Abstract
Mycotoxicoses are usually a consideration in large animal species but can affect companion animals as well. Due to increasing interest and the ease of using rabbits as laboratory models, a growing number of published experimental studies discuss the effects of various mycotoxins on this species. However, the available evidence is fragmented and heterogeneous, and has not recently been collated in a review, to my knowledge. Although mycotoxicoses in rabbits are typically subclinical, clinical signs can include weight loss, anorexia, gastrointestinal disorders, stunted growth, reproductive abnormalities, and susceptibility to infections. An antemortem diagnosis typically relies on a comprehensive clinical history, and assessment of clinical signs and relevant laboratory findings, with confirmation of exposure achieved through the measurement of mycotoxin concentrations in feed or target organs. My review focuses on the clinicopathologic and histopathologic effects of the mycotoxins most important in rabbits, including fumonisins, ochratoxins, aflatoxins, trichothecenes, and zearalenone. This review offers a thorough overview of the effects of mycotoxins in rabbits, serving as a one-stop resource for veterinary practitioners, diagnosticians, and researchers.
Mycotoxins are secondary metabolites of specific filamentous fungi (most commonly Aspergillus spp., Fusarium spp., and Penicillium spp.) that contaminate various agricultural commodities on a worldwide basis. The global mycotoxin prevalence varies significantly depending on the type of mycotoxin or the analytical method used for detection. 36 However, in a 2020 study, the reported prevalence of one or more mycotoxins in food and feed grain crops above detectable levels was 60–80% globally. 36 When consumed by humans and animals, mycotoxins can have severe health implications, leading to a variety of acute or chronic manifestations. Hence, they are regarded as a global threat not only to public health but also to animal production due to the implicated substantial economic losses.
More than 500 mycotoxins are recognized, with notable examples including zearalenone (ZEN), deoxynivalenol (DON), aflatoxin B1 (AFB1), and fumonisins. 67 Contamination with mycotoxins can occur at both pre-harvest (agricultural crops) and post-harvest or storage (agricultural commodities and feedstuff) levels; the prevalence and concentrations of mycotoxins vary, depending on geographic location, environmental conditions (e.g., humidity, temperature, substrate composition), and crop type or years. 15 In reality, more than one mycotoxin is usually encountered in contaminated commodities, which can have synergistic, additive, or antagonistic effects (e.g., ZEN commonly co-occurs with Fusarium toxins such as DON in cereal crops).27,31 Notably, an expanding body of research since ~2010 explores the effects of “masked mycotoxins,” that is, mycotoxin derivatives that can elude conventional analytical methods due to their altered chemical structure in the affected plant. 11 The toxicogenic potential of masked mycotoxins is still under investigation, with some isoforms exhibiting either increased or decreased toxicity. 11
Animals are typically exposed to mycotoxins via the consumption of contaminated feed or rarely via inhalation or skin contact. 133 Mycotoxicoses encompass a wide spectrum of toxicologic effects, with their nature varying based on the chemical structure of the respective mycotoxin. 46 For instance, fumonisins exhibit structural similarities to sphinganine, which is an intermediate product in the production of sphingolipids; the accumulation of sphinganine results in impaired cellular function. 46 Given their chemical diversity, it is challenging to categorize mycotoxins based on their mode of action; in general, they disrupt cellular function through a variety of mechanisms (e.g., inhibition of protein synthesis, perturbation of cell membrane permeability, DNA alterations). 135 The expressions of toxicity differ depending on the quantity consumed and duration of exposure. Documented manifestations of mycotoxicoses in various species, including rabbits, 66 include impaired reproductive function, stunted growth, susceptibility to infections as a result of immunosuppression, and rarely death.
Although mycotoxins are usually discussed in the context of farm animal medicine, they can affect companion animals as well. An outbreak of pancytopenia in cats in the United Kingdom, proposed to be caused by a trichothecene mycotoxin, has highlighted the need to enhance the awareness and understanding of the effects of mycotoxins. 54 Research about mycotoxins in various species tends to emphasize the effects on production animals. The effect of mycotoxins in rabbits, a species employed as a production, experimental, and companion animal, has been a subject of ongoing investigation, commonly in the context of experimental toxicologic studies and infrequently in in vivo studies. Rabbits have also long been employed in the production of antibodies against various mycotoxins, primarily used for the development of detection immunoassays. 88 Rabbits are considered susceptible to at least some types of mycotoxins, presumably due to their prolonged exposure via cecotrophy. 92 The manifestation of mycotoxicoses in rabbits is typically subclinical, and the effects can often be overlooked. Clinical manifestations of mycotoxins in rabbits could include generic signs, such as fever, weight loss, and, in younger animals, stunted growth. 96 However, recurring gastrointestinal issues and susceptibility to infections (e.g., Pasteurella multocida or Aspergillus fumigatus) suggest the possibility of mycotoxicity in rabbits.103,119
Understanding mycotoxicoses in rabbits is of paramount importance, not solely for safeguarding animal health, but also for public health reasons. Mycotoxin carryover from contaminated feed to animal-derived food products is well-documented in production animals 140 and has also been reported in rabbits,5,28 with potential risks to human health. Furthermore, given that rabbits are employed extensively in experiments, the potential presence and impact of mycotoxins, which are often unrecognized, could affect experimental outcomes in unanticipated ways. I provide here an overview of mycotoxicoses in rabbits, with emphasis on clinicopathologic and histopathologic findings that can facilitate their detection. Some clinical and diagnostic considerations are raised; knowledge gaps and areas for future research are also highlighted.
