Abstract
Enterotoxemia caused by Clostridium perfringens type D in sheep is believed to result from the action of epsilon toxin (ETX). However, the sole role of ETX in the intestinal changes of the acute and chronic forms of enterotoxemia in goats remains controversial, and the synergistic action of other C. perfringens toxins has been suggested previously. The current study examined 2 goats that were found dead without premonitory clinical signs. Gross lesions at necropsy consisted of multifocal fibrinonecrotic enterocolitis, edematous lungs, and excess pleural fluid. Histologically, there were multifocal fibrinonecrotic and ulcerative ileitis and colitis, edema of the colonic serosa, and proteinaceous interstitial edema of the lungs. Clostridium perfringens type D carrying the genes for enterotoxin (CPE) and beta2 toxin (CPB2) was cultured from intestinal content and feces of 1 of 2 goats, while C. perfringens type D CPB2–positive was isolated from the other animal. When multiple colonies of the primary isolations from both animals were tested by Western blot, most of the isolates expressed CPB2, and only a few isolates from the first case expressed CPE. Alpha toxin and ETX were detected in ileal and colonic contents and feces of both animals by antigen capture enzyme-linked immunosorbent assay. CPB2, but not CPE, was identified in the small and large intestines of both goats by immunohistochemistry. These findings indicate that CPB2 may have contributed to the necrotic changes observed in the intestine, possibly assisting ETX transit across the intestinal mucosa.
Clostridium perfringens is an anaerobic, Gram-positive, rod-shaped bacterium that is classified into 5 toxinotypes based upon production of 4 major typing exotoxins (alpha [CPA], beta [CPB], epsilon [ETX], and iota [ITX]). 10 All toxinotypes produce CPA; types B and C produce CPB, types B and D produce ETX, and type E produces ITX. Two other toxins of importance, enterotoxin (CPE) and beta2 (CPB2), also can be produced by strains of most types of C. perfringens but are not used in the typing of this microorganism. 8
Enterotoxemia caused by C. perfringens type D has been described mainly in sheep and goats. 3,16 In sheep, the disease is characterized mainly by respiratory and neurological signs and lesions, all of which have been experimentally reproduced by intravenous inoculation of ETX. In goats, however, the subacute and chronic forms of type D enterotoxemia are associated with enterocolitis, with or without respiratory and neurological alterations. 20 The role of ETX in the intestinal changes of goat enterotoxemia, however, remains controversial, and the synergistic action of other C. perfringens toxins with ETX has been suggested in the past as a means of explaining these changes. 20
Beta2 toxin is a recently discovered protein with functional, but not sequence, similarities to CPB. 7,9 Clostridium perfringens strains harboring cpb2 (encoding CPB2) have been isolated from the intestines of several animal species with enteritis, and, therefore, a causal relationship of CPB2 toxigenic C. perfringens has been postulated. 11 However, existence of toxin genes does not necessarily imply production of the toxins, and CPB2 production in vivo has only rarely been documented. 1 To the authors' knowledge, no published studies relate the presence of CPB2 to disease in goats. Therefore, the role of this toxin in caprine intestinal disease remains undetermined.
Enterotoxin is produced by less than 5% of C. perfringens isolates, with the highest prevalence of production by type A strains. This toxin is responsible for human gastrointestinal disease and also has been associated with diseases in dogs, horses, pigs, and several other animal species, including 1 case of enteritis caused by C. perfringens type A cpe-positive in a goat. 6
To the authors' knowledge, no cases of enterotoxemia due to C. perfringens type D producing CPE and CPB2 have been reported in goats. Two cases of subacute caprine enterotoxemia associated with CPE- and CPB2-producing C. perfringens type D and demonstration of the presence of CPB2 in the intestinal tract of those animals are presented in the current study.
A 1.5-year-old Alpine goat doe with no history of health problems on the previous day (case 1) was found dead. This goat belonged to a herd of 30 animals that had been fed a diet of alfalfa hay and grain for the previous 6 months; this goat had delivered twin kids 3 weeks before its death. Vaccination history of the herd included annual clostridial vaccination containing C. perfringens types C and D toxoids, and this doe had reportedly received the latest booster 6 months before death.
