Abstract
Toxoplasma gondii infections are common in a range of mammalian and avian species, but clinical disease has been reported only rarely in domestic rabbits. Two cases of toxoplasmosis in domestic rabbits from the same premises were submitted to a diagnostic pathology facility in Athens, GA, USA. Both rabbits died after exhibiting clinical signs of gastrointestinal stasis. The gross findings observed in both rabbits comprised miliary, random, white-to-tan, necrotic foci throughout the spleen, liver, and lungs. Histologically, tachyzoites were observed within necrotizing inflammatory foci in the spleens of both rabbits, and in various other organs (tracheobronchial lymph node, lung, heart, and cecal appendix) of one rabbit. In both cases, the tachyzoites were immunoreactive with anti–Toxoplasma gondii antibodies. In addition, T. gondii DNA was detected via PCR and sequencing from a fresh lung sample from one rabbit and formalin-fixed, paraffin-embedded spleen, liver, femoral bone marrow, and haired skin from the second rabbit. Given that T. gondii can cause disease in domestic rabbits and is also a concern for other potential intermediate hosts (e.g., humans, other domestic animals), this parasite warrants consideration in the diagnostic evaluation of lagomorph tissues with compatible lesions.
Keywords
Toxoplasma gondii is an apicomplexan coccidian parasite that uses feline definitive hosts and a wide range of intermediate hosts including humans, other mammals, and birds. Within felids, T. gondii replicates sexually in intestinal epithelium, forming oocysts that are shed in feces. Within its intermediate hosts, T. gondii occurs as bradyzoites within tissue cysts or actively dividing zoites (i.e., tachyzoites) within various tissues. Pathogen transmission primarily occurs via the ingestion of infective oocysts in cat feces or via tissue cysts and zoites in contaminated, undercooked meat. 12 In humans and animals, most T. gondii infections are thought to be subclinical; clinical disease often arises secondary to immunosuppression and can be fatal or result in abortion and stillbirth.8,9
Based on seroprevalence data, 25–30% of humans worldwide are suspected to have been infected with T. gondii. 8 The wide host range of T. gondii and potentially severe consequences of infection render this pathogen a significant public and animal health risk, warranting ongoing vigilance by allied healthcare professionals in pathogen surveillance and diagnosis. Although the clinicopathologic manifestations of T. gondii infection in humans and most domestic animal species are well understood, literature specific to the lesion patterns of toxoplasmosis in lagomorphs is scarce.3,4,6 Given the increasing popularity of rabbits as pets and production animals, it is concerning that a clinical T. gondii infection could be overlooked by the veterinary diagnostician in favor of more commonly reported lagomorph pathogens that manifest similar lesions, such as Pasteurella multocida, Francisella tularensis, and Yersinia pseudotuberculosis. Here, we add to the existing pathology literature on T. gondii by critically evaluating the gross, microscopic, and immunohistochemistry (IHC) findings of clinical T. gondii infection in 2 domestic rabbits. The rabbits were submitted for autopsy through the Athens Veterinary Diagnostic Laboratory (AVDL) and the Infectious Diseases Laboratory’s Zoo and Exotic Animal Pathology Service (ZEAPS) of the University of Georgia (Athens, GA, USA).
Both animals originated from a group of 5, indoor, pet domestic rabbits and were presented moribund to a commercial veterinary clinic in John’s Creek, GA, USA. Rabbit 1 (1-y-old, spayed female) was dehydrated, pyrexic (40.3°C), and had a palpable, firm irregularity in the caudal abdominal organs. Abdominal radiographs revealed a small amount of ingesta within the stomach and scant contents within the cecum and colon. The animal died 1 d later despite supportive fluid treatment. Four days later, rabbit 2 (15-mo-old, castrated male) was presented with inappetence, abnormal mentation, and reduced ability to defecate. It exhibited marginal clinical improvement with supportive therapies (fluid treatment, injectable steroids, and antimicrobials), but died 3 d later. No additional clinical signs or mortalities were reported in the group of rabbits.
Autopsies for rabbits 1 and 2 were performed 7 d apart by the AVDL and ZEAPS, respectively. Both rabbits had random, 1–3-mm, flat, well-demarcated, white-to-tan foci throughout the spleen and liver (Fig. 1), both of which were mildly enlarged. The lungs were diffusely congested and edematous, and contained similar, 1–3-mm, well-demarcated, white foci (Fig. 1). Similar white-to-tan foci were scattered throughout the pancreas, peripancreatic adipose tissue, and duodenal mesentery (Fig. 2) in rabbit 1, and on the cut surface of an enlarged tracheobronchial lymph node in rabbit 2. Organ samples (brain, heart, lung, gastrointestinal tract, liver, spleen, kidney, pancreas, adrenal gland, abdominal adipose and mesentery, lymph node, bone marrow, skeletal muscle, haired skin) were collected, fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with H&E.
