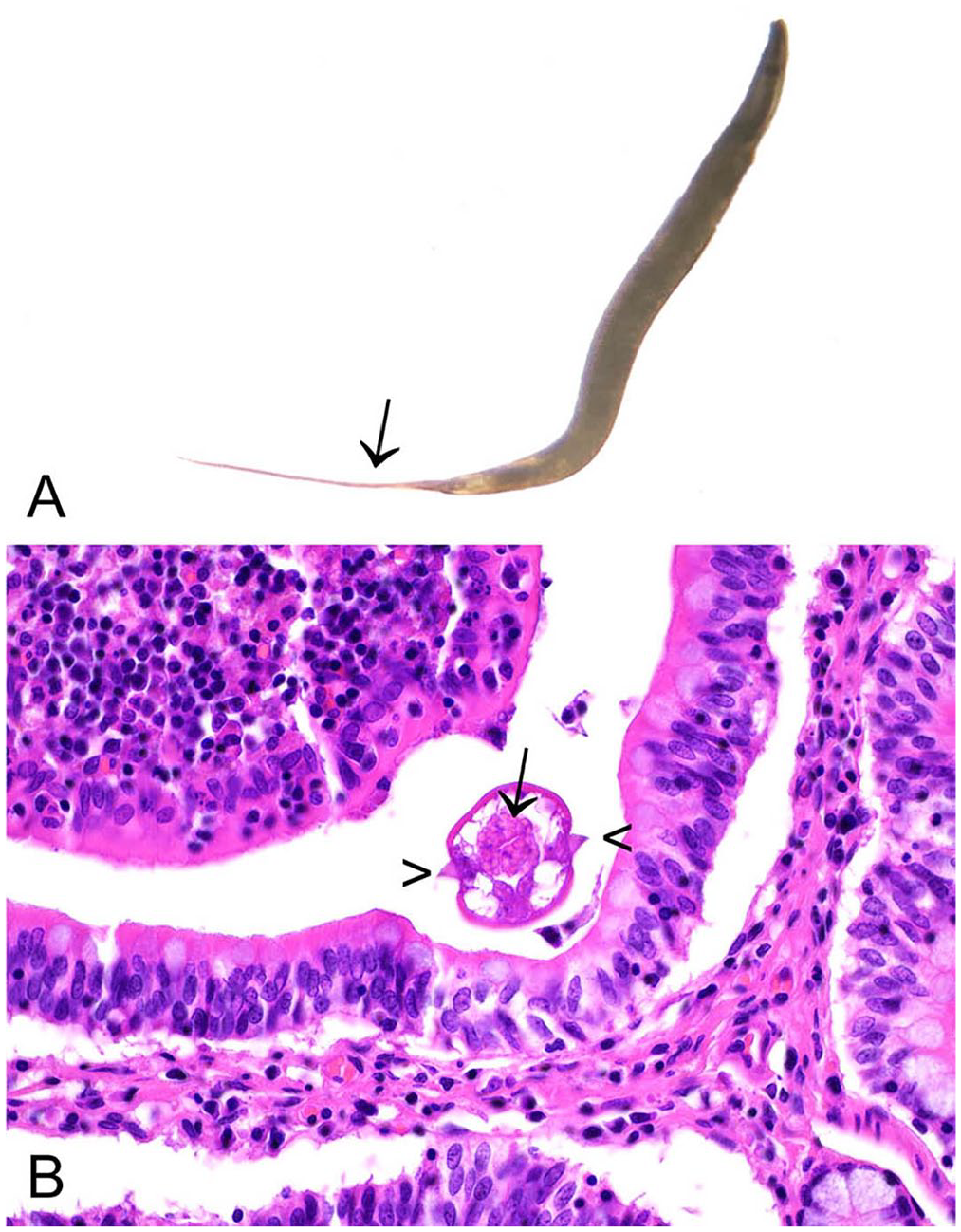
Passalurus ambiguus in the cecum of a European rabbit.
Trichuris spp.
Trichuris spp. are commonly known as whipworms, and they have a direct lifecycle. Adults are present in the cecum and large intestine. Species infecting lagomorphs include T. leporis and T. sylvilagi. Trichuris spp. have been identified in wild European rabbits, 45 eastern cottontails, 39 European brown hares, 101 and Amami rabbits. 49
Macroscopically, T. leporis and T. sylvilagi are ~20 mm long and have a long, hairlike anterior end that is embedded in the mucosa. 134 Trichuris spp. are aphasmid nematodes; thus, they have bacillary bands, and a distinctive stichosome, which refers to a row of esophageal glands surrounding the esophagus. Eggs have bipolar plugs, as do Capillaria hepatica eggs. Like Trichostrongylus spp., Trichuris spp. infections in rabbits receive little attention and do not appear to be commonly associated with disease. Infections appear to be uncommon in pet animals, with 1 study documenting 1 fecal sample containing T. leporis eggs, out of 434 samples, 107 and another study finding 1 positive sample out of 398. 86
Capillaria hepatica (syn. Calodium hepaticum)
As alluded to above, C. hepatica (syn. Calodium hepaticum) is also an aphasmid nematode. The nematode has a direct lifecycle, a wide host range, and is zoonotic.13,48,94,109,114 Embryonated eggs are ingested by natural hosts, and first-stage larvae hatch in the cecum and undergo hepatic migration. In the liver, mature nematodes develop, and fertilized adult females release non-embryonated eggs before dying. Two main mechanisms for continuation of the lifecycle are inferred. Either disintegration of the carcasses of dead hosts facilitates environmental dispersal of non-embryonated eggs, or non-embryonated eggs are consumed by predators and excreted along with the host feces. In favorable conditions, the first-stage larva develops within the egg to an infective stage. 13
Humans may acquire infection by 2 pathways. Embryonated eggs may be ingested from the environment in a process that is referred to as “true infection.” This is a rare occurrence but is frequently fatal. Conversely, consumption of raw or undercooked livers, and thus non-embryonated eggs, from wild hosts, leads to these eggs passing along the gastrointestinal (GI) tract. The non-embryonated eggs are eventually expelled in the feces, in what is termed a “spurious infection.” 13
An 8% prevalence of hepatic capillariosis has been reported in wild European rabbits in the United Kingdom. 16 A 0.9% prevalence of infection has also been reported in European rabbits in La Palma, Canary Islands. 44 Adult female C. hepatica nematodes and non-embryonated eggs cause granulomatous hepatitis, resulting in grossly apparent, multifocal, off-white, hepatic lesions, which may be misdiagnosed macroscopically as hepatic eimeriosis (Fig. 3A). 16 In histologic sections, the granulomas are distributed randomly, rather than as inflammatory lesions focused on the bile ducts, as in hepatic eimeriosis. Profiles of degenerating nematodes, or more commonly bi-operculated eggs, may be found in the necrotic centers of granulomas (Fig. 3B, 3C). Hepatic capillariosis has also been described in European brown hares 10 and eastern cottontail rabbits. 79 As C. hepatica infects a wide species range, the role of lagomorphs in its disease ecology merits further contemporary investigation. For example, it is unclear if the 8% prevalence of C. hepatica in wild European rabbits is representative of the United Kingdom as a whole or if there are significant regional variations in prevalence. Anatomic pathologists are ideally placed to study this parasite because it will not necessarily be detected using fecal egg counts, and differentiation of the macroscopic liver lesions from those of hepatic eimeriosis is required. 16
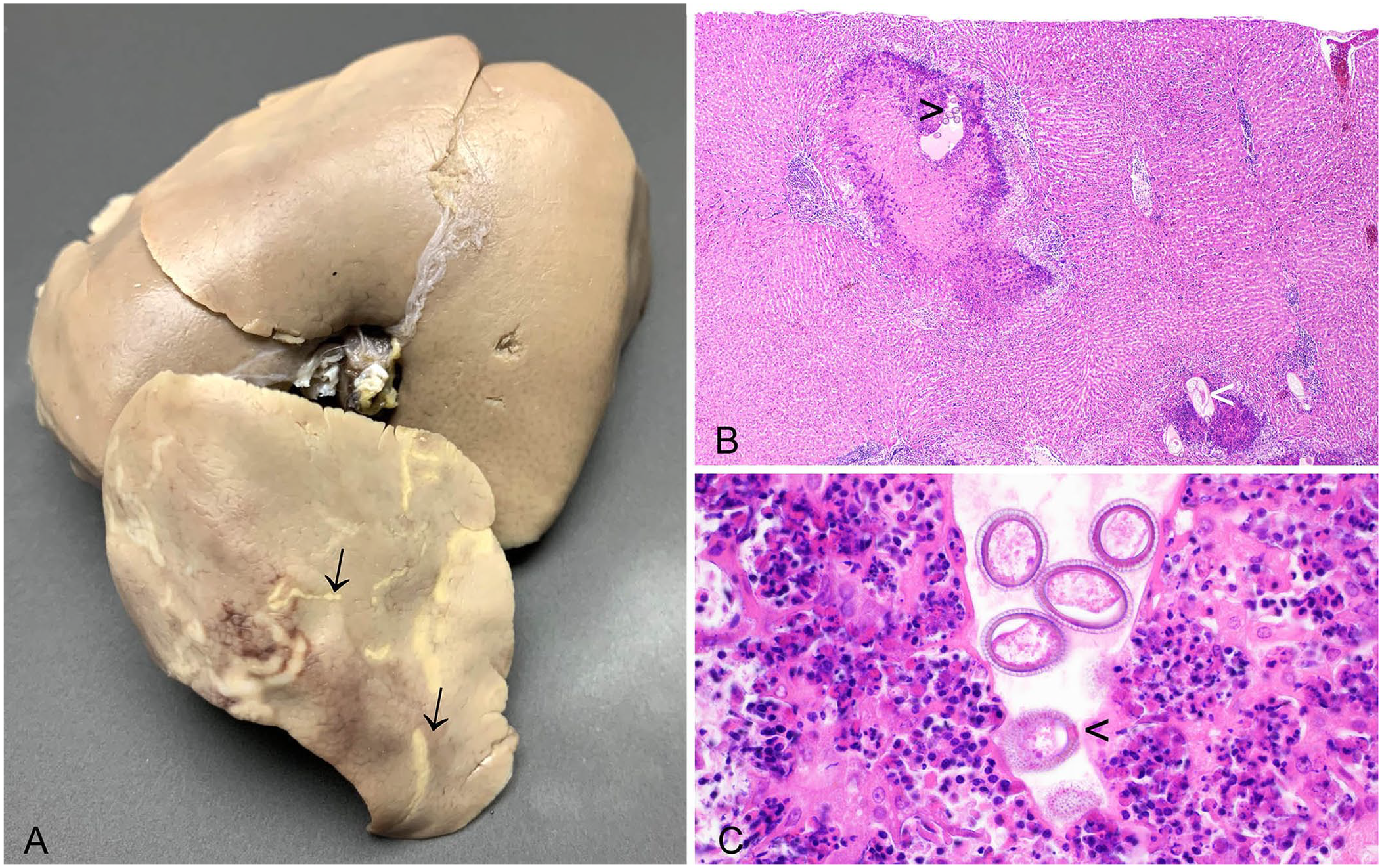
Capillaria hepatica in the liver of a European rabbit.
Protostrongylus spp
Protostrongylus spp. are metastrongyle lungworms with an indirect lifecycle. Finnish mountain hares (Lepus timidus) and European brown hares examined seasonally from September to the end of February 1998–2001 had a high prevalence of lungworm infection, with 96.5% of mountain hares and 60% of European brown hares exhibiting evidence of infection. 77 Protostrongylus spp. infection has also been reported in a domestic rabbit. 124
