Abstract
Gastrointestinal adenocarcinomas are often reported in South American camelids (SAC). We describe here cases of gastroduodenal adenocarcinoma in an adult alpaca (Vicugna pacos) and a llama (Llama glama); both SACs were anorectic and lethargic before death. At autopsy, a prominent and firm caudal C3-pyloric-duodenal junction with stricture and ulceration was present in both animals, as were hemorrhages in various organs and hydrothorax. Microscopically, scattered nests, cords, and tortuous acini of neoplastic epithelial cells were embedded in desmoplastic stroma and invaded the submucosa and muscle layers of the gastroduodenal junction. The mucosa was necrotic, with gram-negative rods in the alpaca and colonies of gram-positive cocci in the llama. No tumor metastases were observed. The neoplastic cells immunolabeled for pancytokeratin. Escherichia coli was isolated from the alpaca and Streptococcus lutetiensis from the llama; septicemia was the cause of death in both animals. Although adenocarcinomas arising from gastric compartments and intestinal segments have been reported in SACs, adenocarcinoma of the caudal C3-pyloric-duodenal junction has not been reported previously in these species, to our knowledge.
The gentle and docile nature of South American camelids (SACs) has contributed to their growing popularity as companion animals, emphasizing the importance of a comprehensive understanding of their biology, care, and medical treatment. 1 This is particularly pertinent when addressing a disease such as neoplasia, which has been reported in SACs since 1974, including cutaneous,13,16,17 ocular, 17 mammary,9,17 and gastric squamous cell carcinoma14 –16; intestinal, 17 uterine,7,16 biliary,3,17 pancreatic, 17 mammary,2,16 and pulmonary adenocarcinoma 12 ; fibrosarcoma;6,17 and melanoma. 1
Of particular concern in SACs is gastrointestinal adenocarcinoma. In human patients, gastroduodenal obstruction due to neoplasia can result in chronic weight loss, decreased appetite, impaired quality of life, and potentially death. 5 Similar effects have been observed in SACs. Despite its significant impact, limited research exists on gastrointestinal adenocarcinoma in camelids, and current knowledge is derived mainly from case reports and a few retrospective studies. 1
We describe here the pathologic features and clinical consequences of infiltrative adenocarcinomas originating from the caudal C3-pyloric-duodenal junction of 2 SACs: case 1, an alpaca (Vicugna pacos), and case 2, a llama (Llama glama). Case 2 was from 2021 in Solano County, CA, USA. Based on this case, we searched the Laboratory Information Management System (LIMS) from the California Animal Health and Food Safety (CAHFS) laboratory for any other caudal C3-pyloric-duodenal adenocarcinoma in SACs. From 688 SAC (llamas, alpacas, guanacos) autopsy cases recor-ded in 2008–2021, we found only 1 alpaca with a caudal C3-pyloric-duodenal adenocarcinoma similar to case 2; case 1 was from 2009 in Sacramento County, CA. Both animals were presented to the CAHFS, Davis branch, for autopsy and diagnostic workup. Both camelids were intact adult females, but their ages were not specified. According to the provided clinical history, the animals were anorectic and lethargic, which ultimately ended in their death within a few days of sign onset. The alpaca was treated with IV fluids including electrolytes and amino acids; the llama was treated for suspected polioencephalomalacia with B vitamins, probiotics, and IV fluids. No specific information regarding the typical daily food and water intake, or the housing or environment, of either animal was available.
Gross examination of both cases revealed pleural effusion with limited fibrin clots, diffuse wet and heavy lungs without collapse, and ascites. The most significant and common finding in both animals was the prominent, firm, and ulcerated area at the caudal C3, pylorus, and cranial duodenal junction with marked stricture of the lumen (Fig. 1). The gastric serosa of the first (C1) and second (C2) compartments of the stomach was edematous. Petechiae were seen throughout the mucosa of the third compartment (C3). In case 1, the small and large intestines had serosal petechiation; case 2 had petechiae, ecchymoses, and rare fibrin clots in the peritoneum. In case 2, a segment of the spiral colon was partially occluded by a 3.5 × 1.5-cm, elongated, dark-green fecalith.