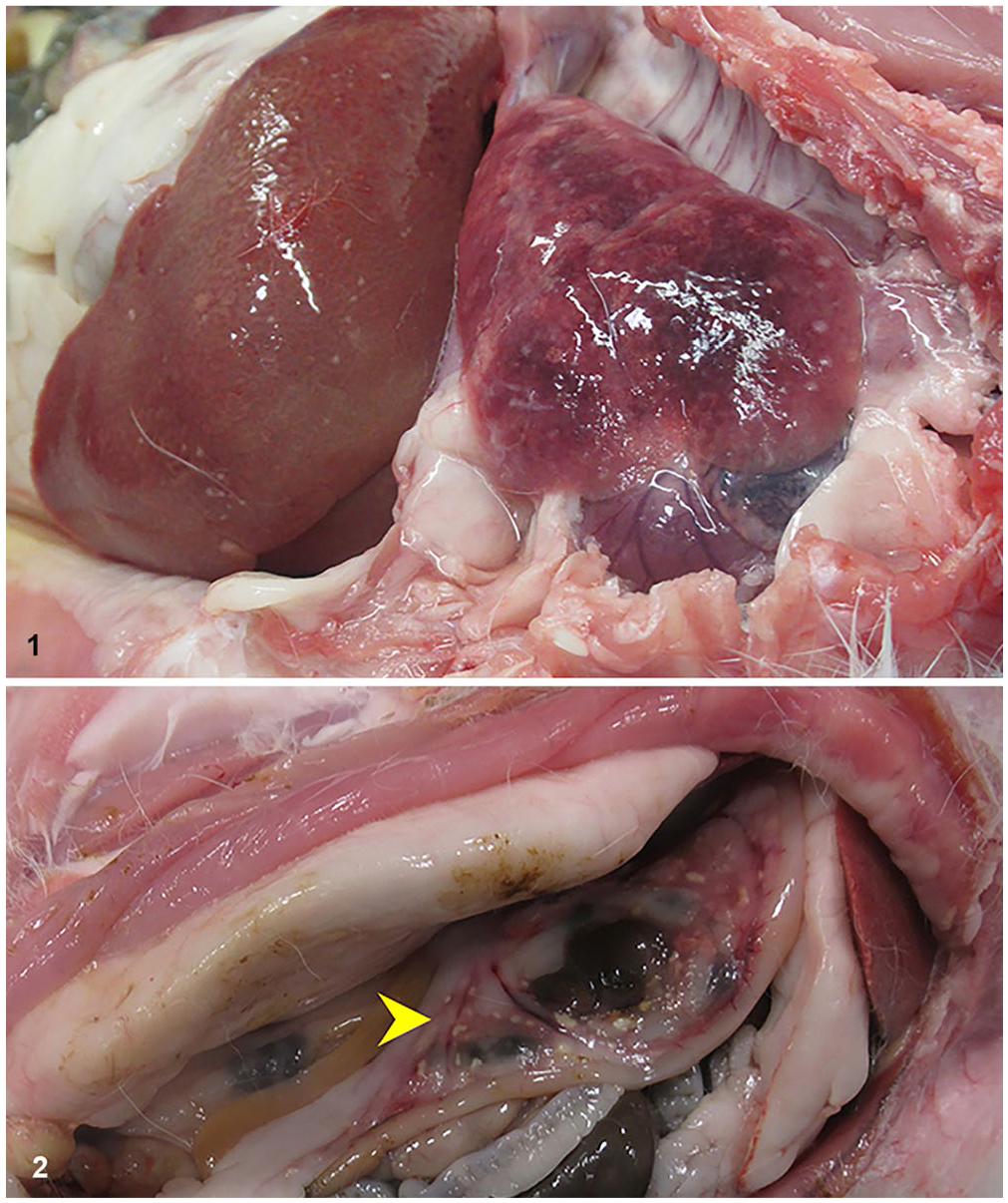
Gross lesions of toxoplasmosis in 2 domestic rabbits.