Prevalence of mycotoxins in rabbit feed
A considerable volume of published research addresses the prevalence of mycotoxins in animal feedstuffs, primarily concerning livestock and infrequently companion animals. Limited studies, conducted at the level of a single country, have addressed the prevalence of mycotoxins in rabbit feed. 56 In a 2012 study, aflatoxins (AFs) were recovered in 100% of rabbit and chinchilla finished-feed samples in Argentina, with the mycotoxin prevalences of 100% for AFs (<1.70–22.6 μg/kg of feed), 95% for DON (222–1,740 μg/kg of feed), 100% for fumonisins (222–6,000 μg/kg of feed), 98% for ochratoxin A (OTA; <5–26.6 μg/kg of feed), 98% for T-2 toxin (<50–130 μg/kg of feed), and 100% for ZEN (<50–178 μg/kg of feed). Mycotoxin co-occurrence was reported in 100% of feedstuff. 56 A 2002 study from Argentina revealed an OTA prevalence of 25% (18.5–25 μg/kg of feed) in the examined samples from finished rabbit feeds. 24 A study from India in 2007 reported a 77% prevalence of AFs in rabbit finished-feed samples. 101
The United States Food and Drug Administration and the European Union have implemented restrictions and guidelines for the daily maximum concentration of mycotoxins that can be safely consumed by humans and animals. 38 The current guidance levels apply to a variety of species, such as swine, ruminants, and poultry. Specifically for rabbit feed, guidance levels have been established only for fumonisins (≤5,000 μg/kg of feed). 37
Mycotoxins and associated effects
The main clinicopathologic and histopathologic effects of some common mycotoxins studied in rabbits are summarized below and tabulated (Table 1). Searches were performed in Google Scholar, PubMed, CAB Direct, Web of Science, and Scopus, using the Boolean search string “(rabbit OR rabbits) AND (mycotoxin OR mycotoxins OR given name of mycotoxin e.g. aflatoxins).” The search retrieved no review on rabbit mycotoxicoses during 2014–2023.
Notable mycotoxins and their clinical, clinicopathologic, and histopathologic effects in rabbits.
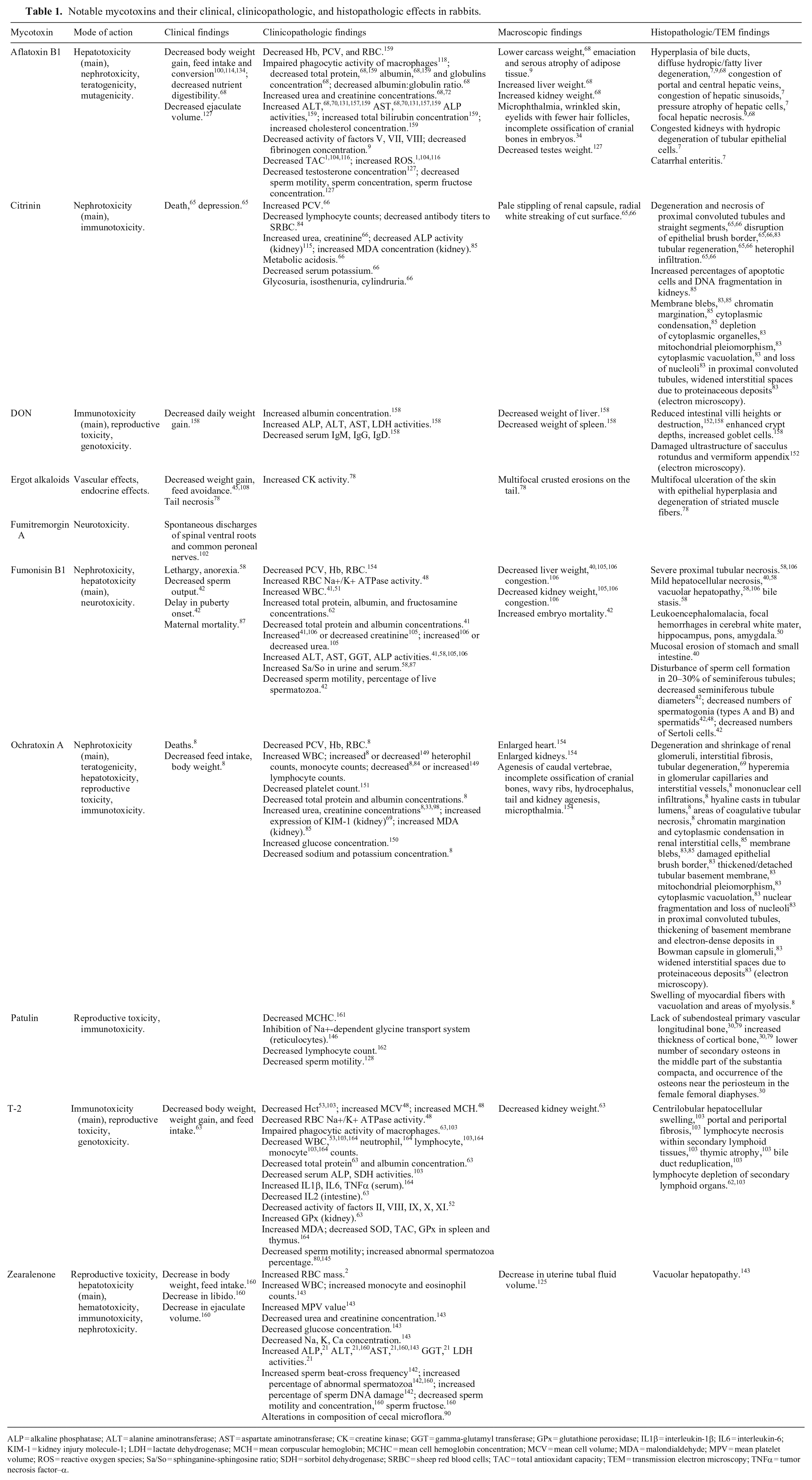
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; CK = creatine kinase; GGT = gamma-glutamyl transferase; GPx = glutathione peroxidase; IL1β = interleukin-1β; IL6 = interleukin-6; KIM-1 = kidney injury molecule-1; LDH = lactate dehydrogenase; MCH = mean corpuscular hemoglobin; MCHC = mean cell hemoglobin concentration; MCV = mean cell volume; MDA = malondialdehyde; MPV = mean platelet volume; ROS = reactive oxygen species; Sa/So = sphinganine-sphingosine ratio; SDH = sorbitol dehydrogenase; SRBC = sheep red blood cells; TAC = total antioxidant capacity; TEM = transmission electron microscopy; TNFα = tumor necrosis factor–α.