Case 2 involved a 2-week-old female Boer goat kid that was also found dead without premonitory clinical alterations. This animal belonged to a herd of 60 goats that had been fed a diet of alfalfa hay and grain for the previous year. The vaccination history of the herd was unknown.
Both animals were submitted for diagnosis to the California Animal Health and Food Safety Laboratory (San Bernardino, CA). Postmortem examination was performed on both carcasses approximately 3 hr after death, and samples of heart, lung, liver, spleen, kidney, skeletal muscle, small and large intestine, mesenteric lymph nodes, thymus, thyroid gland, and the whole brain were collected into 10% neutral buffered formalin. After 24 hr of fixation, samples were processed routinely for histology, sectioned at 4 μm, and stained with hematoxylin and eosin. Selected intestinal sections were also stained with Conn and Hucker Gram.
Four-micron-thick paraffin sections of abomasum, duodenum, ileum, and colon were processed by 2 indirect immunoperoxidase techniques, as previously described for CPB 21 and CPE, 2 using the Dako En Vision Kit, a according to the manufacturer's instructions. Primary antibodies were a rabbit polyclonal anti-CPB2 antibody and a monoclonal anti-CPE antibody prepared in house by one of the authors (B. A. McClane). Rabbit and goat small intestinal ligated loops injected experimentally with purified CPE or CPB2-positive C. perfringens type D, respectively, were used as positive controls. Small intestine from a goat that was culturally negative for C. perfringens carrying cpe and cpb2 genes was used as a negative control. Additional negative controls consisted of tissue sections of the 2 goats with enterotoxemia incubated with normal rabbit or mouse serum instead of the specific antibodies.
Small and large intestinal contents and feces were cultured aerobically and anaerobically on 5% sheep blood agar and aerobically on MacConkey agar. Isolates of C. perfringens were typed by a multiplex polymerase chain reaction (mPCR) technique to amplify segments specific for the genes encoding CPA, CPB, ETX, ITX, CPE, and CPB2, as previously described.4 Briefly, bacteria were grown on brain heart infusion agar plates, incubated anaerobically overnight at 37°C, and then processed for mPCR analysis using colony lysates as template. The mPCR products were then separated in 2% agarose gels, stained with ethidium bromide, and examined by ultraviolet transillumination.
Isolates were also examined by Western blot for production of ETX, CPB2, and CPE in vitro. 12,15 Supernatant from a recombinant C. perfringens type A isolate transformed to overexpress CPB2 was used as a positive control, while a C. perfringens type B isolate was used as a positive control for ETX and purified CPE was used as a positive control for CPE.
Ileal and colonic contents and feces were examined for CPA, CPB, and ETX by way of a commercial capture enzyme-linked immunosorbent assay (ELISA), following the manufacturer's instructions. b Feces were also examined for rotavirus, c and frozen sections of colon were examined by a fluorescent antibody technique for Bovine coronavirus. Feces were also examined by electron microscopy for viral particles and by a flotation technique for parasite eggs and protozoa.
Gross and histological findings were similar in both animals. Postmortem examination revealed that both animals were in good nutritional condition. Strands of fibrin were observed on the small and large intestinal serosa. The spiral colon was distended with liquid green content and gas; severe diffuse serosal edema gave the organ a conical shape. Mucosal surfaces of the rumen, abomasum, ileum, and colon were diffusely red. Multiple shallow, 2 to 3-cm-diameter, irregular, fibrin-covered ulcers (Fig. 1) (more marked around the ileocecal valve) were evident in the terminal ileum and colon. An excess of pleural fluid clotted when the thoracic cavity was opened (Fig. 2). The lungs were diffusely congested and edematous, and abundant froth filled the trachea and bronchi.
Histologically, there was moderate diffuse mucosal congestion throughout the gastrointestinal tract. The mucosa of the terminal ileum and proximal spiral colon had multiple areas of superficial to full-thickness coagulative necrosis with ulceration. These lesions were covered by pseudomembranes of fibrin, mono- and polymorpho-nuclear leukocytes, cell debris, hemorrhage, and many Gram-positive large rods with blunt ends, singly or in clusters (Fig. 3). These bacilli were consistently numerous in the intestinal lumen near the ileal and colonic ulcers, but they were rare in unaffected areas. Small numbers of slender Gram-negative bacilli were also observed throughout the intestinal mucosa. The remaining mucosa was multifocally hemorrhagic, and the lamina propria was infiltrated by neutrophils. There was diffuse severe proteinaceous interstitial and alveolar pulmonary edema.
