Abstract
Clostridium spiroforme has been associated with spontaneous and antibiotic-associated enteric disease (C. spiroforme–associated enteric disease, CSAED) in rabbits, which is clinically characterized by anorexia, diarrhea, or sudden death. Diagnosis is usually based on gross and microscopic lesions, coupled with finding the characteristic coiled bacteria in intestinal smears. Isolation of C. spiroforme is often challenging, and a PCR protocol has been developed. We reviewed 32 cases of CSAED submitted for autopsy to the Davis, Tulare, and Turlock laboratories of CAHFS between 1992 and 2019. The reported gross findings were soiling of the perineum, tail, and/or hind legs with diarrhea (16 of 32); gastric (16 of 32), small intestinal (6 of 32), cecal (15 of 32), and/or colonic (4 of 32) distention with brown-to-green, watery content; and serosal hemorrhages in the cecum (15 of 32). The most common microscopic finding was necrotizing enteritis (19 of 32), followed by cecal mucosal or submucosal edema (8 of 32), necrotizing or pleocellular typhlitis (6 of 32), necrotizing or heterophilic typhlocolitis (6 of 32), and cecal transmural hemorrhages (5 of 32). In all 32 rabbits, typical helically coiled, gram-positive bacilli were observed in fecal or intestinal smears. C. spiroforme was isolated from the intestinal content of 2 of 24 rabbits and detected by PCR assay in 8 of 8 rabbits.
Keywords
Clostridium spiroforme is considered to be the etiologic agent of a spontaneous and antibiotic-associated enteric disease in rabbits, known as C. spiroforme–associated enteric disease (CSAED). This microorganism is a helically coiled, gram-positive, anaerobic, spore-forming bacillus,1,6,17 classified in cluster XVIII of the genus Clostridium together with C. ramosum and C. cocleatum. 1 C. spiroforme was isolated for the first time in 1983 from cecal contents of 27 diarrheic rabbits, including individuals used for research and for commercial production. 5
Most C. spiroforme strains produce an enterotoxin, namely C. spiroforme toxin (CST), which is thought to be the determinant of virulence of this pathogen.7,8,13,15,17 CST is a binary actin-ADP-ribosylating toxin, composed of 2 subunits, namely CSTb, which is responsible for binding to the cell surface receptors of the target cell, and CSTa, which has enzymatic activity and is responsible for destroying the cytoskeleton of the target cell.3,16 –18 CST is structurally similar to other clostridial binary toxins, including C. perfringens type E iota-toxin and C. difficile transferase (CDT).3,7,8,10,13,14,17 Lipolysis-stimulated lipoprotein receptor (LSR) and CD44, the latter a multifunctional surface protein of mammalian cells, have been described as target receptors for the iota family of clostridial binary toxins.10,18
C. spiroforme is thought to be acquired through the fecal–oral route from the environment,11,17 with subsequent proliferation and toxin release in the terminal small intestine, cecum, and colon, where the majority of the changes of CSAED are observed.4,8,12,17 However, the pathogenesis and epidemiologic aspects of the disease have not been fully elucidated. C. spiroforme and CST have been found in diarrheic, but rarely in healthy, rabbits.5,6,11,12,17 In one study, C. spiroforme, but not CST, was found in 2 of 72 healthy rabbits. 5 Most of the strains isolated from rabbits to date have been found to be toxigenic.5,11,12 The disease has been described frequently in domestic rabbits (Oryctolagus cuniculus), especially in commercial facilities, and less frequently in wild cottontail rabbits (Sylvilagus audubonii, Sylvilagus floridanus, Sylvilagus nuttallii) at rehabilitation centers. 11 To date, spontaneous and experimental C. spiroforme infections have been reported in the United States, the United Kingdom, and Europe.2,5,6,11,15 Our review of the literature (Pub Med, Google Scholar, CAB) did not reveal descriptions of cases of CSAED in leporids other than Oryctolagus spp. or Sylvilagus spp.
