Abstract
Infection with Lamanema chavezi, a parasitic nematode of New World camelids, was diagnosed by examination of feces and formalin-fixed liver from a 14-month-old female llama (Lama glama) that died after a 6-week illness. Infection with L. chavezi was initially suspected when a granuloma containing an unidentified nematode was detected microscopically in the hepatic parenchyma from a necropsy specimen. The subsequent diagnosis of L. chavezi infection was based on the morphologic features of 2 immature nematodes dissected from individual hepatic granulomas, characteristics of eggs detected in feces of the llama by centrifugal flotation in sugar solution (specific gravity: 1.30), development of third-stage larvae within the eggs after incubation of the llama feces at room temperature for ≥30 days, and the morphology of third-stage larvae released from the embryonated eggs. Collectively, these findings indicate that the llama, born and raised in Oregon, harbored an autochthonous L. chavezi infection. Eggs identified as L. chavezi were also detected by centrifugal flotation of pelleted feces from 3 of 7 herd mates of the llama indicating this parasite is endemic in the Oregon herd. The findings reported herein serve to alert diagnosticians and veterinary practitioners to the occurrence of L. chavezi in New World camelids in the United States and describe diagnostic features of this potential pathogen.
Lamanema chavezi is a potentially pathogenic nematode of New World camelids (NWCs) that, to the authors’ knowledge, has not been previously reported to occur in the United States.2,3 The direct life cycle of this trichostrongyloid is unusual in that third-stage larvae (L3s) develop within the eggs, hatching of the eggs is stimulated by exposure to low temperatures, and L3s undergo an enterohepatic migration before returning to the small intestine to become adults. 11 Based on the morphologic characteristics of L. chavezi and the unique features of its life cycle, an autochthonous infection with this nematode is reported in a llama (Lama glama) born and raised in the State of Oregon. Furthermore, detection of L. chavezi eggs in feces from herd mates of this llama indicates that this nematode is endemic in the Oregon herd.
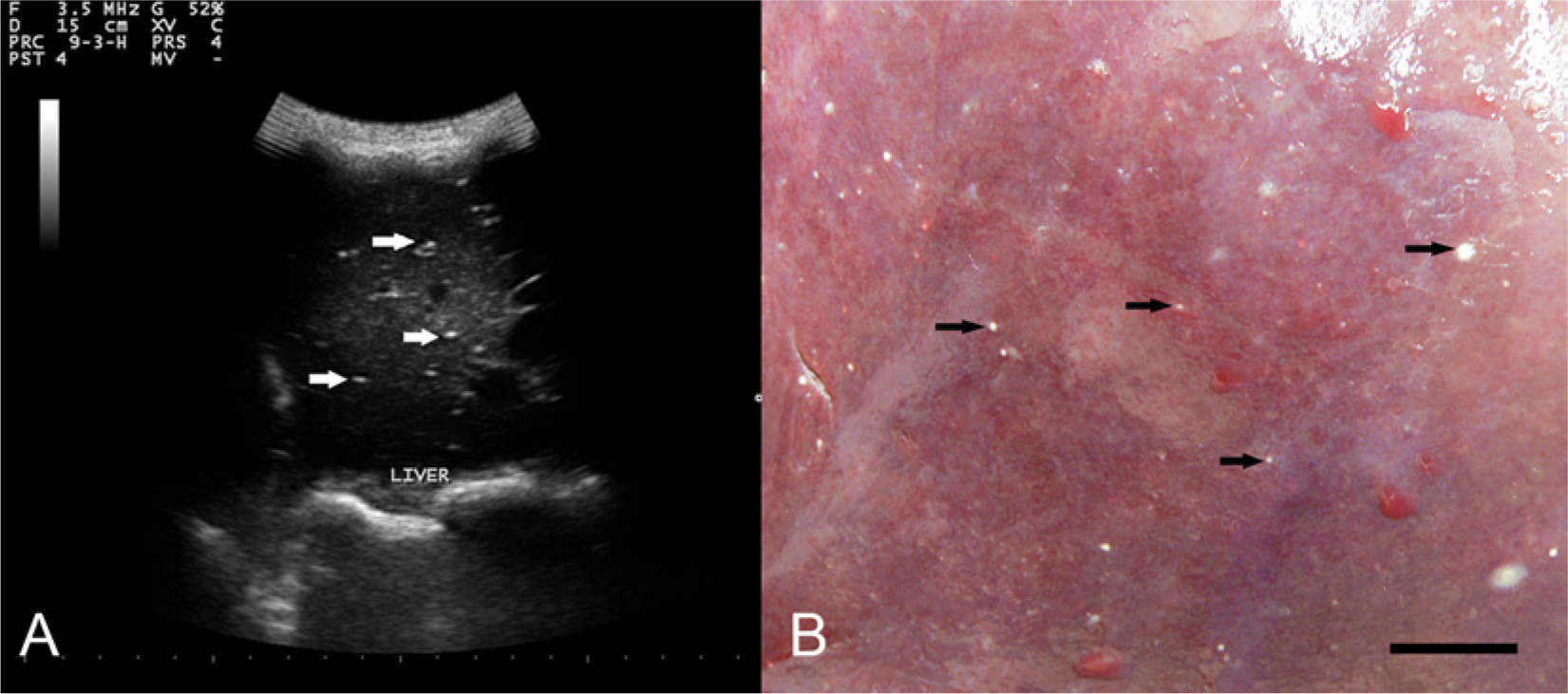
A 14-month-old female llama was transported from Oregon to Iowa on September 9, 2012 to attend a llama event. The owner had treated the llama with fenbendazole at 18.5–40 mg/kg body weight orally for 1–3 days every 2–3 months beginning at 3 months of age. A final dose of fenbendazole (25.5 mg/kg body weight) was given at 13 months of age at the time of transport to Iowa. Four weeks later, on October 8, 2012, the llama was admitted to the Iowa State University (ISU) Lloyd Veterinary Medical Center (Ames, Iowa) with a 3-week history of lethargy, anorexia, and weight loss. Ultrasonographic examination of the liver at the time of presentation to the teaching hospital revealed small, hyperechoic foci scattered throughout the parenchyma (Fig. 1A). During hospitalization, serum hepatic enzymes were not elevated. The llama died on October 22, 2012 following 2 weeks of intensive therapy for suspected septic salmonellosis. A routine, complete necropsy was performed, and systemic salmonellosis and septic peritonitis due to jejunal rupture were diagnosed. Samples of liver and other organs were fixed in 10% neutral buffered formalin, processed routinely, sectioned to 5 µm, and stained with hematoxylin and eosin.