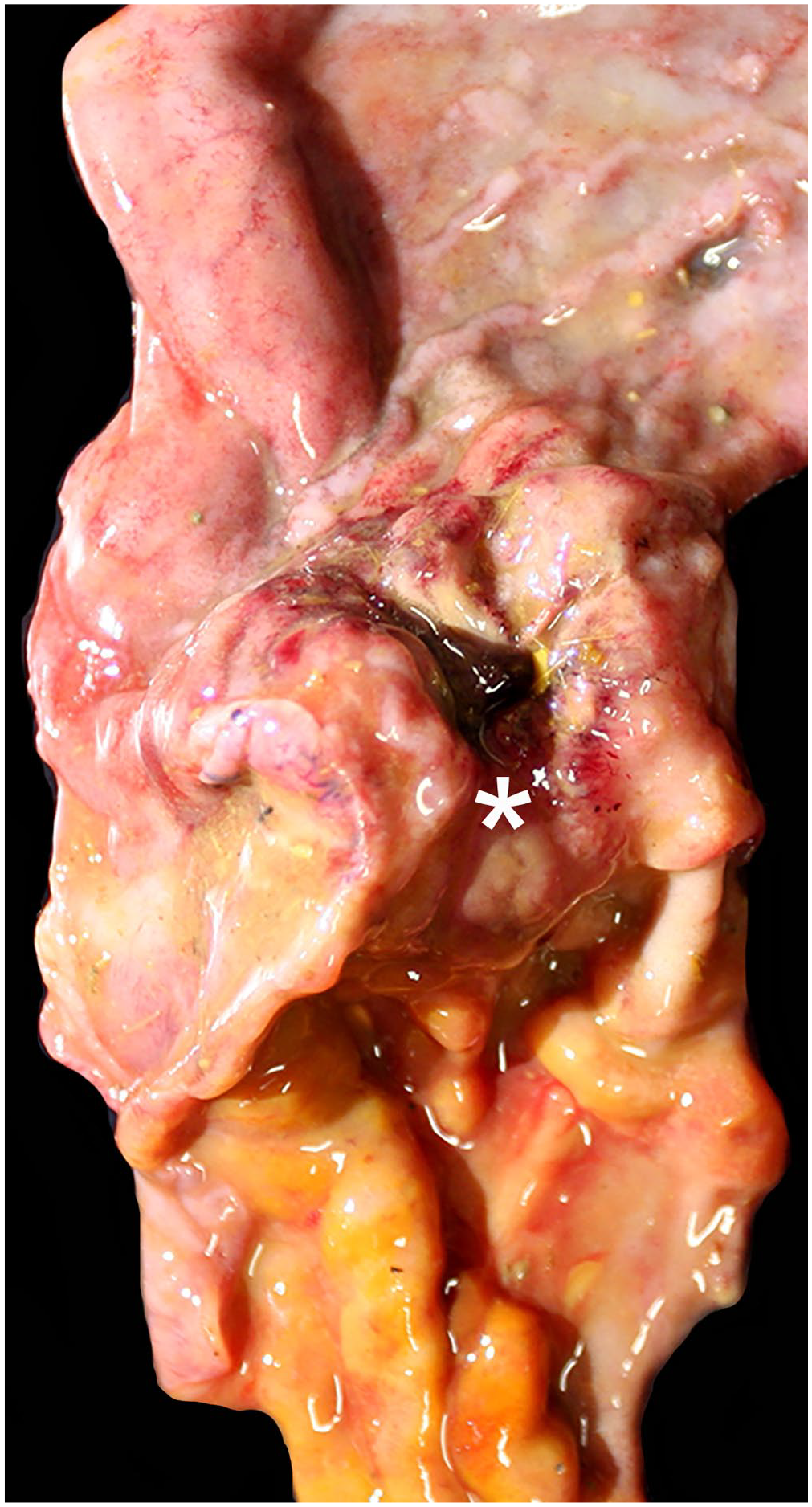
Gross image of a gastroduodenal adenocarcinoma in a llama, case 2. A thickened pyloric-cranial duodenal junction infiltrated by an adenocarcinoma (asterisk) with coalescing ulcers, multifocal reddening, and stricture located between the third compartment (C3) of the stomach (upper organ) and the cranial duodenum (lower organ).
