Abstract
Few routinely available biomarkers are clinically useful in assessing dogs with chronic enteropathy (CE) and aid in CE subclassification. The diagnostic potential of the blood neutrophil-to-lymphocyte ratio (NLR) has not been evaluated in canine CE. We evaluated the NLR in 93 dogs with CE (no steroid treatment for ≥2 wk prior) and tested for an association with clinical, clinicopathologic, and histologic characteristics and also with CE subclassification. NLR was significantly higher in CE dogs with severe clinical disease than dogs with mild clinical disease (
Keywords
Introduction
Chronic enteropathies (CEs) comprise an important group of diseases in dogs, causing chronic gastrointestinal signs for ≥3 wk, with the common denominator of chronic inflammation that can affect some or all segments of the gastrointestinal tract. 44 The pathogenesis of these conditions is complex, and an exaggerated immune reaction to luminal (i.e., dietary and/or microbial) antigens appears to play a role in the pathogenesis of CE. 30
The diagnosis of CE requires exclusion of other diseases that can mimic CE (e.g., endoparasites, exocrine pancreatic insufficiency, atypical hypoadrenocorticism, gastrointestinal neoplasia). 44 Further classification of CE is done retrospectively based on the response to sequential treatment trials as food-responsive enteropathy (FRE) if dogs respond to an elimination diet, antibiotic-responsive enteropathy (ARE) for dogs with a durable response to antibiotic treatment, steroid- of other immunosuppressant-responsive (IRE) or -refractory enteropathy (nonresponsive enteropathy, NRE) if corticosteroids and/or other immunosuppressive medications are needed.10,13 The existence of true ARE has been a matter of debate.10,13
Clinical, clinicopathologic, endoscopic, and histologic characteristics can be similar in dogs with different types of CE. The definitive diagnosis of CE requires histologic evaluation of endoscopic or surgical tissue biopsies of the intestine 10 and, if necessary, also immunohistochemical or other molecular tests. Current consensus regarding the diagnostic algorithm for CE in dogs includes elimination dietary trials prior to more invasive testing.10,13 Thus, dogs with a complete and durable response to an elimination diet (i.e., dogs classified as FRE) might not undergo more invasive testing. Several biomarkers have been evaluated in dogs with CE, but few such markers that can be routinely measured (e.g., serum cobalamin and albumin, fecal calprotectin) appear to be of clinical utility in dogs with CE and can potentially aid in CE subclassification based on the response to treatment (i.e., diagnosis of FRE vs. IRE or NRE based on fecal calprotectin concentrations).23,24
Characteristic leukogram changes expressed as the neutrophil-to-lymphocyte ratio (NLR) have been shown to have diagnostic and prognostic potential in humans with inflammatory bowel disease (IBD),1,5 showing a correlation of the NLR with several clinical parameters and demonstrating that the NLR serves as a potentially useful prognostic marker in human IBD. The NLR also appears to be useful in diagnosing dogs with hypoadrenocorticism 46 and has been evaluated in canine atopic dermatitis, 35 septic and noninfectious systemic inflammatory response syndrome, 27 and various types of cancer.14,34,36,39,41 However, the NLR has not been evaluated in dogs with CE. Thus, we aimed to evaluate the clinical utility of the NLR as a diagnostic marker in dogs with CE. We hypothesized that the NLR correlates (1) with clinical, laboratory, and histological parameters, and (2) with the response to different forms of treatment (i.e., disease subclassification: FRE vs. IRE or NRE) in dogs with CE. Our secondary aim was to determine a reference interval for NLR from healthy controls.
Materials and methods
Sample population
We included data from 93 dogs diagnosed with CE (Fig. 1) enrolled at 2 different veterinary centers: the Gastrointestinal Laboratory at the Small Animal Veterinary Teaching Hospital at Texas A&M University (TAMU, College Station, TX, USA; 2008–2015, cases from the TAMU Small Animal Veterinary Teaching Hospital or other tertiary veterinary centers across the United States) and the Department for Small Animals at the University of Leipzig (UL, Germany; 2013–2018). Data from some of the dogs were reported previously. 23
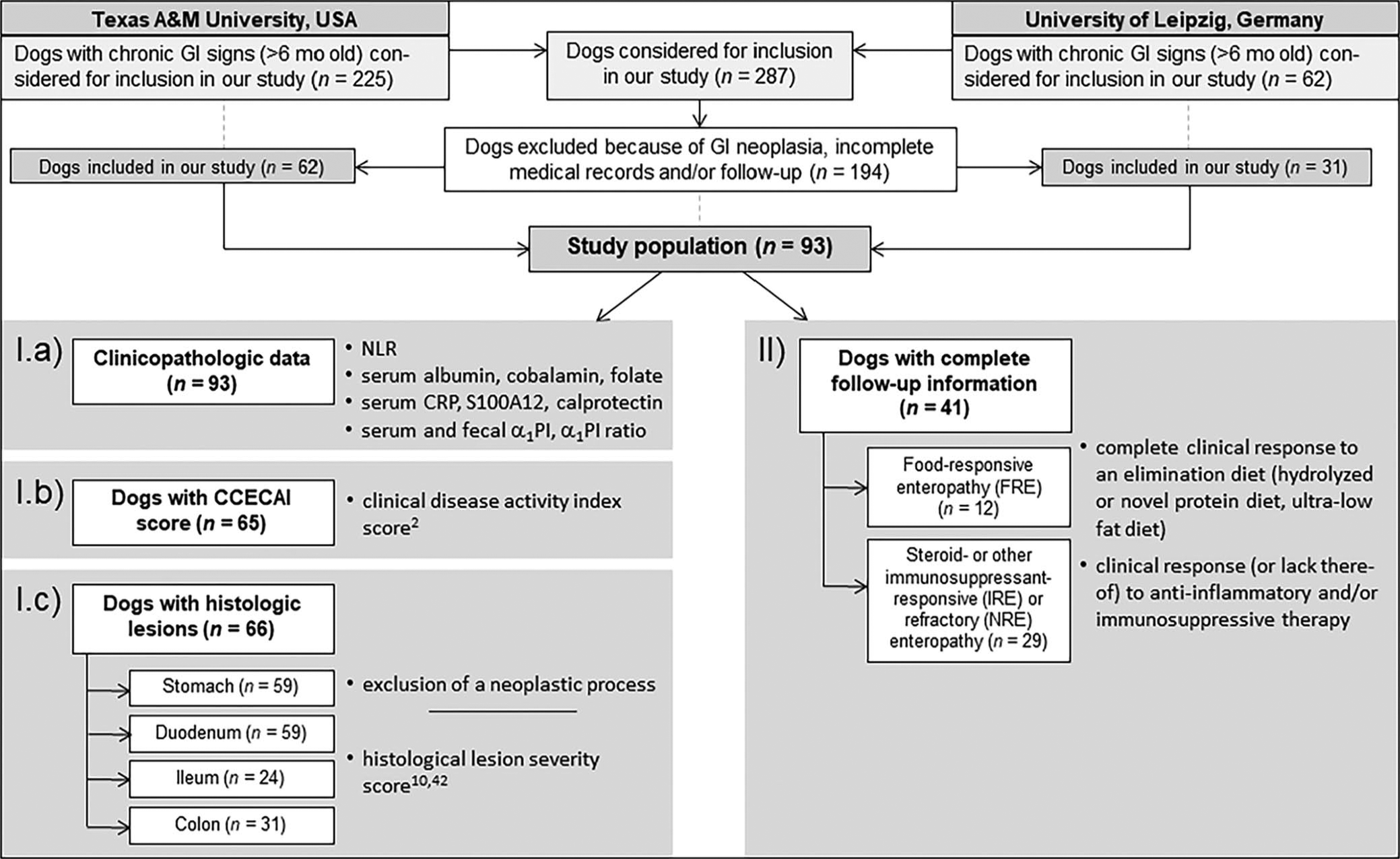
Inclusion flowchart for evaluation of the blood neutrophil-to-lymphocyte ratio (NLR) as a diagnostic marker in dogs with chronic enteropathy. Of the 287 dogs considered for inclusion in the study, we included 93 dogs.