Llama (Lama glama).
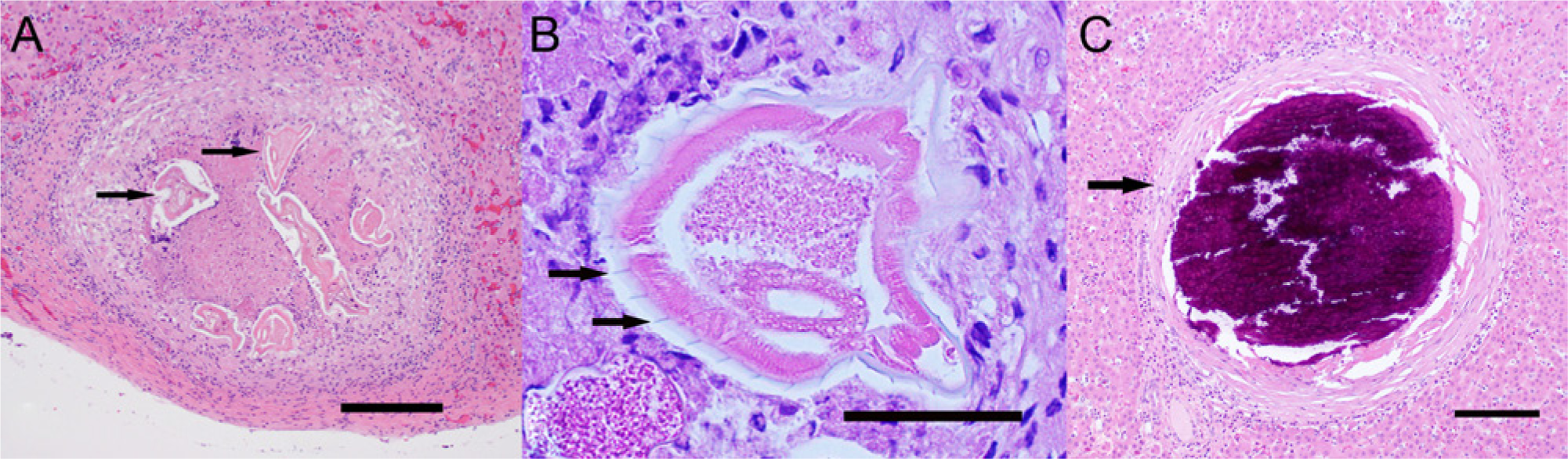
Among the gross lesions observed at necropsy were multiple, small (pinpoint to 2 mm), white-tan foci on the liver surface that extended into the parenchyma (Fig. 1B). The lesions were consistent with the hyperechoic foci previously observed in the ultrasonogram (Fig. 1A). Microscopically, many of the hepatic lesions noted at necropsy were well-organized granulomas that contained a coiled nematode (Fig. 2A, 2B) or were undergoing mineralization (Fig. 2C). Longitudinal cuticular ridges that comprise the synlophe are evident in a section of a single nematode (Fig. 2B). In tissue sections, the lamina propria of the small intestine and colon was expanded by moderate numbers of eosinophils, lymphocytes, and plasma cells, but encysted larvae were not observed.
Hepatic granulomas in a llama (Lama glama).
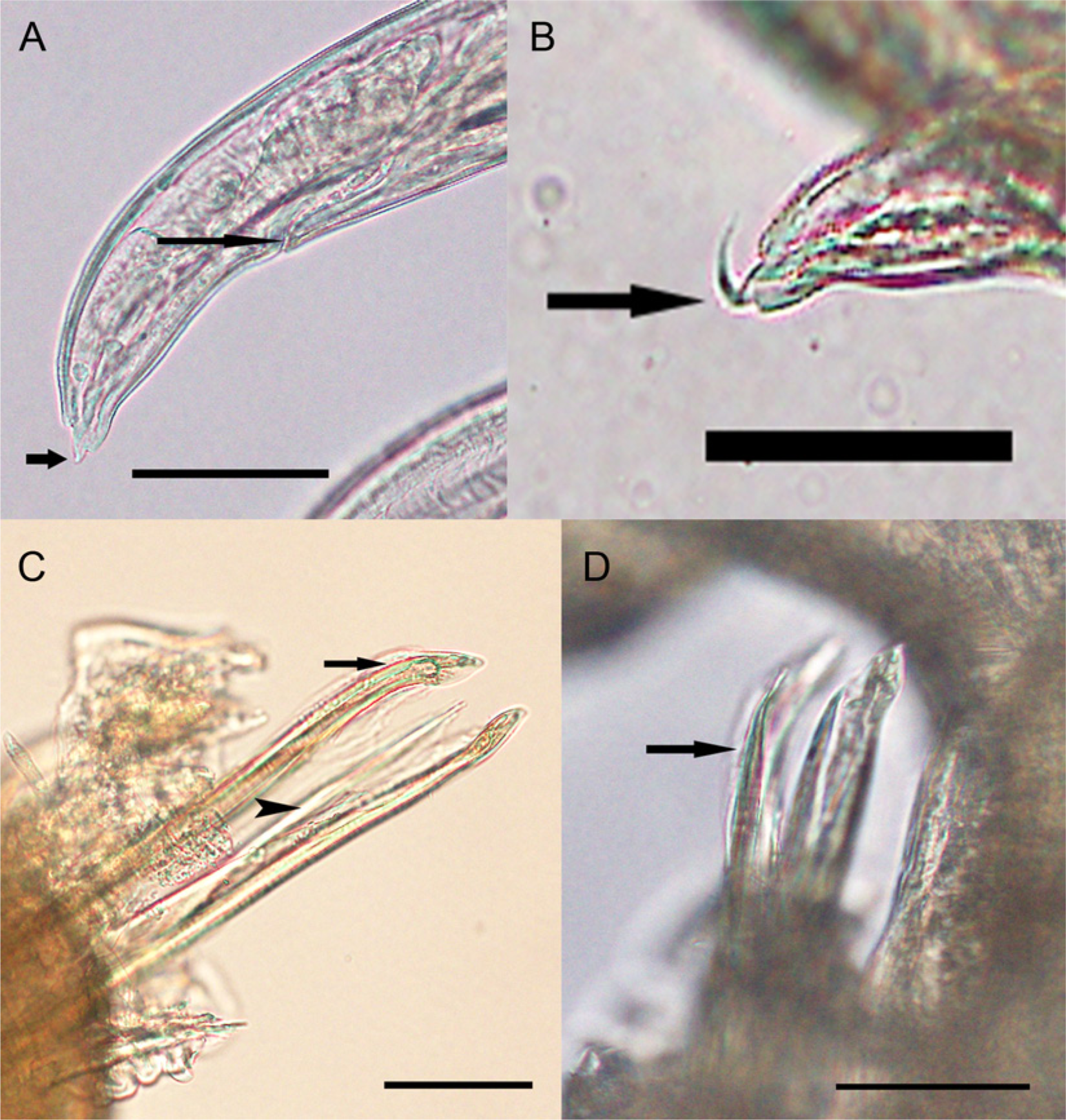
With the aid of a stereoscopic microscope, portions of formalin-fixed liver containing grossly visible pale foci were carefully examined and 2 coiled nematode larvae, 1 intact male and 1 decapitated female, were dissected from individual granulomas. The larvae were initially placed in 70% ethanol–glycerol solution and then transferred to 70% ethanol. The female larva had a subterminal anus and a spine approximately 12 mm long that was directed laterally approximately at a right angle after it emerged from the distal extremity of the tail (Fig. 3A, 3B), features previously described for L. chavezi. 