The terms acute, subacute, subchronic, and chronic follow published definitions. 26 Specifically, acute exposures are defined as single-dose studies, subacute exposures as studies with a duration of 14–28 d, subchronic exposures as those lasting 90 d on average, and chronic exposures as those lasting 270 d on average. An additive mycotoxin effect is defined as the baseline effect that is theoretically expected from the combination of multiple compounds, a synergistic effect is defined as a combination of effects that is greater than the additive effect expected of the individual mycotoxins, and an antagonistic mycotoxin effect is defined as a combined effect that is less than what would be expected. 124
Aflatoxins
AFs are mainly produced by fungi of the genus Aspergillus (primarily A. flavus and A. parasiticus) and can be detected in a variety of feedstuffs, such as wheat, corn, nuts, barley, oats, and alfalfa.86,123 Currently, >20 types of AFs are recognized, with AFB1, AFB2, AFG2, and AFG2 being the most toxic. 105 AFs are primarily metabolized in the liver to a bioactive metabolite (8,9-epoxide) that can react with several cellular constituents, such as proteins, lipids, and DNA, causing oxidative damage and cellular injury. 156 AFB1, in particular, is well-known to be carcinogenic in humans, rodents, non-human primates, fish, and birds, and is implicated particularly in the development of hepatocellular carcinomas. Other documented actions of AFs include nephrotoxicity, neurotoxicity, teratogenicity, and mutagenicity.17,105 Aflatoxicosis is possibly the most important mycotoxicosis in rabbits, and there have been reports of AFB1 outbreaks with clinical signs ranging from anorexia, dullness, weight loss, and impaired productivity to jaundice and high mortality rates at later stages.82,132 Rabbits are considered to be highly susceptible to AFs, with a reported oral LD50 for AFB1 of 300 μg/kg of body weight (BW), which is among the lowest of studied species. 20
The hepatotoxicity of AFB1 has been studied extensively in rabbits with various dosage schemes. The subchronic daily oral exposure of young bucks at 250 μg AFB1/kg of feed resulted in increased activity of serum liver enzymes such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT), although the reported changes were minimal and of questionable clinical significance. More significant changes were noted histologically, including bile duct hyperplasia, diffuse fatty liver degeneration, and focal hepatocellular necrosis. 68 However, such histologic changes might not have been extensive enough to substantially affect serum liver enzyme activities. The pattern of increased liver enzyme activities coupled with histopathologic alterations in liver (particularly hepatocellular vacuolation) appears to be consistent through subchronic daily oral exposures at similar, 72 lower, 7 or significantly higher AFB1 concentrations, 159 and also in subacute daily oral exposures at slightly higher concentrations.3,64,105 This underscores the hepatoxic effect of this mycotoxin; however, the magnitude of changes, and consequently their clinical significance, varies between experiments, with more clinically significant changes noted at the higher concentrations.3,64
The effects of AFB1 on other clinicopathologic analytes of rabbits have also been documented and are generally reported to be mild. Namely, daily subchronic oral exposure of young rabbits to AFB1 at 250 μg/kg of feed resulted in mild decreases in total protein, albumin, and globulin concentrations, mildly decreased glucose concentrations, as well as mildly increased concentrations of urea and creatinine. 68 Although the magnitude of the reported changes in total protein, albumin, globulin, urea, and creatinine concentrations raises doubts about their clinical significance, the mildly decreased mean glucose concentration might be clinically significant. The subchronic daily oral exposure of young rabbits to a slightly higher AFB1 concentration (300 μg/kg of feed) resulted in likely clinically significant changes in total protein, albumin, globulin, and urea concentrations. 72 Mild changes in glucose and protein concentrations were found in a 2003 study addressing slightly higher daily oral dosages of AFB1 (15 and 30 μg/kg BW), along with mildly decreased Hb, RBC, and PCV, and mildly increased cholesterol concentration. 159 Subacute daily oral exposure to relatively higher dosages of AFB1 (60 and 90 μg/kg BW) was reported to significantly affect coagulation in exposed rabbits by decreasing the activity of factors V, VII, and VIII, as well as fibrinogen concentration. 9 Consumption of AFB1 was also found to affect the redox status of rabbits, by decreasing total antioxidant capacity (TAC) and increasing reactive oxygen species (ROS) concentration, both at serum and organ levels.1,104,116 Finally, AFB1 was found to depress the phagocytic activity of rabbit macrophages, indicating an adverse effect on the immune system, which might account for the clinically reported susceptibility to infections. 118
As is the case with several mycotoxins, AFB1 was found to have a detrimental effect on rabbit growth. AFB1 negatively affected the daily body weight gain, feed intake, and feed conversion rate of growing rabbits orally exposed to concentrations of 50–500 μg/kg of feed daily for 2 mo.100,114,134
The developmental toxicity of AFB1 has been established in several species, including rabbits. Daily oral exposure to 50 μg/kg BW AFB1 on days 6–18 of gestation caused an array of significant developmental abnormalities in embryos including microphthalmia, wrinkled skin, and incomplete ossification of cranial bones. 34 Similar results were noted in a 2005 study, which identified the minimum oral teratogenic dosage of AFB1 in rabbits as 100 μg/kg BW. 155 The reproductive toxicity of AFB1, in turn, has been less well established, with sporadic reports. Subchronic oral exposures to AFB1 on a daily basis resulted in decreased testes weight and serum testosterone concentration, as well as significantly decreased ejaculate volume, sperm concentration and motility, and semen fructose concentration. 127
Citrinin
Citrinin is produced by fungi of the genera Aspergillus and Penicillium and commonly contaminates cereals, such as wheat, barley, oats, corn, and beans. 60 Citrinin has mainly nephrotoxic and immunosuppressive actions,94,110 and acts by inducing mitochondrial pore permeability transition 23 and interfering with the respiratory chain. 18 In rabbits, the 72-h LD50 was reported to be 50,000 μg/kg BW by intraperitoneal administration and 134,000 μg/kg BW when administered orally. 65
The experimental citrinin dosages used in vivo in rabbits are generally higher than those reported in relevant dietary concentrations on a worldwide basis. 74 Acute oral exposure of rabbits to various high dosages of citrinin (120,000–150,000 μg/kg BW) or subacute daily oral exposure to multiple sublethal dosages (33,500 or 77,000 μg/kg BW) caused variable degeneration and necrosis of the renal tubules, reflecting its potent nephrotoxic action. 65 Similarly, a single oral dosage of 120,000 μg citrinin/kg BW caused marked azotemia and metabolic acidosis, as well as hemoconcentration and mild hypokalemia in male rabbits. In the same study, the single oral administration of 80,000 μg/kg BW or 100,000 μg/kg BW caused marked azotemia, a decreased creatinine clearance rate, as well as renal tubular dysfunction and necrosis, with glucosuria, isosthenuria, and cylindruria when observed for 7 d; these changes were mostly reversible by the end of the experiment (day 7). 66 In a 1977 study, the intraperitoneal bi-daily exposure to significantly lower citrinin concentrations (2,000 μg/kg BW) for 7 d caused a decrease in kidney alkaline phosphatase (ALP) activity, possibly due to its conversion to an inactive form; no effect was observed on serum ALP activity. 115 Subchronic daily oral exposure of young bucks to 15,000 μg citrinin/kg of feed caused increased percentages of apoptotic cells with DNA fragmentation in kidney tissue 85 and depressed humoral immune responses. 84 These changes were also reflected in electron microscopy findings of nuclear crenation and fragmentation, loss of nucleoli, depletion of cytoplasmic organelles, mitochondrial pleomorphism, uniform folding of cell membranes, and cytoplasmic vacuolation in proximal convoluted tubules. 83 Finally, the in vitro exposure of rabbit blood samples to citrinin caused significantly decreased glucose concentration at the highest mycotoxin concentration 14 ; however, this finding has not been confirmed in an in vivo study.
