Abstract
Mannheimia granulomatis was first isolated from pneumonic European hares in the 1980s and has since been reported sporadically in pneumonic Swedish roe deer and Australian cattle. Although the pneumonic lesions caused by M. haemolytica in livestock have been extensively studied and reported, little is published with regard to the pneumonic lesions associated with M. granulomatis infection in any species. We describe the histopathology of purulent bronchopneumonia associated with M. granulomatis in a Belgian hare (Lepus europaeus) resident in British Columbia, Canada, and compare the lesions with those caused by M. haemolytica in livestock.
Mannheimia granulomatis is a gram-negative, non-motile, small rod or coccobacillus. 2 The bacterium was initially isolated in the 1980s from European captive and wild brown hares with purulent bronchopneumonia and conjunctivitis, and classified on a phenotypic basis as Pasteurella haemolytica Bisgaard taxon 20, a member of the P. haemolytica complex. 9 At the same time, bacteria with similar phenotypic characteristics were isolated from chronic progressive subcuticular granulomas on the neck, chest, and shoulders of Brazilian cattle and classified as P. granulomatis, another member of the P. haemolytica complex. 15 In 1999, a polyphasic molecular investigation of the taxonomy of all bacteria classified within the P. haemolytica complex led to the proposal for a new genus, Mannheimia. 2 Five species were proposed based on phylogenetic relationships, M. haemolytica, M. glucosida, M. ruminalis, M. granulomatis, and M. varigena. 2 P. haemolytica taxon 20 and P. granulomatis were reclassified as Mannheimia granulomatis. 2
Since the reclassification, M. granulomatis has been isolated occasionally in other situations. The bacterium was isolated from a tongue abscess, a jaw abscess, and purulent bronchopneumonia in 3 Australian cattle, 3 from the mammary gland of an Israeli cow with subclinical mastitis, 4 and from glossitis, stomatitis, keratoconjunctivitis, purulent pneumonia, pleuritis, and a neck abscess in Swedish roe deer. 5 Although purulent bronchopneumonia associated with M. granulomatis infection has been reported in European hares, 9 European roe deer, 5 and Australian cattle, 3 the histopathology has not been described previously, to our knowledge. We describe bronchopneumonia associated with M. granulomatis infection in a Belgian hare (Lepus europaeus) resident in British Columbia, Canada.
In November 2014, a 4-mo-old Belgian hare, raised in a rabbitry, developed noisy breathing, as described by the owners. The noisy breathing persisted for 6 wk without any evidence of sneezing, coughing, or nasal or ocular exudates. The hare maintained a normal appetite and activity. None of the littermates were affected. In January 2015, the hare developed respiratory distress and an elevated respiratory rate that did not respond to 72 h of antibiotic treatment. The hare was euthanized because of the poor prognosis and was presented to the Animal Health Centre in Abbotsford, British Columbia for autopsy.
At autopsy, the hare was in fair-to-good body condition with a bodyweight of 2.9 kg. Abnormal findings were restricted to the lungs. There was bilateral, generalized, mottled, tan-pink discoloration of the pleural and cut surface of the cranioventral lung lobes. Thick pale yellow purulent material ran from large airways on cut surface. The discolored lung lobes were firm and sank in formalin. No other gross abnormalities were noted. Organs were fixed in 10% buffered formalin, routinely processed, and stained with hematoxylin and eosin for histologic examination.
Lung, liver, and spleen were cultured. Using aseptic technique, all tissues were inoculated onto Columbia blood agar with 5% sheep blood (Oxoid, Nepean, Ontario, Canada) and MacConkey agar (Oxoid), and incubated at 35°C ± 2°C in 5–10% CO2 and aerobic conditions, respectively. Agar plates were observed at 24 h and 48 h for bacterial growth. All bacterial isolates were subcultured to obtain a pure isolate for identification purposes. Bacterial identifications were based on colony morphology, growth characteristics, Gram stain, and biochemical testing, as well as molecular techniques.
Virus isolation was attempted by 2 blind passages in African green monkey kidney epithelial (Vero) cells, Madin–Darby kidney cells (MDKC), and rabbit kidney (RK-13) cells. Briefly, 10% w/v lung tissue homogenate was prepared in minimum essential medium (Sigma-Aldrich, St. Louis, MO), supplemented with 0.1% cycloheximide, gentamicin, and amphotericin B (Sigma-Aldrich), inoculated into Vero, MDKC, and RK-13 cell monolayers, and evaluated for cytopathic effect (CPE) every second day. At 7 d post-inoculation, cultures were blind-passaged onto fresh cell monolayers and observed for CPE for an additional 7 d.