The collection of specimens from dogs with CE for the study of several biomarkers was approved by the Clinical Research Review Committee (CRRC approval TAMU 2009-06, approved 01-15-2009), the Institutional Animal Care and Use Committee at TAMU (IACUC approval TAMU 2012-083, approved 05-22-2012), and the Veterinary Authority of the German Free State of Saxony (TVV 06/17, approved 04-11-2017). Written informed consent was obtained from the owner of each dog that was prospectively enrolled in the first part of the study (correlation analyses: part I.a–c,
As an inclusion criterion, dogs must have been diagnosed with CE based on the current World Small Animal Veterinary Association (WSAVA) consensus statement.10,44 Dogs were excluded from further consideration in our study if they had received corticosteroids and/or other anti-inflammatory or immunosuppressant medication within 2 wk before diagnostic evaluation or if the information about previous administration of such medications was not available.
Dogs prospectively included in the first part of the study (Fig. 1) had a canine chronic enteropathy clinical activity index (CCECAI) score calculated at first presentation by either a veterinarian or veterinary staff as a semi-objective determination of the clinical disease severity (part I.b;
Follow-up data (part II) were included from dogs (
Data from 44 healthy pet dogs enrolled in a blood donor program and presented at UL (2012–2019) for their annual check-up and vaccination, or pre-screening for inclusion in the blood donor program, served as a retrospective control group to establish a reference interval (RI) for NLR.
Sample analyses
Whole blood was used for routine hematology, which was performed at the Texas Veterinary Medical Diagnostic Laboratory (Cell-Dyn; Abbott), for cases enrolled at TAMU, or the Small Animal Veterinary Diagnostic Laboratory at the UL College of Veterinary Medicine (ProCyte Dx; Idexx), for dogs enrolled at UL. Neutrophil counts (RIs: TAMU = 3.0–11.5 × 109/L, UL = 3.0–11.6 × 109/L) and lymphocyte counts (RIs: TAMU = 1.0–4.8 × 109/L, UL = 1.0–5.1 × 109/L) were extracted from the routine hematologic profile, and the NLR was calculated as (neutrophil count)/(lymphocyte count).
Serum (fasted sample) was used for a serum chemistry profile (LiquiColor, Sirrus clinical chemistry analyzer, Stanbio Laboratory for TAMU cases; Fuji DRI-CHEM NX500i, Scil for UL cases) and for measurement of serum cobalamin (Immulite 2000, vitamin B12; Siemens), folate (Immulite 2000, folic acid; Siemens), canine-specific pancreatic lipase (Spec cPL assay; Idexx), and C-reactive protein (Phase CRP; Tri-Delta) concentrations. Serum concentrations of the pro-inflammatory molecules calprotectin (RI: 0.6–11.8 mg/L) and S100A12 (RI: 49–320 μg/L), as well as the anti-inflammatory decoy receptor sRAGE (soluble receptor for advanced glycation end products), were determined by species-specific ELISAs (only TAMU cases).20,22,25
Fecal concentrations of calprotectin (preliminary RI: 3.2–18.4 μg/g) and S100A12 (RI: 2–484 ng/g) were also measured by species-specific ELISAs (only TAMU cases),22,25 and the 3-d mean fecal concentrations of both analytes were calculated 18 and used for statistical analyses. Alpha1-proteinase inhibitor (α1PI) concentrations in serum (RI: 732–1,800 mg/L), fecal extracts (RI for the 3-d sample mean: 2.2–13.9 μg/g), and the serum-to-fecal α1PI ratio were determined as markers of gastrointestinal protein loss (only TAMU cases).17,19,21
Urine samples were used for routine urinalysis (performed at the TAMU Clinical Pathology Laboratory for cases enrolled at TAMU or the Small Animal Veterinary Diagnostic Laboratory at UL for dogs enrolled from UL) and, if indicated, for further testing (i.e., urine culture, urine protein-to-creatinine ratio).
Histologic evaluation of gastrointestinal tissue biopsies was performed by 1 of 8 board-certified pathologists using the WSAVA Gastrointestinal Standardization grading system.11,44 The severity of structural and inflammatory lesions in the duodenum, ileum, and colon were recorded based on a 4-point grading scheme (0 = normal, 1 = mild lesions, 2 = moderate lesions, 3 = severe lesions; Suppl. Table 3),11,44 and the maximum and mean cumulative lesion scores (calculated as the sum of individual lesion scores from each segment) were considered for statistical analysis. Gastric biopsies were evaluated to exclude neoplasia. 3
Statistical analyses
Data were analyzed for the assumptions of normality and equal variances using a Shapiro–Wilk W and Levene test, respectively. Summary statistics for continuous variables are presented as medians and interquartile ranges (IQRs), and for categorical data are reported as counts (
A RI for the NLR in dogs was established using the robust method after Box–Cox transformation (Reference Value Advisor v.2.1; http://www.biostat.envt.fr/reference-value-advisor) of the single-sample NLR (using the ProCyte Dx) determined in the 44 healthy adult pet dogs that were of various breeds and ages (median age: 4.2 y, range: 1.0–9.3 y; 28 males, 16 females).15,16
Results
The NLR was 0.2–54.0 (median: 5.5) in the 93 dogs with CE included in our study (Table 1); NLRs varied within this group of dogs, but most dogs had an NLR <10. Age was moderately correlated with the magnitude of the NLR (Table 2). Most dogs with CE had a normal neutrophil count (
Characteristics of 93 dogs with chronic enteropathy included in our study.
