Abstract
A 14-y-old intact female llama (Lama glama) was presented for evaluation of a right maxillary swelling of 3-mo duration. Clinically, the animal had mild nasal discharge, abnormal retropulsion of the right eye, and moderate gingival disease. An incisional biopsy of the maxillary mass revealed pleomorphic and mitotically active neoplastic spindle-to-stellate cells organized in haphazard lacunae embedded in abundant chondroid matrix. Given the poor prognosis, euthanasia was elected. Postmortem examination and sectioning of the head exposed a large solid, white, firm mass that vastly expanded the right infraorbital region, extending to the maxilla, effacing the right nasal conchae and ipsilateral zygomatic bone. Collectively, postmortem dissection, cytology, and histopathology of the primary mass supported a diagnosis of sinonasal chondrosarcoma. To our knowledge, this entity had not been reported previously in this species and should be considered a differential for facial deformities in New World camelids.
A 14-y-old intact female llama (Lama glama) was admitted to the Auburn University Large Animal Teaching Hospital (Auburn, AL, USA) for evaluation of a right maxillary swelling of 3-mo duration. Clinically, the animal had mild nasal discharge, abnormal retropulsion of the right eye, moderate gingival disease, and abscessation around the upper right third molar tooth. No hair loss, heat, or draining tracts were noted on examination of the swelling at clinical admission. A CBC revealed mild leukocytosis with mature neutrophilia and mild monocytosis. Radiographs demonstrated severe maxillary bone destruction and involvement of 3 molar tooth roots. Sonographic findings of the facial mass included mixed echogenicity with evidence of gas, roughened maxillary bone, and involvement of the ventral orbit. A fine-needle aspirate was consistent with infection, and an incisional biopsy was obtained.
Histologically, the biopsy sample contained a dense population of spindle-to-stellate cells arranged in sparse loose streams and bundles, and haphazard lacunae embedded in abundant myxomatous-to-chondroid matrix. Neoplastic cells had indistinct cell borders and small amounts of pale eosinophilic wispy-to-flocculent cytoplasm. Nuclei were round-to-elongate with finely to coarsely stippled chromatin and variably prominent nucleoli. Anisocytosis and anisokaryosis were marked, with frequent multinucleation and 14 mitotic figures in 10 standardized 400× fields (2.37 mm2). Small foci of coagulative necrosis and hemorrhage were throughout the neoplasm. These changes were consistent with chondrosarcoma. Given the poor prognosis, the owners opted for euthanasia, followed by postmortem examination.
Grossly, a 6 × 14 × 9-cm mass markedly expanded the right infraorbital region and maxilla (Fig. 1A). A cross-section at the level of 3rd–4th molar teeth exposed a solid, tan, firm mass with multifocal areas of lytic necrosis that completely occluded the right nasal passages and effaced the right maxillary sinus, nasal conchae, and adjacent zygomatic bone (osteonecrosis; Fig. 1B). No metastasis was detected. Postmortem impression smears and scraping cytology showed individual or aggregated mesenchymal cells with marked anisocytosis, anisokaryosis, and occasional binucleation. The cells were surrounded by abundant pink fibrillary extracellular matrix. Histologically, the facial mass was similar to the incisional biopsy (Fig. 1C, 1D). Postmortem dissection, cytology, and histopathology of the primary mass were diagnostic for sinonasal chondrosarcoma.