22 The spicules of the male larva (Fig. 3C, 3D) were consistent with descriptions of adultL. chavezi.3,22 Other features of the formalin-fixed larvae could not be evaluated because they were obscured by superimposition of larval coils on one another and by tissue adherent to the larval surfaces that could not be removed without compromising the integrity of the larvae. The presence of adult L. chavezi, previously described as 8–18 mm long, red, and with a corkscrew-like appearance of the anterior two thirds of the body, 11 could not be determined because gross samples of the intestinal tract of the llama had been discarded and were not available for examination.
Lamanema chavezi larvae dissected from hepatic granulomas in a llama (Lama glama).
The ISU Clinical Pathology Laboratory initially examined feces collected from the llama on October 8 and 9, 2012, and reported 300 trichostrongyle and <100 Nematodirus eggs per gram of feces by the McMaster method, no Fasciola hepatica eggs by fecal sedimentation, and no Eimeria macusaniensis oocysts by either method. The remaining feces were refrigerated until examined by centrifugal flotation in sugar solution (specific gravity: ≥1.3) on November 2, 2012. Low numbers of yellow-brown, cylindrical eggs with parallel sides and rounded poles measuring 170.2 µm × 73.4 µm (n = 2) and containing a highly cellular internal mass were present in the llama feces that had been refrigerated for 24 days (Fig. 4A). In comparison, Nematodirus eggs present in the same fecal sample had 8–16 large, round dark cells.