CSAED is clinically characterized by anorexia, diarrhea, or sudden death.1,3,10,11,15,17 The main gross lesions reported are soiling of the perineum and hindlimbs with liquid feces; dilation of the cecum with watery, dark-green to almost-black content and gas; thickened and edematous cecal wall; and cecal serosal ecchymoses.6,11,12 Microscopic lesions are typically most severe in the ileum, cecum, and colon, and are characterized by mucosal necrosis with villus blunting and atrophy, lamina proprial edema, lamina proprial and submucosal infiltration by granulocytes and mononuclear inflammatory cells, and proteinaceous fluid, fibrin, leukocytes, and erythrocytes in the intestinal lumen.11,12,17 However, these lesions may not be specific to C. spiroforme infection, because other concomitant pathogens, including rotavirus A, Eimeria spp., and enteropathogenic Escherichia coli are often present simultaneously.9,11,12,15 Traditionally, high morbidity and mortality have been reported in farmed rabbits affected by CSAED.1,15
We present here a retrospective study of 32 cases of CSAED, with special emphasis on gross and microscopic findings, diagnostic criteria, and literature review of the condition in rabbits.
Materials and methods
We queried the archives of the California Animal Health and Food Safety (CAHFS) Laboratory, including the Davis, San Bernardino, Tulare, and Turlock branches, for cases that included: 1) rabbits submitted for autopsy between 1989 and 2022, and that 2) had received a diagnosis of CSAED, which was considered the primary cause of death or clinical signs reported in these animals. As a result, we included in our study 32 rabbits from 12 different rabbitries submitted for autopsy between 1992 and 2019 to the Davis, Tulare, and Turlock CAHFS laboratories with a diagnosis of CSAED (Suppl. Table 1). Briefly, the ages of the animals ranged from 3 wk to 3 y; the exact age was not reported in 24 of 32 rabbits. Both females and males were included. Reported breeds were New Zealand White (12 of 32), Rex (4 of 32), Lop-eared (2 of 32), and Californian White (1 of 32); the breed was not specified in 13 rabbits. The clinical histories included diarrhea, sudden death, and increased mortality in the rabbitry. No previous antibiotic treatments were reported in any of the submitted animals. In all cases, a diagnosis had been established based on gross and microscopic lesions coupled with finding the characteristic helically coiled, gram-positive bacteria (Fig. 1) compatible with C. spiroforme, in fecal or intestinal mucosal smears, and in 2 and 8 cases, also by isolation and PCR detection of C. spiroforme, respectively. In addition, other causes of enteric disease had been ruled out in all cases.