Macroscopically, these metastrongyles may be difficult to identify as they are frequently embedded in lung tissue and are brown and hair-like. In the Finnish study, lesions were most severe in the caudal lung lobes. Macroscopic changes included well-demarcated foci of green and brown pulmonary parenchyma. Histologic lesions comprised mixed inflammation including granulocytes. Lungworm infection did not appear to impact the condition and weight of the hares. 77
Baylisascaris spp.
Baylisascaris spp. are ascaridoid nematodes. 118 B. procyonis infects common raccoons (Procyon lotor) and has a direct lifecycle, although rabbits, hares, and other species can act as paratenic hosts. B. procyonis is zoonotic and can cause neurologic disease in humans and wildlife, including eastern cottontails 66 and European rabbits.26,119 Neurologic disease arises due to larval migration (cerebral larval migrans). Histologic lesions comprise malacic migration tracts and variable degrees of encephalitis, granuloma formation, or gliosis. Nematode larvae are inconsistently detected in histologic sections.26,119 Raccoon feces are a potential source of infection for domestic rabbits; hence, pets maintained outside, or in proximity to raccoons, are more at risk than house rabbits.26,81 The European rabbit has also been experimentally infected with Ascaris columnaris (now designated B. columnaris). 23
Dirofilaria spp. and other filarial nematodes
Dirofilaria spp. are filarial nematodes. The lifecycle includes a vertebrate definitive host and an arthropod vector, such as a mosquito. Adult female nematodes in the definitive host produce microfilariae that are disseminated in the blood. A feeding arthropod vector ingests the microfilariae. The microfilariae mature in the vector, and the resulting infective larvae migrate to the mosquito’s mouth-parts. From here, the microfilariae enter a new host when the vector feeds. In the new definitive host, the microfilariae mature, migrate to the site where the adult parasites live, and mate. The ensuing microfilaremia continues the lifecycle. 126
D. immitis infection is typically associated with dogs and cats; recorded infections involving lagomorphs are sparse. Experimentally, D. immitis infection causes pulmonary nodules in European rabbits. 120 Two adult D. immitis nematodes have been recorded in the lungs of a Japanese hare (Lepus brachyurus angustidens). The histologic pulmonary lesions included corrugated, proliferative intimal fibrosis, endoarteritis, and thrombosis. 99 Similar lesions have been associated with an incidental finding of suspected D. immitis in a pet rabbit with spinal lymphoma. 110
D. scapiceps is primarily reported in North American lagomorphs4,11,73 but has also been recorded in an individual European brown hare from Macedonia, Greece. 28 Adult D. scapiceps nematodes are found associated with the tendon sheath, most commonly in the tarsal region, or less commonly near the stifle. Adult nematodes can be detected grossly. Histologically, there may be minimal reaction to the presence of the nematodes or there may be a granulomatous response. Microfilariae may also be seen in spaces near adult nematode profiles. 11
Filarial nematodes have also been detected in 2 adult Iberian hares (syn. Granada hare; Lepus granatensis). Sequencing suggested that these filariae were genetically related to Micipsella numidica, and they have been provisionally assigned the name Micipsella iberica n. sp. The authors of the study highlighted the potential for filarial nematode infections to cause population declines in already fragile lagomorph populations, such as that of the Iberian hare. 32
Toxocara spp.
The potential for T. canis and T. cati to cause visceral larval migrans in rabbits has been demonstrated experimentally. 141
Cestodes
The cestodes discussed in this section have a 2-host lifecycle. Adult tapeworms are present in the GI tract of the definitive host. Proglottids containing eggs are passed in the feces of the definitive host, and these are ingested by the intermediate host, which is usually a different species. In the intermediate host, eggs hatch into larvae and encyst in skeletal muscle or other foci (see below). When the intermediate host is consumed by the definitive host, the cysts are digested in the intestine of the definitive host, releasing the scolex, that forms the anterior aspect of the adult cestode, and allowing attachment to the intestinal wall. Development of the adult cestode completes the cycle. 63 Rabbits and hares may be affected by a range of cestodes, both as an intermediate and definitive host (Table 1).
