Abstract
Clostridium perfringens produces enteric diseases, generically called enterotoxemias, in sheep, goats, and other animals. This microorganism can be a normal inhabitant of the intestine of most animal species, including humans, but when the intestinal environment is altered by sudden changes in diet or other factors, C. perfringens proliferates and produces potent toxins that act locally or are absorbed into the general circulation with usually devastating effects on the host. History, clinical signs, and gross postmortem findings are useful tools for establishing a presumptive diagnosis of clostridial enterotoxemia in sheep and goats. Definitive diagnosis requires laboratory confirmation. Isolation of some types of C. perfringens (e.g., B and C) can be of diagnostic value, but other types (e.g., A) are so commonly found in the intestine of normal animals that isolation is meaningless from a diagnostic point of view. The most accepted criterion in establishing a definitive diagnosis of enterotoxemia is detection of C. perfringens toxins in intestinal contents. Also, histopathological examination of brain is very useful for diagnosis of type D disease, as lesions produced by epsilon toxin in the brains of sheep and goats are pathognomonic for type D enterotoxemia. Ancillary tests, such as measuring urine glucose or observing Gram-stained smears of intestinal mucosa, can be used. However, although such tests have a presumptive diagnostic value when positive, they cannot be used to rule out a diagnosis of enterotoxemia when negative.
Introduction
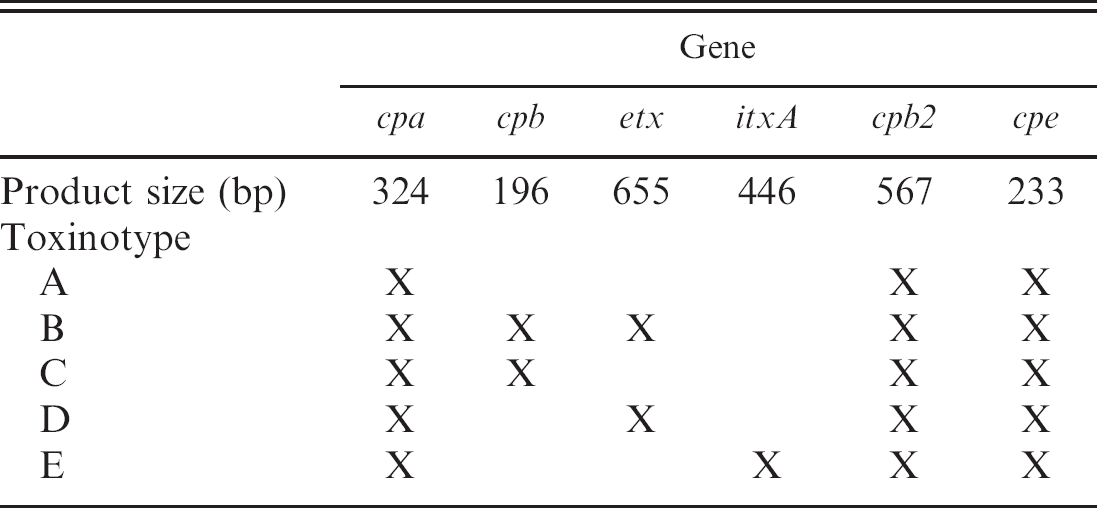
Clostridium perfringens is classified into 5 toxinotypes (A, B, C, D, and E) according to the production of 4 major toxins, namely alpha (CPA), beta (CPB), epsilon (ETX), and iota (ITX; Table 1). 52,54 However, C. perfringens can produce up to 15 toxins in various combinations, including lethal toxins such as perfringolysin O (PFO), enterotoxin (CPE), and beta2 toxin (CPB2). 33,55,59 Colonies of all toxinotypes of C. perfringens after 24 hours of incubation are usually 3–5 mm in diameter, grayish, and circular. An inner, complete zone of hemolysis is caused by PFO and the less complete outer zone is caused by CPA (Figs. 1, 2).
Enteric C. perfringens infections in sheep, goats, and other species are generically called enterotoxemias because toxins produced in the intestine may be absorbed into the general circulation (Table 2). However, although this is true for many of the C. perfringens toxins, some toxins produced in the intestine act only locally (Table 2). Proliferation and toxin production often follow sudden changes in diet or other factors. On smears of intestinal mucosa of animals with enterotoxemia, C. perfringens appears as Gram-positive bacilli with rounded edges, a few of which may have spores.
History, clinical signs, and gross postmortem findings are useful in establishing a presumptive diagnosis of clostridial enterotoxemia, but confirmation requires laboratory testing (Table 3). In particular, detection of C. perfringens toxins in intestinal content and quantitative culture followed by genotyping are, as explained later in this review, particularly useful to confirm a diagnosis of enterotoxemia.
Toxin detection can be performed by several techniques, although only a handful of commercial methods are currently available for this purpose. In recent years an enzyme-linked immunosorbent assay (ELISA) kit a that detects CPA, CPB, ETX, and C. perfringens (the bacterium itself) has been used extensively in the author's laboratory. At least one other commercial kit b is available for detection of CPE. To the authors' knowledge, no systems are commercially available for detection of ITX or CPB2.
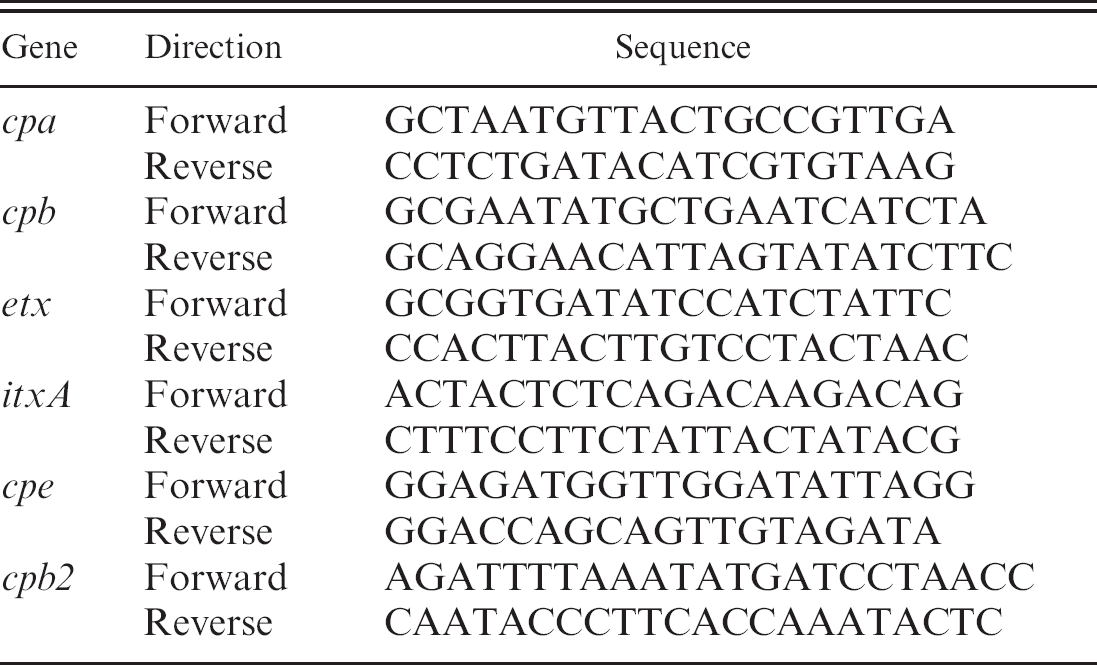
Polymerase chain reaction (PCR) genotyping 44 provides a useful alternative to in vivo toxin neutralization tests for typing of C. perfringens isolates. Genotype can, in many cases, provide the final piece of information needed to establish a diagnosis. Sequences for forward and reverse primers for detection of toxin genes are presented in Table 4 (detailed instructions for performing the multiplex PCR assay can be found at http://microvet.arizona.edu). Template is prepared by suspending 1 or 2 colonies in 200 ml of high-performance liquid chromotography–grade water, boiling for 20 min, and centrifugation (5 min, 13,000 X g). Each 50-μl reaction contains 10X assay buffer (100 mM of Tris-HCl, pH8.3 [at 25°C], 500 mM of KCl, 15 mM of MgCl2, 0.01% gelatin [wt/vol]), 0.12 mM of dNTP, 0.34 mM of each cpe primer, 0.36 mM of each cpb primer, 0.36 mM of each cpb2 primer, 0.44 mM of each etx primer, 0.5 mM of each cpa primer, 0.52 mM of each itxA primer, and 5 U Taq DNA polymerase. c After 35 cycles (1 min at 94°C, 1 min at 55°C, 1 min at 72°C), with an additional 5 min extension at 72°C, products are separated by electrophoresis in a 1.5% agarose gel. Staining is with ethidium bromide, followed by observation via ultravioled transillumination. Product sizes and genotypes are given in Table 5. The main criteria for diagnosis of infections by C. perfringens intestinal infections in sheep and goats are presented in the current review.
Major toxins produced by Clostridium perfringens.

Clostridium perfringens type A
Clostridium perfringens type A is implicated in ovine and caprine enterotoxemia in some parts of the world, 2,35 but it is rarely responsible for intestinal diseases in sheep and goats in North America. Type A causes yellow lamb disease, 43 a rare form of acute enterotoxemia in lambs. Clostridium perfringens type A produces CPA and can also produce several of the nontyping toxins, including CPE and CPB2. Information about pathogenesis of type A enteric infections in ruminants is minimal and often contradictory, but it is generally assumed that most clinical signs and lesions are due to the effects of CPA.
The CPB2-producing C. perfringens type A has also recently been linked to disease in several animal species, including sheep and goats. 12,24,34,75 However, most evidence implicating CPB2 in pathogenesis of ovine or caprine infections is based on isolation of CPB2-positive C. perfringens from sick animals. The condition has not been reproduced experimentally with CPB2-producing strains, and the importance of including the gene encoding for CPB2 production detection (in vivo or in vitro) in the diagnostic definition of type A infections is unknown.