Immunohistochemistry (IHC) of ileal and colonic sections showed that most large bacilli in the sections with areas of mucosal ulceration or necrosis were positive for CPB2 (Fig. 4), but none were positive for CPE. Positive staining for CPB2 was observed consistently around the rods and occasionally in the cytoplasm of these bacterial cells. Small clusters of slender bacilli diffusely present in the ileal and colonic lumen, but not specifically associated with areas of ulceration, were negative for both toxins by immunostaining. Goat intestinal loops inoculated with CPB2-positive C. perfringens showed staining with this antibody around or inside microorganisms, while rabbits inoculated with CPE showed positive immunostaining as a thin line on the apical membranes of the superficial epithelium and surrounding necrotic sloughed epithelial cells in the intestinal lumen. Positive immunostaining was not observed in the negative controls.
Clostridium perfringens was isolated from ileal and colonic contents and feces of both animals. Eleven isolates (4 from ileum, 6 from colon, and 1 from feces) from case 1 and 4 isolates from ileum of case 2 were mPCR genotyped as CPB2-positive C. perfringens type D. In addition, all isolates from case 1 carried the cpe gene that encodes CPE. All isolates from both cases expressed ETX, while 7 of the isolates from case 1 and 2 isolates from case 2 expressed CPB2 by Western blot (Fig. 5). Five isolates from case 1 also expressed CPE by Western blot. The 5 CPE-producing strains sporulated in vitro, while the remainder did not.
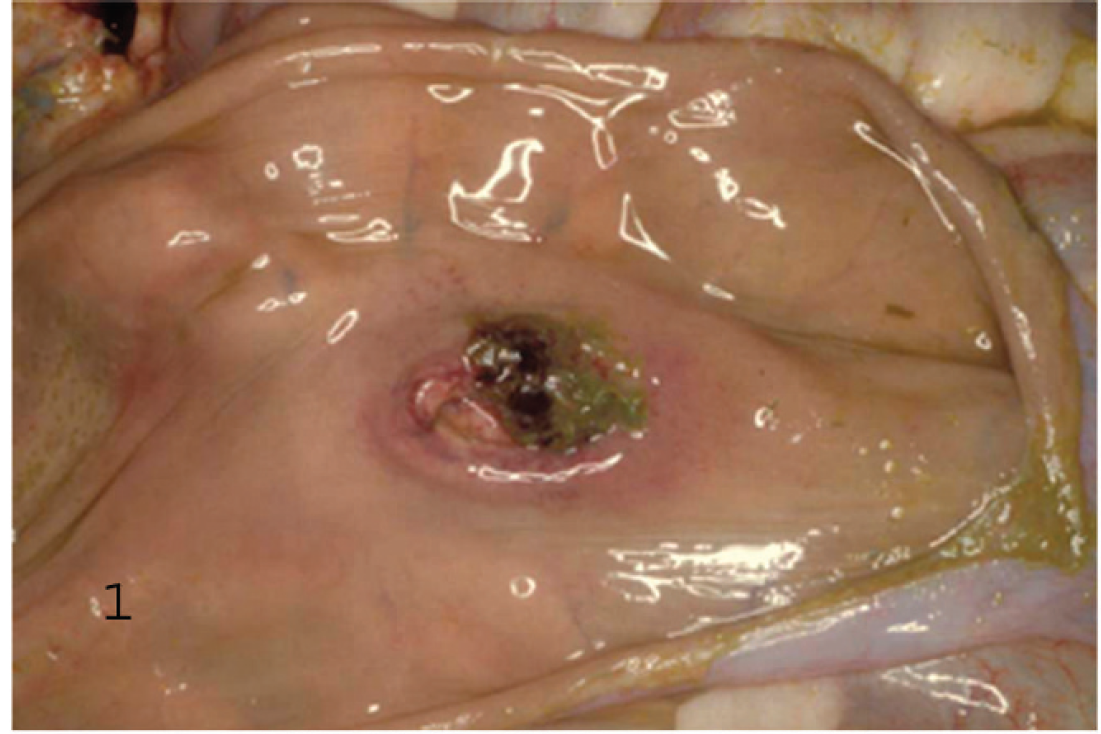
Focal ulcerative, fibrinonecrotic ileitis in a goat with beta2 toxin/enterotoxin–positive Clostridium perfringens type D enterotoxemia (case 1).