Selected cestode parasites of lagomorphs.
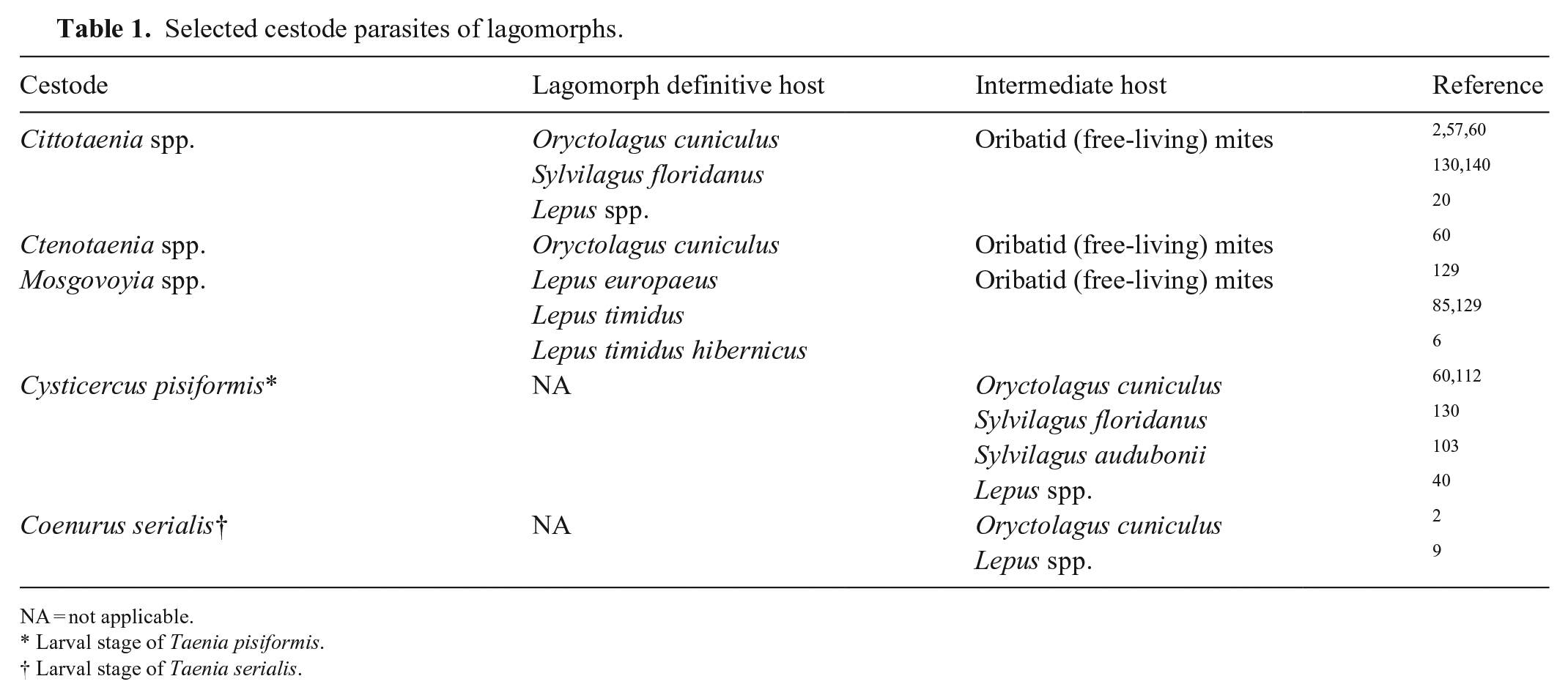
NA = not applicable.
Larval stage of Taenia pisiformis.
Larval stage of Taenia serialis.
Lagomorphs as the cestode definitive host
Theoretically, lesions due to adult cestodes may arise from the physical presence of the cestodes, or due to erosions resulting from the scolex hooks. Hypersensitivity reactions are also theoretically possible. 63 Rabbits and hares are the definitive hosts for several species of tapeworm, including Cittotaenia spp., Ctenotaenia spp., and Mosgovoyia spp. However, lesions are rarely recorded.
Macroscopic identification of cestode infection is straightforward, with tapeworms frequently visible through the relatively thin-walled small intestine even prior to incision (Fig. 4). Gravid proglottids containing eggs may be noted in the feces. 57
Cestodes in the small intestine of a European rabbit (definitive host).
Myxomatosis virus increases the prevalence and intensity of infection of wild European rabbits infected with Mosgovoyia pectinata. 15 This is similar to the case with Trichostrongylus spp.
Lagomorphs as the cestode intermediate host
Lagomorphs are an intermediate host for the carnivore cestodes Taenia pisiformis and T. serialis. A peritoneal location is typical for Cysticercus pisiformis, 116 whereas the larval form of T. serialis, Coenurus serialis, typically encysts in the musculature. 1 Cysts are easily identifiable macroscopically and comprise fluctuant structures with translucent contents. C. pisiformis tends to form multiple grape-like clusters of cysts, each with a single inverted scolex recognizable as an off-white nodule. 104 C. serialis forms numerous, off-white, ~1-mm nodules, arranged in characteristic radiating clusters and rows (Fig. 5A), 1 which are the invaginated scolices adhering to the cyst interior. In histologic sections, the armed scolex or scolices of the cestode larval stage may be visualized (Fig. 5B, 5C). Basophilic calcareous corpuscles may also be seen, the function of which is incompletely elucidated. T. serialis infection can be zoonotic, with humans acting as an intermediate host. 38
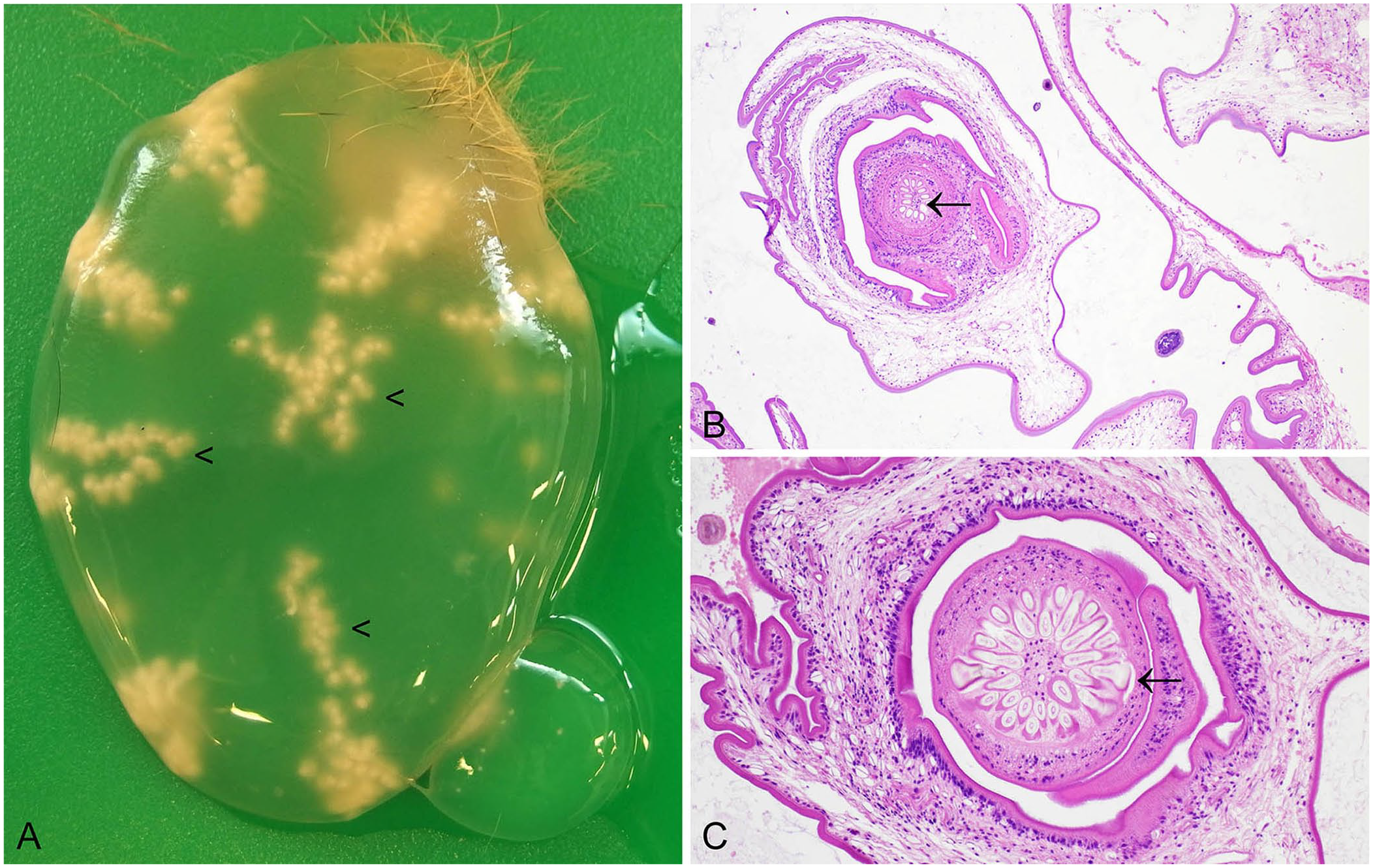
Coenurus serialis dissected from skeletal muscle of a European rabbit.