Histologic findings were similar for both rabbits and comprised either multifocal or coalescent, random, necrotizing inflammatory lesions involving the spleen (Fig. 3), liver (Fig. 4), lung, pancreas, peripancreatic adipose tissue (Fig. 5), and myocardium. Similar lesions were found in the tracheobronchial lymph node (Fig. 6), femoral bone marrow, adrenal gland, kidney, and skeletal muscle of the ventral abdomen of rabbit 2. Lesions were foci of lytic necrosis with cellular, karyorrhectic, and karyolytic debris interspersed with variable numbers of heterophils, macrophages, and lymphocytes. In addition, livers in both rabbits had multifocal, occasionally bridging, paracentral or centrilobular-to-midzonal coagulative necrosis characterized by cells that retained cellular outlines but had homogeneously eosinophilic cytoplasm and pyknotic or faded nuclei (Fig. 4). Within the lungs of both rabbits, the lytic necrosis involved widespread, random segments of alveolar septa and was part of the overall lesion pattern of acute interstitial pneumonia (Fig. 7), whereby septa were extensively thickened by numerous heterophils, fewer macrophages, and admixed fibrin interspersed with a few thrombosed septal blood vessels. Similar inflammatory cells often occupied alveoli, which varied from shrunken to collapsed. Apicomplexans in all tissues were scant-to-absent and were in 20–40-µm pseudocysts within inflamed areas containing scant-to-numerous, 1–2-µm, ovoid-to-crescentic, basophilic tachyzoites. In rabbit 2, pseudocysts were within macrophages in the spleen, tracheobronchial lymph node, alveoli (Fig. 7), cecal appendix and, infrequently, in type II pneumocytes and cardiomyocytes. In rabbit 1, pseudocysts were scarcer than in rabbit 2; 2 macrophages within 1 necrotic focus in the spleen of rabbit 1 contained pseudocysts; pseudocysts and free extracellular zoites were not observed in the other tissues.
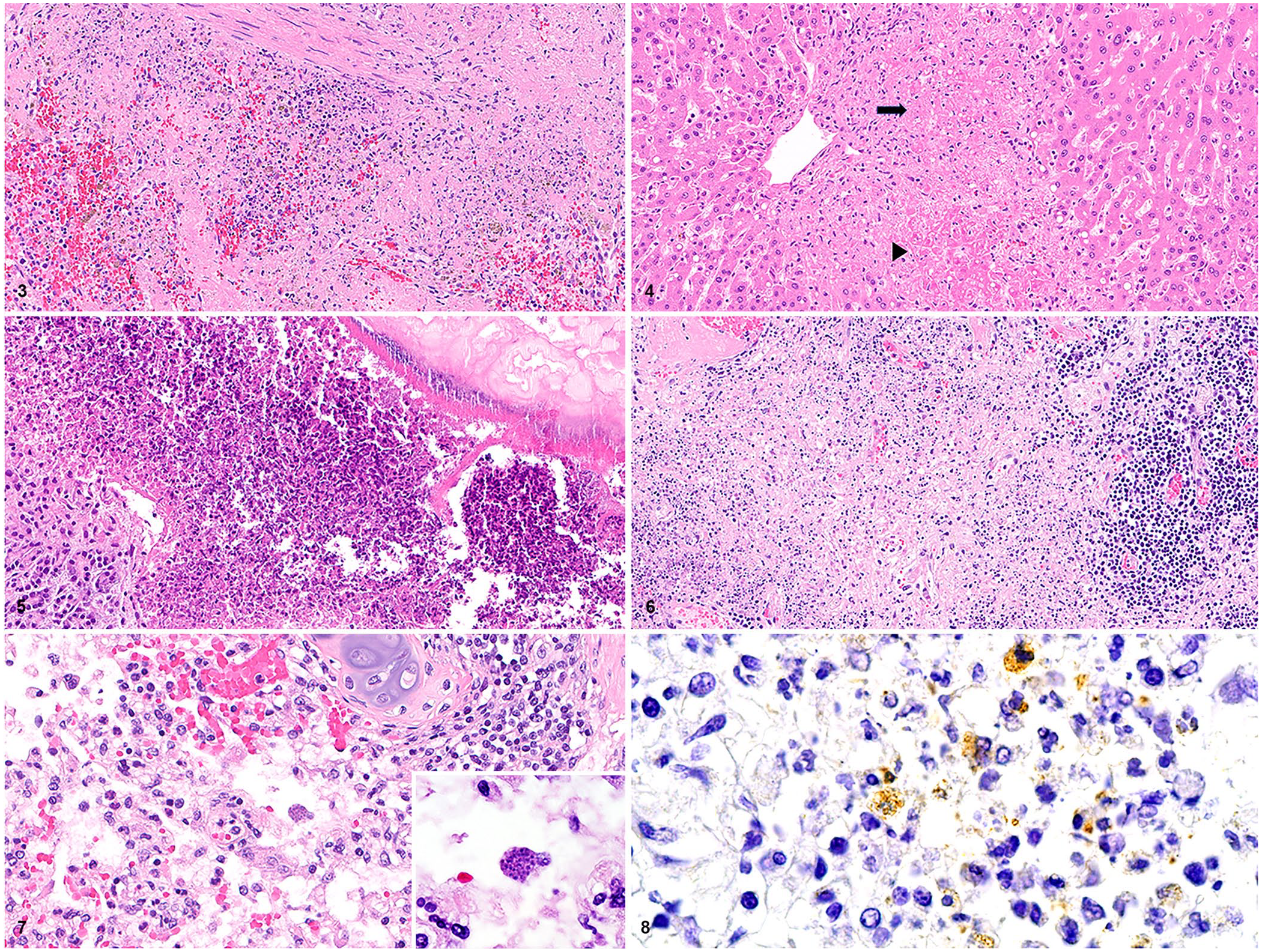
Microscopic, multi-organ lesions of toxoplasmosis in 2 domestic rabbits.
IHC with an anti–T. gondii antibody was performed on formalin-fixed, paraffin-embedded (FFPE) spleen and liver sections from rabbit 1 and on FFPE spleen, liver, lung, and tracheobronchial lymph node sections from rabbit 2. All FFPE sections were subjected to antigen retrieval induced by undiluted Protease 3 (0.24 casein U/mL; Ventana, Roche) at room temperature, a 10-min incubation with a feline polyclonal anti–T. gondii antibody (210-70-TOXO; VMRD) diluted 1:5,000 (rabbit 1) and 1:10,000 (rabbit 2), a subsequent streptavidin–biotin–horseradish peroxidase complex detection method, and immunohistochemical reaction with the 3,3′-diaminobenzidine chromogen. Overall, positive immunostaining of apicomplexans was rare and weak-to-moderate; scant, immunoreactive, intrahistiocytic tachyzoites were within the spleens of both rabbits and the tracheobronchial lymph node and lung (Fig. 8) of rabbit 2.