Ergot alkaloids
Ergot alkaloids are produced mainly by the fungi of the genus Claviceps (primarily C. purpurea) and are further classified into clavines, lysergic acid amides, and ergopeptines, according to their chemical structure. 129 Ergot alkaloids are implicated in the oldest reported mycotoxicosis in humans and are known to cause ischemic acral necrosis in several species, such as ruminants.6,78 A few studies have addressed the effects of ergot alkaloid in rabbits. The sensitivity of rabbits to ergot alkaloids has been suggested 57 ; other authors have reported decreased weight gain and feed avoidance in exposed animals.45,108 Daily oral exposure of young animals to relevant dietary ergot alkaloid concentrations (410 ± 250 μg/kg of feed) was hypothesized to cause tail necrosis, likely due to peripheral ischemia. 78 Notably, mildly increased creatine kinase (CK) activity (1,000–2,000 IU/L) was noted in the affected rabbits, which indicated mild acute muscle damage and possibly reflected the rhabdomyolysis observed in the tail. 78
Fumonisins
Fumonisins are produced mainly by the fungi of the genus Fusarium (primarily F. verticillioides and F. proliferatum) 91 and typically contaminate grains (e.g., maize) and hay.147,148 There are 4 main chemical analogs of fumonisins (A–C and P). Fumonisins B1 and B2 (FB1, FB2) are the most abundant and toxic variants, with FB1 being far more prevalent.19,117 Fumonisins have complex toxicity that is exerted mainly via the disruption of cell membrane sphingolipid metabolism (through the inhibition of ceramide synthase) and induction of oxidative stress.76,121 In rabbits, FB1 is rapidly eliminated from circulation after ingestion and is primarily excreted in feces. 106
Fumonisins have been reported to primarily exert nephrotoxic and hepatotoxic effects in several species. In rabbits, the nephrotoxic and hepatotoxic effects of FB1 have also been established and reported with various exposure durations and doses. Specifically, the acute oral exposure of adult rabbit bucks to a high FB1 concentration (31,500 μg/kg BW) affected serum ALP, ALT, AST, and gamma-glutamyl transferase (GGT) activities, with moderate-to-marked, likely clinically significant, increases noted for ALT, AST, and GGT. Marked azotemia and increased urine protein concentrations were observed, reflecting FB1 nephrotoxicity. Subacute oral exposure of weaned rabbits to lower FB1 concentrations (10,000 μg/kg BW) resulted in altered hepatic and mitochondrial membrane lipid profiles and increased Na+/K+ ATPase activity in erythrocytes, which was deemed a potential mechanism for its nephrotoxic action.136,137 The subacute daily intravenous administration of FB1 to adult rabbits at significantly lower dosages (150, 300, 500, or 1,000 μg/kg BW) induced lethargy, anorexia, and hepatotoxicity demonstrated by significantly increased serum liver enzyme activities and vacuolar hepatopathy, hepatocellular necrosis, and bile stasis on histopathology. 58 Nephrotoxicity was also observed at these exposure levels, as evidenced by decreased urine production and severe proximal tubular necrosis on histopathology. Importantly, increased sphinganine-to-sphingosine ratios were found in liver and kidney tissue, as well as in serum and urine, which reflects the toxin’s detrimental effect on sphingolipid metabolism and the proposal to use this ratio as a potential FB1 exposure marker. 58 Subacute daily oral exposures to FB1 dosages at similar (1,500 μg/kg BW) or subchronic daily oral exposures to lower (100 to 10,000 μg/kg feed) dosages similarly caused a mild increase in serum liver enzymes activities, a mild decrease in serum urea concentration, and variable mild changes in serum creatinine.41,105 Histologically, hepatic and renal congestion and vacuolar hepatopathy were noted, as with other exposures. 105 A chronic oral daily exposure of rabbit bucks to FB1 caused significantly lower liver and spleen weight, higher kidney and testes weights, dose-dependent liver necrosis, Sertoli cell degeneration, and mucosal erosion of small intestine and stomach. 40 These findings might indicate a wider anatomic distribution of FB1 toxicity during chronic exposures.
The effect of Fusarium toxins on other clinicopathologic analytes is not documented adequately in rabbits. Subacute daily oral exposure of weaned rabbits to FB1 resulted in mildly increased serum total protein, albumin, and fructosamine concentrations, 62 which are likely of little clinical significance and could reflect a degree of dehydration. The subchronic daily oral exposure of adults to similar dosages of FB1 (7,500 or 10,000 μg/kg of feed) caused, in turn, a mild decrease in PCV, RBC, and Hb values, a mild increase in WBC, as well as mildly decreased total protein and albumin concentrations. 41 These results were further supported by a study investigating similar daily oral dosages in pregnant does and reflect the influence of exposure duration on toxicologic effects. 51 In contrast to those results, when bucks were orally exposed to slightly higher FB1 concentrations on a daily basis for a similar amount of time (5 wk), no significant differences were observed in the examined hematologic and serum biochemical analytes. 44 With regard to the effect of fumonisins on the redox status, there are very few reports in rabbits. In particular, the subchronic daily oral exposure at 5,000 μg FB1/kg of feed caused a significant decrease in malondialdehyde (MDA) concentration in the RBC hemolysate of adult rabbit bucks, 48 which however is of questionable clinical significance.