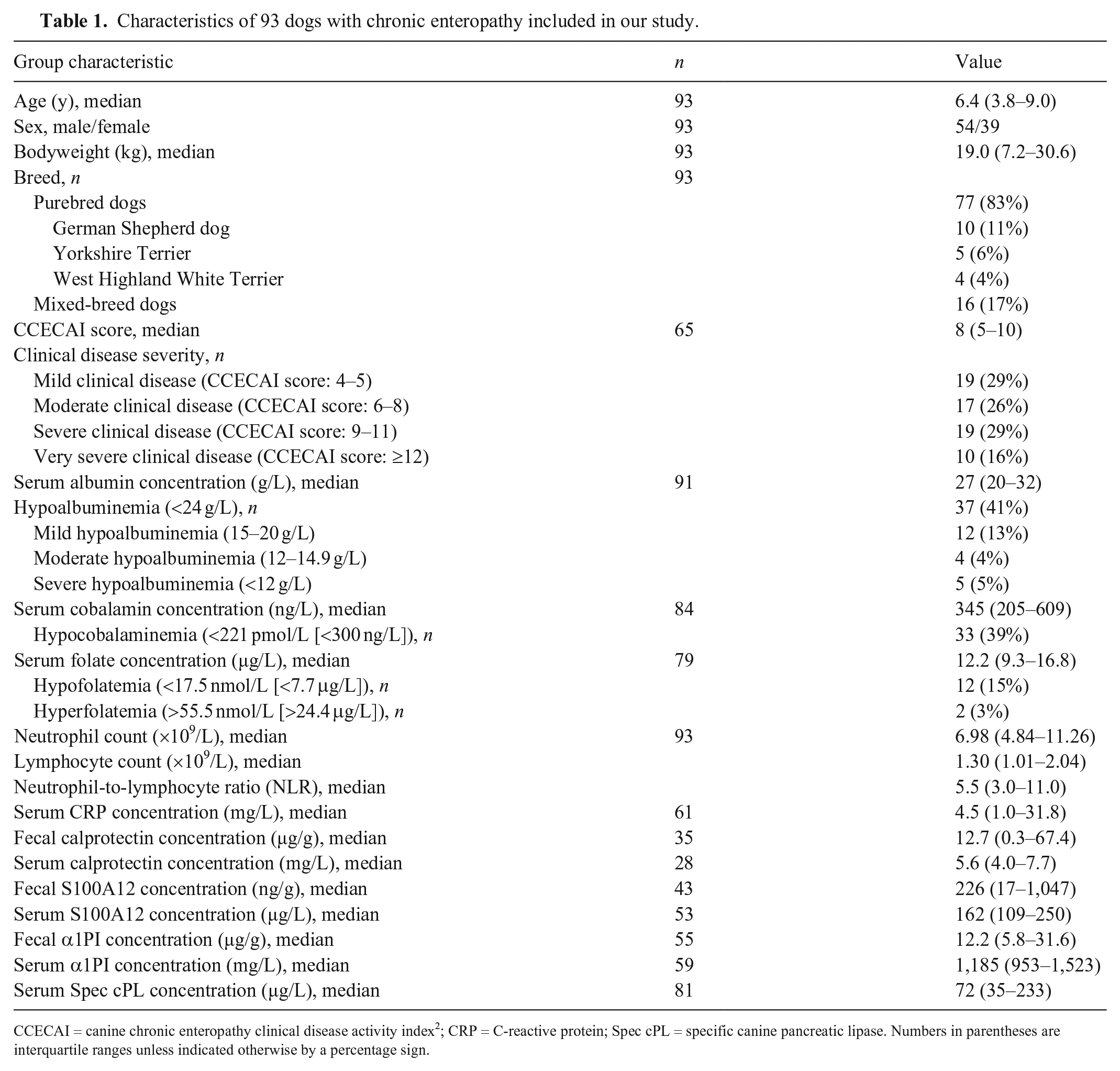
CCECAI = canine chronic enteropathy clinical disease activity index 2 ; CRP = C-reactive protein; Spec cPL = specific canine pancreatic lipase.
Numbers in parentheses are interquartile ranges unless indicated otherwise by a percentage sign.
Association between neutrophil-to-lymphocyte ratio (NLR) and demographic and clinicopathologic parameters in 93 dogs with chronic enteropathy.