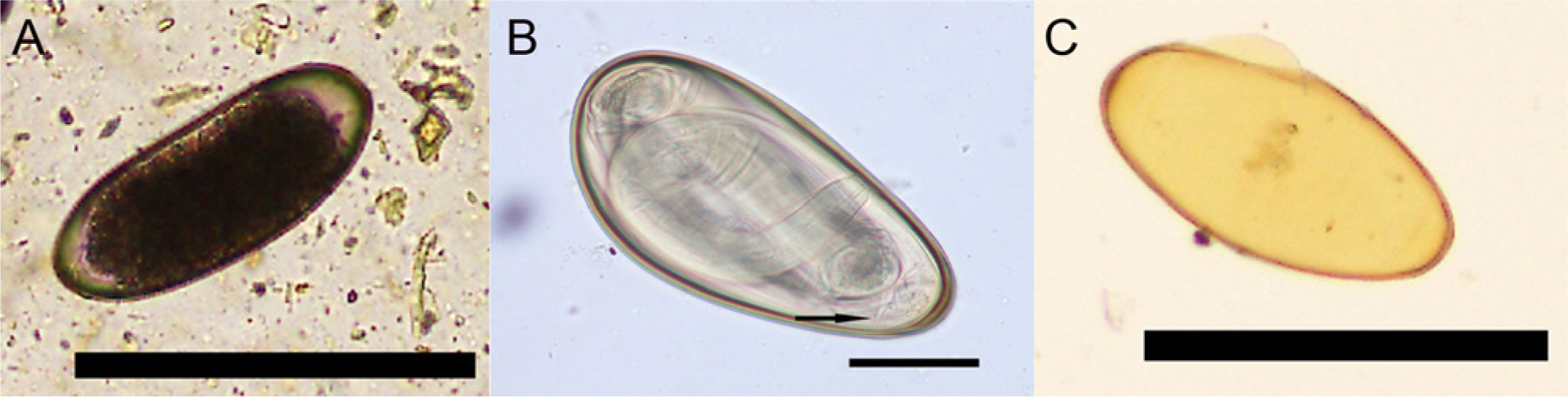
All remaining feces from the samples were thoroughly mixed with tap water and incubated for 38 days at room temperature. During this time, aliquots of the cultured feces were periodically removed and examined by centrifugal flotation to monitor larval development. By 30 days of incubation of the llama feces, L3s with 2 retained cuticle sheaths were present within the L. chavezi (Fig. 4B) and Nematodirus eggs. When eggs containing an L3 were ruptured on glass microscope slides by applying pressure to the coverslip, larvae actively emerged. The egg shells remaining after L3s had emerged from the L. chavezi eggs were yellow-brown with a darker golden-brown outer wall (Fig. 4C). Third-stage larvae from 2 L. chavezi eggs averaged 697.2 µm × 29.8 µm, had 8 large intestinal cells, an unnotched larval tail tapering abruptly to a relatively broad blunt point, and a short (26.2 µm), sharply pointed sheath tail (Fig. 5A–5C).
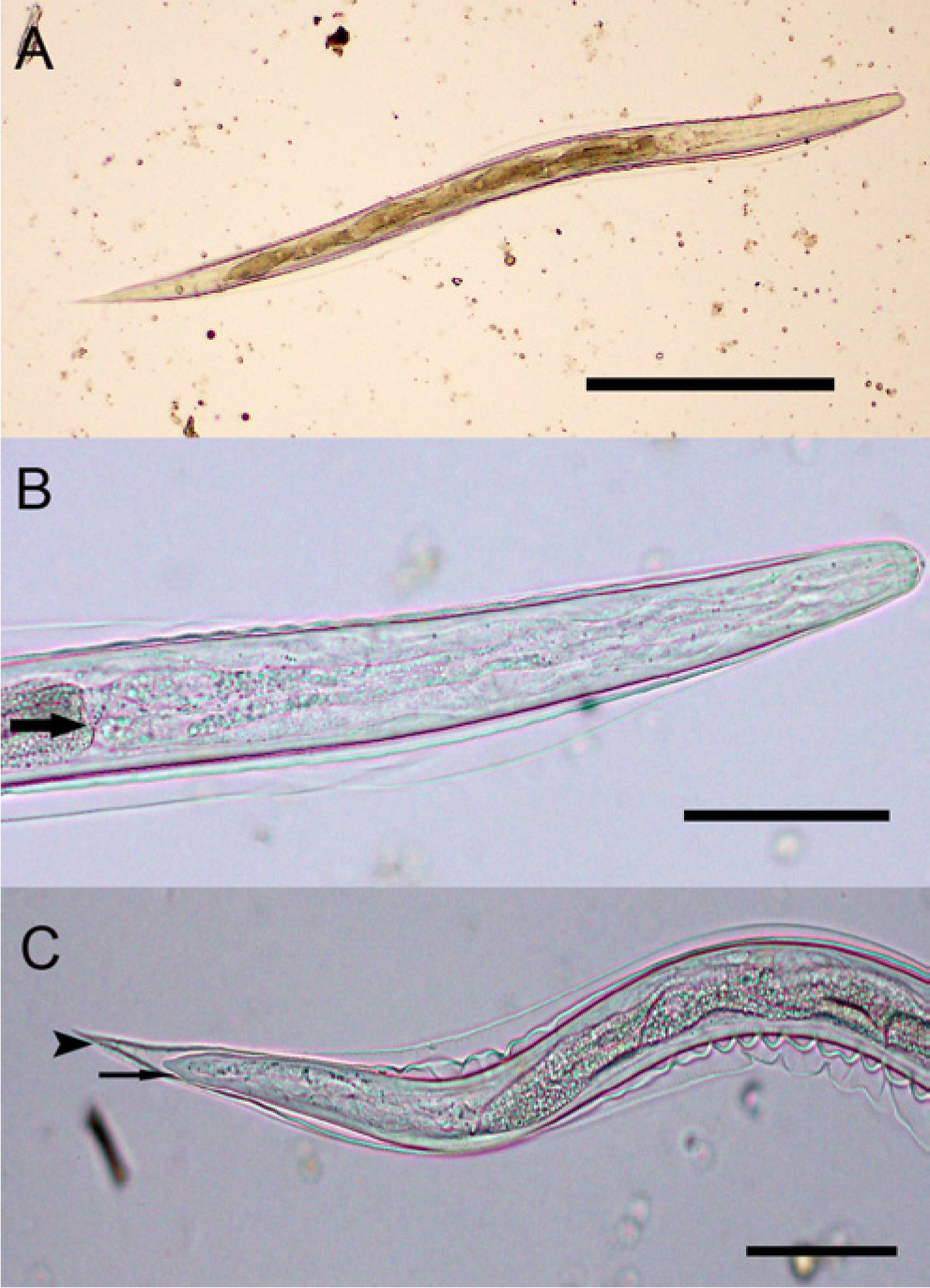
Third-stage larvae (L3) from Lamanema chavezi eggs.
On November 27, 2012, fecal samples were obtained from 7 healthy Oregon herd mates of the llama and low numbers of eggs consistent with those of L. chavezi were detected in feces from 3 llamas. The eggs averaged 171.2 µm × 77.3 µm (n = 11) and contained a developing first-stage larva.
In South America, L. chavezi is a common, small intestinal parasite of NWCs with reported prevalence rates of 18.5% in Argentinian llamas, 6 48.2% in Peruvian alpacas (Vicugna pacos), 7 13–61.3% in Chilean llamas,1,20 and 64% in Bolivian llamas (Sporndly E, Nissen AM: 2008, Prevalence of parasites in llamas in the Andean Bolivia. Faculty of Life Sciences, University of Copenhagen, Denmark. Master’s thesis. Abstract available at http://www.ddrn.dk/filer/forum/File/Lamas_Bolivia_ES_AMN.pdf. Accessed June 9, 2013). Although L. chavezi is generally considered to be a host-specific parasite of NWCs, it has also been described in 2 species of South American rodents.9,22 Consistent with the age of the llama in the present case, the greatest prevalence of L. chavezi in South America occurs in NWCs under 2–3 years of age.7,11 Unusual eggs detected in feces of alpacas and llamas in New Zealand provided the first indication that L. chavezi might parasitize NWCs outside of South America. 16 Subsequently, identification of adults expelled in feces of a llama following anthelmintic treatment confirmed the occurrence of L. chavezi in New Zealand. 17 Further studies are needed to determine the prevalence of L. chavezi in NWCs in the United States and New Zealand.