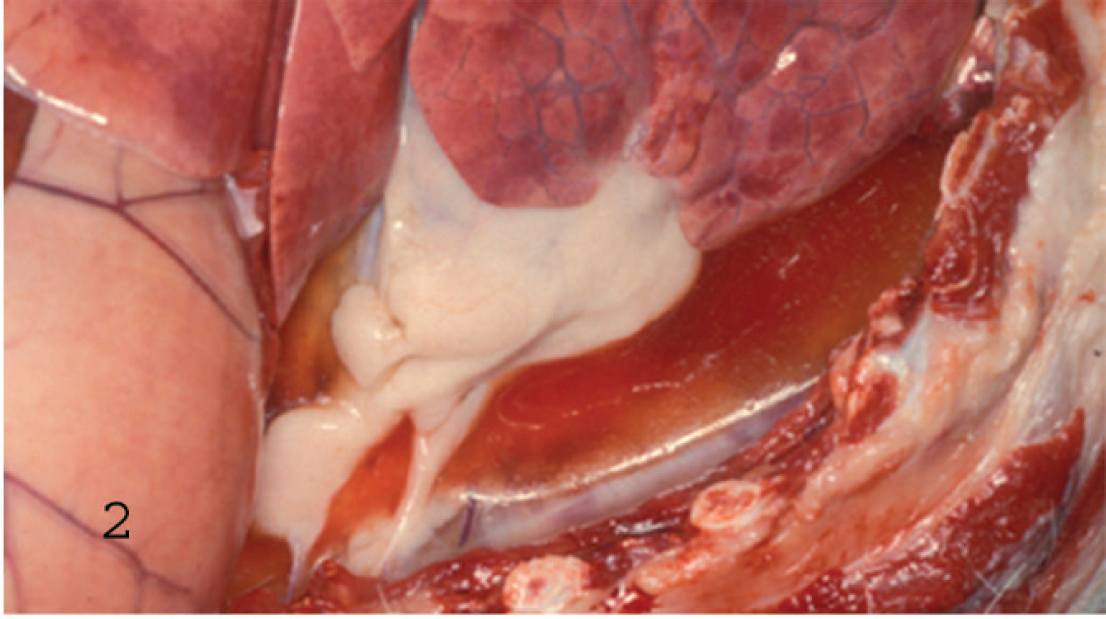
Abundant clotted, protein-rich pleural fluid in a goat with beta2 toxin/enterotoxin-positive Clostridium perfringens type D enterotoxemia (case 1).