α1PI = alpha1-proteinase inhibitor; CCECAI = canine chronic enteropathy clinical activity index
2
; CRP = C-reactive protein; Spec cPL = specific canine pancreatic lipase; NS = not significant;
In the healthy control group (
Association of NLR with the severity of clinical disease and histological lesions
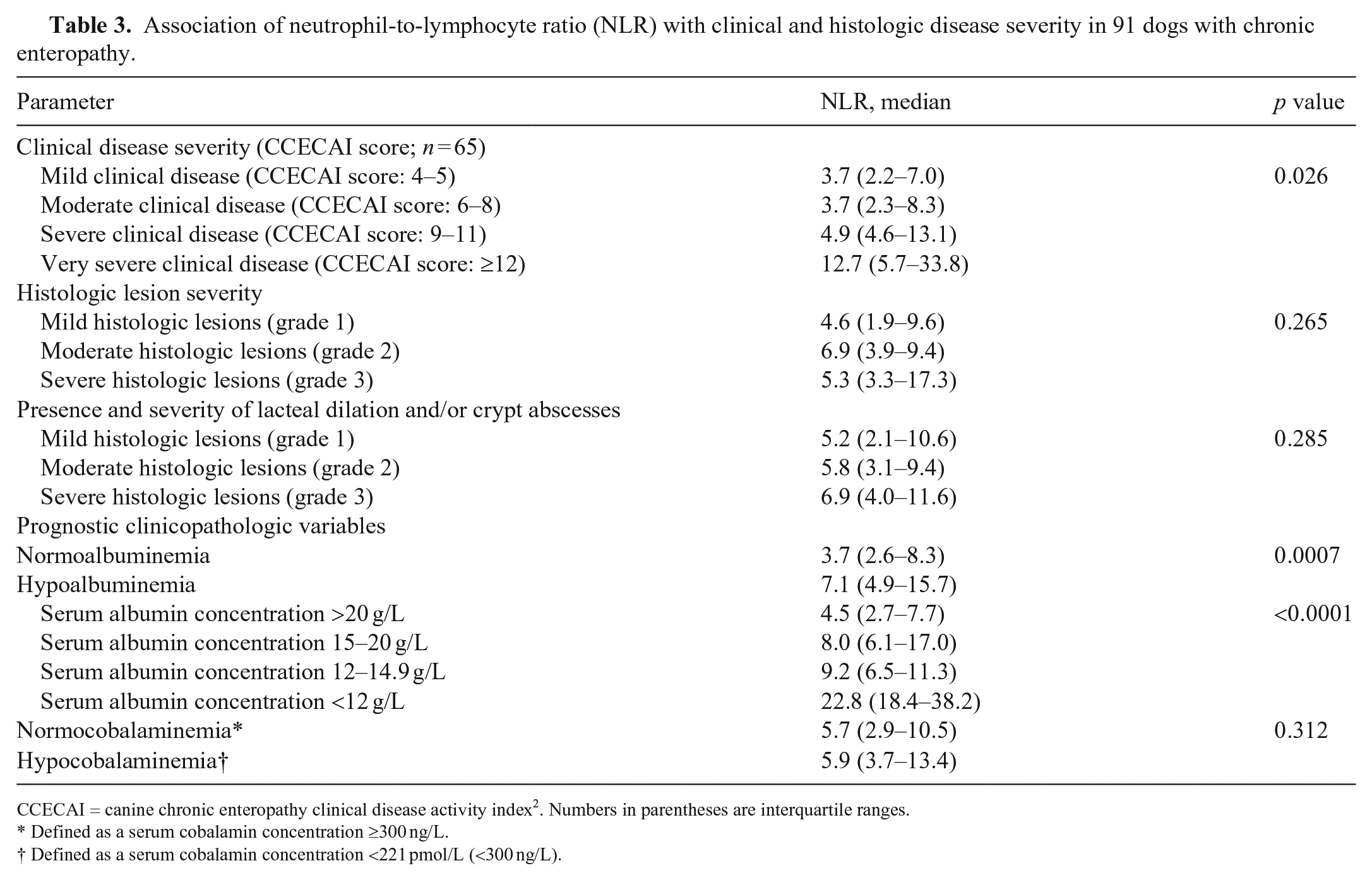
NLR was moderately positively correlated with CCECAI scores (Table 2) and differed significantly (
Association of neutrophil-to-lymphocyte ratio (NLR) with clinical and histologic disease severity in 91 dogs with chronic enteropathy.
CCECAI = canine chronic enteropathy clinical disease activity index 2 .
Numbers in parentheses are interquartile ranges.
Defined as a serum cobalamin concentration ≥300 ng/L.
Defined as a serum cobalamin concentration <221 pmol/L (<300 ng/L).
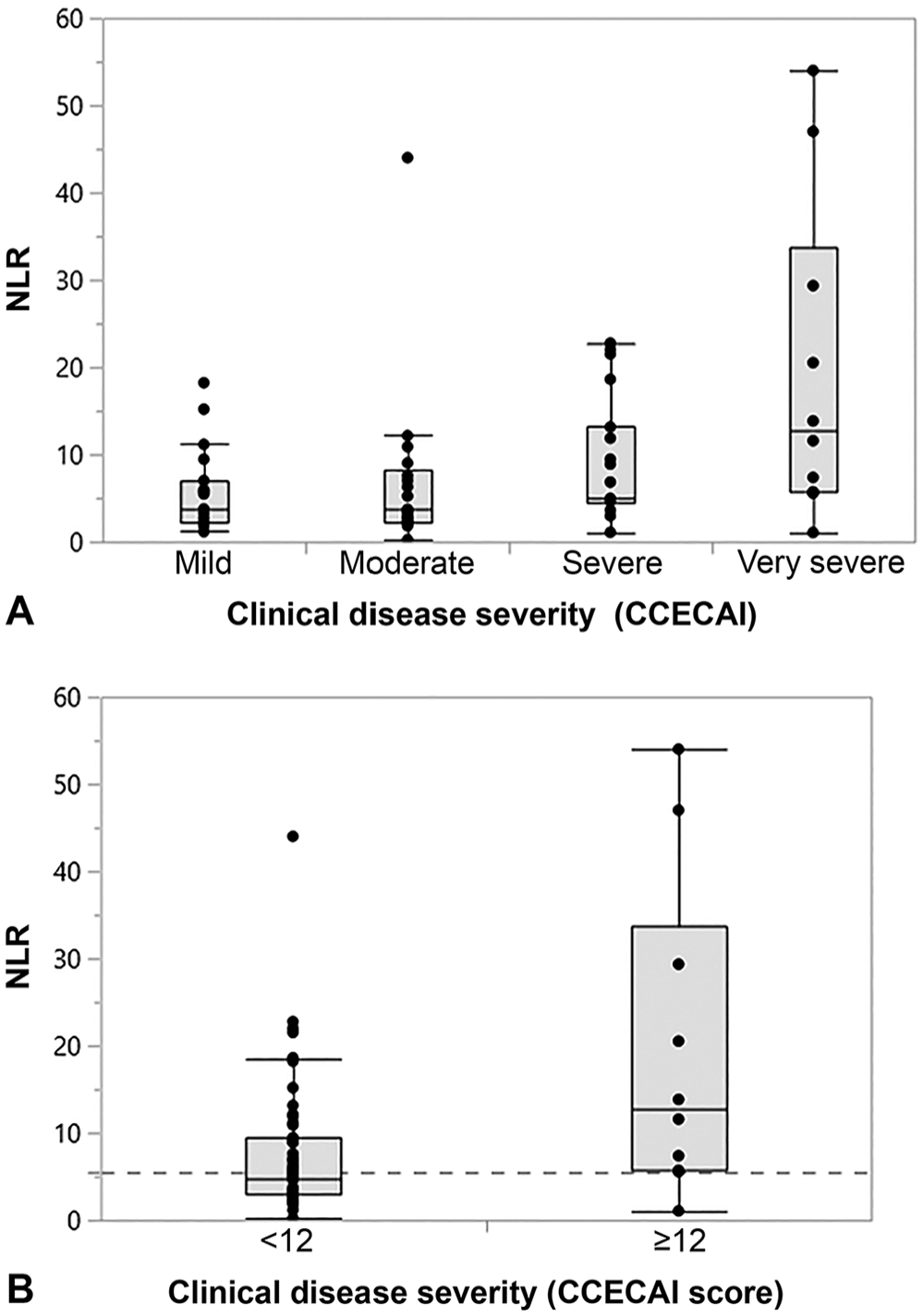
Association of neutrophil-to-lymphocyte ratio (NLR) with the severity of chronic enteropathy in 93 dogs.