Selected tissues were fixed in 10% neutral-buffered formalin and processed routinely; 3-µm sections were mounted on slides and stained with H&E. Additionally, histochemical stains (periodic acid–Schiff [PAS], Brown–Hopps Gram, Masson trichrome) and immunohistochemistry (pancytokeratin, vimentin) were performed following CAHFS laboratory standard operating procedures (SOPs). In brief, formalin-fixed, paraffin-embedded tissue sections mounted on charged slides were deparaffinized and rehydrated using xylene and graded alcohols. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide in water. After heat-induced epitope retrieval using a citrate buffer, the slides were incubated with either a mouse monoclonal antibody directed against vimentin (clone V9; Sigma-Aldrich) or a pancytokeratin mouse monoclonal antibody (clone AE1/AE3; Sigma-Aldrich) for 45 min, and then detected with a horseradish peroxidase–conjugated anti-mouse polymer (K4001, Envision +; Dako/Agilent) for 30 min before visualized by incubating (SK-4805, ImmPACT NovaRed substrate; Vector) for 10 min. The sections were counterstained with Mayer hematoxylin, blued, and coverslipped.
Lung, liver, and peritoneal swabs were collected during the autopsies and submitted for aerobic culture. Lung swabs were inoculated onto MacConkey, 5% sheep blood, and chocolate agar plates (Hardy); the liver and peritoneal swabs were plated on MacConkey and sheep blood agar. Culture plates were incubated at 35–37°C with 5% CO2 for 24–48 h, following CAHFS SOPs for aerobic culture. Aerobic bacterial culture was performed (lung, liver: cases 1, 2; peritoneum, pleura: case 2); quantitative real-time PCR (qPCR) was performed on spleen for bovine viral diarrhea virus (BVDV; cases 1, 2) and bluetongue virus (BTV; case 2). Fecal flotation was performed in both cases. Liver samples from both cases were submitted for selenium and heavy metal screening, including arsenic, copper, iron, lead, manganese, mercury, molybdenum, and zinc.
Microscopically, the mucosa of the caudal C3-pyloric-duodenal junction was multifocally ulcerated (more in the llama than the alpaca), with fibrin thrombi, neutrophils, and ectatic glands filled with neutrophils and lined by bacterial colonies that were predominantly gram-negative rods (alpaca) and gram-positive cocci (llama). Within the submucosa and muscle layers of the caudal C3-pyloric-duodenal junction was a non-encapsulated, poorly demarcated, moderately cellular, and infiltrative neoplasm formed by tortuous acini (Fig. 2A), cords, or nests of polyhedral neoplastic cells surrounded by dense, trabecular desmoplastic stroma (more in the alpaca than llama; Fig. 2B). Neoplastic cells had distinct cell borders and scant, pale eosinophilic and occasionally finely vacuolated cytoplasm. The nuclei were round-to-oval with finely stippled chromatin and 1–4 nucleoli and moderate anisokaryosis; 8 mitotic figures were counted in 2.37 mm 2 (10 high power fields, 40× objective). Some lymphatics and blood vessels contained emboli of neoplastic cells. The desmoplastic stroma contained scattered lymphocytes and plasma cells. Positive immunolabeling for pancytokeratin was observed in the cytoplasm of the mucosal epithelial cells and the invading neoplastic cells (Fig. 2C); vimentin immunolabeling was negative. The cytoplasm of some neoplastic cells was filled with moderate amounts of PAS-positive mucus (Fig. 2C, inset), and some acini and nests were filled with cellular debris.