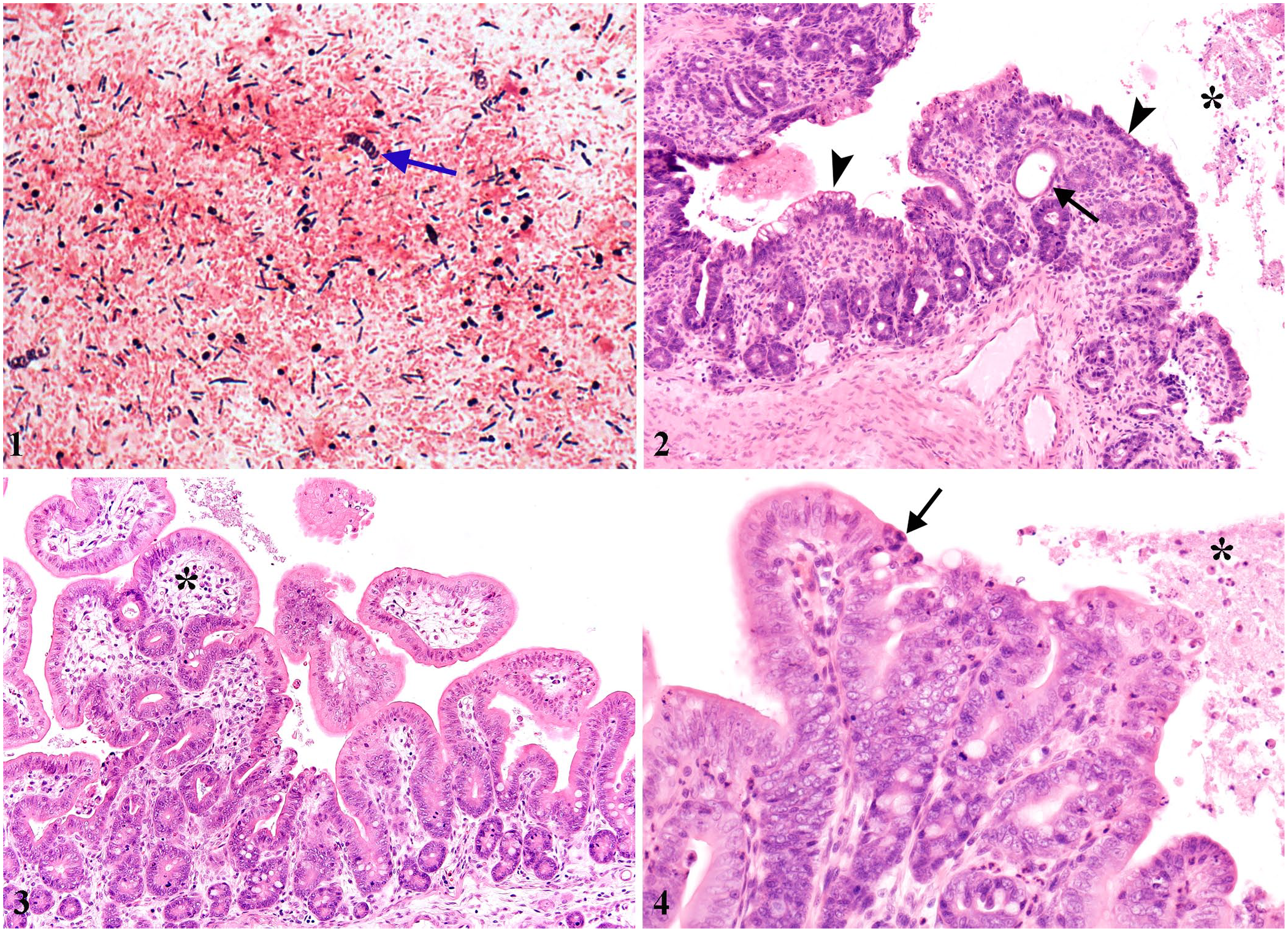
Intestinal smear from a rabbit with Clostridium spiroforme infection. Gram-positive, helically coiled rods (arrow) compatible with C. spiroforme admixed with large numbers of gram-negative straight rods and lesser numbers of gram-positive straight rods. Gram stain.
An autopsy had been performed on each carcass. Samples of heart, liver, kidney, spleen, stomach, small intestine, colon, cecum, brain, and lungs were collected in all of the cases and fixed by immersion in 10% neutral-buffered formalin (pH 7.2), for 24–72 h; 4-µm sections were routinely stained with H&E. Several ancillary tests were performed following CAHFS standard operating procedures (Suppl. Table 2). Briefly, these tests included examination of Gram-stained or wet fecal or intestinal mucosal smears, fecal flotation, aerobic and anaerobic culture of liver, lung, small and large intestinal content, Salmonella culture and/or PCR assay, and/or virus isolation, and/or direct electron microscopy (DEM) of intestinal content or feces. Aerobic and anaerobic culture was done on Columbia blood agar (Hardy) and incubated for 48 h in aerobic or anaerobic conditions, respectively. Isolated colonies were first identified by morphology and confirmed by conventional biochemical tests or MALDI-TOF MS (Bruker). The final diagnoses of the cases were evaluated based on the autopsy reports, with re-evaluation of histologic slides available by one of the authors (L Tuomisto). Also, a C. spiroforme PCR assay was performed on formalin-fixed, paraffin-embedded (FFPE) tissues containing small and large intestine, which were available from 8 rabbits from 6 different farms (Suppl. Table 2). DNA was extracted (QIAamp DNA FFPE tissue kit; Qiagen), according to manufacturer’s instructions. The extracted DNA was used as template for PCR amplification of a 254-bp fragment of the C. spiroforme toxin genes, using the primers F (5′-CGGAGATCTGGACCCCAAGA-3′) and R (5′-TACTCGCCTGCATAACCTGG-3′). PCR was performed in a total volume of 25 µL containing 0.5 µL of each primer (0.5 µM), 5 µL of extracted DNA, 7 µL of nuclease-free water, and 12 μL of master mix (2×, Platinum II hot-start green PCR master mix; Thermo Fisher). Thermocycler profiles were as follows: 95°C for 10 min, 35 cycles of 95°C for 45 s, 60°C for 45 s, and 72°C for 45 s, and a final extension step at 72°C for 5 min. Samples were held at 4°C. PCR amplicons were visualized in ethidium bromide–stained 1.5% agarose gels (Agarose SFR; Amresco). DNA extracted from C. spiroforme strain NCTC 11211 was used as a positive control, and DNA extracted from intestinal tissue of a healthy rabbit was used as a negative control.