Fusarium toxins are a well-established cause of primary leukoencephalomalacia in horses. 47 Similar neurotoxic action was exhibited in pregnant does after subacute daily oral exposures to a relatively high dosage of FB1 (1,750 μg/kg BW). Specific histopathologic findings included leukoencephalomalacia and focal hemorrhages in cerebral white matter and hippocampus. 16 Subchronic daily oral exposures at FB1 concentrations >5,000 μg/kg of feed caused a significant decrease in acetylcholinesterase concentrations in hippocampus, pons, and amygdala areas. 50
Fumonisins were also reported to exert reproductive toxicity in rabbits. Chronic daily oral exposure of pubertal bucks at higher FB1 concentrations (5,000, 7,500, and 10,000 μg/kg of feed), which have generally been reported in agricultural commodities, caused a decrease in daily sperm production, sperm mass activity, motility and percentage of live spermatozoa, as well as an increase in embryo mortality in a dose-dependent manner.42,43 Similar results were found with subchronic daily oral exposures at similar concentrations; intensity of spermatogenesis was decreased by 43%. 48 In contrast to those results, no effects were reported on testicular and sperm parameters of subchronically orally exposed bucks at similar or higher concentrations (10,000 and 20,000 μg/kg of feed) on a daily basis. 138 In females, subacute daily oral exposure of does at lower FB1 concentrations (500 or 1,000 μg/kg BW) was associated with maternal deaths and increased urine, serum, and kidney sphinganine-to-sphingosine ratios; however, embryotoxicity was not observed at the studied doses. 87
Ochratoxins
Ochratoxins are produced by fungi of the genera Aspergillus and Penicillium and are commonly encountered in cereals, dried fruit, nuts, and animal byproducts. 89 There are 3 recognized ochratoxins (A–C), with OTA being far more prevalent. 60 OTA is reported to primarily induce nephrotoxicity in several species, but can also cause hepatotoxicity, reproductive toxicity, teratogenicity, carcinogenicity, and immunotoxicity. 60 Its main mechanisms of action include oxidative stress, disturbance of calcium homeostasis, and inhibition of protein synthesis.60,139 Rabbits are more susceptible to OTA compared to mice, rats, and guinea pigs, but are generally considered less susceptible than other species such as pigs, possibly due to differences in pharmacokinetics.49,113 The reported LD50 for OTA in rabbits is 10,000 μg/kg BW. 97 After ingestion, the bioavailability of OTA is 56%; wide tissue distribution occurs, with the maximum concentration observed 1 h after ingestion.49,60 OTA is subsequently excreted in urine or feces, with rabbits demonstrating rapid metabolism and elimination rates.49,60 A degree of OTA glucuronidation was noted in rabbit liver S9 fractions, suggesting a detoxification pathway in this species. 25
In rabbits, the studies addressing the toxicity of ochratoxins are relatively limited and primarily focus on the nephrotoxic action of OTA. Specifically, acute intraperitoneal exposure to 500 μg OTA/kg BW caused increased expression of kidney injury molecule-1 (KIM-1), degeneration and shrinkage of renal glomeruli, interstitial fibrosis, variable degrees of tubular degeneration and atrophy, and loss of renal epithelial cell nuclear and cytoplasmic definition. 69 In a comparable fashion, the subchronic daily oral exposure to 750 μg OTA/kg of feed, which has generally been reported in agricultural commodities, significantly increased the MDA concentration in kidney tissue; it increased the percentage of apoptotic cells, the incidence of nuclear fragmentation, and the cytoplasmic blebbing in renal interstitial cells when studied by electron microscopy. 85 Similar results were reported for subchronic daily oral exposure to 1,000 μg OTA/kg of feed, which is a concentration that is generally higher than those reported in agricultural commodities; OTA exposure also caused kidney enlargement and discoloration, degeneration of the proximal convoluted tubules, disruption of the mitochondrial membrane, and swelling of the endoplasmic reticulum. 114 Subchronic daily oral exposures at similar or significantly lower OTA concentration caused variable degrees of azotemia.8,33,98 The effect of OTA on redox status of rabbits, which is implicated in its nephrotoxicity, might be mediated by the downregulation of nuclear factor erythroid 2–related factor 2 (NRF2) transcription factor and heme oxygenase-1 (HO-1) gene. 8
Published reports on the hepatotoxicity of OTA in rabbits are relatively scarce. Subchronic daily oral exposure to relevant dietary concentrations (30 μg OTA/kg of feed) caused mild increases in serum AST and ALP activities. 33 Similarly, subchronic daily oral exposures at generally higher reported relevant dietary concentrations (300 μg/kg of feed) resulted in mild-to-moderate, likely clinically significant, increases in ALP, AST, and lactate dehydrogenase (LDH) activities, although the latter were attributed to cardiac injury. 8 Mild effects were reported for ALT and ALP activities, as well as a mild but possibly clinically significant increase in AST activity throughout the experimental period for subchronic daily oral exposures higher than relevant dietary concentrations (1,000 and 2,000 μg/kg of feed), 98 indicating a dose-dependent element to OTA-induced hepatotoxicity.