NLR was strongly correlated with the blood neutrophil count in the group of dogs with a CCECAI score ≥12 (ρ = 0.88,
NLR was not correlated with the mean cumulative histologic lesion score from all intestinal segments evaluated (ρ = 0.07,
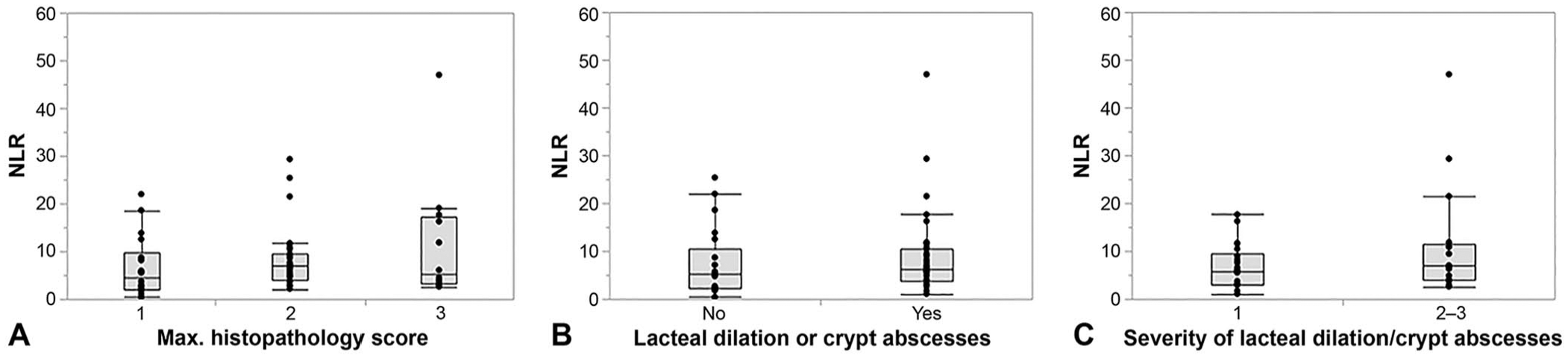
Association of neutrophil-to-lymphocyte ratio (NLR) with the severity of histologic lesions in 66 dogs.
Magnitude of the NLR was slightly higher in dogs with microscopic lesions typically seen in dogs with protein-losing enteropathy (PLE; i.e., lacteal dilation and/or crypt abscesses), and particularly in dogs with more severe such lesions (histologic lesion score ≥2). However, these associations were not statistically significant (Fig. 3B, 3C; Table 3). Neither NLRs nor neutrophil or lymphocyte counts differed between dogs with characteristics of a PLE [i.e., (1) moderate or severe lacteal dilation and/or crypt abscesses, and/or (2) serum albumin concentration <20 g/L) and those without (
Association of NLR with prognostic clinicopathologic factors
A weak inverse correlation was detected between NLR and serum albumin concentration (Table 2). Dogs with hypoalbuminemia (defined as a serum albumin concentration <24 g/L) had significantly higher NLRs than normoalbuminemic dogs (Table 3, Fig. 4A), with a sensitivity of 78% and a specificity of 61% (AUC = 71%) to distinguish hypoalbuminemic dogs with CE from those dogs with normoalbuminemia when using a cutoff NLR of 4.9. Severe hypoalbuminemia (serum albumin concentration <12 g/L) was associated with significantly higher NLRs than having a serum albumin concentration ≥12 g/L (Table 3, Fig. 4B). Hypoalbuminemia was not associated with hyperglobulinemia (
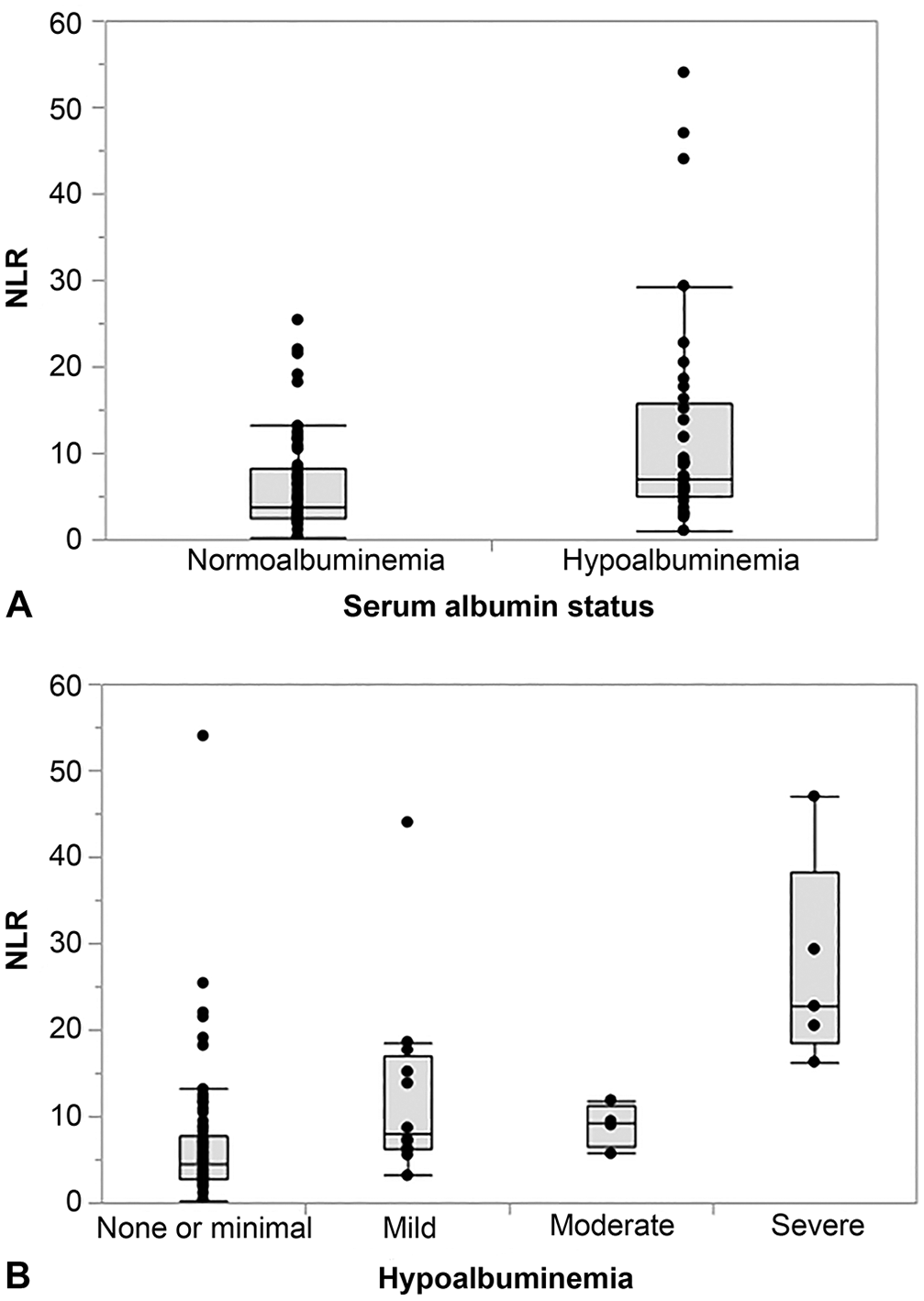
Association of the serum albumin status and neutrophil-to-lymphocyte ratio (NLR) in 91 dogs with chronic enteropathy.