Clinical history and clinical signs
It is generally assumed that factors predisposing to onset of other C. perfringens type enterotoxemias (i.e., sudden changes to diets rich in carbohydrates) can also predispose type A infections. Yellow lamb disease is characterized clinically by depression, anemia, icterus, and hemoglobinuria. Descriptions of type A caprine enterotoxemia are few and poorly documented; definitive confirmation of the role of this organism in disease production is uncommon. 2,35,60
Necropsy findings
Necropsy findings in type A enterotoxemia of sheep are not specific, and a diagnosis of this condition should not be based solely on gross changes. To the authors' knowledge, there are only 2 published reports describing gross and histopathological changes in lambs with type A enterotoxemia. 22,43 According to these authors, ovine type A enterotoxemia is characterized by generalized icterus and enlarged, pale, and friable liver and spleen. Red urine can be found in the urinary bladder because of hemolysis and hemoglobinuria. Histopathological changes include periacinar necrosis of the liver, splenic congestion, nephrosis with hemoglobin casts, and pulmonary congestion and edema. 43
Enterotoxemia associated with CPB2-positive C. perfringens type A was recently described in a goat kid. 24 Gross and histologic lesions included congested and edematous small and large intestine, but pathology of type A disease in goats is otherwise not well documented.
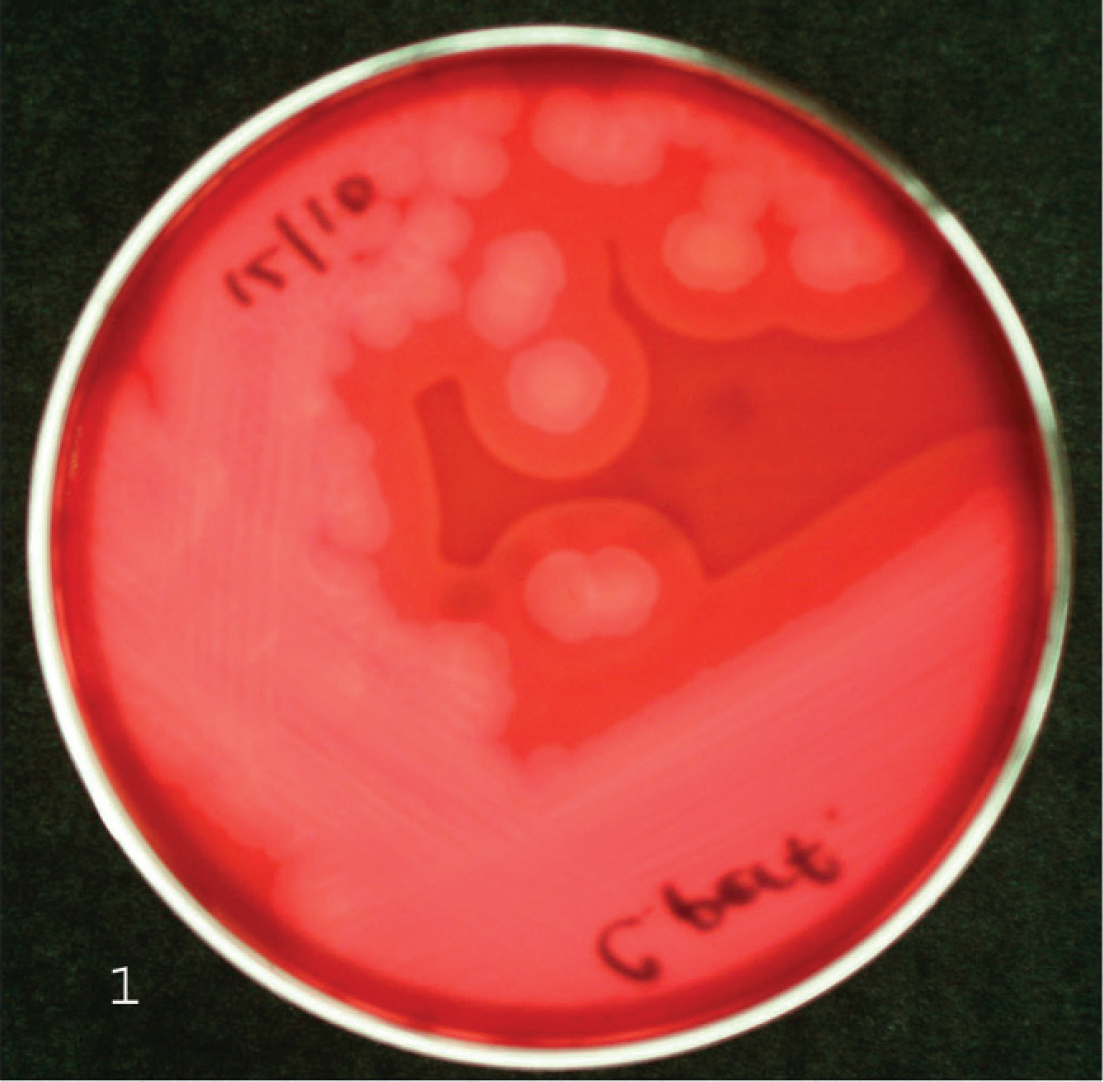
Clostridium perfringens cultivated on blood agar. Surrounding the colonies, an inner, complete zone of hemolysis is caused by perfringolysin O and the less complete outer zone is caused by C. perfringens alpha toxin.

Clostridium perfringens cultivated on egg yolk agar. The left area of the streak has been treated with C. perfringens alpha toxin (CPA) antibodies before inoculation with C. perfringens, hence inhibiting the lecithinase effect of CPA on the egg yolk. This effect can be observed on the right side of the streak, where no antibodies were applied.
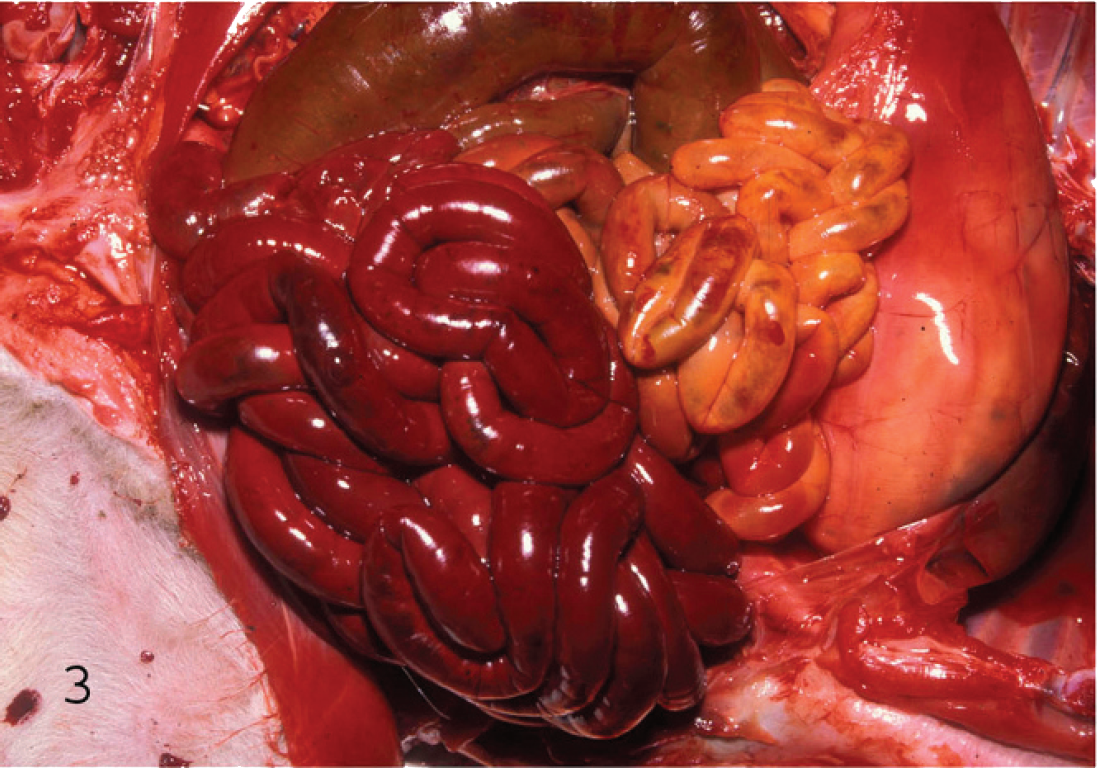
Serosal view of the small intestine of a lamb with hemorrhagic enteritis due to Clostridium perfringens type C. The intestine is distended with gas and hemorrhagic fluid.
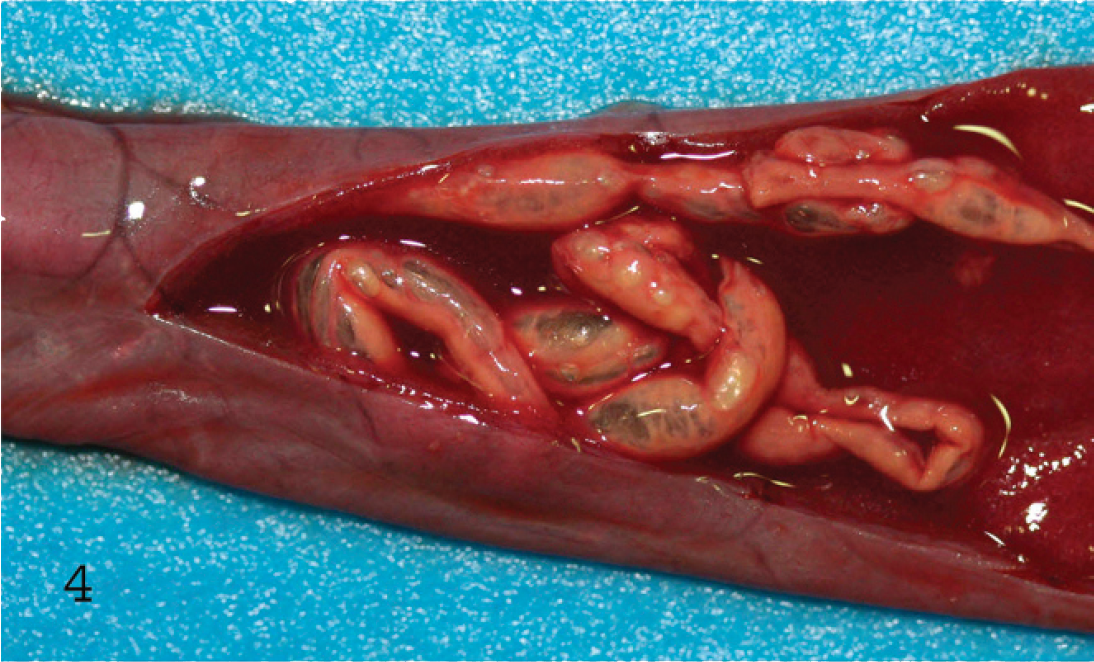
Mucosal view of the small intestine of a lamb with hemorrhagic enteritis due to Clostridium perfringens type C. Observe the dark red necrotic mucosa and the large amount of fibrin present in the lumen.