Nucleic acids for polymerase chain reaction (PCR) assays were extracted from lung tissue following the manufacturer’s instructions (Qiagen, Toronto, ON, Canada). Conventional PCR was performed using a 25-µL reaction–containing bead (GE Healthcare, Piscataway, NJ) on a thermocycler (Bio-Rad, Mississauga, ON, Canada). For Toxoplasma gondii, PCR was performed using a forward primer (5’-GGAACTGCATCCGTTCATGAG-3’) and reverse primer (5’-TCTTTAAAGCGTTCGTGGTC-3’) that target the B1 gene. 13 For mycoplasmas, a hemi-nested PCR was performed that targets the 16S ribosomal (r)RNA gene of mycoplasmas.19,20 PCR products were electrophoresed on 2% agarose gels, stained with ethidium bromide, and analyzed using an ultraviolet photodocumentation system (AlphaImager HP Imaging System, ProteinSimple, Santa Clara, CA).
On bacterial culture, a pure, light growth of a clear, non-hemolytic organism on Columbia blood agar with 5% sheep blood was isolated from the lung. The organism was identified as a gram-negative, oxidase- and indole-negative coccobacillus. Biochemical testing (Biolog, Hayward, CA) produced an identification of M. granulomatis. Confirmation of the identification was performed using 16S rRNA sequencing. Nucleic acids were extracted from a well-isolated colony of the organism following the manufacturer’s protocol (Qiagen). The 16S gene was amplified using a forward primer (5’-AGAGTTTGATCMTGGCTCAG-3’) and reverse primer (5’-GWATTACCGCGGCKGCTG-3’) to obtain a 492-bp product. 11 The product was sequenced and compared to known sequences in GenBank, which confirmed the biochemical identification of M. granulomatis. No bacteria were cultured from the liver and spleen. Molecular tests for mycoplasmas and T. gondii were negative, and no virus was isolated from cell culture.
On histologic examination, there was multifocal-to-generalized consolidation and congestion of the lung characterized by large numbers of macrophages with neutrophils and edema fluid within alveoli. There was similar exudate in bronchiolar lumina, some of which were distended and occasionally exhibited foci of epithelial attenuation and/or erosion. Type 2 pneumocyte hyperplasia (Fig. 1), Langhans, epithelial syncytial, and foreign-body type multinucleate cells (Fig. 2), and scattered small foci of mineralized material were observed in association with the largest aggregates of inflammatory cells within alveoli. There was mild-to-moderate multifocal perivascular, peribronchiolar, and occasionally subpleural mixed mononuclear inflammation dominated by plasma cells and lymphocytes. Bronchial-associated lymphoid tissue was moderately hyperplastic in areas of greatest inflammation (Fig. 1). Plant material was occasionally intermixed with exudate in a few bronchioles and alveoli (Fig. 1). Less-affected parenchyma exhibited alveolar septal thickening and hypercellularity with margination of intravascular leukocytes and scattered areas of intermixed partial atelectasis and alveolar overdistension. Purulent bronchopneumonia associated with primary M. granulomatis infection was diagnosed in the Belgian hare based on histologic examination, aerobic bacterial culture, and negative test results for mycoplasmas, T. gondii, and viruses.