Results
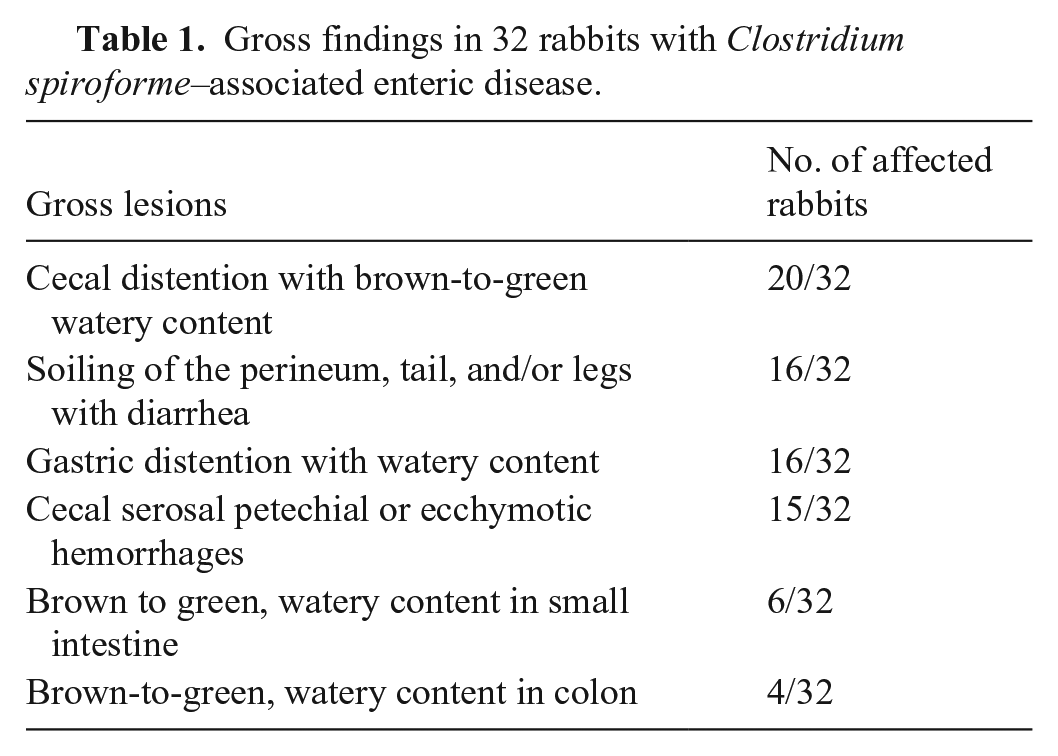
Briefly, gross findings included cecal distention with brown-to-green, watery content (20 of 32); soiling of the perineum, tail, and/or hind legs with diarrhea (16 of 32); distention of the stomach with watery content (16 of 32); serosal petechiae or ecchymoses in the cecum (15 of 32); and brown or green, watery content in the small intestine (6 of 32), colon (4 of 32), or both (1 of 32; Table 1; Suppl. Table 3). Gross findings in 2 rabbits were not detailed, although inflammatory changes in the cecum and liquid feces consistent with diarrhea were mentioned in the report. Seven rabbits were in good nutritional condition; for the remainder, nutritional condition was not reported.