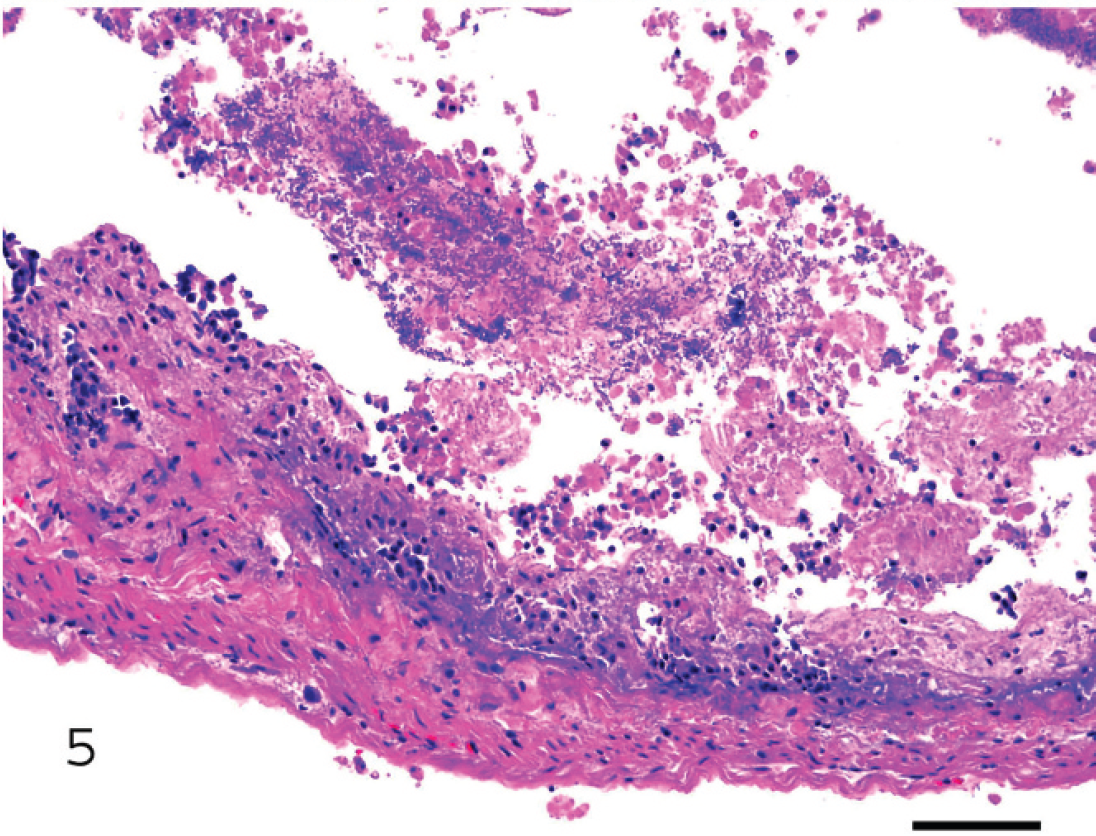
Small intestine of a lamb with hemorrhagic enteritis due to Clostridium perfringens type C. Observe the almost complete necrosis of the epithelium and the presence of bacilli mixed with fibrin, desquamated cells, and leukocytes in the lumen. Bar = 100 μm.
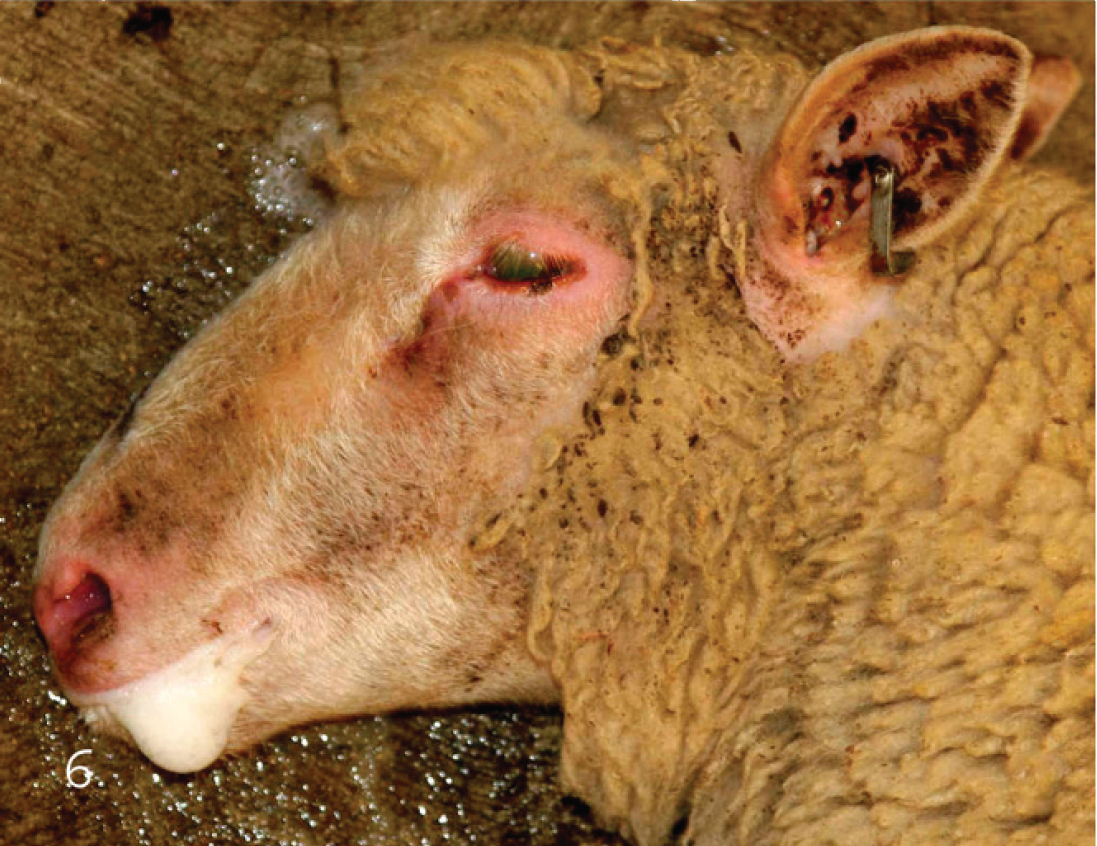
Recumbent lamb with acute type D enterotoxemia. Observe frothing by the mouth.
Main intestinal diseases produced by Clostridium perfringens toxinotypes in sheep and goats.

Very little information has been published on this disease in this animal.
NR = not reported.
Bacteriologic culture
Toxinotype A is the C. perfringens type found most frequently in the intestine of healthy sheep and goats (Uzal FA, Marcellino RB: 2002, Clostridium perfringens in clinically healthy sheep of Patagonia, Argentina. In: Proceedings of the 6th Biennial Congress of the Anaerobe Society of the Americas, p. 116. Anaerobe Society of the Americas, Los Angeles, CA), so mere isolation from intestinal contents of sheep and goats is not diagnostic for type A disease. However, quantitation of C. perfringens in intestinal content may provide information useful in diagnosis. 21,64,65,68 This seems to be true for type A and type D enterotoxemias of lambs, in which elevated numbers of C. perfringens are found in intestinal contents (usually 10 4 –10 7 colony-forming units [CFU]/g). 53,59 However, there is no evidence to support this assertion for infections by type A in goats.
Toxin detection
In most cases of C. perfringens enterotoxemia, detection of preformed toxins in intestinal content is very helpful in establishing a final diagnosis, but this is not usually the case for type A disease. Methods to detect C. perfringens toxins vary greatly in sensitivity and specificity. For instance, some ELISAs have relatively high sensitivity and can detect small amounts of CPA in intestine of clinically normal animals (authors' unpublished observation). This increases the difficulty in interpreting results, because threshold CPA concentrations, which allow distinction between clinically normal sheep and goats and those with type A enterotoxemia, are unknown. In these cases, use of the conventional mouse neutralization test (MNT) seems to be more appropriate; it is less sensitive and therefore less likely to detect small amounts of CPA in the intestine of healthy animals (authors' unpublished observation). In summary, a positive CPA test in intestinal content is consistent with, but by no means confirmatory of, a type A infection. A negative CPA test in intestinal content rules out a diagnosis of type A infection. In general, diagnosis of type A enterotoxemia is best based on the confluence of clinical and laboratory findings and may be most effective when approached on a herd, rather than an individual animal basis.