No differences were detected between the NLRs in dogs with hypocobalaminemia (defined as a serum cobalamin concentration <221 pmol/L [<300 ng/L]
31
) and normocobalaminemic dogs (
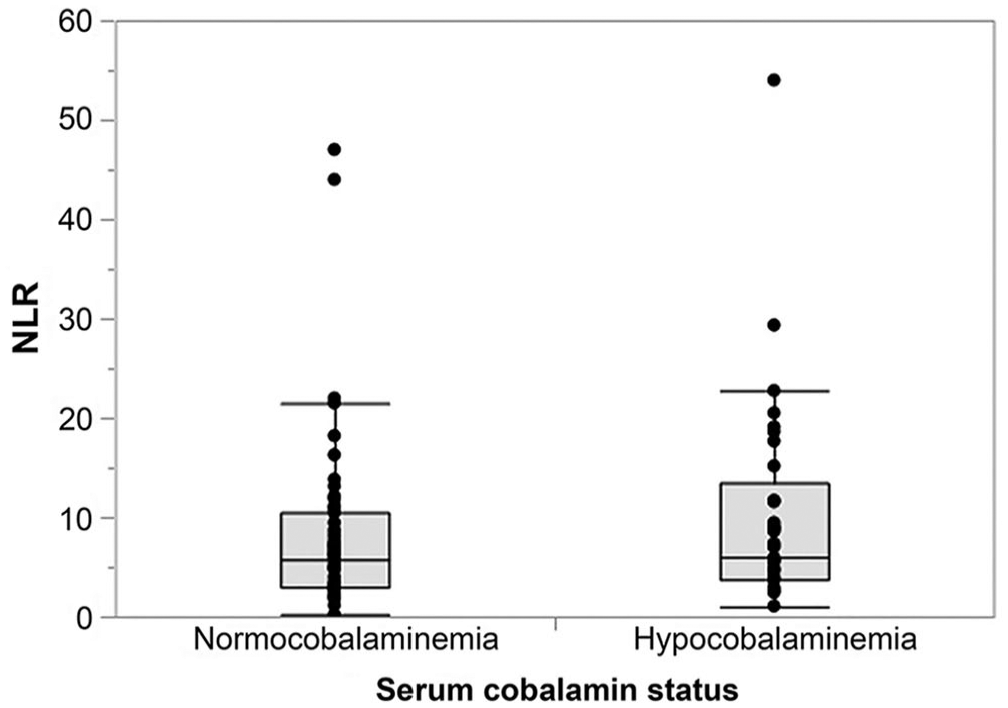
Association of serum cobalamin status and neutrophil-to-lymphocyte ratio (NLR) in 84 dogs with chronic enteropathy. No difference in NLR was detected between dogs with hypocobalaminemia (serum cobalamin concentration <221 pmol/L [<300 ng/L]; median: 5.9, interquartile range [IQR]: 3.7–13.4) and normocobalaminemic dogs (median: 5.7, IQR: 2.9–10.5;
Association of NLR with biomarkers of inflammation and gastrointestinal protein loss
Of the circulating biomarkers of inflammation that we evaluated, serum CRP and S100A12 concentrations were weakly positive, and serum sRAGE was moderately negatively correlated with the NLR in univariate analysis, but there was no statistical significance after correction for multiple comparisons (Table 2).
Fecal calprotectin and S100A12 concentrations were also not significantly correlated with the corresponding NLR, but serum Spec cPL concentration was moderately correlated with NLR (Table 2). In addition to the serum albumin concentration, fecal α1PI concentrations (positively) and the serum-to-fecal α1PI ratio (inversely) were moderately correlated with the NLR (Table 2).