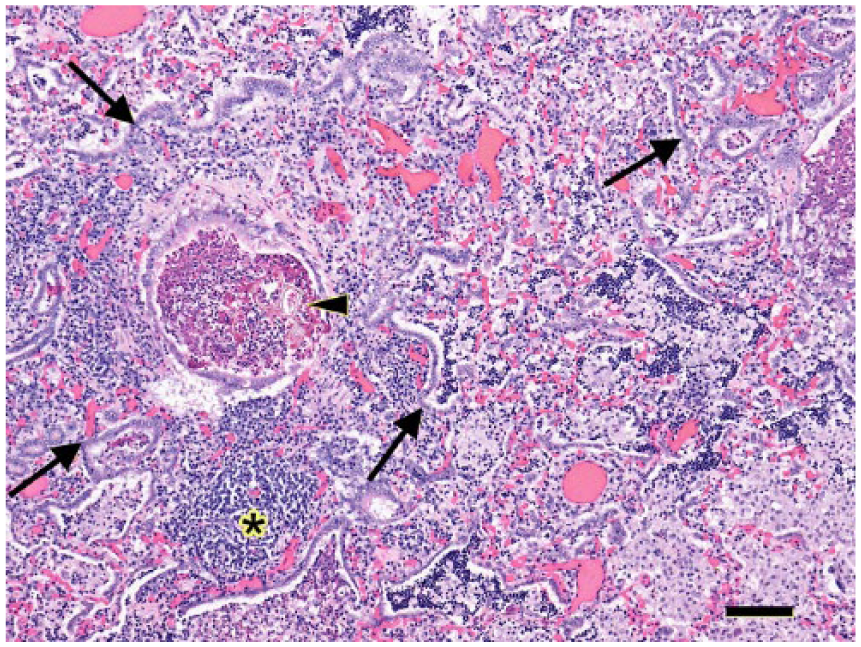
Plant material is present in a bronchiolar lumen (arrowhead) in the pneumonic lung of a Belgian hare (Lepus europaeus). Widespread hyperplasia of type 2 pneumocytes is evident throughout the field (arrows). Bronchial-associated lymphoid tissue was hyperplastic (asterisk). H&E. Bar = 100 μm.
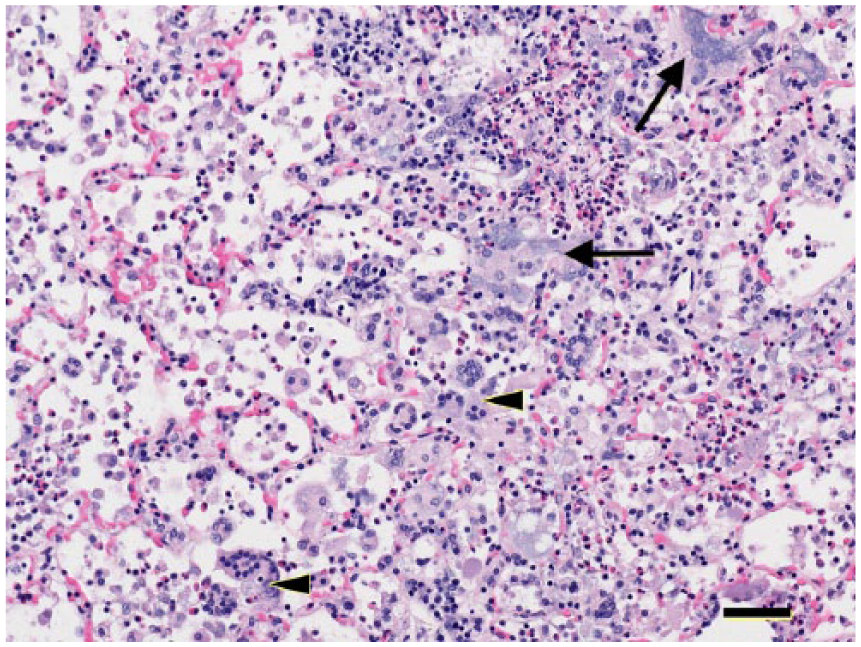
Clusters of Langhans, foreign-body type (arrowheads), and epithelial syncytial multinucleate (arrows) cells are scattered in this field in the pneumonic lung of a Belgian hare (Lepus europaeus). H&E. Bar = 50 μm.