Clostridium perfringens types B and C
Clostridium perfringens beta toxin is considered the main virulence factor in type C infections 31,56 (CPB and ETX seem to be necessary for type B disease) 27 ; it is very sensitive to trypsin digestion, and animals with low levels of intestinal trypsin (such as newborns) are usually the most susceptible to infection by C. perfringens isolates producing CPB. 56,59 In contrast, ETX requires proteolysis via trypsin, or other intestinal or bacterial proteases, to become completely active. 46 These opposing effects of trypsin on ETX and CPB activity may indicate that when both toxins are present in the intestine during type B–associated infections, different intestinal conditions might select for the predominant activity of ETX over CPB or vice versa. In sheep, C. perfringens types B and C produce similar diseases that are characterized by sudden death or acute neurologic signs with or without hemorrhagic diarrhea. 40,59,60
Samples, condition for submission, and tests for the diagnosis of Clostridium perfringens enterotoxemias in sheep and goats.*
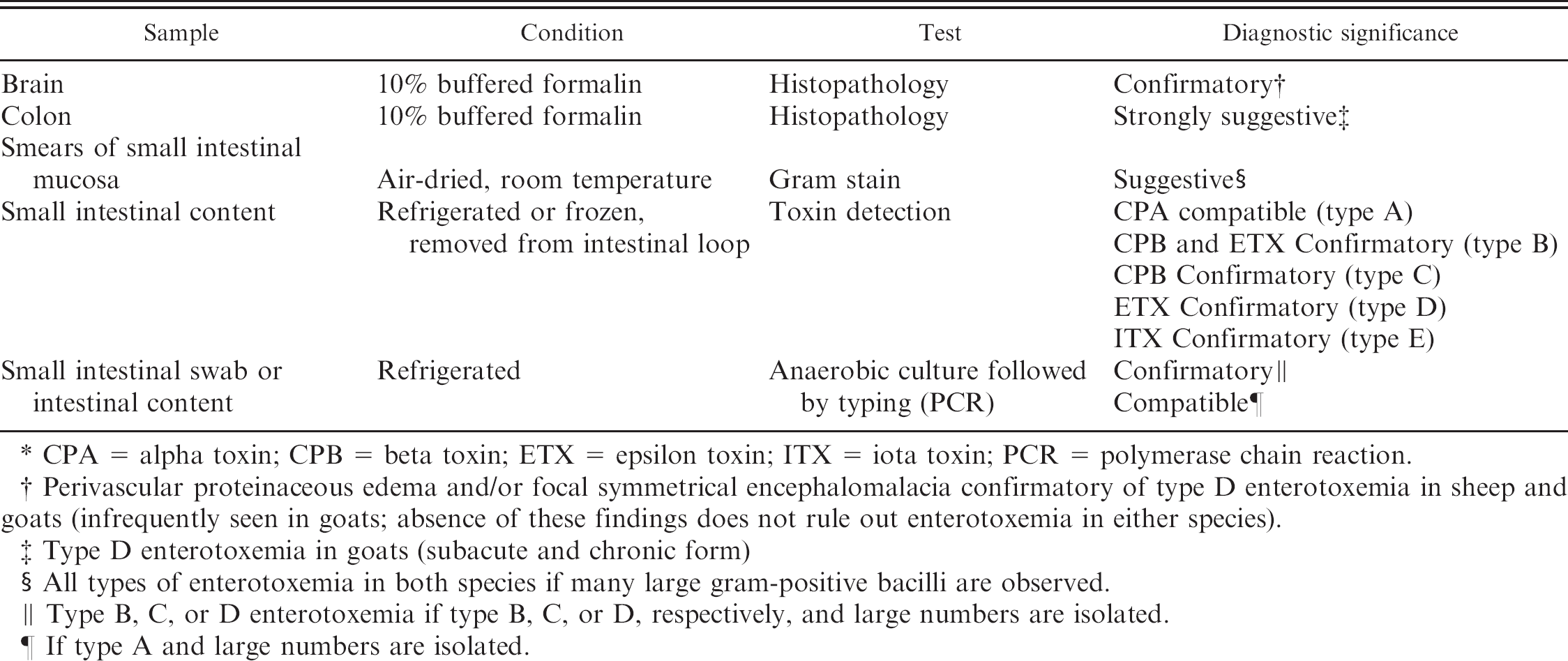
CPA = alpha toxin; CPB = beta toxin; ETX = epsilon toxin; ITX = iota toxin; PCR = polymerase chain reaction.
Perivascular proteinaceous edema and/or focal symmetrical encephalomalacia confirmatory of type D enterotoxemia in sheep and goats (infrequently seen in goats; absence of these findings does not rule out enterotoxemia in either species).
Type D enterotoxemia in goats (subacute and chronic form)
All types of enterotoxemia in both species if many large gram-positive bacilli are observed.
Type B, C, or D enterotoxemia if type B, C, or D, respectively, and large numbers are isolated.
If type A and large numbers are isolated.
Primers for detecting Clostridium perfringens toxins of primary importance.
Clinical history
Type B infections have been reported in Europe, South Africa, and the Middle East, usually in lambs younger than 14 days, and the condition is referred to as lamb dysentery. 1 A more chronic form in older lambs has been described in England, where it is known as pine. 1 Most type C infections occur in neonatal lambs and are referred to as hemorrhagic enteritis. It is usually assumed that this age predisposition is related to low levels of trypsin in the intestinal tract. Another form of type C infection (struck) occurs in adult sheep, usually beginning as an infection of damaged ruminal mucosa. 40 Reports of type B and C disease in goats are scanty and poorly documented. 2,35,60
Clinical signs
In lambs with dysentery, death may occur without premonitory signs, although there is usually abdominal pain with distended tympanic abdomen and bloody diarrhea. 1 The more chronic disease of older lambs caused by type B (pine) is characterized by loss of condition and depression, reluctance to suckle, and a peculiar stretching when the animals rise 1 ; these clinical signs are usually attributed to the action of ETX on the central nervous system. In goats, few descriptions of enterotoxemia by C. perfringens types B and C have been documented, 2,35,60 and the clinical signs described are much like those in lamb dysentery.
Polymerase chain reaction product sizes and genotypes.
Necropsy findings
Infections by types B and C in sheep result in similar intestinal lesions consisting of diffuse or multifocal hemorrhagic and necrotizing enteritis, predominantly in ileum, which can be observed grossly as blood- and fibrin-filled loops (Figs. 3, 4). 1 An excess of serosanguineous fluid in the abdominal cavity is frequently seen. 1 Older animals surviving type B hemorrhagic enteritis for a few days may develop focal symmetrical encephalomalacia (FSE; see below) due to the action of ETX. 41 These gross postmortem changes can be very striking and strongly suggest the corresponding forms of enterotoxemia. However, these changes are not specific, and a diagnosis of type B or C enterotoxemia should not be based solely on gross changes. Pathology of caprine type B and type C infection is not well documented. 2,11,23,35
Microscopic lesions
Histopathological changes in caprine and ovine type B and C enterotoxemia are not specific but can be highly suggestive of these infections. The changes consist of acute necrosis of the intestinal mucosa. In most cases this is coagulative necrosis, but if the animals survive for a longer period, complete loss of the mucosa is observed (Fig. 5). Thrombosis of mucosal and submucosal vessels is a common finding, and inflammation is usually not a striking component, although it might be more marked in subacute cases. Diffuse or multifocal distribution of Gram-positive bacilli in the intestinal lumen is common (Fig. 5); although they may be in close contact with the mucosa, and attachment has been suggested, no definitive evidence of attachment has been demonstrated in C. perfringens infections in ruminants. Recently, it has been shown that C. perfringens can move by attachment and retraction of type IV pili, which pull the bacterium toward the site of attachment; this provides C. perfringens with an unusual form of gliding motility. 74
Bacteriologic culture
Some still believe isolation of types B and C from intestinal content has diagnostic value. 62 This belief is undermined to an extent by the fact that types B and C have been found in the intestinal content of a small proportion of healthy sheep (Uzal FA, Marcellino RB: 2002, Clostridium perfringens in clinically healthy sheep). 64 However, isolation from clinically normal animals is rare and in low numbers, so isolation of large numbers of types B or C, especially in pure culture, has excellent diagnostic value in the presence of appropriate clinical signs and lesions.
Toxin detection
Detection of CPB in intestinal content of sheep and goats is diagnostic for types B and C enterotoxemia. 62 Differentiation between these two should be based on detection of ETX in cases of type B enterotoxemia. 62 Failure to detect these toxins does not eliminate a diagnosis of enterotoxemia. This is particularly true for CPB, which is very sensitive to the action of trypsin and is destroyed rapidly in the intestine. 62 Because of the sensitivity of CPB to trypsin, it is possible that a type B infection (produces CPA, CPB, and ETX) could be diagnosed as type D (produces CPA and ETX) if CPB has been destroyed by the action of intestinal trypsin and the diagnosis is based solely on toxin detection. That is why a final diagnosis of type B and C infections should not be based solely on toxin detection and should be accompanied by pathological as well as microbiological findings.
Clostridium perfringens type D
Toxinotype D is by far the most common form of clostridial enterotoxemia in sheep and goats. Pathogenesis of disease caused by C. perfringens type D is mostly mediated by ETX. This metabolite is one of the most potent bacterial toxins, with a mouse lethal dose of 100 ng/kg. 55 It is produced as an inactive prototoxin 42 and converted to the >1,000-fold more toxic form when cleaved by a protease from C. perfringens 45 or from the host. 5,78 Epsilon toxin binds with high affinity to rat brain synaptosomes, 49 and limited evidence suggests that the receptor is a sialoglycoprotein. 47,48,49 Vascular endothelial tight junctions in the brain degenerate, 18 causing swelling and rupture of perivascular astrocyte processes. 28 Increased capillary permeability, 29 rapid extravasation of fluid, 30 elevated intracerebral pressure, and parenchymal necrosis follow. 18
Clinical history
Most cases of type D enterotoxemia in sheep relate to sudden changes in diet, usually to feeds rich in highly fermentable carbohydrates 7,40,68 ; as a consequence, large amounts of undigested carbohydrate enter the small intestine. This is often associated with introduction of animals to feedlots without progressive adaptation to grains or concentrated rations and access to lush grass after rains in spring and autumn. 14 Other factors that disturb the intestinal environment (i.e., heavy parasite infestation [King NB: 1980, Pregnancy toxaemia and enterotoxemia. In: Proceedings, Refresher Course for Veterinarians, pp. 209–212. Post-Graduate Foundation, Sydney, Australia], 26,63 dosing with phenothiazine, 38 overdosing with netobimin 72 ) may predispose to type D enterotoxemia, but convincing evidence has not been published. Even less is known about factors that predispose goats to type D enterotoxemia, and cases have occurred in goats fed a regular hay diet for several months (authors' unpublished observation). Thus, a history of sudden dietary change is a useful indicator of possible type D enterotoxemia, but the absence of this precedent does not preclude a diagnosis of this disease in sheep or goats.