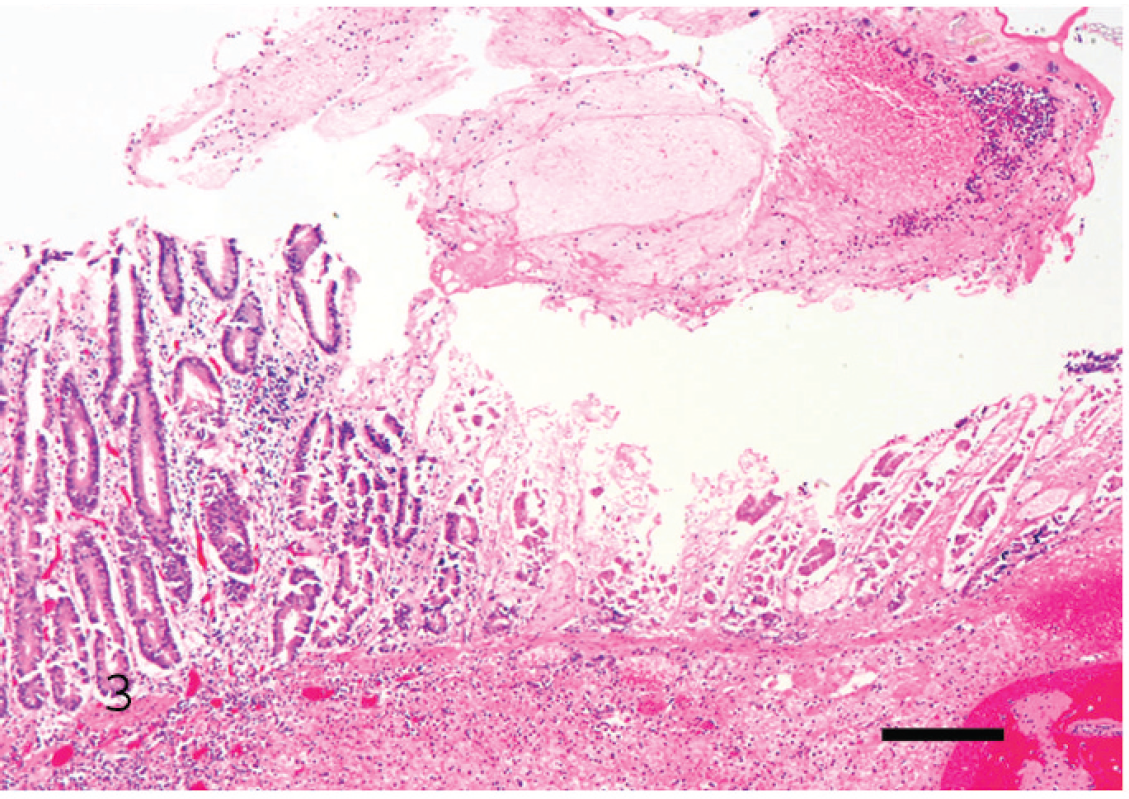
Focal ulcerative fibrinonecrotic ileitis in a goat with beta2 toxin/enterotoxin-positive Clostridium perfringens type D enterotoxemia. Hematoxylin and eosin. Bar = 100 μm (case 1).
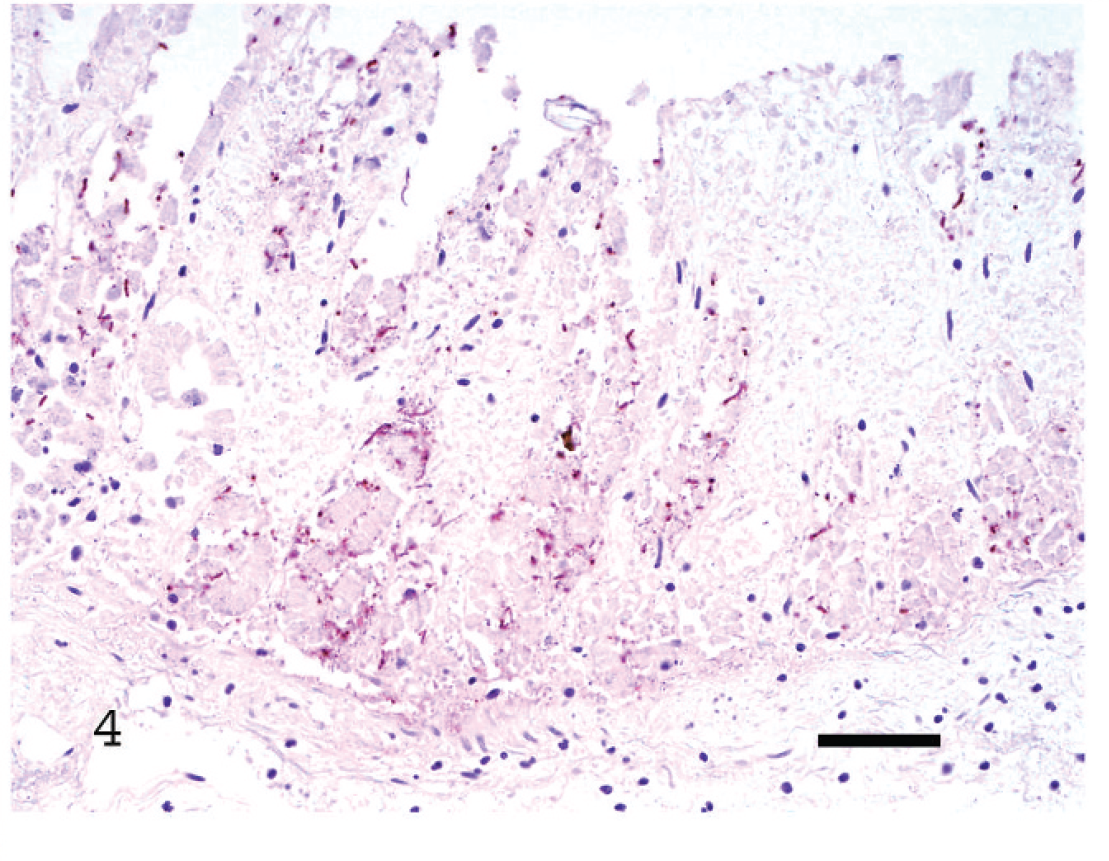
Beta2 toxin (CPB2)-positive Clostridium perfringens in the colon of a goat with