To confirm T. gondii infection, PCR was performed on scrolls of FFPE blocks (spleen, liver, femoral bone marrow, and haired skin; all on the same paraffin block) from rabbit 1 and a fresh lung sample from rabbit 2. Genomic DNA was extracted from the scrolls from rabbit 1 and lung tissue from rabbit 2 (QIAamp DNA FFPE tissue kit, DNeasy blood & tissue kit; Qiagen) according to the manufacturer’s protocols. The genomic DNA extract from rabbit 1 was subjected to a pan-apicomplexan PCR targeting the 18S rRNA gene 11 ; subsequently, amplicons were sequenced bi-directionally (Genewiz; Azenta), analyzed using Geneious Prime v.2019.0.4 (Biomatters) to generate a consensus sequence, trimmed of their primers, and analyzed by BLASTn (https://blast.ncbi.nlm.nih.gov/Blast.cgi). The genomic DNA extract from rabbit 2 was subjected to a T. gondii PCR assay using a standard quantitative PCR master mix (Qiagen) and primers targeting a repetitive 529-bp DNA fragment. 7 As was the case with rabbit 1, amplicons were sequenced bi-directionally (Genewiz), analyzed using Geneious Prime v.2019.2.3 to generate a consensus sequence, trimmed of their primers, and analyzed by BLASTn. The DNA sequences obtained from both rabbits were 100% identical to numerous T. gondii sequences in GenBank.
Ancillary assays were performed to rule out other pathogens that can cause lesions similar to those of systemic T. gondii infection. A F. tularensis PCR assay performed on liver (rabbits 1 and 2) and spleen (rabbit 2) was negative. 10 Fresh liver tissue from rabbit 1, submitted to the AVDL for rabbit hemorrhagic disease virus 2 (RHDV2; Caliciviridae, Rabbit hemorrhagic disease virus) PCR assay and subsequently transferred to the USDA–NVSL–Foreign Animal Disease Diagnostic Laboratory (Plum Island Animal Disease Center, Greenport, NY, USA) for RHDV1 PCR and RHDV2 PCR assays, was negative for RHDV. Bacteria did not grow in an aerobic culture of a liver sample from rabbit 1. Warthin–Starry, Ziehl–Neelsen, and Brenn and Brown Gram stains (rabbit 1) and a Brenn and Brown Gram stain (rabbit 2) targeting the histologic lesions did not highlight other infectious agents. During the diagnostic workup for both rabbits, systemic Y. pseudotuberculosis infection was considered based on the gross lesions, but subsequently deemed less likely given the absence of large colonies of gram-negative bacteria (characteristic of Y. pseudotuberculosis) within the histologic lesions.