Vaccines against type D are among the most frequently used biologics in sheep and goats worldwide. Therefore, animal owners and veterinarians frequently use vaccination history to rule out infections by type D. However, vaccine quality varies greatly among countries and manufacturers, and vaccines are not always correctly transported, stored, and/or administered. In addition, individual variation in antibody responses between animals occurs frequently in sheep and goats. 8,15,16,66 History of vaccination as an indicator of possible disease is even less useful in goats than in sheep because antibody titers are believed to be of lower levels and shorter duration in goats. 66
Clinical signs
Clinical signs are, at best, suggestive of type D infections in sheep and goats, and no final diagnosis can be based on clinical grounds only. Type D produces an acute, subacute, or chronic neurologic condition in sheep, characterized by sudden death or neurologic and respiratory signs, including blindness, opisthotonos, convulsions, bleating, frothing by the mouth (Fig. 6), and recumbency with paddling immediately before death. Diarrhea is occasionally observed, although this is not a common clinical sign in sheep. 40
In goats, type D produces acute, subacute, or chronic disease as well. The acute form occurs more frequently in young, unvaccinated animals and is clinically similar to the acute disease in sheep. 7,9 The subacute form is more frequently seen in adult goats, 58 vaccinated or not, and is characterized by hemorrhagic diarrhea, abdominal discomfort, severe shock, opisthotonos, and convulsions. The disease may result in death 2–4 days after onset, 3,9 but some animals recover. Adult animals, often vaccinated, can also exhibit chronic disease, 58 which is characterized by profuse, watery diarrhea (often containing blood and mucus), abdominal discomfort, weakness, anorexia, and agalactia in milking does. 7 This chronic form may last for days or weeks 57 and may culminate in either death or recovery. 10
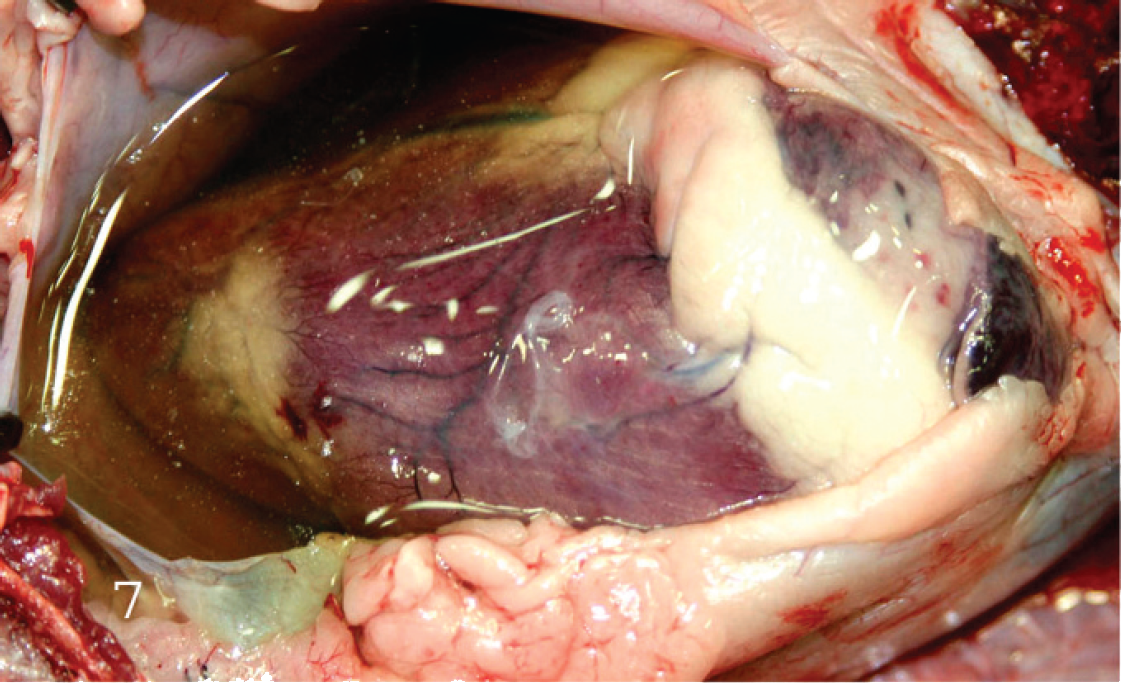
Heart of a lamb with Clostridium perfringens type D enterotoxemia. The pericardial sac has been opened and there is a large amount of pericardial fluid and presence of fibrin strands.
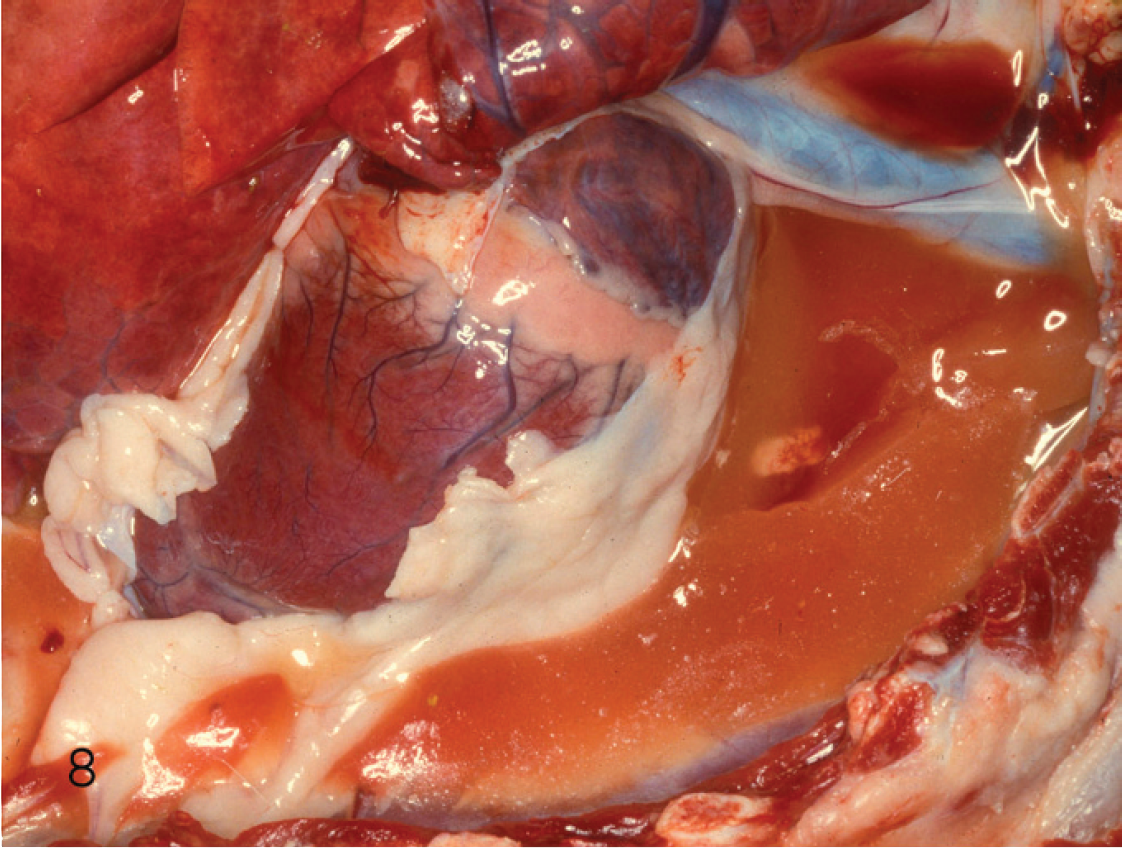
Clotted pleural fluid of a goat kid with acute type D enterotoxemia.
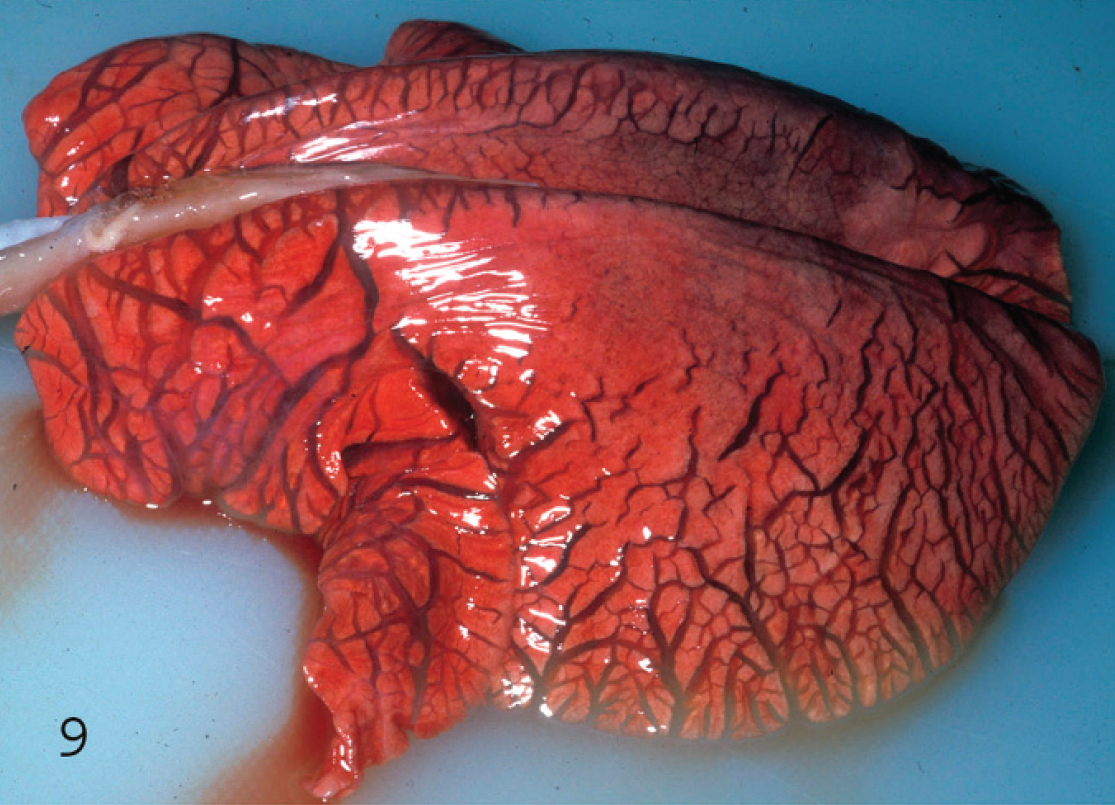
Interstitial lung edema in a goat kid with acute Clostridium perfringens type D enterotoxemia. Notice the widened interlobular septa.
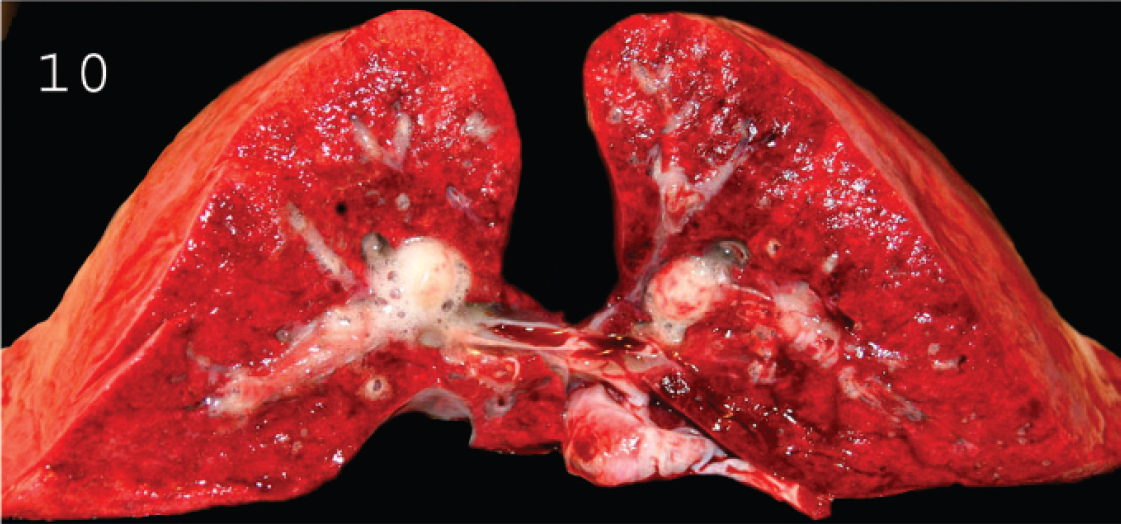
Large amount of froth in the airways of a lamb with type D enterotoxemia, indicating lung edema.