CPB2/enterotoxin-positive C. perfringens type D enterotoxemia. EnVision. Bar = 50 μm (case 1).
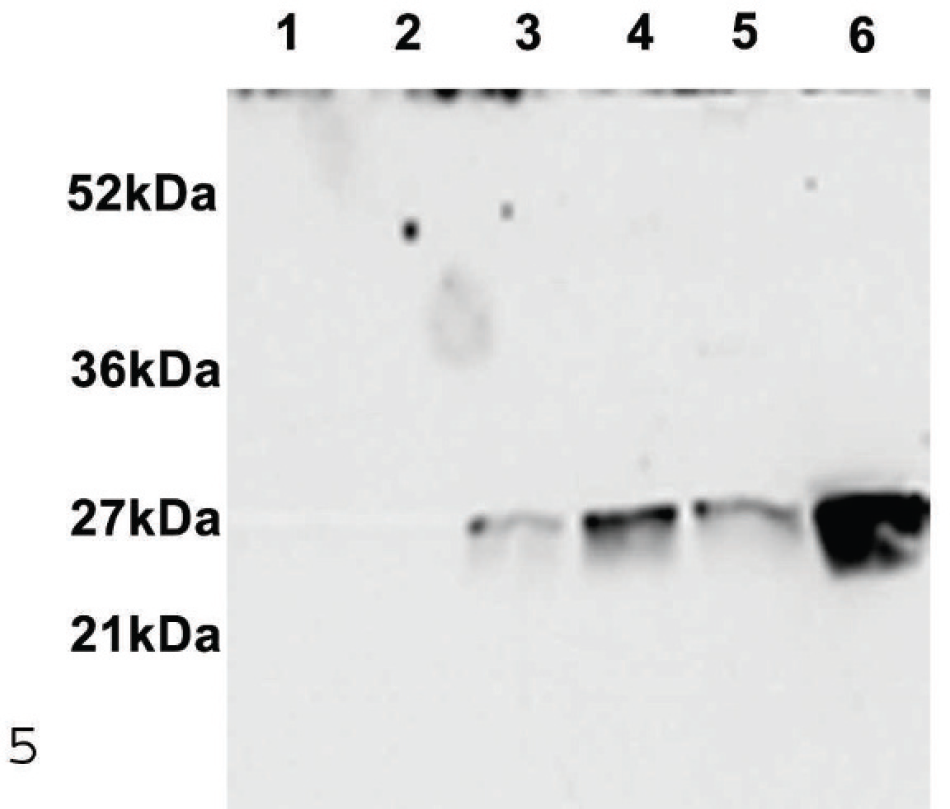
Western blot for beta2 toxin (CPB2) in Clostridium perfringens isolates obtained from a goat with enterotoxemia. Lane 1: Clostridium perfringens CPB2-negative; lanes 2–6: strains 10, 5, 8, 7, and 6 (case 1), respectively.