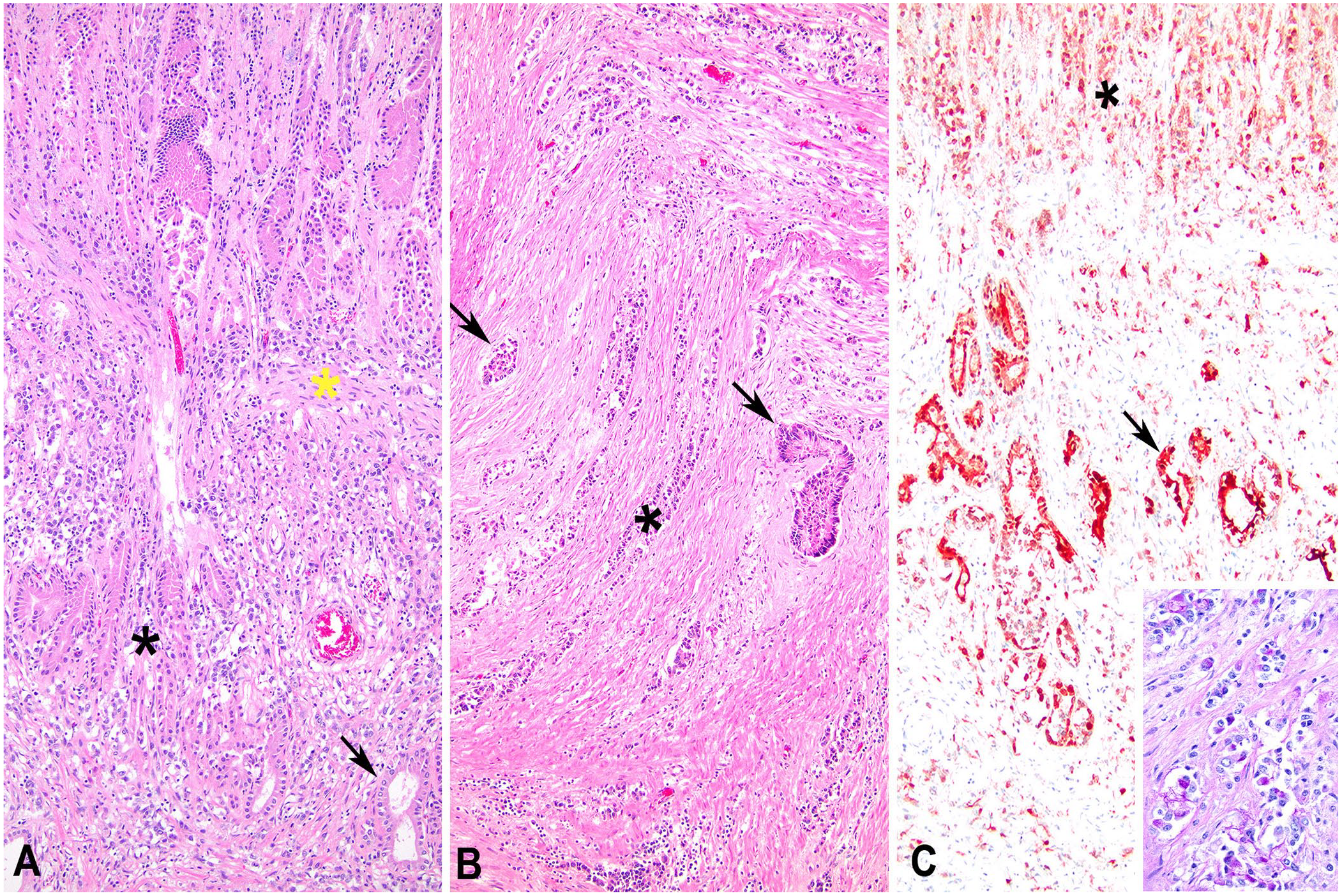
Histology of the pyloric-cranial duodenal adenocarcinoma in an alpaca, case 1.