The effects of OTA on other clinicopathologic analytes are relatively less well documented. A 1996 study indicated that in vitro exposure of rabbit RBCs to OTA resulted in their lysis in a concentration-dependent manner. 167 Indeed, the toxic effect of OTA on rabbit RBCs is reflected in the results of subsequent in vivo studies, in which RBC indices appear to be negatively affected. Specifically, the subchronic daily oral exposure to a relevant dietary OTA concentration (30 μg/kg of feed) caused a mild, possibly clinically significant, decrease in RBC and Hb concentrations, as well as in total protein, albumin, catalase, and superoxide dismutase (SOD) concentrations, whereas glucose and serum MDA concentrations were significantly increased. 33 These results were confirmed for subchronic daily oral exposures to higher reported relevant dietary concentrations (300 μg/kg feed), with the addition of likely clinically significant increases in WBC, heterophil, and monocyte counts. 8 Meanwhile, significant decreases were noted in body weight, weight gain, the phagocytic activity of macrophages, lymphocyte count, sodium and potassium concentrations, as well as serum TAC and glutathione peroxidase (GPx) activities. 8 Similar results in cell-mediated immune responses were reported for subchronic daily oral exposures at a slightly higher OTA concentration (750 μg/kg of feed). 84 Subchronic daily oral exposure of young rabbits at a significantly higher than the reported relevant dietary OTA concentrations (10,000 μg/kg of feed) resulted in decreased granulocyte and increased lymphocyte counts, hyperglycemia, decreased platelet counts, and prolonged clotting times149 –151; however, this OTA dosage is generally higher than relevant dietary concentrations reported. 29
OTA was also found to exert developmental toxicity in rabbits. Daily oral exposure of pregnant does (days 6–18 of gestation) to 100 μg OTA/kg BW was associated with an increased incidence of fetal developmental abnormalities, such as agenesis of caudal vertebrae, incomplete ossification of cranial bones, hydrocephalus, and tail agenesis. 154 Finally, there is some evidence that OTA might exert reproductive toxicity in rabbits; however, this is not currently established. Subchronic daily oral exposure to nephrotoxic doses of OTA caused disruption in the spermatogonial cell pattern, severe atrophy of the epididymal duct, and increased testicular interstitial connective tissue in bucks. 114
Patulin
Patulin is produced by fungi of the genera Penicillium, Aspergillus, and Paecilomyces (syn. Byssochlamys) and is typically detected in apples and their derived products. 128 Patulin has been reported to exert its toxic effects on the reproductive and immune systems, as well as neurotoxicity, mutagenicity, and carcinogenicity. 128 Relatively limited research is available on the effect of patulin in rabbits, and existing studies have primarily reported its impact on the male reproductive system, immune system, and bone structure. Notably, the aforementioned studies have been conducted either in vitro or in vivo using only parenteral administration routes, posing a challenge when extrapolating to a natural mycotoxicosis scenario. The subacute semi-weekly exposure of adult bucks to a relatively low dosage of patulin (10 μg/kg BW intramuscularly) caused a significant decrease in total and progressive sperm motility. 128 The semi-weekly intramuscular exposure of adult rabbits to the same low dosage of patulin for 5 wk caused a decrease in peripheral lymphocyte count without any effects on other CBC analytes 162 and oxidative stress. 163 A decrease in mean corpuscular hemoglobin concentration was noted in a similar study, 161 which is however of questionable clinical significance. In a combined in vitro and in vivo experiment, the subacute daily intraperitoneal exposure of rabbits to 2,500 μg patulin/kg BW was found to induce reversible immunotoxicity. 35 This was demonstrated by a decrease in immunoglobulin concentration and suppression of peripheral lymphocyte function. 35 In vitro exposure of rabbit peritoneal leukocytes to patulin caused significant suppression of the oxidative burst. 35 These results correlate with the previously reported decrease in lymphocyte counts. 162 In a 1976 in vitro study, patulin also appeared to affect cell membrane integrity, as it was found to be a potent inhibitor of the Na+-dependent glycine transport system in rabbit reticulocytes. 146 Finally, the subacute intramuscular bi-weekly administration of patulin in a relatively low dosage (10 μg/kg BW) was found to affect bone microstructure in rabbits in a sex-dependent manner, possibly reflecting a subsequent sex hormone imbalance. In particular, enhanced density of the cortical bone was noted in both sexes, with treated males manifesting evidence of bone remodeling and in females indications of altered bone turnover. 30 These results were further supported by a similarly designed study in subacutely exposed male rabbits. 79
Tremorgenic toxins
Tremorgenic mycotoxins are mainly produced by the fungi of genera Penicillium, Aspergillus, Claviceps, and Neotyphodium and can cause intermittent or sustained tremors in vertebrates. 39 These mycotoxins include penitrems, roquefortine A, verruculogen, aflatrem, and paxilline, among others. 39 In companion animal medicine, intoxication with tremorgenic mycotoxins is most common in dogs, although other species, including rabbits, can rarely be affected. A single experimental study has addressed the effect of fumitremorgin A in rabbits, which is a secondary metabolite of A. fumigatus that commonly contaminates silage and hay13,102; a single intravenous injection of 100,000–200,000 μg fumitremorgin A/kg BW caused spontaneous discharges of spinal ventral roots and common peroneal nerves in exposed animals. 102 However, given the high mycotoxin dosages and the intravenous administration route employed, it is difficult to extrapolate these findings to a natural mycotoxicosis. No reports have been published addressing potential clinicopathologic or histopathologic effects of tremorgenic mycotoxins in rabbits, to my knowledge.
Trichothecenes
Trichothecenes are produced by fungi of several genera such as Fusarium, Stachybotrys, and Trichothecium, with the genus Fusarium being the most common. 61 Trichothecenes commonly contaminate grains, such as barley, oats, maize, and rye. 61 Four main types of trichothecenes are recognized: A (e.g., T-2), B (e.g., DON, nivalenol), C (crotocin), and D (satratoxin H, roridin A) depending on their chemical structure. 61 Their main mechanisms of action include polyribosomal disaggregation, inhibition of protein, RNA, and DNA synthesis, as well as oxidative damage to cellular membranes. 22 After consumption, trichothecenes are mainly metabolized in the liver and are excreted mostly via urine. 61 In monogastric animals, with the exception of rabbits and horses, vomiting is reported at high trichothecene concentrations; at lower concentrations, feed refusal is noted. These toxins are widely known to exert immunotoxicity in several species; reproductive toxicity, genotoxicity, alteration of the intestinal barrier, and carcinogenesis have also been reported.22,109 Rabbits were reported to have variable sensitivity to the effects of trichothecenes, depending on their type. For instance, rabbits are considered sensitive to T-2 toxin (type A), with the oral no observed adverse effect level (NOAEL; i.e. the highest dose or exposure level of a substance at which no detectable adverse effects are observed in an exposed population) for adult bucks being relatively low (<20 μg/kg BW), 81 whereas rabbits were found to be less susceptible to DON (type B) toxicity than were other species. 71