Clostridium perfringens, CPA, and ETX were detected in ileal and colonic contents and feces of both goats by competitive ELISA. No aerobic or other anaerobic bacterial pathogens were isolated from either of the goats, and neither rotavirus nor coronavirus was detected. No parasites, parasite eggs, or viral particles were detected in feces.
Clostridium perfringens type D enterotoxemia was suspected in both cases based on the history (sudden death), pathological findings (fibrinonecrotic ileocolitis, pulmonary edema, and excess of protein-rich pleural fluid), and isolation of CPB2-producing C. perfringens type D. Definitive diagnosis was confirmed by detection of ETX in intestinal contents, which is considered confirmatory for this condition. 17
Beta2 toxin was detected in the intestine of these goats by IHC. CPB2 has been associated with enteritis in several animal species, including piglets, 8 horses, 8 calves, 13 and lambs. 14 Clostridium perfringens type A carrying the cpb2 gene was isolated from the intestinal content of a goat kid with hemorrhagic enteritis, 5 and it was suggested that CPB2 was associated with the pathogenesis of the mentioned condition. However, even though the cpb2 gene was detected, no information was provided about expression of CPB2 in vivo or in vitro.5 There is no direct evidence (such as fulfillment of molecular Koch's postulates) in goats to indicate that CPB2 has a role in enteric disease.
Immunohistochemistry has been used to detect clostridial toxins in tissues, including CPB2, in sections of the gastrointestinal tract of animals. 1 In the current study, CPB2 was detected in the intestine of both goats by IHC. In addition, most isolates from both goats produced CPB2 in vitro. Beta2 toxin was detected around bacteria and was associated with intestinal lesions. Thus, results of the present study have confirmed the findings of others, 1 namely, that CPB2 is produced in vivo, which lends support to the hypothesis that this toxin plays a part in the pathogenesis of some forms of enterotoxemia and other forms of enteric disease. Beta2 toxin is reportedly necrotizing and may have been at least partially responsible for the intestinal necrosis in both goats. Necrotizing colitis is frequently seen in goats infected with type D, but this lesion is usually diffuse; the deep, acute, multifocal ulcers in the terminal ileum of the goats described in the current study are uncommon. Furthermore, large numbers of CPB2-positive rods were detected by IHC in close association with the ileal ulcers but rarely in other areas of the gastrointestinal mucosa. These findings may indicate that CPB2 was at least partly responsible for the intestinal ulcers observed in the goats. If CPB2 caused intestinal epithelial damage, it may have facilitated uptake of ETX into systemic circulation and could explain the peracute nature of the deaths of these animals.
Enterotoxin was not detected in the intestine of either goat, although most of the type D C. perfringens isolates from case 1 were CPE positive. Enterotoxin production is co-regulated with sporulation and probably did not occur in the intestine of this goat. It is, therefore, unlikely that CPE played a major role in the pathogenesis of enterotoxemia in the goat.
No lesions were observed in the brains of the 2 goats. Perivascular edema and focal symmetrical necrosis of the brain are diagnostic for type D enterotoxemia in sheep, 17 and these lesions have been described less frequently in goat enterotoxemia. 19 Absence of brain lesions in the goats in the current study does not, therefore, preclude a diagnosis of enterotoxemia.
One of the goats in the current study (case 1) had been vaccinated against types D and C enterotoxemia 6 months before death, indicating that this animal may not have been protected. Goats should receive a booster immunization every 3 to 4 months. 18 In addition, commercial enterotoxemia vaccines usually contain beta and epsilon toxoids, but none for CPB2. This may explain why conventional enterotoxemia vaccines did not protect the goat. The results of the present study indicate a role for a CPB2-producing C. perfringens type D variant in caprine enterotoxemia, and they indicate as well that further study is needed.
Acknowledgements. The authors thank Dr. Michel Popoff (Pasteur Institute, Paris) for supplying antibodies against CPB2, Dr. P. Hauer (NVSL, Ames, IA) for providing antibodies against epsilon toxin, Ms. E. J. Hurley for excellent technical assistance, and Ms. S. Fitisemanu for excellent work and dedication in typing this report.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Bio-X Diagnostics SPRL, Jemelle, Belgium.
c.
Rotaclone®, Meridian Bioscience Inc., Cincinnati, OH.