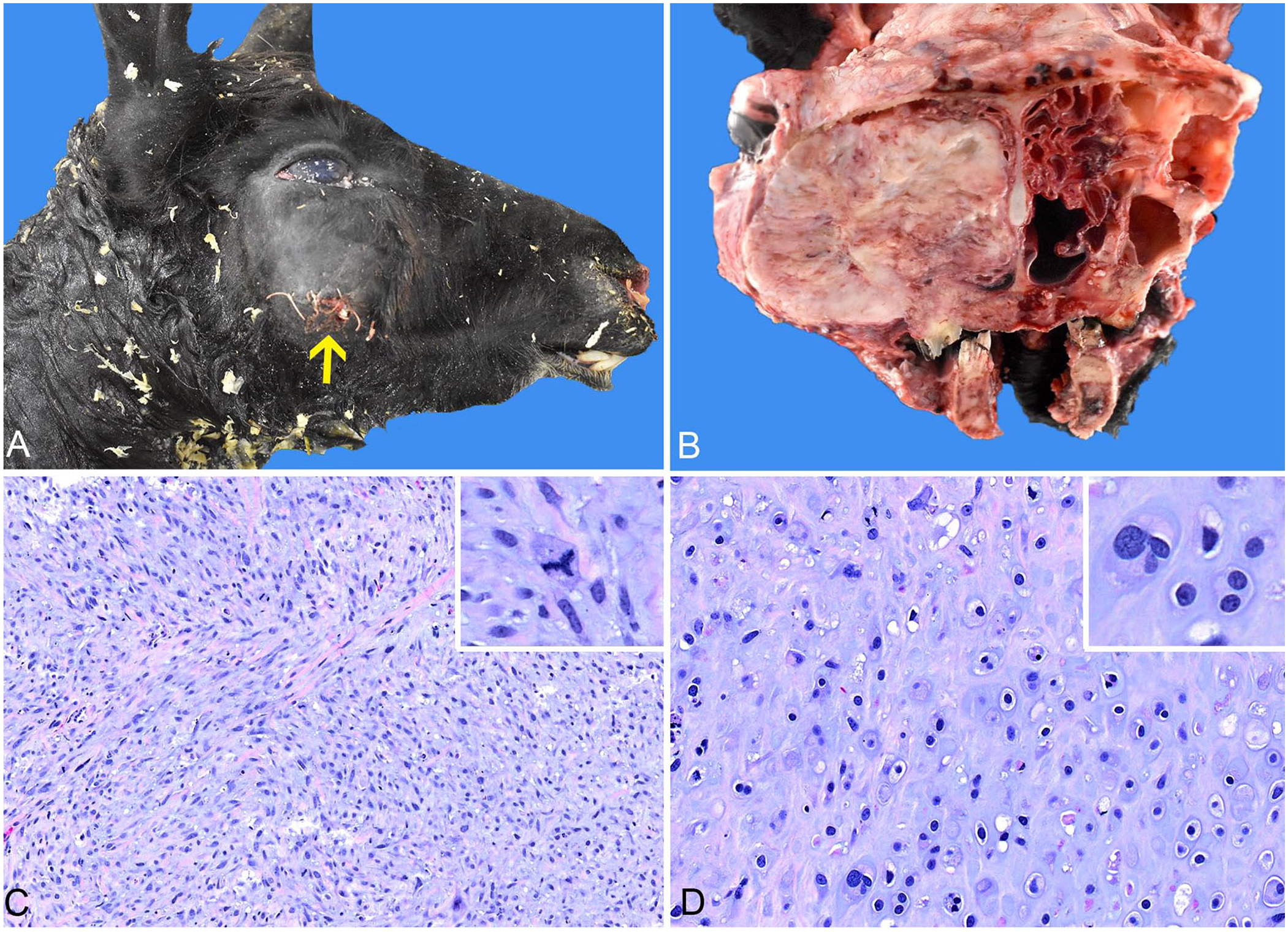
Sinonasal chondrosarcoma in a llama.
South American camelids (SACs; Camelidae) consist of Llama and Vicugna genera, including alpacas (Vicugna pacos), llamas, guanacos (L. guanicoe), and vicuñas (V. vicugna). 8 In North America, the uses of llamas include breeding, showing, packing, companion, guarding, and fiber. 8 Even though literature about the pathology of SACs has become progressively available to the veterinary community, it is still modest, counting on selected resources, retrospective studies, and isolated case reports.
Given their relatively small and narrow nasal passages, the airflow in camelids is restricted, and diagnostic procedures, such as endoscopy and intubation, can be challenging. 4 In addition, these animals are considered obligate nasal breathers; hence, diseases of the upper respiratory tract can lead to severe dysfunction and potentially death.4,6 Assorted common upper respiratory diseases of camelids include pharyngeal collapse, malformations (choanal atresia, wry face, cleft palate), and rhinosinusitis from bacterial infection.4,6,9 Bot flies have been reported as a cause of localized upper airway swelling and respiratory signs, such as sneezing, nasal discharge, stridor, and reduced airflow.7,10 Cephenemyia spp. (deer nasopharyngeal bots) and Oestrus ovis (small ruminant sinonasal bots) are the identified species; Cephenemyia spp. are associated with more pronounced granulomatous inflammation in SACs given that SACs are aberrant hosts. 7 In such cases, endoscopy is often diagnostic, and treatment with anthelmintics is successful.7,10 One case of Aspergillus spp.–like hyphae eliciting severe granulomatous rhinitis, sinusitis, and osteomyelitis occurred as a large soft-tissue mass causing unilateral exophthalmos, deviation of the ear, and destruction of several facial bones, mimicking a neoplastic growth; diagnosis was only reached with postmortem examination. 3
Inflammatory extension from tooth root abscesses is also common in SACs and is typically the result of direct trauma from rough or stem-filled forages during tooth eruption, but fractures, trimming, decay, or periodontal disease can also predispose to local abscessation. 6 Affected animals often have painful oral swelling, draining tracts, altered mastication, and malodorous sinus discharge, which are clinical signs familiar to SAC veterinarians. 6 Given that tooth root abscesses, especially at the premolar and molar teeth, are common in llamas and alpacas, osteomyelitis was also a clinical differential in our case. Other considerations for this expansile lesion were abscessation, granulomatous or pyogranulomatous inflammation (foreign body, actinobacillosis, actinomycosis), and neoplasms, such as fibroma, sarcoma, lymphoma, squamous cell carcinoma, and adenocarcinoma.