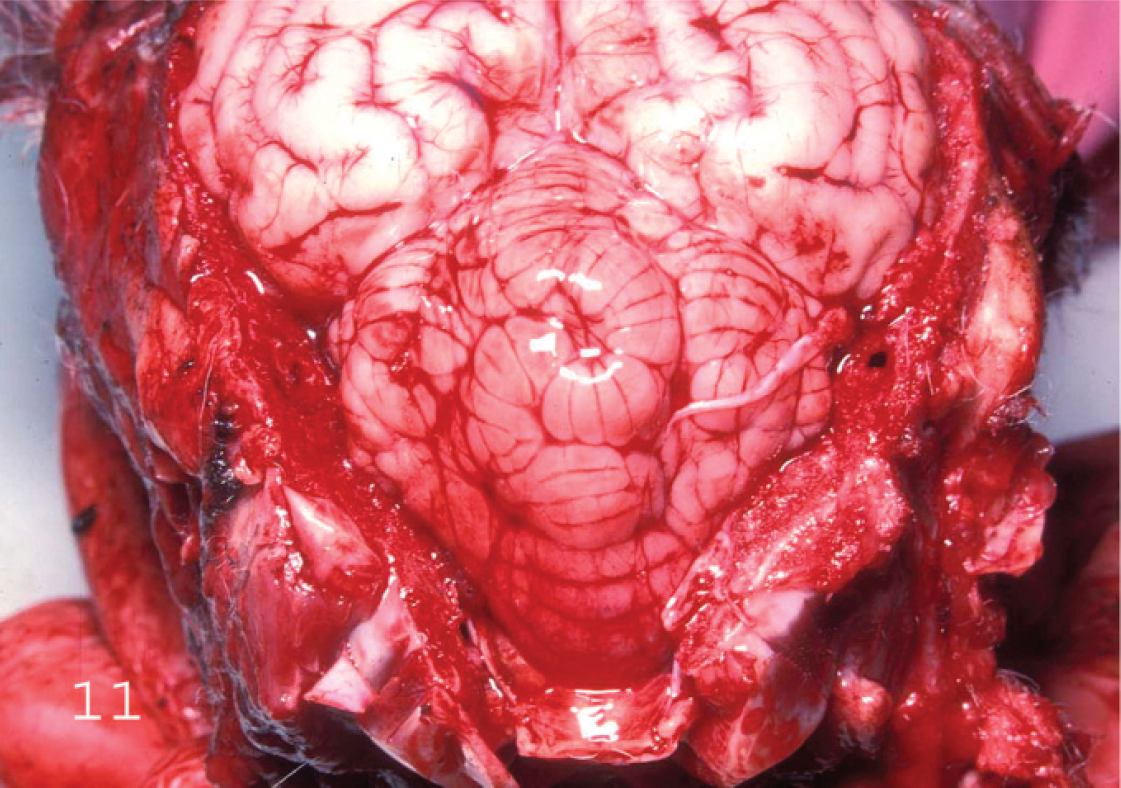
Brain of a lamb with Clostridium perfringens type D enterotoxemia showing cerebellar vermis hernia. Notice that the posterior end of the cerebellar vermis extends backward into the foramen magnum of the occipital bone. There is also superficial hemorrhage on the posterior end of the cerebellar vermis caused by the rubbing of this structure against the occipital bone at the foramen magnum.
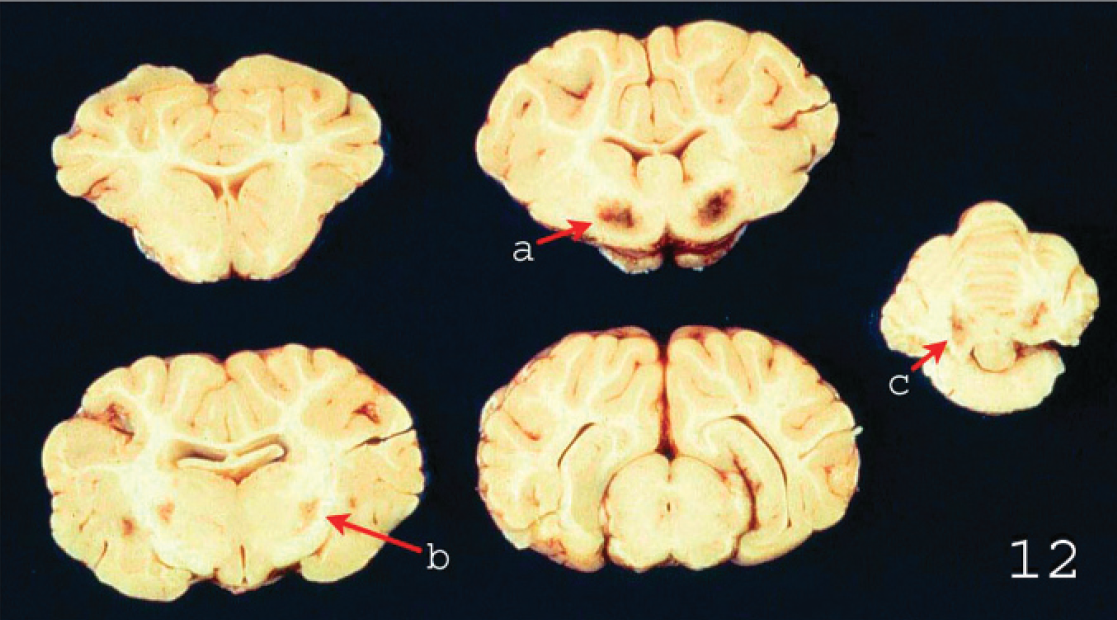
Focal symmetrical encephalomalacia in the brain of a lamb with Clostridium perfringens type D enterotoxemia. These changes are most consistently seen in
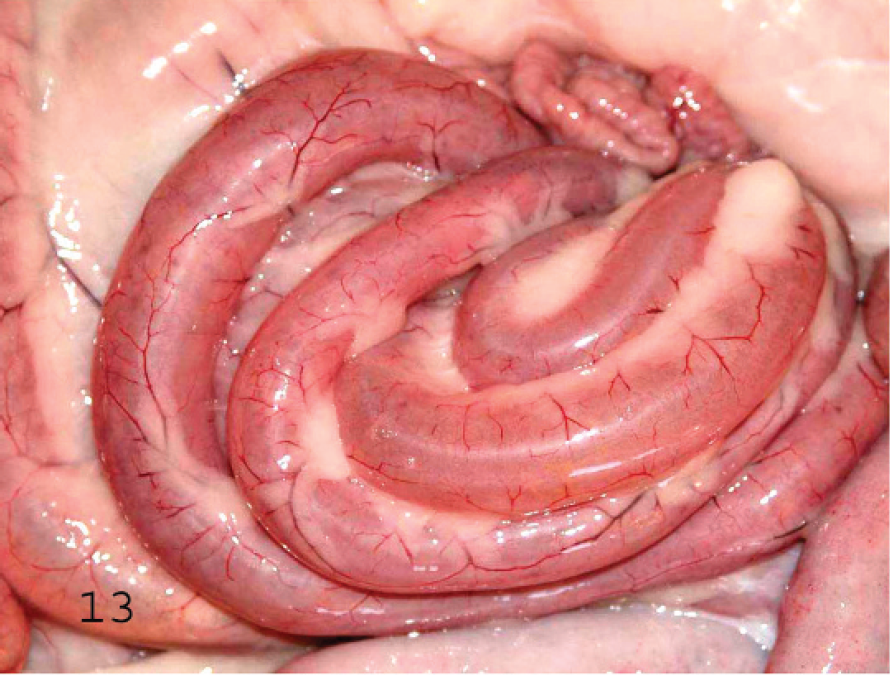
Serosal view of the spiral colon of a goat with subacute type D enterotoxemia. The serosa is hyperemic and the colon is distended by gas, fluid, and fibrin.
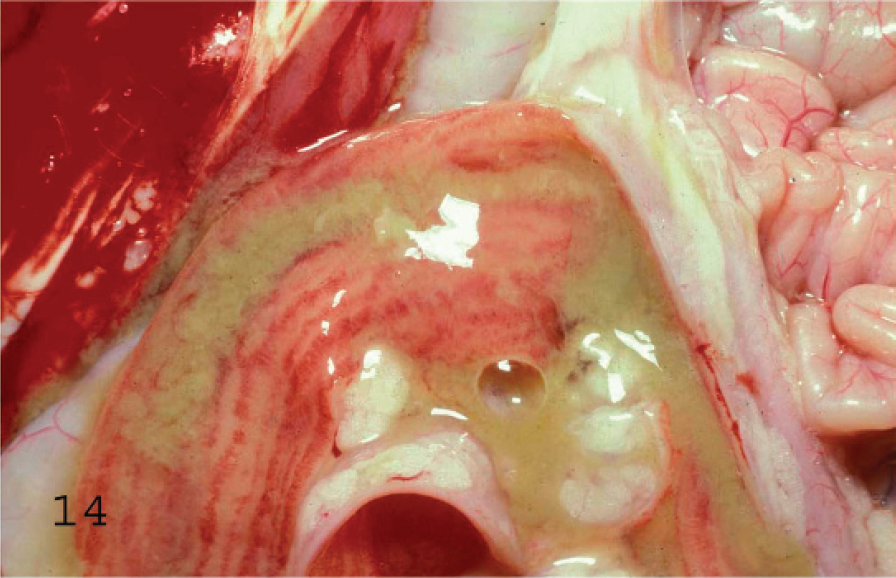
Mucosal view of the spiral colon of a goat with subacute type D enterotoxemia. The mucosa is highly hyperemic and there are multiple strands of fibrin in the lumen. Observe the apparently unaffected small intestine (right top corner).