Neutrophil counts, but not lymphocyte counts, were correlated with serum S100A12 (ρ = 0.50,
Association of NLR with CE subclassification
A diagnosis of FRE was established in 12 dogs, and 29 dogs were categorized as IRE or NRE. The follow-up time was 29–2,945 d (median: 171 d) in dogs with FRE, and 7–586 d (median: 69 d) in dogs with IRE or NRE. Dogs with FRE received either a hydrolyzed diet (
CCECAI scores were higher in dogs with IRE or NRE (median: 9, IQR: 6–11) than in dogs with FRE (median: 6, IQR: 3–7), but the difference did not reach statistical significance (
Neutrophil counts were significantly higher in dogs with IRE or NRE than in dogs with FRE (
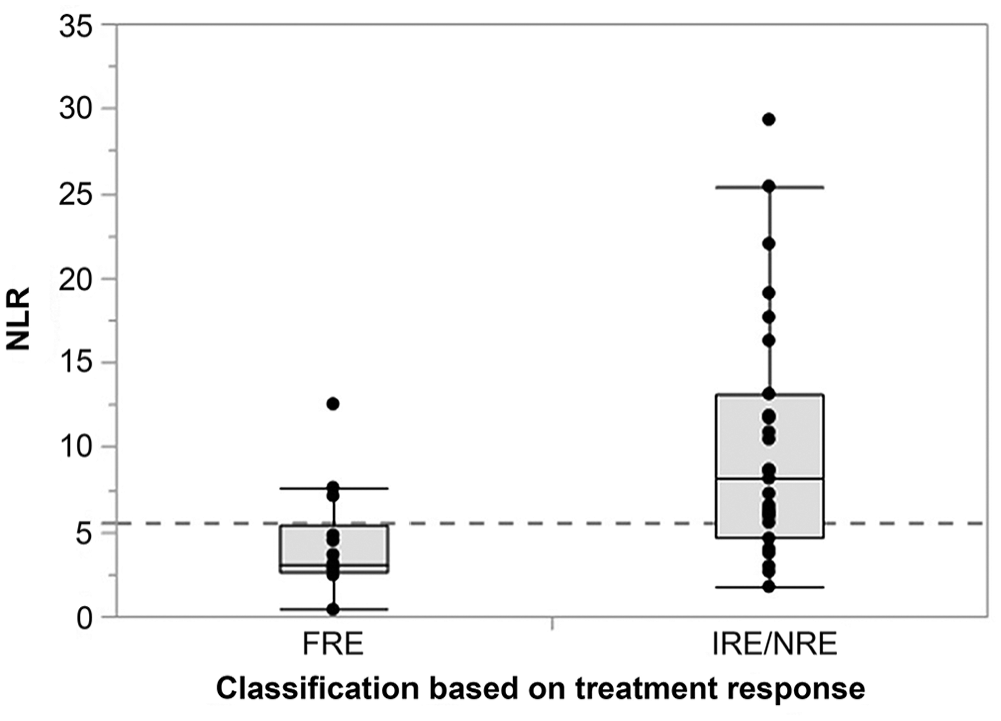
Disease subclassification and neutrophil-to-lymphocyte ratio (NLR) in 41 dogs with chronic enteropathy. Dogs that were diagnosed with steroid- or other immunosuppressant-responsive or -refractory enteropathy (IRE or NRE,
There was no difference in the NLR between dogs with IRE (median: 7.7, IQR: 5.3–12.1, range: 1.8 –29.3;
Investigation for a confounding effect of the different hematology analyzers used at TAMU and UL on variables that affect NLR (CCECAI score, hypoalbuminemia, disease subclassification), neutrophil count (CCECAI score, disease subclassification), and lymphocyte count (CCECAI score) revealed no confounding effect on the association between CCECAI score and NLR (
Discussion
Dogs in our study appeared to have only slightly higher NLR values and a larger variation of the NLRs than human patients with active IBD.1,5 The NLR was strongly correlated with the neutrophil count in dogs with CE and very severe clinical disease. In contrast, the correlation between NLR and lymphocyte counts in clinically severely affected dogs, and the correlations in dogs with less severe clinical signs, were only moderate. This might explain the slightly higher NLRs (calculated as the ratio of both variables) in dogs with CE compared to people.1,5 Further, it suggests species-specific differences in leukogram changes. However, interleukin-8 (the primary regulator of neutrophil responses and recruitment 4 ) is not dysregulated in canine CE. 6
CE is characterized by changes localized to the intestinal wall. However, this condition can also be associated with systemic effects, such as an increase in inflammatory markers (e.g., serum CRP concentration) or hypoalbuminemia (e.g., from PLE). Characteristic leukogram changes, reflected by an increase in the NLR, in dogs with CE in our study suggest that canine CE is associated with systemic stress (stress leukogram) and/or inflammatory response (inflammatory leukogram). This could reflect an increased production of endogenous cortisol associated with chronic inflammation, and it suggests that neutrophils might play a role in the systemic inflammatory response associated with canine CE. Neutrophils are an essential element of innate immunity and, beyond immune surveillance, are essential to bridge innate and adaptive (T cell–mediated) immune responses. Increased neutrophil counts in dogs with CE could also result from TNF-α– or NF-κB–mediated inhibition of neutrophil apoptosis,6,33 interleukin-17–mediated increase in granulopoiesis,
43
increased recruitment and decreased clearance associated with chronic sterile inflammation,12,26 or TLR4 ligand–mediated platelet activation.
8
In line with this, TLR4 ligands calprotectin and S100A12 were also correlated with the neutrophil counts (but not NLR) in our study. Also, despite canine CE (and particularly IRE or NRE) being a diagnosis by exclusion, an effect of comorbidity on increased NLR values (e.g., concurrent pancreatitis) cannot be excluded in all of the dogs in our study. Increased serum Spec cPL concentrations (compatible with a diagnosis of pancreatitis) were also associated with increased NLRs and with a diagnosis of IRE or NRE (
There was moderate correlation between the NLR and the clinical severity of CE as assessed by the CCECAI score in the dogs in our study, but there was also a significant degree of overlap in the NLR between various CCECAI scores. This finding agrees with the results of a study in people, 1 showing that the NLR is associated with the clinical activity of Crohn disease. The cutoff to distinguish dogs with a CCECAI score > 12 from dogs with mild or moderate clinical signs (NLR of 5.5) was also higher than those cutoffs determined in humans (Crohn disease: 3.2, ulcerative colitis: 3.1) to distinguish patients with active IBD from patients in clinical remission. 1
Lymphoplasmacytic enteritis is the most common histologic form of canine CE, and severe inflammation can cause protein-losing enteropathy (PLE). We found no significant correlation between the NLR and the overall severity of histologic lesions (comprised of structural and inflammatory changes). NLRs only tended to be higher in dogs with histologic lesions characteristic of a PLE, i.e., lacteal dilation and/or crypt abscesses. This finding is interesting, given the inverse correlation of the NLR with serum albumin concentration and the serum-to-fecal α1PI ratio, the positive correlation between NLR and fecal α1PI concentrations in our study, and also the fact that hypoalbuminemia was previously shown to be a negative prognostic factor in dogs with CE. 2 Lymphopenia is also a common finding in dogs with intestinal lymphangiectasia and PLE,9,13 and this could further contribute to an increase in the NLR in affected dogs. Lack of a significant relationship between the NLR and the presence of histologic lesions typically seen with PLE could be explained by the fact that lymphangiectasia can either be caused by idiopathic inflammation compromising lymphatic flow or obstructing lymphatic vessels within the intestinal mucosa, or can be a primary condition that in turn might result in intestinal mucosal infiltration of inflammatory cells.9,13 Hypocobalaminemia, another negative prognostic factor in dogs with CE,2,31 was not associated with a high NLR in our study. However, we cannot exclude the possibility of supplementation of cobalamin in all of the dogs before enrollment in the study.