In a span of 19 y (2004–2023), 272 camelid cases were submitted to our Pathology laboratory [45 (17%) biopsies; 227 (83%) autopsies], mostly from alpacas (70%). Of all of the cases that required surgical biopsy or had postmortem examination performed, only in 2 (0.7%) individuals (both alpacas) was there a primary concern for upper respiratory tract disease with nasal or maxillary soft-tissue swellings. These animals were ultimately diagnosed with bacterial necrosuppurative or fibrinonecrotizing rhinitis and intra-lesional foreign plant material, representing the common inflammatory and septic entities affecting this anatomic site in camelids.
Reported neoplasms directly involving the nasal cavity of camelids include sarcoma of soft tissues and fibrosarcoma of the nasal septum.1,4 In addition, tumors arising from the upper alimentary system can also lead to the obliteration of adjacent air passages, such as oral squamous cell carcinoma, odontogenic neoplasm, and ossifying fibroma. In such instances, affected animals can develop significant clinical respiratory signs such as nasal discharge, facial distortion, and reduced airflow. 4
Although neoplasia has been reported in llamas and alpacas, neoplasia accounted for <10% of all camelid cases received in a North American reference diagnostic laboratory. 1 Disseminated lymphoma was the most frequent neoplasm, followed by squamous cell carcinoma, fibroma, and adenocarcinoma. 1 Mesenchymal neoplasms were not as prevalent, and benign entities outnumbered malignant tumors, most of which were unspecified sarcomas on the lip, nose, urethra, trachea, pancreas, third compartment, and elbow. 1 The diagnosed sarcomas retrospectively recorded in llamas and alpacas include fibrosarcoma, leiomyosarcoma, hemangiosarcoma, osteosarcoma, myxosarcoma, and liposarcoma, affecting the subcutis, mucocutaneous sites, abdominal viscera, maxilla, jaw, or humerus.1,11
In our institution, we collected 15 records from 2004 to 2023 (~6% of total camelid samples) of incidental or clinically relevant neoplasms, including our case (Table 1). Cases diagnosed at our laboratory reflected the most prevalent entities reported to affect SACs, such as lymphoma and alimentary squamous cell carcinoma, or included similar diagnoses listed in the veterinary literature.1,11 Although distal appendicular and mandibular chondrosarcomas have been documented in camels (Camelus dromedarius), 5 chondrosarcoma was not listed in a retrospective study of neoplasms in SACs. 1
Incidental and clinically significant neoplasms in South American camelids diagnosed histologically at Auburn University, 2004–2023 (15 of 272 camelid cases).