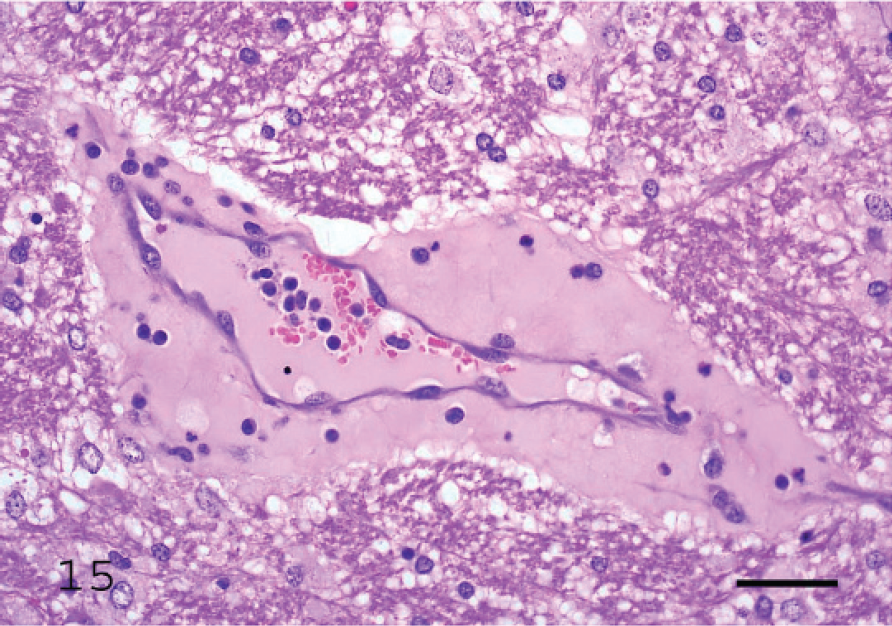
Proteinaceous edema surrounding a vein in the internal capsule of a lamb with acute Clostridium perfringens type D enterotoxemia.
Necropsy findings
Gross changes may be absent from some cases of acute or subacute type D ovine enterotoxemia, and lack of such lesions should not, therefore, be considered sufficient to rule out this condition. Intestinal changes, although rarely present, consist of hyperemic small intestine mucosa with slight to marked red fluid contents. Colitis may occur, but it is not a consistent finding in sheep enterotoxemia. Several gross findings, such as excess pericardial (Fig. 7), pleural (Fig. 8), and/or abdominal fluids (with or without fibrin strands), which may clot on exposure to air, serosal petechiation, and lung edema (Figs. 9, 10), suggest, but are not specific for, type D enterotoxemia. Pathognomonic gross changes in sheep are rarely observed in brain and consist of herniation of the cerebellar vermis (cerebellar coning) in acute or subacute cases (Fig. 11) and FSE, in chronic cases, although this is a misnomer as the lesions are generally multifocal. The FSE is characterized by dark hemorrhagic foci in corpus striatum, thalamus, midbrain, and cerebellar peduncles and white matter (Fig. 12). 18,19,36 The kidney lesion from which one of the common names of the disease is derived (pulpy kidney disease) is likely to be a postmortem change. 1 It is widely believed that autolysis occurs faster in animals dying of enterotoxemia than in animals dying of other causes, but no data support or refute this contention. This change in kidney is highly subjective, and relying on presence of these lesions can lead to diagnostic errors.
Gross changes in acute caprine type D enterotoxemia are similar to those in the ovine disease. In the chronic form of the disease in goats, fibrinohemorrhagic colitis (Figs. 13, 14) with occasional involvement of the distal portion of the small intestine seems to be the most consistent lesion described. 7 A combination of the findings in acute and chronic forms of disease are frequently seen in subacute forms. The so-called pulpy kidney has not been reported in caprine enterotoxemia, nor has cerebellar herniation or FSE.
Microscopic changes
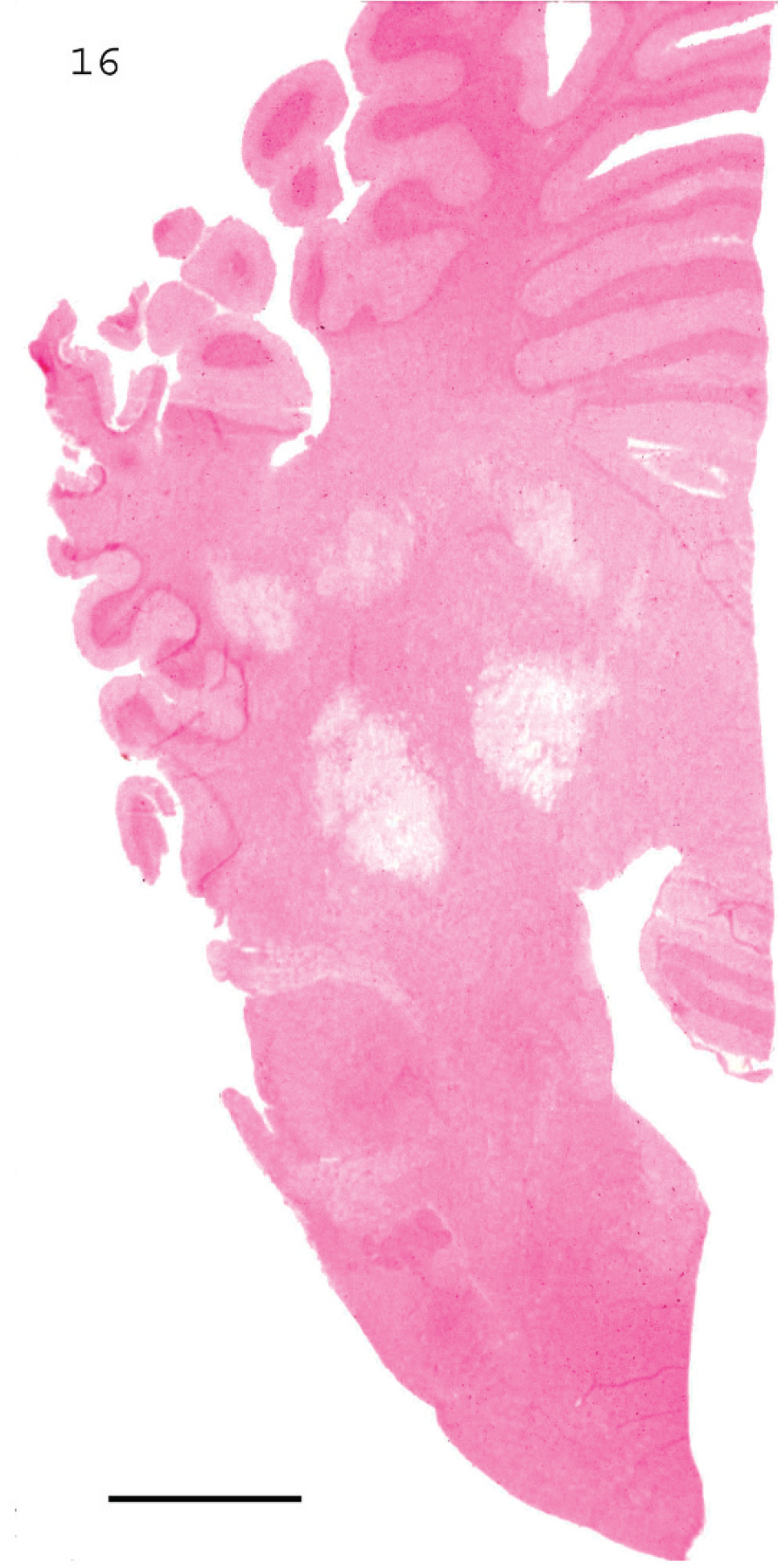
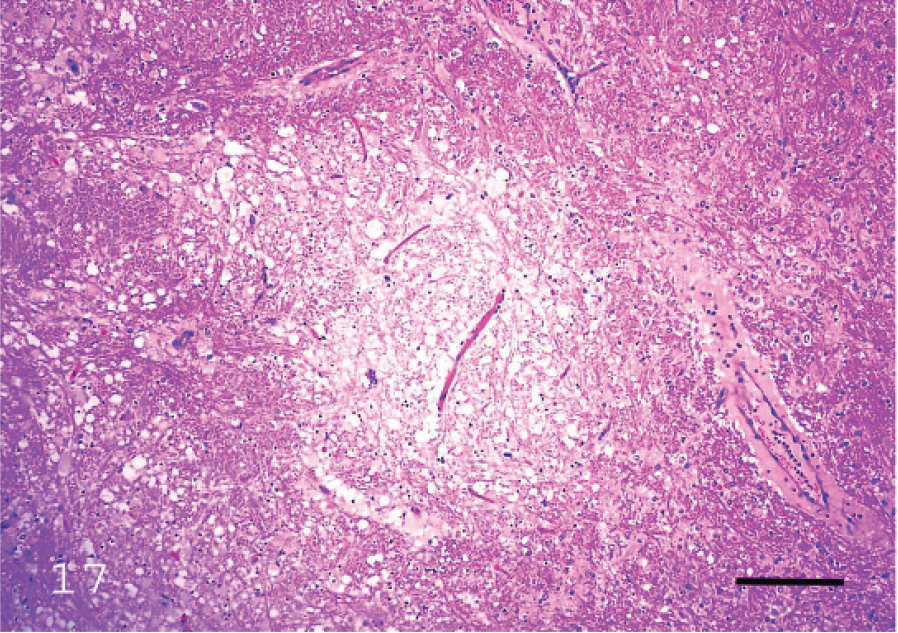
Microscopic changes in the brain of sheep with type D infection are unique and pathognomonic, although they are not present in all cases. 19,70 The most consistent change, observed in approximately 90% of cases, 70 is perivascular proteinaceous edema (microangiopathy) in the brain, which presents as acidophilic accumulations of protein surrounding small and medium-sized arteries and veins (Fig. 15). 19 These lesions are first evident a few hours after onset of clinical signs. Apparently, no other conditions of sheep produce this highly proteinaceous perivascular edema in brain, and this change is therefore diagnostic for type D enterotoxemia. In chronic disease, necrosis of white matter, grossly known as FSE, can be observed (Figs. 16, 17). 36 This lesion is usually multifocal and characterized by degeneration of white matter, hemorrhage, and astrocyte and axonal swelling (Fig. 17). Perivascular edema and degeneration, and necrosis of brain parenchyma, are always bilateral and symmetrical, and they have been described most frequently in corpus striatum, thalamus, midbrain, cerebellar peduncles, and cerebellar white matter. 18,19 These areas are not exclusively affected, and lesions can sometimes be seen in other parts of brain, such as cortex and hippocampus. 19 Most descriptions of microscopic lesions in brain of sheep with enterotoxemia are based on experimental inoculations, 18,19 and there is little information about distribution of lesions in natural cases. This change is a valuable criterion for diagnosing enterotoxemia, particularly when intestinal content is not available for examination for ETX. However, although histologic changes are useful indicators of enterotoxemia, absence of these lesions does not preclude a diagnosis in sheep. Usually, no significant histologic changes are found in intestine of sheep dying from enterotoxemia. 15,70 Histologic changes were not observed in kidneys of experimentally inoculated lambs necropsied immediately after death, 70 supporting suggestions that these lesions are due to postmortem change. Thus, microscopic lesions in kidney should not be considered a diagnostic indicator of ovine or caprine enterotoxemia.