The space-occupying effects of Cysticercus pisiformis and Coenurus serialis may have location-dependent pathologic consequences for the lagomorph. For example, exophthalmos has been reported as a consequence of a T. serialis coenurus located in the conjunctival fornix of a pet dwarf lop rabbit 97 ; facial swelling and reduced appetite were reported in a pet lionhead rabbit with mandibular C. serialis. 106 Although C. serialis typically encysts in the musculature in rabbits, GI stasis has also been reported due to the atypical presence of a mid-abdominal C. serialis cyst. 1
The migration of T. pisiformis larvae in eastern cottontail rabbits can cause hepatic inflammation and necrosis. 116 Similarly, granulomatous hepatitis has been recorded in European rabbits associated with numerous T. pisiformis cysticerci of various developmental stages.54,117
Coccidia
Coccidia are intracellular obligatory parasites in the phylum Apicomplexa. Some are monoxenous (utilizing a single host species), such as Eimeria spp. in most instances, whereas other species are heteroxenous (use 2 hosts).
Eimeria stiedai
Eimeria stiedai infects biliary epithelium and is a major cause of white-spotted liver in European rabbits. In wild rabbits in the United Kingdom, white-spotted liver caused by E. stiedai is associated with the juvenile age class. 16 E. stiedai is a common pathogen of young farmed rabbits. 40 Sporadic, clinically significant, infections in pet rabbits are documented. 95 Cottontail rabbits do not appear to develop natural infections of E. stiedai, but may be infected experimentally. 62
E. stiedai transmission is fecal-oral. Unsporulated Eimeria oocysts pass into the GI tract and are shed in the feces. Sporulation occurs within 1–4 d, and results in the oocysts becoming infective. Oocysts are resistant to a range of environmental conditions. Sporulated oocysts may be ingested via contaminated food or water. Auto-infection through cecotrophy does not occur because cecotropes contain unsporulated Eimeria oocysts. 127 Schizogony is the phase of asexual reproduction, and gametogony is the phase of sexual reproduction that leads to oocyst formation, and these occur in biliary epithelial cells.
E. stiedai infections result in off-white, frequently bosselated, hepatic foci (Fig. 6A). We devised a grading scheme to delineate the extent of the lesions. 16 Although there is a high prevalence of infection in wild rabbits, most animals have mild gross lesions that are likely not clinically significant. 16 Histologically, various developmental stages of the parasite, including meronts (merozoites), macro- and microgamonts, and oocysts may be observed in the biliary epithelium, with destruction of parasitized cells, and in the bile duct lumen. Lesions are frequently accompanied by periductular fibrosis, infiltration of inflammatory cells, including macrophages and heterophils, and bile duct hyperplasia (Fig. 6).7,16,133
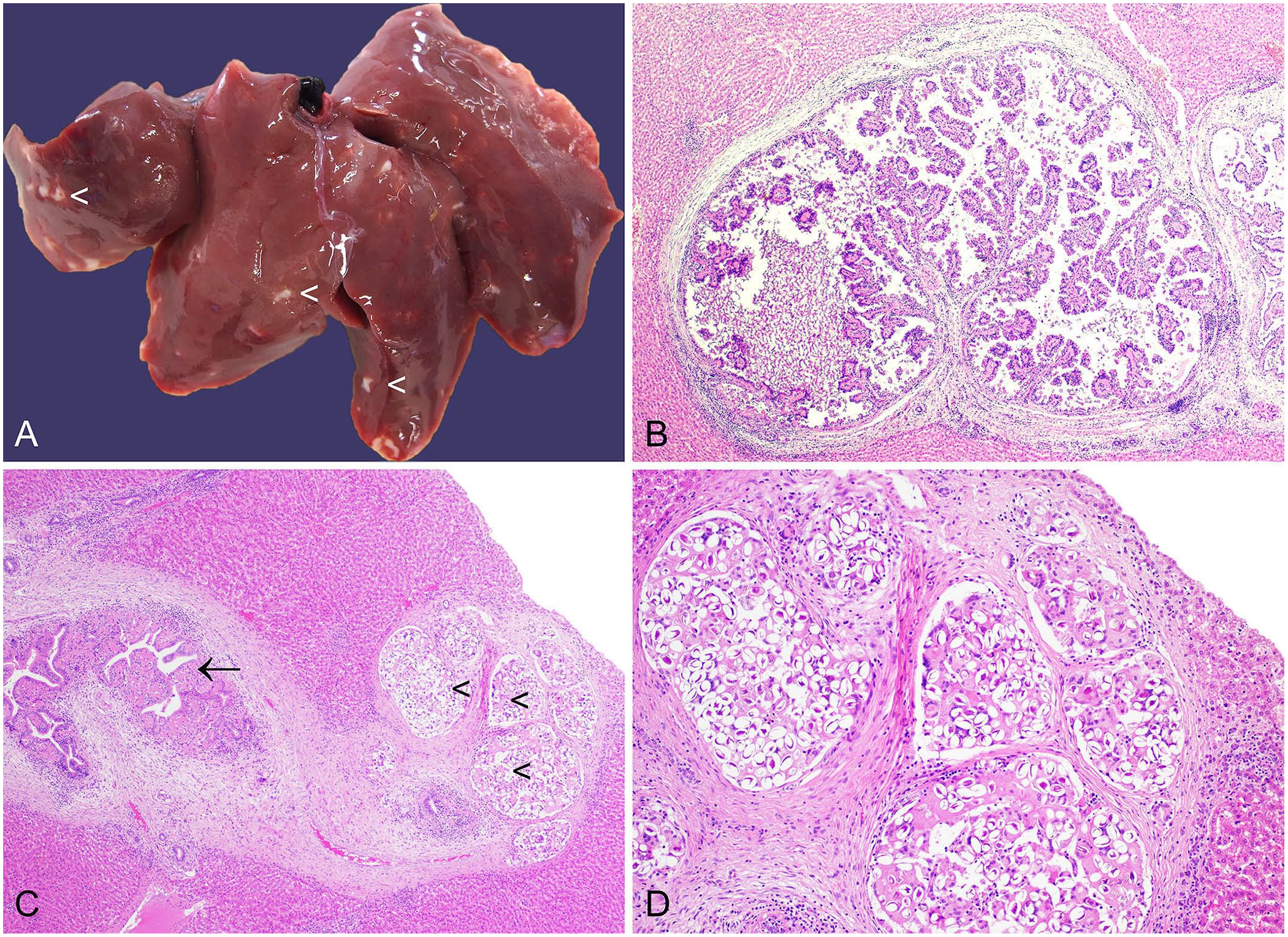
Eimeria stiedai in the liver of a European rabbit.