Within the genus Mannheimia, the lesions caused by, and virulence of, M. haemolytica have been most widely studied and documented. M. haemolytica is a commensal of the ruminant nasopharynx and tonsil, which under adverse conditions can multiply and enter the lungs.8,18 Concomitant infection with respiratory viruses and/or stress can impair pulmonary defense mechanisms and facilitate bacterial colonization of the lung leading to pneumonia.8,18 Clinical disease is mediated by several virulence factors and characterized by infiltration of alveoli by large numbers of neutrophils and macrophages with heavy fibrin exudation, foci of coagulative necrosis, and presence of necrotic leukocytes. 8 Necrotic leukocytes are associated with secretion of high concentrations of leukotoxin during the log phase of bacterial growth. 8 By-products of leukocyte necrosis further damage pulmonary parenchyma. 18
Leukotoxin is a member of the family of RTX (repeats in toxin) exotoxins produced by gram-negative bacteria, which have a wide variety of dose-dependent effects on target cells. 12 Although numerous bacteria produce genetically related RTX toxins, target cell specificity differs significantly. 18 Ruminant leukocytes are the primary target of leukotoxin secreted by M. haemolytica. 12 Different strains of M. haemolytica express different amounts of leukotoxin with differing activity, and thus pathogenicity will vary. 18 In calves with bronchopneumonia caused by Mannheimia species cluster V, leukotoxin is expressed, and the lesion is characterized by fibrinopurulent inflammation with necrotic leukocytes and prominent hemorrhage but without areas of coagulative necrosis. 6
Lipopolysaccharide (LPS) is another important virulence factor of M. haemolytica that triggers dose-dependent damage via vasodilation, vascular leakage, the production of pro-inflammatory cytokines, and other bioactive compounds by leukocytes, complement activation, activation of the coagulation cascade, and direct cell damage. 18 LPS acts synergistically with leukotoxin to stimulate alveolar macrophages to recruit neutrophils, which then degranulate, produce oxidative compounds, and undergo lysis. 18 LPS can also produce focal areas of coagulative necrosis; the mechanism is poorly understood but is thought to be the result of thrombosis and/or the direct effects of bacterial toxins or leukocyte activity on lung tissue. 8
The pulmonary lesions in the Belgian hare in our report lack the areas of coagulative necrosis, heavy fibrin exudation, and necrotic leukocytes associated with leukotoxin activity caused by M. haemolytica in ruminants. Given that the lkt gene has been demonstrated to be present in M. granulomatis, 5 the absence of leukotoxin-associated lesions in the hare described herein suggests that either the gene was not expressed, a sufficient concentration of leukotoxin was not achieved, or that leporine leukocytes are not target cells of the toxin. Given that the culture was non-hemolytic and hemolysis is highly correlated with activity of the lkt gene, it is most likely that the gene was not expressed in this case. 12
Although the Belgian hare lacked lesions caused by leukotoxin, numerous multinucleate cells were present. These cells are not described in pneumonia caused by either M. haemolytica or M. species cluster V in cattle.6,8 However, multinucleate cells are described with pneumonic pasteurellosis caused by Pasteurella multocida in rabbits, 14 suggesting that they may be a species-specific response of rabbits with bacterial bronchopneumonia. Alternatively, the multinucleate cells may represent a chronic inflammatory response to the presence of M. granulomatis. Although epithelioid macrophages were described encircling granulomas and abscesses in the chronic fibrogranulomatous panniculitis (lechiguana) of Brazilian cattle caused by M. granulomatis, Langhans, foreign-body, and epithelial syncytial multinucleate cells were not described. 16
Plant material was observed in the pneumonic lung of this hare and the foreign-body type multinucleate cells may have developed in response to the presence of this plant material. Although not extensive, the material was present in a few scattered airways and alveoli. No bacteria or alteration in the overall character of the inflammation was noted in association with the plant material, suggesting that it was not a recent event superimposed on a pre-existing pneumonia. Most Mannheimia spp. are commensals of mucous membranes in animals, being particularly prevalent in cattle and sheep. 3 If M. granulomatis is a commensal of the leporine oropharynx, aspiration may have introduced the bacterium to the lower respiratory tract and triggered the pneumonia.
Hyperplasia of type 2 pneumocytes is indicative of pneumocyte injury commonly observed with lentiviral infection in small ruminants, 8 bovine respiratory syncytial virus infection in cattle, 17 horses with multinodular pulmonary fibrosis associated with equid herpesvirus 5 infection, 7 and Mycoplasma ovipneumoniae infection of muskox. 10 Hyperplasia of type 2 pneumocytes was observed in the Belgian hare, but without any evidence of viral or mycoplasma infection. Thus, this lesion most likely represents pneumocyte injury, either caused directly by M. granulomatis or indirectly by chronic inflammation. Hyperplasia of type 2 pneumocytes is reported following experimental induction of P. multocida bronchopneumonia in rabbits, 1 suggesting that, similar to the multinucleate cellular response, this may be a leporine-specific response to chronic bacterial bronchopneumonia. M. granulomatis appears to be a significant respiratory pathogen in leporids and should be considered in the differential diagnosis of hares and rabbits with purulent bronchopneumonia.
Footnotes
Acknowledgements
We recognize the technical expertise of Sandra Etheridge, Joanne Taylor, Fiona Downer, Kristen Younie, Jaime Osei-Oppiah, Giselle Hughes, Erin Graham, Melissa Trapp, and Petra Szathmary.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