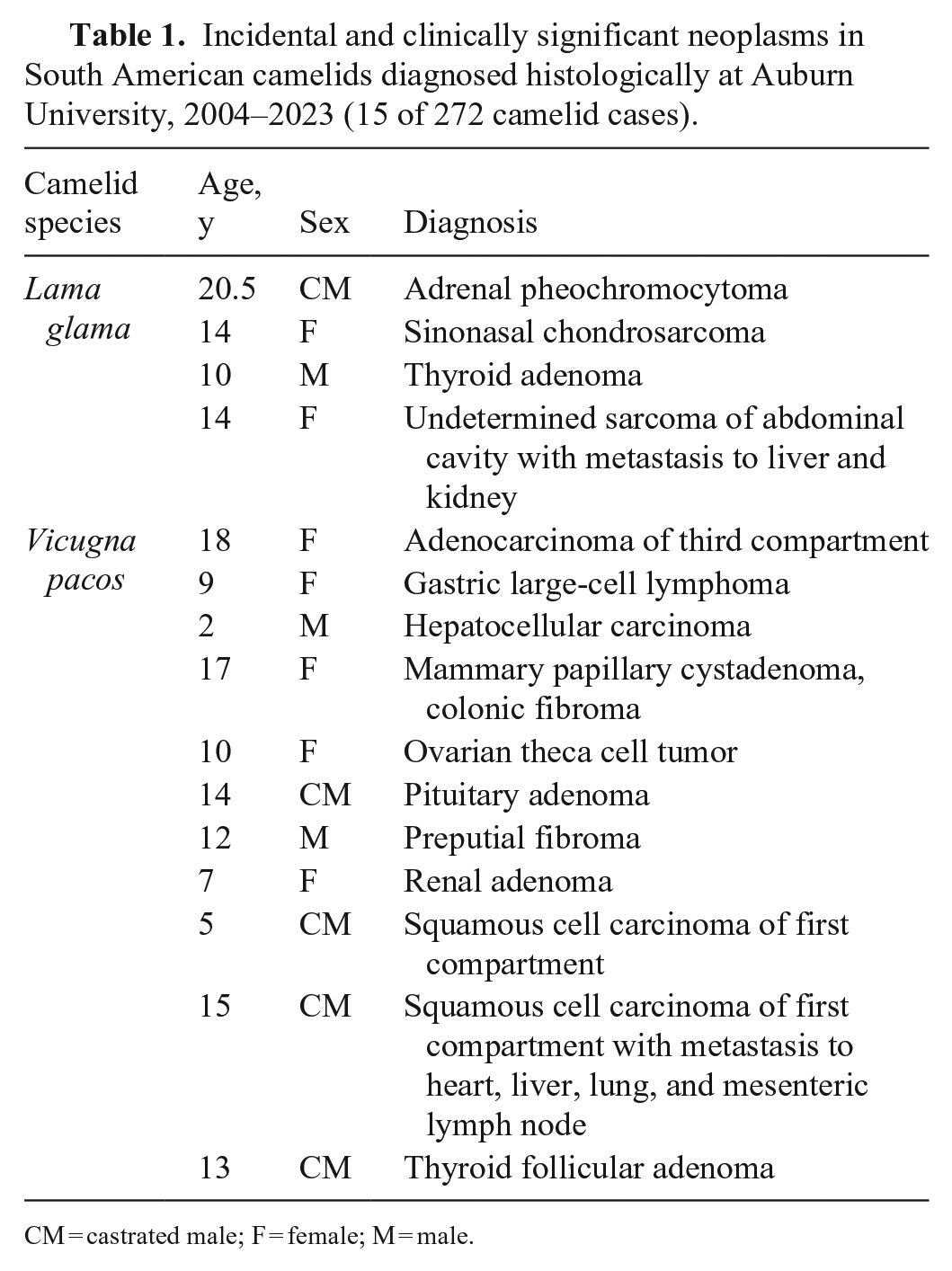
CM = castrated male; F = female; M = male.
Skeletal and sinonasal chondrosarcomas are well-documented in small companion animals, especially dogs. 12 When affecting the upper airways, these chondrosarcomas have a typically insidious space-occupying effect, potentially leading to nasal discharge, local deformities, exophthalmos, or neurologic signs. These signs can be confused with rhinitis, foreign body impaction, osteomyelitis, or periodontal abscesses; thus, definitive diagnosis relies on different diagnostic modalities, such as thorough physical examination, radiographs, ultrasound, bacterial or fungal culture, and surgical biopsy, for which multiple tissue core samples are often needed. In dogs, radiotherapy remains the most beneficial treatment for this neoplasm, with or without surgery. 12 Surgical removal of a nasal chondrosarcoma with a 15-mo disease-free interval has been reported in a cow 2 ; however, given the degree of airway obstruction, bone, soft tissue involvement, and therapeutic limitations in our case, the prognosis was poor regardless of metastatic status.
Our case further corroborates the low incidence of neoplastic diseases in older camelids and contributes an additional record to the literature. We found no cases of chondrosarcoma diagnosed in SACs in a comprehensive search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using the search terms “chondrosarcoma”, “camelid”, “llama”, and “alpaca”, suggesting that this condition has not been reported in SACs. Incisional biopsy was pivotal for diagnosis, prognosis, and decision-making in our case. Sinonasal neoplasms, including chondrosarcomas, should be included in the differential list for evaluation of facial deformities, nasal discharge, and upper respiratory distress in adult camelids.
Footnotes
Acknowledgements
We thank Cynthia Hutchinson and Lisa Parsons (Histology Laboratory, Pathobiology, College of Veterinary Medicine, Auburn University) for preparing the histopathology slides.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