The immunosuppressive properties of T-2 were suggested by a 1988 study in rabbits, in which exposure to the toxin appeared to impair the phagocytic activity of alveolar macrophages against A. fumigatus. 103 The negative effect of T-2 on cellular immunity has long been established, as this mycotoxin induces apoptosis in rapidly proliferating cells, such as lymphocytes. 63 Acute exposures to T-2 concentrations of 500 (oral) to 15,000 (intravenous) μg/kg BW resulted in a transient, likely clinically significant, mild-to-moderate decrease in WBC, 53 as well as necrosis within the mucosa-associated lymphoid tissue in the gastrointestinal system and the mononuclear phagocyte system, lymphocyte depletion in lymphoid organs, and depletion of myelopoietic cells in the bone marrow. 55 The high mycotoxin dosages administered and the intravenous administration route employed render it difficult to extrapolate those results to a natural mycotoxin exposure scenario. The subacute daily oral exposure to 400 μg T-2/kg BW was reported to similarly cause moderate, likely clinically significant, decreases in WBC, heterophil, lymphocyte, and monocyte counts. 164 The immunotoxic effect of the mycotoxin was further demonstrated in the increased levels of MDA and decreased TAC, SOD, and GPx in the spleen and thymus, along with areas of hemorrhage, alterations in splenic structure and lymphoid follicles, inflammatory cell infiltration, and decrease in thymic cells on histopathology. 164 Similar results were reported for subacute daily oral exposures at slightly higher concentrations (750 μg/kg BW) 103 ; depletion of splenic lymphocytes was also observed at higher reported relevant dietary T-2 concentrations (2,000 μg/kg of feed) during subacute daily oral exposures. 62 There is equivocal evidence with regard to the effect of T-2 on humoral immunity, as subacute exposures at a relatively low concentration caused an increase in serum cytokines (interleukin-1β, interleukin-6, and tumor necrosis factor–α) expression 164 ; subacute daily oral exposure at a higher reported relevant dietary concentration (2,000 μg/kg of feed) resulted in a decrease in cytokine expression in the intestinal mucosa. 63
The effects of T-2 on other clinicopathologic analytes of rabbits are rarely reported. The acute intravenous exposure of rabbits to T-2 caused a significant decrease in Hct and coagulation factors II, VIII, IX, X, and XI52,53; subacute daily oral exposures caused a mild decrease in serum total protein and albumin concentrations. 63 Subacute daily oral exposure of weaned rabbits to higher reported relevant dietary T-2 concentrations (2,000 μg/kg of feed) caused a decrease in RBC Na+/K+ ATPase activity, as well as a mild, likely clinically insignificant, increase in mean corpuscular volume (MCV) and mean corpuscular hemoglobin (MCH). 136
The reproductive toxicity of T-2 is less well documented in rabbits compared to other species. The subacute daily oral exposure to 780–990 μg T-2/kg BW caused a decrease in progressive forward motility of spermatozoa and testosterone concentration; it increased the percentage of abnormal spermatozoa. 80 The detrimental effect of T-2 on rabbit spermatozoa was confirmed in an in vitro study. 145 In turn, mycotoxin HT-2 (T-2 metabolite) was found to have an in vitro dose-dependent negative effect on ovarian steroidogenesis in rabbits, suggesting a negative effect on the female reproductive system. 77
Few studies have addressed the effect of DON on rabbits, which might reflect decreased research interest due to the lesser sensitivity observed in this species. As a general observation, higher dosages of DON have been tested, which reflects the generally high natural contamination levels compared to other mycotoxins. 29 In particular, the subacute daily oral exposure of weaning rabbits to 500 or 1,500 μg DON/kg BW had a mild hepatotoxic action that was indicated by mild increases in serum ALP, ALT, AST, and LDH activities; histopathologic alterations occurred in intestines (significant reduction in villus heights and enhancement of crypt depths, increased numbers of goblet cells, mucosal injury), and intestinal inflammatory cytokine expressions were significantly enhanced. 158 The toxic effect of DON in the intestine of rabbits was confirmed in a study in which the subacute daily oral exposure to DON affected the ultrasonographic appearance of intestinal segments, compromised the integrity of the intestinal barrier, and altered the composition and diversity of intestinal flora. 152 Interestingly, the subacute daily oral exposure at a higher reported dietary relevant DON concentration (10,000 μg/kg of feed) did not exert any hepatotoxic, nephrotoxic, or genotoxic effects in rabbits, but it caused immunotoxicity as indicated by lymphoid tissue depletion and apoptosis. 73 DON has not been reported to exert developmental toxicity in rabbits; there are conflicting reports in terms of its reproductive toxicity.75,95,145 Finally, very high doses of DON were found to cause hemolysis in vitro, but that effect was not proven in vivo at naturally occurring contamination levels. 122
Zearalenone
ZEN is produced by fungi of genus Fusarium (primarily, F. graminearum and F. culmorum) that commonly contaminate cereal crops, such as wheat, barley, corn, maize, and sorghum. 125 ZEN has worldwide prevalence and mainly acts as an endocrine disruptor; it has also been implicated in the induction of endoplasmic reticulum stress and cellular apoptosis.4,10,166 Although it is primarily recognized as a reproductive toxicant in several species, ZEN’s toxic action can also include hepatotoxicity, immunotoxicity, and nephrotoxicity. 111 In rabbits, ZEN is primarily metabolized in the liver to 2 major metabolites: alpha-zearalenol (alpha-ZOL) and beta-zearalenol (beta-ZOL). These both have estrogenic action and are excreted in the urine. 143 Most rabbit urine ZEN metabolites are in conjugated form, with 29% involving alpha-ZOL and 25% beta-ZOL. 90
ZEN exerts a dose- and exposure-dependent reproductive toxicity in other species. 141 However, only a few studies have addressed its effect on the rabbit reproductive system, particularly in females, in contrast to other species such as swine. Specifically, the subacute daily oral exposure of rabbit bucks at a relatively low dosage of ZEN (50 μg/kg BW) caused mild changes in sperm quality parameters, with the most notable being increased percentages of spermatozoa with head and midpiece abnormalities and DNA fragmentation. However, the clinical significance of these findings was unclear; no histopathologic abnormalities were noted in testes and epididymides at the end of the experiment, which might indicate a reversible mycotoxin action. 142 Similarly, subchronic bi-daily oral exposure to even lower concentrations of ZEN (0.5, 5, or 10 μg/kg BW) caused a decrease in libido, ejaculate volume, sperm concentration and motility, and semen fructose concentration. ZEN increased the percentages of abnormal and dead spermatozoa, implying an adverse effect on the reproductive system; however, the clinical significance of these findings was not defined. 160 The adverse effects of ZEN on rabbit sperm quality were also confirmed in an in vitro study. 145 Regarding rabbit does, the subacute daily oral exposure to ZEN at dosages of 100, 1,000, or 2,000 μg/kg BW was found to exert potent estrogenic action. 112 Subacute daily oral exposure at an even higher dosage (11,500 μg/kg BW), in turn, was reported to affect fertility factors of the early pre-implantation period, such as uterine tubal fluid volume and amino acid composition. 107 However, most of the aforementioned mycotoxin dosages are very high and it is unclear whether these could reflect relevant dietary concentrations, especially in the context of the latest world mycotoxin prevalence surveys. 29