Diagnosis is readily achieved by a combination of gross and microscopic examination. Infection with C. hepatica constitutes a differential diagnosis for the gross lesions, and a number of other potential or reported causes of white-spotted liver should also be considered, including Cysticercus pisiformis, and bacteria including Yersinia pseudotuberculosis and Francisella tularensis. 16 Impression smears have been used to aid in the diagnosis of hepatic eimeriosis. 5
Enteric Eimeria spp
Many species of Eimeria, of varying pathogenicity, infect enterocytes. The lifecycle is as described for E. stiedai but with infection of enterocytes rather than biliary epithelial cells. Level of pathogenicity and localization in the intestine vary and have been reviewed.36,100 Although their sites of development within the intestine overlap, it is likely that each species of Eimeria has its own niche within the intestinal microenvironment. 100
Authors vary slightly in their classification of Eimeria spp. as high, intermediate, or low pathogenicity, but in general, for European rabbits, E. flavescens, E. intestinalis, E. irresidua, E. magna, and E. piriformis are considered of “high” or “intermediate” pathogenicity, and some authors also include E. media as “mildly pathogenic” or “pathogenic.” 100 E. brachylagia has been described in the pygmy rabbit. E. brachylagia has been suggested to be potentially extremely pathogenic, with the caveat that the affected animals may have experienced stress due to captivity. 37 E. furnessi n. sp., E. hilleri n. sp., and E. sagentae n. sp. have been identified in fecal samples from the Amami rabbit. Further research is required to definitively establish their pathogenicity. 136 Of the 32 species of hares, only 10 have been examined for coccidia and, from these species, 43 Eimeria spp. have been identified, with some overlap with the domestic rabbit and cottontail rabbits. 35 Thus, across genera of rabbits and hares, a plethora of enteric Eimeria spp. exist and are of varying clinical significance. However, some general patterns of pathology emerge across lagomorphs infected with pathogenic Eimeria spp.
Enteric coccidiosis causes unthriftiness and diarrhea in young, recently weaned animals. This can be a particular issue in breeding units, primarily affecting European rabbits, but also where hares are kept in captivity.21,142 Young weanling wild cottontail rabbits that are presented to wildlife rehabilitation centers may also have enteric eimeriosis. This manifests as diarrhea, potentially with additional GI stasis and cecal impaction. 102 Enteric coccidiosis is also a major cause of mortality in young, wild European brown hares. 41
Infected juveniles may have low body condition score and extensive fecal soiling in perianal foci and affecting the hindlimbs. High burdens of enteric Eimeria spp. manifest as numerous white nodules expanding the intestinal wall and grossly visible from both serosal and mucosal aspects (Fig. 7A, 7B). Various developmental stages of coccidia are microscopically detectable within enterocytes (Fig. 7C, 7D), together with necrosis and inflammation.102,104 Fecal examination provides quantification of fecal oocyst numbers and is therefore a valuable adjunct to diagnosis. However, it is important to note that oocysts can be detected in the feces of clinically normal animals because not all Eimeria spp. are pathogenic. In addition, the Eimeria asexual reproduction phase can lead to necrosis and inflammation prior to shedding of oocysts. 102 Fecal oocyst counts should therefore be interpreted in parallel with gross and microscopic findings.
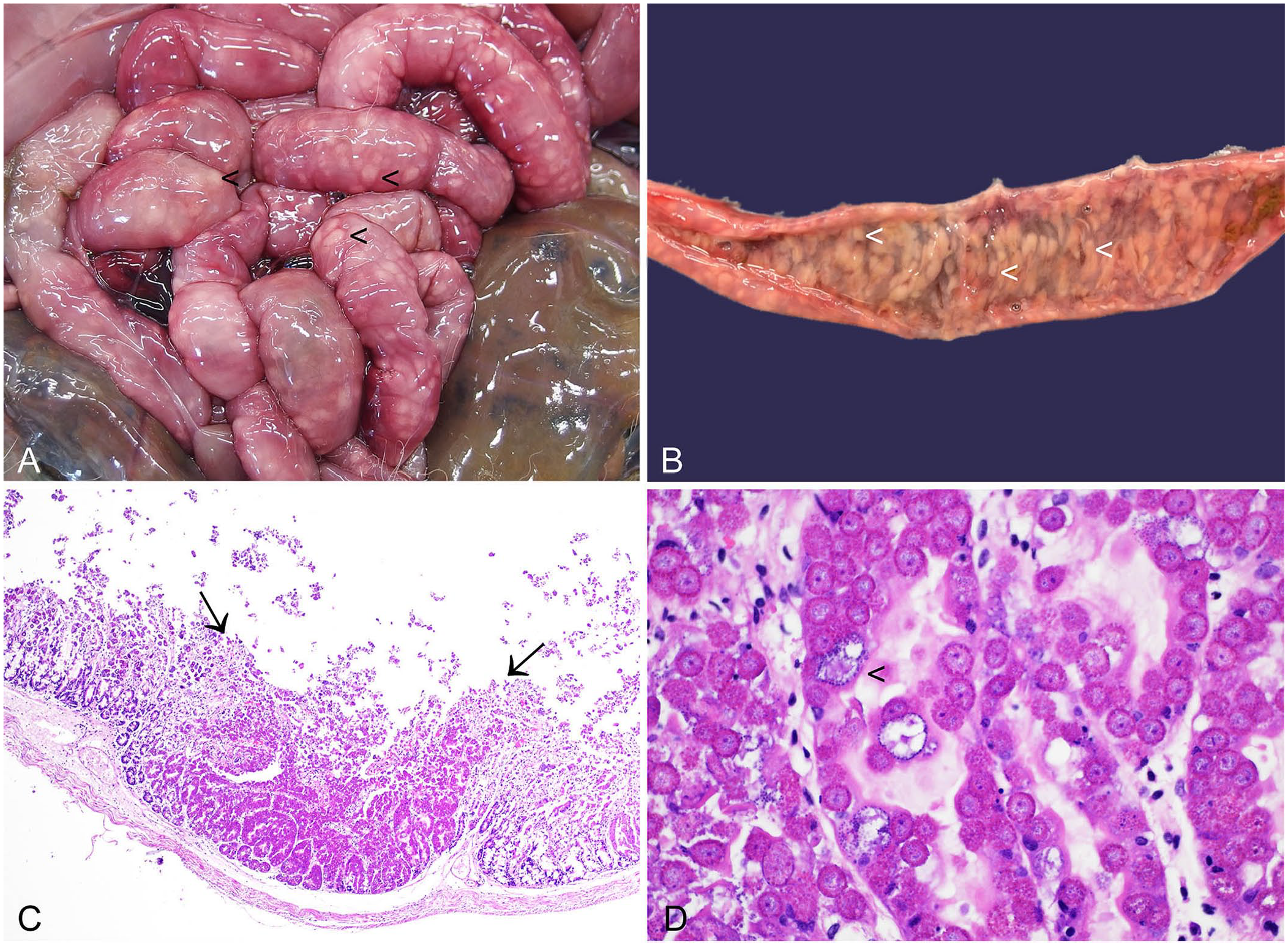
Enteric coccidiosis in the small intestine of a European brown hare.