The gross, microscopic, and IHC findings strongly implicate systemic toxoplasmosis as the cause of death in both rabbits; the widespread inflammation and necrosis are similar to previously described lagomorph toxoplasmosis cases.3,4,6 Grossly, the most consistent lesions were hepatosplenomegaly and pulmonary congestion and edema, with intraparenchymal necrotic foci in these and other sites including the pancreas, peripancreatic tissue (adipose and duodenal mesentery), and regional lymph node(s). The corresponding microscopic lesions most consistently included necrotizing hepatitis, splenitis, acute interstitial pneumonia, pancreatitis, peripancreatic steatitis, and lymphadenitis, although similar lesions were observed in several other sites to which T. gondii was presumed to have disseminated hematogenously. Although the inflammatory lesions were widespread and florid, the scant-to-absent tachyzoites in affected tissues differed from previous reports3,4 and necessitated additional IHC and molecular testing to confirm the diagnosis. Molecular testing was also instrumental in ruling out other potentially pathogenic apicomplexan protozoa (e.g., Sarcocystis spp.) that can have a morphology and lesion pattern similar to that of T. gondii in infected animals.
The gross and microscopic lesion patterns of toxoplasmosis can be similar to those of other, more common lagomorph infectious diseases. The latter include any potential cause of bacterial sepsis (e.g., pasteurellosis, salmonellosis), but also high-impact diseases such as rabbit hemorrhagic disease (RHD) and tularemia. In rabbit 1, initial interpretations from gross and microscopic evaluation had raised concerns for RHD and tularemia, both of which are state-reportable diseases. Moreover, F. tularensis is a Tier 1 biological select agent. As characteristic tachyzoites were microscopically inconspicuous in rabbit 1, a systemic infectious etiology was suspected but could not be confirmed. The subsequent postmortem diagnosis of T. gondii in rabbit 2 prompted re-evaluation, additional T. gondii–targeted testing, and, finally, confirmation of T. gondii infection in rabbit 1. Although all viable differentials for a given lesion pattern must be considered, T. gondii infections with a paucity of histologically discernible zoites may pose a diagnostic challenge to the pathologist unaware of the parasite’s disease-causing potential in lagomorphs or its broader gross and microscopic lesion patterns.
T. gondii infection can likewise pose a diagnostic challenge to clinicians. Although suggestive of a systemic disease process, the clinical signs in both rabbits (i.e., pyrexia, dehydration, obtunded mentation, diminished gastrointestinal tract motility) were nonspecific, mirroring the clinical presentation of T. gondii infection as has been reported in other domestic animal species. 2 Considerations that might raise clinical suspicion for T. gondii involvement include recent seroconversion, 5 potential exposure to cat feces, and, as with our cases, a definitive postmortem diagnosis in a related animal.
The rabbits were indoor pets housed in a room with no direct contact with cats or feline excrement, thus implicating an indirect transmission route via fecal-contaminated fomites and/or feed from outside sources. Interestingly, clinical signs and mortality were not reported in the other 3 rabbits within this group despite similar husbandry and food sources. Subclinical infection remains a possibility in this and other infected lagomorph populations; a study of T. gondii seroprevalence in domestic rabbits in Durango State, Mexico, reported a seroprevalence of 16.3% in 423 clinically healthy animals. 1 Stress and/or immunosuppression could predispose subclinically infected animals to clinical disease; however, no overt cause of immunosuppression was identified in rabbits 1 and 2.
Footnotes
Acknowledgements
We thank Dr. Elizabeth Howerth and Nicole Young for furnishing the information on the anti–T. gondii antibody IHC protocol, and Infectious Diseases Laboratory staff for performing the T. gondii PCR and genomic sequencing for rabbit 2. We thank the histology and laboratory technicians and staff of the Department of Pathology and AVDL for their assistance with slide processing and ancillary diagnostic testing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