Gross findings in 32 rabbits with Clostridium spiroforme–associated enteric disease.
Briefly, the main microscopic findings were in small intestine, cecum, and colon (Table 2; Suppl. Table 4). The most common finding was necrotizing enteritis (19 of 32), which was characterized by mucosal necrosis and variable heterophilic inflammation. Other histologic findings included necrotizing or pleocellular typhlitis (6 of 32), necrotizing or heterophilic typhlocolitis (6 of 32; Figs. 2–4), cecal mucosal or submucosal edema (8 of 32), and cecal mural hemorrhages (5 of 32). Small-to-large numbers of intraluminal, coiled, gram-positive bacilli were detected in the intestinal lumen of 7 rabbits. Small numbers of coccidian organisms were observed in the cecum of 2 rabbits, and large numbers of coccidian oocysts and schizonts in the small intestine of 5 rabbits.
Microscopic findings in 32 rabbits with Clostridium spiroforme–associated enteric disease.

Fecal or intestinal mucosal smears from all of the rabbits were examined. In all cases, there were coiled gram-positive bacilli without recognizable spore formation (Fig. 1). Bacterial culture in 14 rabbits yielded mixed bacterial growth. C. spiroforme was isolated from the intestinal content of 2 rabbits. Untyped E. coli was isolated from the intestinal content of 7 rabbits. No Salmonella spp. were isolated or detected by PCR in any of the cases. Coccidian oocysts were detected in wet intestinal smears or fecal flotation in 18 rabbits. The number of coccidian oocysts was small in 4 rabbits, moderate in 9, and large in 1 rabbit; in 4 rabbits, the number was not specified. Coccidian organisms were detected only in wet intestinal smears or fecal flotation in 13 of 32 rabbits, only in histologic sections in 2 of 32 rabbits, and both in wet intestinal smears or fecal flotation and histologic sections in 5 of 32 rabbits. Calicivirus-like particles were detected by DEM in feces of one rabbit. No viral agents were isolated from any animal. ELISA for rotavirus performed on intestinal content of one rabbit was negative.
C. spiroforme DNA was detected by PCR in FFPE samples of small and large intestine in rabbits from 6 different rabbitries. Histologically, 6 of these rabbits had typhlitis with necrosis or mixed inflammation and 2 of the rabbits had necrotizing or heterophilic typhlocolitis. Five rabbits also had cecal mucosal and/or submucosal edema or cecal mural hemorrhages or both.
Discussion
C. spiroforme typically infects young rabbits, most frequently preweaning and growing individuals, although CSAED has been reported occasionally in adults.9,12,15 In our study, more than half of the cases were juvenile (17 of 32), but a few adults (6 of 32) were also affected. However, in a large number of the rabbits (24 of 32), an exact age was not reported. Typical gross findings reported in our study are similar to those described in previous reports and included soiling of the perineal region, tail, and/or hindlimbs with diarrhea; gastric and cecal distention with brown-to-green, watery content; and cecal hemorrhages.6,11,12 Microscopic changes were most common in the small intestine and consisted of necrotizing enteritis. In the large intestine, the most common findings were necrotizing, pleocellular, and/or heterophilic typhlitis or typhlocolitis; and cecal mural hemorrhages and mucosal and/or submucosal edema. These gross and microscopic changes are compatible with CSAED produced by C. spiroforme.6,11,12 Gram-positive helically coiled bacteria were detected in the intestinal or fecal smears of all rabbits. This typical morphology of C. spiroforme can be better observed in smears than in histologic sections. Therefore, it is critical to prepare intestinal mucosal smears for Gram staining during the autopsy of rabbits with presumptive CSAED. The curved gram-positive bacilli observed in histologic sections are suggestive of, but not specific for, C. spiroforme.