Intussusception has been reported in an adult female European brown hare with high levels of E. leporis infection; the intussusception may have resulted from intestinal hypermotility due to high levels of endoparasitism. 89
Cryptosporidium spp.
Cryptosporidium spp. can infect intestinal and/or respiratory epithelial cells. A study including modest numbers of European rabbits and European brown hares identified C. parvum in feces from 7% of wild rabbits in central mainland Britain, but in neither of 2 hares. 131 Fecal positivity has also been documented in wild Iberian hares from southern Spain. 111 Cryptosporidia have been visualized histologically in a wild juvenile female eastern cottontail rabbit found dead in Illinois. 116
As in other species, cryptosporidiosis is predominantly of clinical consequence in younger rabbits and is more likely to constitute a clinical problem in facilities in which rabbits are reared intensively.71,125 Infection with Cryptosporidium spp. may contribute to a multifactorial syndrome of enteritis. 57 Histologic diagnosis is based on the presence of apical, weakly basophilic, ~2-µm, round protozoa that are present within a parasitophorous vacuole. 116 Villus atrophy may be observed.
Toxoplasma gondii
Toxoplasma gondii is a heteroxenous apicomplexan parasite. Sexual reproduction occurs in the intestine of the definitive host, a felid. Infection of intermediate hosts is by ingestion of food or water contaminated with oocysts from the definitive host. Asexual reproduction occurs in tissue cysts of intermediate hosts. Intermediate hosts are birds and mammals, including rabbits and hares. The carnivorous definitive hosts are infected by consumption of tissue cysts in the intermediate hosts.3,132 Infection may also occur in utero or via milk.
Toxoplasmosis has been a noted cause of mortality in hares.56,70,105,121 In contrast, lesions associated with toxoplasmosis are rarely diagnosed in European rabbits. 31 Antibodies to T. gondii have been detected in eastern cottontail rabbits from Missouri and Kansas. 128 The apparent variable level of susceptibility to clinical infection of the hares compared to European rabbits and cottontail rabbits merits further investigation. 3
Hares experimentally infected with T. gondii oocysts develop hemorrhagic enteritis, mesenteric lymphadenopathy with hyperemia, splenomegaly, and necrotic hepatic foci. 121 Diagnosis of toxoplasmosis is usually achieved via histopathology. Necrotic lesions in multiple organ systems are associated with protozoal tachyzoites or bradyzoite cysts and variable levels of attendant inflammation.31,121 Immunohistochemistry (IHC) may aid diagnosis. 31
T. gondii is zoonotic. Rabbits and hares are hunted for meat for human consumption in some countries, which has potential implications for zoonotic disease transmission.3,55 Consumption of undercooked meats, including game, is an established risk factor for toxoplasmosis in pregnant women. 24
Sarcocystis spp.
Sarcocystis spp. are also heteroxenous. Rabbits and hares act as intermediate hosts, and Sarcocystis spp. bradyzoite cysts can be found in skeletal and cardiac muscles. S. cuniculi has been detected in European rabbits, 19 and S. leporum in cottontail rabbits. 43 In both cases, the definitive host is the cat. Prevalence appears regionally variable. 55 Sarcocystis spp. cysts have also been documented in European brown hares. 98
Mild infections are usually undetectable grossly, but severely parasitized muscles may have macroscopically visible streaks. Histologically, sarcocysts are round to elongate-oval and ~50 µm in diameter, with a thin hyalinized wall. They contain abundant crescent-shaped zoites each measuring ~4 × 10–15 µm (Figs. 8, 9). 122 It has been suggested that adiaspiromycosis could be misdiagnosed as protozoal cysts. 50 We have identified adiaspores in rabbit lung and lymph node as either an incidental or potentially clinically relevant finding. The bi- or trilaminar wall of adiaspores, together with the granular-to-foamy, basophilic core and location, aid in distinction from Sarcocystis spp. cysts.64,65
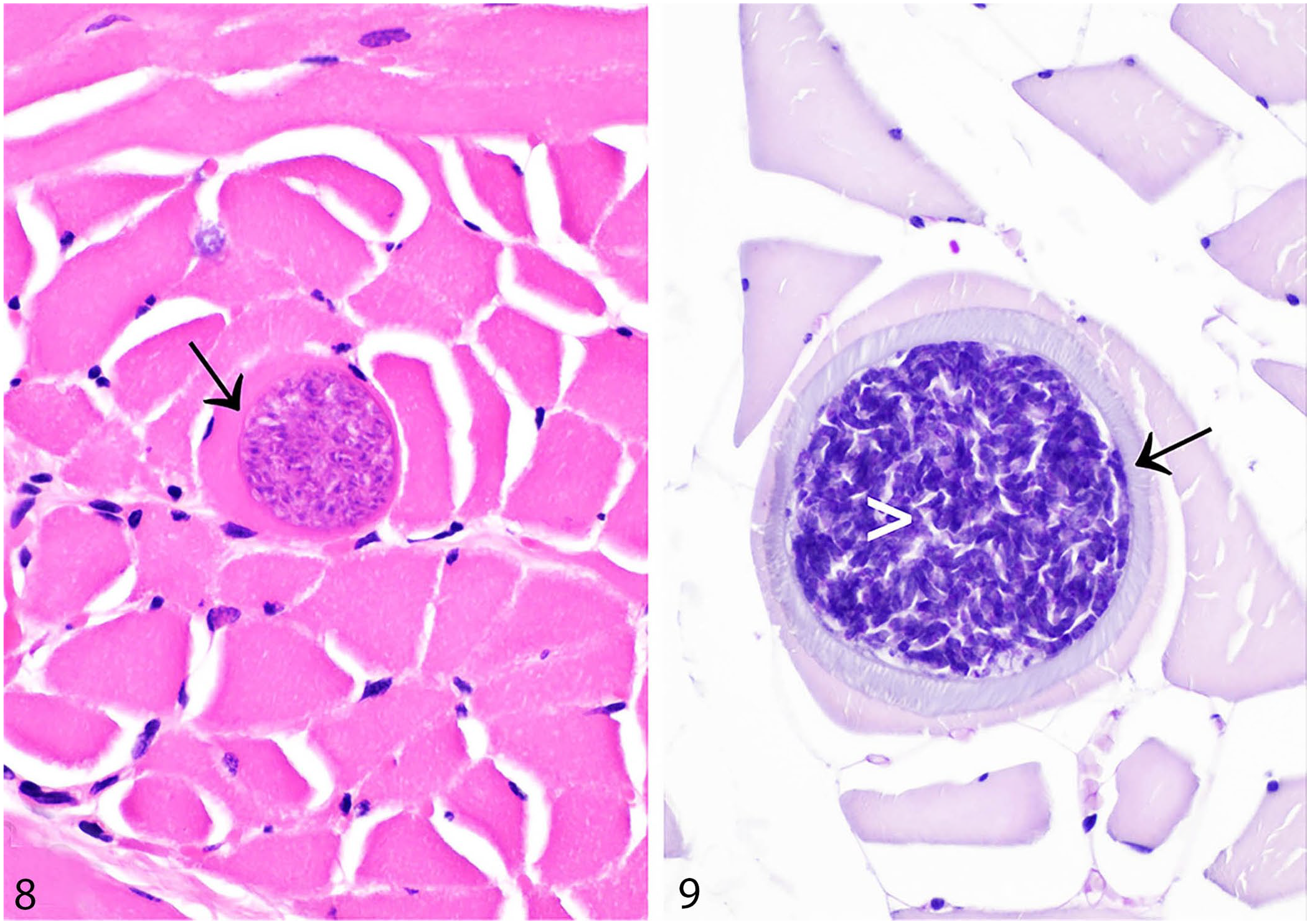
Sarcocystis spp. in skeletal muscles of rabbits and hares.