In the alpaca, the C1 of the stomach was ulcerated with numerous neutrophils and cellular debris and colonized by gram-negative filamentous bacteria and gram-positive short rods. The liver had random necrotic foci with clusters of neutrophils, some of them associated with gram-negative rods. Portal triads with marked biliary hyperplasia were infiltrated by large numbers of neutrophils. In the liver of the llama, fibrin thrombi were observed within random hepatic sinusoids and pulmonary capillaries accompanied by clusters of cocci.
Bacterial aerobic culture was positive for E. coli (liv-er: cases 1, 2; lung: case 2) and Streptococcus lutetiensis (liver, lung, peritoneum, pleura: case 2). qPCR was negative for BVDV and BTV in both cases. No parasites or parasitic eggs were detected in the feces of either animal. The liver of case 1 had markedly decreased copper (3.3 ppm; RI: 25–100 ppm); the iron concentration in the liver of case 2 was slightly elevated (250 ppm; RI: 70–200 ppm). Other heavy metals and selenium in both cases were between RIs. S lutetiensis was identified using MALDI-TOF MS, and the scores for duplicate spots of isolated colonies of 2.16 and 2.15 met the criteria for species-level identification (score > 2.0).
We conduct a thorough search using various sources, including Google, PubMed, CAB Direct, Web of Science, and Scopus, using keywords such as tumor, adenocarcinoma, cancer, camelids, stomach, duodenum, and intestine to find cases of caudal C3-pyloric-duodenal adenocarcinomas in llamas or alpacas. However, we found no reported cases of this condition in these animals. This suggests that caudal C3-pyloric-duodenal adenocarcinomas have not been documented in llamas or alpacas before.
In both SACs, the neoplasm in the caudal C3-pyloric-duodenal junction caused pyloric stenosis and resulted in partial obstruction of the lumen, contributing to weight loss, lethargy, and eventual death. In humans, cases with pyloric stenosis had a poor survival rate. 18 Additionally, damage to the mucosa of the gastroduodenal tract with subsequent ulceration might lead to bacterial invasion and septicemia, which was the cause of death in our cases. Human patients with pyloric stenosis caused by adenocarcinoma invasion experienced more complications when the cancer invaded deeper layers. 4
We believe that E. coli and S. lutetiensis were the bacteria involved in fatal septicemia in the studied alpaca and llama, respectively. In SACs, colibacillosis is primarily reported as a cause of diarrhea and septicemia in neonates and is secondary to failure of passive transfer; other gastrointestinal diseases might predispose to E. coli invasion and dissemination in older animals. 19 Thus, the colonies of gram-negative bacterial rods in the areas of superficial necrosis of the adenocarcinoma and the areas of necrosis in the liver suggest septicemic colibacillosis. In the llama, S. lutetiensis was isolated from the liver, lung, pleura, and peritoneum. These bacteriologic results and the histologic evidence of septicemia in the llama suggest that this bacterium was likely the cause of the fatal septicemia. S. lutetiensis has been reported to cause clinical mastitis in dairy cows, 10 and it has been isolated from a cat with intestinal lymphoma. 11 To our knowledge, S. lutetiensis has not been reported previously in SACs. The extraintestinal isolation of this bacterium and the cocci colonies in the superficial necrotic area of the tumor suggests that the source of the S. lutetiensis septicemia was the necrotic focus in the caudal C3-pyloric-duodenal adenocarcinoma. The severe necrosis in the caudal C3-pyloric-duodenal adenocarcinoma might have predisposed to secondary bacterial colonization and dissemination in both camelids.
In case 1, copper deficiency was present, which can lead to a cascade of detrimental effects, including reduced productivity, vulnerability to other diseases, gastrointestinal distress, and anemia. 8 This deficiency may have been primary or secondary to the malnutrition or anorexia of the alpaca. In case 2, the slightly elevated liver iron concentration likely had no clinical significance. Neither variation in element concentration could be directly associated with the deaths in these cases.
Footnotes
Acknowledgements
We thank Dr. Alex Loretti for his work on one of the cases and the CAHFS laboratory staff in general for their technical assistance, and in particular Karen Sverlow.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