C. spiroforme has been described both as an only pathogen and with other concurrent pathogens, including enteropathogenic E. coli, Eimeria spp., and rotavirus A in cases of CSAED.9,11,12,15 Although concurrent infections with other pathogens occur, C. spiroforme is thought to play a significant role in the development of CSAED. 11 The most common concurrent pathogen in our study was Eimeria spp., which was detected in more than half of the rabbits; most of the affected rabbits had mild-to-moderate infection. Coccidial infections could have partly contributed to intestinal changes in these rabbits. E. coli was the second most common microorganism detected. Molecular serotyping of E. coli isolates was not performed, but histologically typical gram-negative coccobacilli adhering to intestinal mucosa suggesting attaching-effacing E. coli (AEEC) were not observed in any of the animals, suggesting that the isolates were most likely commensal strains. With one exception, no viruses were detected in intestinal samples. Absence of other significant intestinal pathogens in several cases suggests that the intestinal changes described were primarily associated with C. spiroforme infection. In our study, although C. spiroforme–like bacteria were detected on intestinal smears of all 32 rabbits, C. spiroforme could only be isolated from 2 rabbits, which highlights the low sensitivity of anaerobic culture for the isolation of this microorganism. However, PCR detection of C. spiroforme performed on FFPE tissues from 8 rabbits yielded positive results in all 8 cases tested. Unfortunately, FFPE tissues from the remaining cases were no longer available, and PCR could not be performed. Based on our results on a limited number of samples, PCR could be a sensitive method of detection of C. spiroforme.
Normal intestinal microbiota is thought to act as a microbial barrier, and any disbalance of this microbiota secondary to antimicrobial treatment, lactation, weaning, concurrent infections, or altered diets (including high carbohydrate intake) may trigger CSAED.4,12,15,16 It is important to recognize risk factors for CSAED, especially when large numbers of animals are affected. In young animals, a less-developed immune system, higher gastric pH, and carbohydrate-dense diets, especially in farmed rabbits, could contribute to intestinal dysbiosis. 9 No seasonal variance of C. spiroforme infections has been observed in previous studies. 9
A diagnosis of CSAED can be made based on clinical signs, gross and microscopic postmortem findings, and visualization of characteristic helically coiled, gram-positive bacilli microscopically in fecal or intestinal mucosa smears.2,4,7,11 Isolation of C. spiroforme in culture or detection of C. spiroforme DNA with PCR supports the diagnosis.7,11 However, consistent with our results, C. spiroforme is a fastidious microorganism and is difficult to culture, which can result in unsuccessful isolation even if characteristic bacteria have been visualized in the intestinal content.1,11 As demonstrated in our study, in addition to anaerobic culture, PCR, even from FFPE tissues, can be used for detection of C. spiroforme. However, larger sample sizes are required to compare the sensitivity of PCR and anaerobic culture. Although detection of CST in intestinal content is also considered confirmatory for the disease, no commercial tests are available for detection of this toxin. 7 Thus, PCR detection in fresh or FFPE tissues should be included in the diagnostic workup of suspected CSAED cases.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241257676 – Supplemental material for Clostridium spiroforme–associated enteric disease in domestic rabbits: a retrospective study of 32 cases in California, 1992–2019, and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241257676 for Clostridium spiroforme–associated enteric disease in domestic rabbits: a retrospective study of 32 cases in California, 1992–2019, and literature review by Laura Tuomisto, Mauricio A. Navarro, Fábio S. Mendonça, Arturo Oliver-Guimerá, M. Isabel Casanova, Kevin Keel, Javier Asin, Denise Imai, Simone Stoute, Aslı Mete and Francisco A. Uzal in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Seana M. Fitisemanu (CAHFS San Bernardino, UCDavis) for data collection and our histotechnology team for preparing the histologic samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