Humans infected with non-human Sarcocystis spp. (e.g., through consumption of undercooked game) can become aberrant intermediate hosts, or so-called “dead-end hosts,” with possible attendant clinical signs. However, the potential for this in the case of consumption of rabbit and hare Sarcocystis spp. cysts is, to my knowledge, poorly characterized. 55
Other protozoa
European rabbits may be infected with Trypanosoma nabiasi, a hemoflagellate protozoan. It is believed to have minimal effects on most hosts. 93 Hepatozoon spp. have been detected in 1 of 171 (0.6%) European brown hares from Croatia. 137 The flagellate Giardia duodenalis has been identified in wild, farmed, and pet European rabbits and wild Iberian hares.8,68,82,111
Microsporidia
Microsporidia are basic eukaryotic organisms that are obligate intracellular parasites. Their cellular structure is characterized by an absence of typical mitochondria, Golgi apparatus, and peroxisomes, and the presence of small, prokaryote-like, ribosomes. Microsporidia are phylogenetically closely related to fungi. 84
Encephalitozoon cuniculi
The European rabbit is the natural host for the zoonotic microsporidian parasite E. cuniculi, which is present worldwide and has a wide host range. The lifecycle is direct, with both horizontal and vertical (transplacental) transmission occurring in rabbits.84,138
In rabbits, postnatal infection usually occurs within 6 wk following transmission from an infected dam or other infected animal. Transmission is via ingestion or inhalation of spores that are present in the urine of infected rabbits in large numbers for 1–2 mo following infection. Spores are relatively resistant to environmental conditions, with survival times outside the host of up to 6 wk. By 3 mo post-infection, most spore dissemination is complete, although there may be future intermittent periods of shedding. 76
E. cuniculi spores infect cells via a polar tube or filament that extrudes during germination. Initial target organs for infection are highly vascularized organs, such as the lungs, liver, and kidney. 76 Nervous system involvement may occur at a more advanced disease stage, with a historic study demonstrating that brain lesions occur at least 8 wk after a positive antibody titer. 25 However, other authors detected E. cuniculi in the medulla oblongata, cerebellum, and leptomeninges at 2 wk post-infection. 67 These 2 findings are not necessarily contradictory as the latter study detected the organism using IHC, whereas the earlier study assessed lesions that would postdate the presence of organisms.
Once within a host cell, the infective sporoplasm forms a parasitophorous vacuole. It then matures into meronts that undergo an asexual reproduction phase called merogony. In a stepwise process, meronts differentiate into sporonts, and subsequently mature spores. This process is termed sporogony. Spores have a resistant spore wall and develop a polar tube. Eventually, when engorged with spores, the parasitophorous vacuole ruptures to release the spores, which can then disseminate locally and to more distant sites via the vasculature.76,84 In an immunocompetent rabbit, cell rupture is associated with an inflammatory response, and thus chronic, subclinical infections can ensue. 76
Serologic studies of healthy pet rabbits suggest that there may be relatively wide contact with the pathogen, with seroprevalence of 23–52% of animals in the United Kingdom, and 68% of animals in Italy.29,59,72 In a German study, 18% of animals with no clinical signs detected upon physical and neurologic examination were positive for E. cuniculi antibodies, with an equivalent figure of 48% for rabbits with clinical signs of disease. 61 Although it might be tempting to ascribe a role in the dissemination of infection to wild rabbits, we found that most of a cohort of U.K. wild rabbits did not have renal lesions consistent with E. cuniculi infection. 78 Similarly, a different U.K. population of wild rabbits did not have E. cuniculi antibodies. 18 This trend is seen in other countries as well. A cross-sectional assessment of wild European rabbits and Iberian hares from southern Spain did not detect E. cuniculi by PCR, 87 and wild lagomorphs examined from northern Spain did not have lesions consistent with encephalitozoonosis. 40 It is therefore important to appreciate that E. cuniculi has a broad host range and that other species may be important in infection dynamics.75,78,84,92
The pathology picture in cases of rabbits with encephalitozoonosis may be variable. Patients may be presented with polyuria and polydipsia, poor body condition, and inappetence, and have severe renal disease secondary to infection with the organism. In this case, the kidneys may appear macroscopically multifocally red and irregularly pitted, or may have minimal gross changes. Granulomatous or pyogranulomatous interstitial nephritis may be observed histologically. Organisms are small, round-to-oval, amphophilic-to-basophilic structures on H&E. Organisms may be detected in the tubular lumina, or in the surrounding interstitium. On H&E, organisms may be variably distinct, and detection of spores may be enhanced by use of additional histochemical stains. Chronic interstitial nephritis, together with renal fibrosis causing macroscopic sub-capsular indentations, may also be detected at autopsy. 108 These more chronic cases may also have attendant histologic neurologic lesions, and animals may become latent carriers.
Rabbits with neurologic lesions may have a history of subtle behavior change, a minor head tilt, or severe vestibular disease. 58 Lesions observed in the CNS may include perivascular cuffing, meningitis, and multifocal granulomatous encephalitis, with organisms visible (Fig. 10).25,80 A 6-point grading scheme has been described for brain lesions associated with encephalitozoonosis, reflecting the apparent level of organization and chronicity of the lesion. 80 In that study, the most frequently observed lesion was the type 4 lesion, comprising a necrotic center, with epithelioid macrophages, gemistocytic astrocytes, occasional multinucleate giant cells, and less prominent lymphocyte infiltration. 80 In rabbits infected with E. cuniculi, granulomatous infiltrates may also be observed in the liver, lungs, and heart. 80
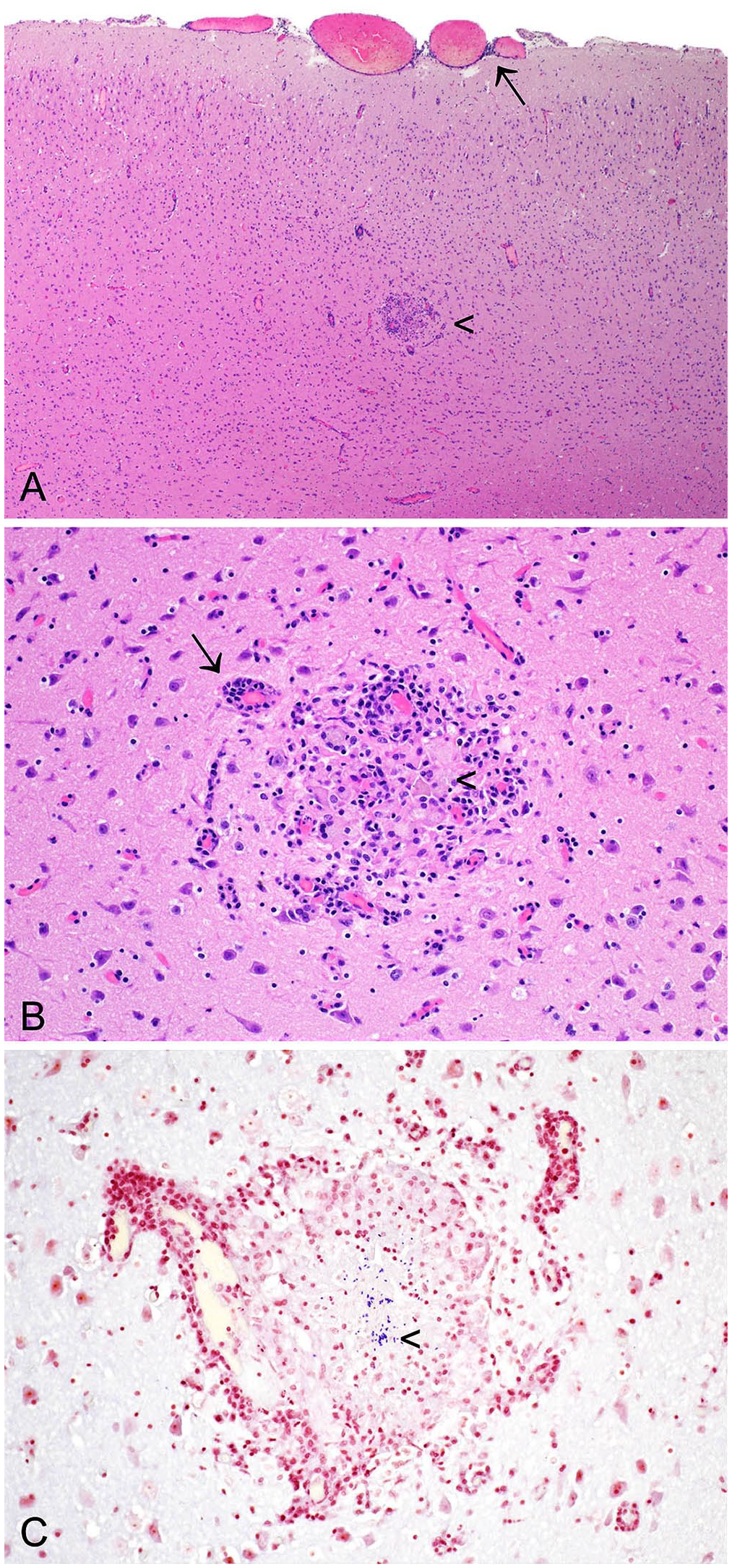
Encephalitozoon cuniculi in the cerebrum of a European rabbit.