Clinical severity of CE in dogs can be characterized by clinical, histologic, and endoscopic findings.10,44 Also, noninvasive variables such as serum CRP, fecal calprotectin, or fecal α1PI concentrations might be useful surrogate markers to diagnose, characterize, and/or monitor this condition. 24 Weak-to-moderate correlations between the NLR and other biomarkers of systemic inflammation (i.e., serum S100A12 and CRP concentrations, serum sRAGE concentration) that did not remain significant after correction for multiple comparisons might indicate spatial and/or temporal differences in the expression of these markers. This is further supported by the lack of a correlation with fecal calprotectin and S100A12 concentrations as markers of gastroenteritis. Similar to our findings, there was no significant correlation between NLR and CRP in human patients with IBD. 1
Eliminating a diagnosis of FRE as a cause of the chronic gastrointestinal signs requires thorough dietary elimination trials, which is time-consuming and can be frustrating for both the client and the veterinarian. 10 Further patient evaluation, if dietary trials have failed, includes the histologic examination of gastrointestinal biopsies to document inflammation, further characterizing the histologic picture, and excluding a diagnosis of alimentary lymphoma.10,44 Thus, markers that can help differentiate IRE or NRE from FRE early in the diagnostic process of canine CE would be very useful in clinical practice. Such markers might also guide the clinician’s decision whether a dietary trial should be performed first or if further testing (i.e., intestinal biopsy) should be pursued, followed by a more aggressive approach to treatment. Our study showed that NLR values in dogs diagnosed with IRE or NRE were significantly higher and more variable than in dogs with FRE, and that an NLR of ≥5.5 detected dogs with IRE or NRE with moderate sensitivity and specificity. However, the possibility of an additive effect of concurrent pancreatitis cannot be excluded definitively. Thus, the NLR might be a further surrogate marker to help differentiate FRE from IRE or NRE. Lack of a difference in NLR between dogs with IRE and dogs classified as NRE in our study contrasts with the utility of the NLR to predict the response to immunomodulatory treatment in human patients with IBD. 5 Further studies are warranted to determine whether NLR could be useful to differentiate between CE and alimentary lymphoma in dogs.
A significant correlation between NLR and the patient’s age was observed in dogs with CE and in healthy controls in our study. In contrast, the effect of age on NLR values was not seen in a previous study evaluating hematologic and serum biochemical variables in puppies. 40 The age effect seen in our study might be explained by the increased probability of metabolic and inflammatory diseases with increasing age. An effect of breed on NLR has not been reported and cannot be evaluated based on the results of our study given the large number of canine breeds represented. Further, an effect of season on the neutrophil count has been demonstrated in dogs. 42 The possibility of this effect on the NLR cannot be excluded entirely from our study, although we saw no seasonal differences in the NLRs among CE dogs and healthy controls (data not shown). Also, an effect of exercise, which has been demonstrated to cause an increase in both neutrophil and lymphocyte counts, 29 appears to be unlikely.
We acknowledge that the clinical and partially retrospective nature of this investigation is a limitation. Another shortcoming of our study is that not all white blood cell counts were manually differentiated, thus inherent errors in automated cell counting cannot be excluded. For this reason, platelet counts or the platelet-to-lymphocyte ratios (PLRs) were also not evaluated. Furthermore, the study includes data obtained from 2 different hematology analyzers. A correlation and regression (Passing–Bablok and/or Deming) and/or agreement (kappa) analysis could have determined the agreement between both methods, but patient samples could not be tested on both machines. However, the RIs determined for the neutrophil (TAMU: 3.0–11.5 × 109/L; UL: 3.0–11.6 × 109/L) and lymphocyte (TAMU: 1.0–4.8 × 109/L, UL: 1.0–5.1 × 109/L) counts used for the 2 machines were almost identical, and multivariate analyses for selected variables detected no confounding effect for using 2 different hematology analyzers in our study. The severity of endoscopic lesions in the duodenum was also shown to be associated with a negative prognosis. 2 However, we did not include or evaluate endoscopic lesion scores given the inclusion of dogs from several veterinary centers. Thus, the relationship between NLR and the severity of endoscopic lesions in dogs with CE warrants further study. Further, gastrointestinal biopsies were evaluated by several pathologists rather than a single pathologist. This presents a limitation of our study because of the known interobserver variability among pathologists evaluating intestinal biopsies, 45 even using the WSAVA guidelines.11,44 The absence of ileal biopsies in some of the dogs (6 of 27 dogs [22%] in which a lower gastrointestinal endoscopy was performed) is another limitation of our study because histologic lesion severity can differ between intestinal segments (e.g., duodenum and ileum).7,38 Also, diffuse gastrointestinal neoplasia (e.g., alimentary lymphoma) cannot be ruled out in some of the dogs in our study in which gastrointestinal biopsies were not performed (e.g., with a complete response to an elimination diet). Lastly, the reduced numbers in some comparisons and the lack of complete information from some dogs are limitations of our clinical study.
Supplemental Material
sj-pdf-1-vdi-10.1177_1040638721992057 – Supplemental material for Blood neutrophil-to-lymphocyte ratio (NLR) as a diagnostic marker in dogs with chronic enteropathy
Supplemental material, sj-pdf-1-vdi-10.1177_1040638721992057 for Blood neutrophil-to-lymphocyte ratio (NLR) as a diagnostic marker in dogs with chronic enteropathy by Anja Becher, Jan S. Suchodolski, Jörg M. Steiner and Romy M. Heilmann in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Part of the data was presented as a poster at the 29th Annual Congress of the European College of Veterinary Internal Medicine (ECVIM), Milano, Italy (19–21 September 2019), for which the 2019 ECVIM-ESCG poster award was received.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support from a formal grant for the research, authorship, and/or publication of this article. Materials and supplies for the study were generously provided by the Gastrointestinal Laboratory at Texas A&M University; publication costs were paid by the Leipzig University Department for Small Animals.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