In addition to eggs of L. chavezi, feces of the llama in the present case also contained eggs of trichostrongyles, Nematodirus spp., and Nematodirus battus. Of nematodes infecting NWCs, only Nematodirus spp. and Marshallagia spp. produce large eggs (>130 µm) that need to be differentiated from eggs of L. chavezi. The eggs of Nematodirus spp. in the present case were readily identified by the presence of a morula composed of ≤16 large dark cells within a clear to colorless shell, by their greater size compared with L. chavezi, and/or by their shape (i.e., wide centrally and narrow at the poles).2,23 In fresh feces, eggs of L. chavezi and Marshallagia both have flat parallel sides with rounded poles, contain a morula comprised of >64 cells, and their size ranges overlap, but the eggs of L. chavezi are yellow-brown while those of Marshallagia have a colorless shell.2,6,13,16,19 The eggs of N. battus and L. chavezi are similar in size and shape, but N. battus eggs are brown or coffee-colored, have a thickened shell with an irregular pitted internal surface at the poles, and fewer and larger morula cells in comparison to L. chavezi.2,6,16
Larvae develop to second-stage larvae within the eggs of Marshallagia spp. and to L3s in Nematodirus spp. andL. chavezi. 19 In the present case, L3s of Nematodirus spp. were easily identified by their long filamentous sheath tail; those of N. battus had a long tapering notched larval tail. 24 In contrast, L3s obtained from the L. chavezi eggs had a short sheath tail, an unnotched larval tail, and were comparable in size to L3s previously reported. 16
Small, mineralized granulomatous or fibrotic lesions are frequently reported as incidental findings in the livers of NWCs in the United States and are attributed to parasite migration, but the specific parasites causing these lesions are commonly not identified. 4 Migration of L. chavezi, Fasciola hepatica, Fascioloides magna, Dicrocoelium dendriticum, and tapeworm larvae can produce lesions in the liver of NWCs, but, to date, only F. hepatica and F. magna are reported to cycle in NWCs in the United States.2,8,12 In the present case, no fluke eggs were found when feces from the llama were examined by sedimentation, and no immature or adult liver flukes or tapeworm larvae were found in the liver or bile ducts at necropsy. Although the llama feces contained N. battus eggs, infection with this nematode would not be expected to cause hepatic lesions because it remains in the intestinal tract and does not migrate in the liver. 14 The occurrence and appearance of trichostrongyloid larvae in hepatic granulomas of the llama in the present case are consistent with descriptions of L. chavezi infection. 11
Lamanema chavezi is considered the most pathogenic nematode of NWCs, and pathogenicity is typically greatest in younger hosts infected with large numbers of larvae. Anemia, anorexia, emaciation, prostration, colic, and pale feces were noted in a 4-month-old alpaca that died 20 days after it was inoculated with 200,000 L3s. 10 Inoculation with 10,000 L3s resulted in anemia, eosinophilia, focal granulomatous liver lesions, and parasites in the bile ducts 30 days after inoculation of a 20-day-old alpaca 10 and an increase in serum aspartate transaminase (serum glutamic oxaloacetic transaminase) levels 14 days after inoculation of two 3-month-old alpacas. 21 In contrast, a 6-month-old alpaca inoculated with 10,000 L3s became anemic but showed no clinical signs while neither anemia nor clinical signs occurred in a 12-month-old alpaca inoculated with 7,000 L3s. 10 A naturally infected, 2-year-old llama in poor body condition that was shedding large numbers of L. chavezi eggs had small hepatic abscesses and hemorrhagic enteritis at necropsy, while a second naturally infected 3-year-old llama herd mate shed lesser numbers of L. chavezi eggs and exhibited no clinical signs. 5 Natural L. chavezi infections of NWCs in Argentina 6 and New Zealand16–18 also had no overt clinical signs. In the present case, detection of L. chavezi eggs in feces indicates that the llama had been infected with this parasite for at least the length of the pre-patent period, which is 30–32 days, and mineralization of the granulomas suggests a chronic infection. 11
Anthelmintics with demonstrated efficacy against L. chavezi in NWCs include levamisole 11 and ivermectin, 15 but resistance to ivermectin has also been reported (Ciro-Marino TA: 2011, Determinacion de resistencia anthlemintica frente a ivermectina de nematodos gastrointestinales en alpacas (Vicugna pacos) Puno-Peru [Determination of anthelmintic resistance against ivermectin of gastrointestinal nematodes in alpacas (Vicugna pacos) in Puno-Peru]. In Spanish. Abstract in English. Available at: http://www.buscagro.com/www.buscagro.com/biblioteca/Ciro-Traverso-Arguedas/nematodos-en-alpacas.pdf. Accessed July 15, 2013). It is of interest that a patent L. chavezi infection was present in the llama of the current study even though it had been repeatedly treated with fenbendazole at 1–3-month intervals beginning at 3 months of age with the most recent treatment given 6 weeks prior to death. Whether or not infection with L. chavezi might have predisposed this llama to Salmonella infection or otherwise contributed significantly to its illness is unknown.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