Encephalitozoonosis may cause intraocular lesions, including cataracts, uveitis, and hypopyon. Intraocular lesions are typically unilateral. The pathogenesis is considered to be in utero (vertical) infection, with subsequent sporogony leading to lens rupture, and ensuing phacoclastic uveitis. 58 In these cases, spores may be identified in lens epithelial cells and macrophages using IHC. 52
Although the clinical history, and macro- and microscopic findings, may lead to a strong suspicion of encephalitozoonosis, spores may be subtle and hard to detect in H&E-stained sections. This difficulty is exacerbated by the presence of necrosis and inflammation. In a study comparing 14 histochemical stains for the detection of microsporidian spores, it was concluded that modified trichrome stain and Gram stain were optimal for use with light microscopy (Fig. 10C), and calcofluor white stain was beneficial for detection of spores by ultraviolet light microscopy. 113 IHC, if available, is likely to be more sensitive than histology for the detection of spores, and real-time PCR is likely even more sensitive.19,80 T. gondii is a differential for E. cuniculi histologically. 19 The morphology of T. gondii cysts and tachyzoites distinguishes them from the spores of E. cuniculi.
Although most consideration of E. cuniculi infection in rabbits and hares has focused on infection in European rabbits, PCR has demonstrated E. cuniculi DNA in eastern cottontails in northern Italy. 143
Encephalitozoon intestinalis
Encephalitozoon intestinalis (formerly Septata intestinalis) is the second most common human microsporidian and primarily infects enterocytes, but may also infect macrophages, fibroblasts, and endothelial cells.17,139 It causes diarrhea in both immunocompetent and immunocompromised hosts. E. intestinalis DNA has been amplified, by nested PCR, from 2.7% of fecal samples from rabbits from pet shops in Sichuan province, China. 27 Another study that also examined fecal samples, this time from wild European rabbits and Iberian hares from the Autonomous Region of Andalusia in southern Spain, detected DNA from E. intestinalis in 1 of 438 rabbits examined. 111 The microsporidian has also been documented in kidney from 1 wild rabbit and in 2 brains and 1 kidney from 3 wild Iberian hares, from southern Spain. The latter study did not include samples from the GI tract as it was focused on potential zoonotic transmission through consumption of organs from lagomorphs. 87
In future studies, it would be informative to examine histologic sections from positive lagomorphs, to attempt to correlate disease-associated lesions with amplification of microsporidian DNA.
Enterocytozoon bieneusi
Enterocytozoon bieneusi can infect enterocytes, biliary, pancreatic, and respiratory epithelial cells, as well as macrophages. 139 It is the most frequently diagnosed microsporidian infection in humans and is linked to cases of diarrhea that may be persistent in immunocompromised hosts. 83 Transmission is via the fecal-oral route. Although E. bieneusi DNA was not detected in fecal samples from wild European rabbits and Iberian hares from southern Spain, 111 E. bieneusi DNA was detected in kidney samples from 3 wild European rabbits sampled from the same region. 87 E. bieneusi DNA has also been identified in 15.4% of fecal samples from rabbits from pet shops in Sichuan province, China. 27 As with E. intestinalis, correlation of microsporidian genetic material with any lesions in target organs would aid in assessment of the significance of these findings.
Pentastomes
Class Pentastomida are arthropods. The adults reside in vertebrate upper respiratory passages and resemble segmented worms.
Linguatula serrata
Linguatula serrata adults infect the nasal cavities and sinuses of domestic carnivores and foxes. Adults are 1.5–2.0 cm long (males) to 8.5 cm long (females), tongue-shaped, and have transverse striations. Eggs are disseminated from the host via nasal secretions, or are swallowed and expelled in the feces. Herbivores, including rabbits and hares, can become infected via ingestion of eggs. In the intermediate host, larvae migrate through the intestinal wall and encyst in the viscera. Several molts ensue before the formation of infective nymphs that can infect the definitive host. In the carnivore definitive host, the nymphs migrate to the nasal cavity and mature into adult pentastomes. Disease is zoonotic and humans can be infected as either intermediate or definitive hosts. 47
L. serrata larvae were detected in 16 of 84 (19%) European brown hares from Macedonia, Greece, where they were present on the GI tract serosal surface. 28 Nymphs have also been characterized from European rabbits from Australia 12 and European brown hares from Romania. 69 Although pathology descriptions are somewhat limited, it appears that nymphs present in the lung may elicit a granulomatous response. 69 Research on L. serrata in wildlife is limited but is vital to understanding the disease ecology of this parasite. 47
Future perspectives
Accurate gross and microscopic identification of endoparasites in rabbits and hares is an important element of the diagnostic work of veterinary anatomic pathologists. There are also abundant opportunities for pathologists to contribute to research in this field. For example, studies detecting microsporidia in wild lagomorphs have capitalized on the availability of molecular techniques to identify microsporidian genetic material, but there is a need to clarify whether microsporidia such as E. intestinalis and E. bieneusi cause disease in these hosts. Furthermore, given the zoonotic nature of rabbit and hare endoparasites, such as C. hepatica, E. cuniculi, and others, veterinary pathologists are ideally poised to make valuable contributions to the understanding of the disease ecology of these pathogens in the context of a One Health research program.
Footnotes
Acknowledgements
As a veterinary anatomic pathologist with a keen interest in parasitology, I thank Emeritus Senior Lecturer in Veterinary Parasitology, Dr. Sheelagh Lloyd, Department of Veterinary Medicine, University of Cambridge, for her tutelage in the field of veterinary parasitology and particularly in the identification of parasites from rabbits and other small mammals. I also thank my collaborator Emeritus Professor of Conservation Biology, Diana Bell, University of East Anglia. I thank the citizen scientists who report sightings of deceased hares. I also thank Hugh Balmer, Paul Crompton, Yvonne Pratt, Mathew Rhodes, Andrea Starling, and Emma Ward of the Department of Veterinary Medicine, University of Cambridge, for excellent technical expertise in the preparation of tissue sections and for excellent assistance in the postmortem room.
Declaration of conflicting interests
Katherine Hughes is an Editorial Board Member for the Journal of Veterinary Diagnostic Investigation.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