Submicroscopic view of focal symmetrical encephalomalacia in the cerebellar peduncles and cerebellar white matter of a lamb with type D enterotoxemia. Bar = 800 μm. Photo courtesy of Dr. Bill Hartley.
Degeneration of white matter in the internal capsule of a lamb with type D enterotoxemia. Bar = 150 μm.
Fibrino-necrotic (pseudomembranous) colitis in a goat with subacute type D enterotoxemia. Observe almost complete loss of epithelium and pseudomembrane composed mostly of sloughed epithelial cells, leukocytes (mostly neutrophils), fibrin, and bacilli. Bar = 70 μm.
In goats, there are few descriptions of histologic changes in type D enterotoxemia, and changes in brain are not considered a consistent feature of caprine enterotoxemia. Nevertheless, perivascular edema and degeneration of white matter, similar to those observed in sheep, can be observed in cases of acute and subacute caprine enterotoxemia. 67
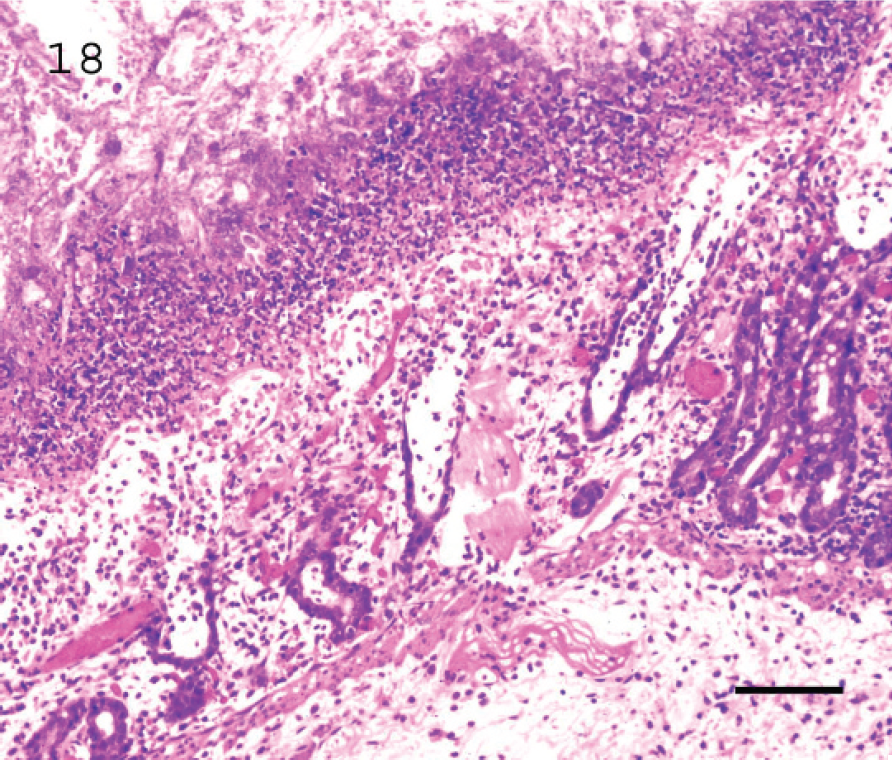
In addition, subacute and chronic type D enterotoxemias in goats are characterized by fibrinonecrotic (pseudomembranous) colitis (Fig. 18) with large numbers of intralesional Gram-positive bacilli. 58 However, this lesion is suggestive but not specific, and it cannot be used to establish a definitive diagnosis of enterotoxemia. Microscopic lesions in kidney are not characteristic of goat enterotoxemia.
Culture and/or polymerase chain reaction detection
Toxinotype D can be a normal inhabitant of the intestine of a moderate number of sheep and goats, so isolation of the organism, especially in low numbers, is not by itself diagnostic (Uzal FA, Marcellino RB: 2002, Clostridium perfringens in clinically healthy sheep). 73 However, as in the case of type A enterotoxemia, quantitation of type D in intestinal content may provide an indication of disease in lambs, 21,64,77 in that elevated numbers of type D are found in intestinal contents of affected animals (usually 10 4 –10 7 CFU/g). 53,59 No equivalent information is available for goats.
Toxin detection
The most widely accepted criterion on which to base definitive diagnosis of type D enterotoxemia in both sheep and goats is detection of ETX in intestinal contents. 62,65 Low concentrations of ETX (≤250 lethal dose 50/ml) may be found in small intestine of clinically healthy sheep, 13 although in other studies, no ETX was found in intestinal content of healthy sheep (authors' unpublished observations). 71 There are no reports of presence or absence of ETX in the intestine of normal goats. If diagnosis is based solely on toxicologic results, it is potentially possible to misdiagnose type B (produces CPA, CPB, and ETX) infections as type D (produces CPA and ETX) if CPB is destroyed by the action of intestinal trypsin. However, type B infections are rare, and gross and histologic lesions of type D disease can be very characteristic of this form of enterotoxemia.
Several techniques are available for detecting ETX in intestinal contents or other body fluids and in culture supernatants. Among these are the MNT, 62 several ELISAs, 25,47,50,71,76 counterimmunoelectrophoresis, 37 and latex agglutination. 41 Comparison of 4 techniques for ETX detection in intestinal contents and other body fluids of sheep and goats revealed marked differences in sensitivity, but the ELISAs were the most sensitive. 71 Yet ELISAs are not necessarily better diagnostic techniques and may, in fact, be detecting concentrations of toxin that are clinically irrelevant. Most conventional methods of toxin detection do not, in fact, detect these amounts of ETX. 71 Thus, detection of ETX by any method in small intestinal contents of a suspect case of ovine or caprine enterotoxemia should be considered a strong indicator of this condition. However, failure to detect toxin in intestinal contents of sheep with appropriate pathological changes (i.e., microangiopathy and/or focal symmetrical degeneration of the brain) does not rule out a diagnosis of enterotoxemia. 19
Addition of chloroform to ileal contents before shipment for laboratory confirmation of enterotoxemia has been recommended (Oxer DT: 1932, “Pulpy kidney”: or acute infectious enterotoxemia of sucking lambs due to B. ovitoxicus [Bennetts]. Australia Inst Sci Ind Res, Pamphlet No. 35, 9–25), and this practice is still widespread. However, maintaining the toxicity of ETX-containing filtrates depends on prompt cooling, 51 and chloroform as a preservative is unnecessary. 4 Furthermore, chloroform at concentrations >0.5% can be toxic to mice, interfering with biological detection of ETX. To the authors' knowledge, no information is available on possible interference by this reagent with other diagnostic methods, such as ELISA or latex agglutination. Toxicity was conserved for approximately 12 months in an aliquot of intestinal content kept at 4°C and for 12 weeks in aliquots kept at room temperature without chloroform. 4 However, the concentration of ETX in ileal contents decreased markedly when left in situ after death of the animal. 17 It is, therefore, advisable that ileal contents from suspected enterotoxemia cases be collected and chilled as soon as possible postmortem.
Detection of C. perfringens toxins can be performed on intestinal content, serum, or serous exudates. 20,64 However, results of a recent study 39 revealed a marked difference in outcome when different bodily fluids were analyzed by a capture ELISA for ETX. Epsilon toxin was detected in most samples of duodenal and ileal content but in very few other body fluids, including urine, pericardial fluid, aqueous humor, and serum (Table 3). Contents of the small intestine are, therefore, the samples of choice for ETX detection.
Ancillary tests
Ancillary tests for diagnosis of type D enterotoxemia in sheep 32,68 and goats 68 have been described. The most useful is detection of urinary glucose, the presence of which in any amount is strongly suggestive of enterotoxemia in both sheep and goats. However, this finding is not consistent, 69,70 and although glycosuria is a useful positive predictor of type D enterotoxemia, absence of glycosuria does not rule out a diagnosis of enterotoxemia in either species. Care should be taken to consider the clinical history, in particular any previous parenteral administration of glucose, which may produce high levels of glycosuria.
Other ancillary tests, such as examination of Gram-stained smears of intestinal mucosa, can also be used to establish a presumptive diagnosis of enterotoxemia. To be considered a positive indicator, Gram-stained smears should reveal large numbers of Gram-positive bacilli with rounded ends. Other bacterial forms can be seen, but there should be a preponderance of bacilli. 20 If negative, Gram-stained smears do not rule out enterotoxemia. Distribution of C. perfringens in sheep and goats with enterotoxemia can be multifocal, and several smears of intestinal mucosa should therefore be made. 40
Hemoconcentration, acidosis, and hyperglycemia are frequent changes in lambs with type D enterotoxemia. 32 However, they are not specific and are rarely used in evaluating affected animals.
Clostridium perfringens type E
Toxinotype E enteric infection of domestic animals was first reported in the late 1940s and has generally been considered a rare occurrence. Toxinotype E is not an uncommon cause of hemorrhagic enteritis and sudden death in beef calves (authors' unpublished observations), 6 having been found in approximately 10% of calves submitted for diagnosis of these symptoms. However, descriptions of clinical disease, gross and microscopic lesions, and bacteriologic findings in sheep and goats are scant. 61 The extent of type E–associated disease should be revealed by careful attention to genotyping of isolates from sheep, goats, and other ruminants.
Acknowledgements
This manuscript was prepared in part through the financial support of the USDA-CSREES-NRICGP, Novartis Animal Health, Pfizer Animal Health, Schering-Plough Animal Health, Intervet, and Boehringer-Ingelheim Vetmedica. The authors thank Dr. J. Ortega Porcel, Ms. P. Yant, and Ms. S. Fitisemanu for help with different aspects of preparing this manuscript. Selected photographs were kindly provided by Drs. B. Hartley and R. Kelly.
Footnotes
a.
Enterotoxaemia (alpha, beta, epsilon, Clostridium perfringens) ELISA Kit, Bio-X Diagnostics SPRL, Jemelle, Belgium.
b.
C. perfringens Enterotoxin Test, TECHLAB, Inc., Blacksburg, VA.
c.
Fisher Scientific Company, Rochester, NY.