Only a few studies have investigated the effect of ZEN in rabbit clinicopathologic analytes, particularly focusing on liver enzymes due to its well-established hepatotoxicity. ZEN was found to exert dose-dependent hepatotoxicity in subacutely orally exposed rabbits, as demonstrated by increased serum liver enzyme activities. Exposure to a low ZEN concentration (10 μg/kg BW) caused a mild increase in serum ALP activity; exposure to a significantly higher dosage (100 μg/kg BW) affected more serum enzyme activities such as ALP, ALT, AST, GGT, and LDH, possibly reflecting a more extensive yet mild effect. 21 Similarly, the subchronic daily oral exposure of rabbit bucks at a dosage in the intermediate range (50 μg/kg BW) caused mild hepatocellular damage and dysfunction, as indicated by the mildly increased serum AST activity and total bilirubin concentration, which was supported by the mild vacuolar hepatopathy noted on histopathology. 143 The subchronic bi-daily oral exposure to even lower ZEN concentrations caused a decrease in body weight and feed intake, as well as in serum AST, ALT, and acid phosphatase activities, reflecting an even milder effect. 160 In a rare report of the effect of ZEN in rabbit CBC analytes, a significant increase in RBC mass, 2 as well as WBC, monocyte, and eosinophil counts, and mean platelet volume were noted during a subchronic daily oral exposure 143 ; the reported changes, however, were of questionable clinical significance. Lastly, ZEN was suspected to cause renal damage as seen in a Fanconi-like syndrome in exposed bucks. 143
ZEN may have an impact on rabbit cecal microflora, which could potentially affect intestinal function. Specifically, subacute daily oral exposure to generally high ZEN dosages of 400, 800, or 1,600 μg/kg BW significantly affected the composition of intestinal microflora in weaned rabbits, as reflected by the increased numbers of Proteobacteria and Cyanobacteria and decreased numbers of Lactobacillus spp. 90
Combined mycotoxin exposure
There are few reports of research conducted on combined mycotoxin exposure in rabbits. Combined mycotoxin exposures can cause severe adverse effects in exposed animals, especially through additive or synergistic actions, particularly if they share the same mode of toxicity. 153 Frequently investigated pairs of mycotoxins include natural co-occurrences in crops, such as ZEN and DON, FB1 and AFB1, or OTA and citrinin. The subchronic daily oral exposure of adult rabbit bucks to 1,500 μg FB1/kg BW and 30 μg AFB1/kg BW induced anorexia, lethargy, and marked, likely clinically significant, increases in urea and creatinine concentrations, as well as in liver enzyme activities with prominent increases noted for AST and GGT, which demonstrate the synergistic hepatotoxic and nephrotoxic action of these mycotoxins. 105 Accompanying histopathologic findings included congestion of liver, kidneys, lungs, and parts of the digestive tract, as well as focal hepatic necrosis, duct hyperplasia, sinusoidal leukocytosis, vacuolar degeneration of the renal tubules, and lymphocyte depletion in the spleen. 105 Synergistic, antagonistic, and additive effects on various reproductive endpoints were reported in rabbit bucks subchronically exposed to ZEN, DON, and FB1 orally. 48 A combined 4-wk oral exposure of weaned rabbits to higher reported relevant dietary concentrations of T-2 and FB1 (2,000 μg/kg and 10,000 μg/kg of feed, respectively) caused a significant decrease in body weight and a mild, likely clinically insignificant, increase in MCV and MCH; it revealed an antagonistic action on erythrocyte Na+/K+ ATPase activity, which was similar to the controls. 136 In a combined subacute daily oral exposure of weaned rabbits to 10,000 μg FB1/kg and 2,000 μg T-2/kg of feed, FB1 and T-2 had a synergistic effect on decreasing blood antioxidant parameters (glutathione, GPx) and increasing an oxidative marker (MDA) at 2 wk of exposure; both mycotoxins caused DNA damage in lymphocytes, but no synergy or additivity was observed. 62 Lastly, the subchronic daily oral combined exposure to relatively low concentrations of OTA and citrinin resulted in significantly increased cellular apoptosis and lipid peroxidation in kidney tissue with observed additive effects. 85
Clinical and diagnostic considerations
A diagnosis of mycotoxicosis is often reached through exclusion, given that this is typically a “silent syndrome,” lacking any distinctive features. In that sense, acute mycotoxicosis might be easier to identify, as chronic manifestations might mimic other chronic diseases; however, the former are typically rare occurrences in clinical practice. In any case, due to the immunomodulatory effects of most mycotoxins, suspicion of mycotoxicosis is always relevant in rabbits with recurrent infections or peripheral cytopenias, particularly leukopenias. The clinical presentation and impact of mycotoxicosis in each animal varies depending on the consumed dose, duration of exposure, age, sex, overall health status (e.g. concurrent infections), and exposure to other environmental toxicants (e.g., lead, pesticides). 99 Mycotoxins can have immunomodulatory effects that can result in susceptibility or exacerbation of underlying infections. 130 Furthermore, the co-occurrence of some mycotoxins with other environmental toxins in feedstuff (e.g., heavy metals, pesticides) can result in alterations in the exhibited toxicity through the mechanisms of synergy, additivity, or antagonism. 59
The diagnostic approach to mycotoxicoses varies between a companion animal clinic and a farm setting. The farm setting often involves larger population sizes and offers more opportunities for comprehensive testing such as postmortem and histologic evaluations, as well as determination of mycotoxin residues in target organs, although the latter are not ideal samples (limited service by the laboratories, difficult to differentiate intoxication from exposure). However, it is crucial to bear in mind that false-negative results are quite likely because mycotoxins exist in feed at low concentrations and are often eliminated rapidly, leading to concentrations in tissues that are frequently insufficient for residue detection at the time of death or sample collection. 12
When a mycotoxicosis is suspected in practice, a CBC, serum biochemistry profile, and urinalysis should be ordered as a bare minimum to assess for common alterations, such as increased liver enzymes or peripheral cytopenias. Ultimately, the determination of mycotoxin concentrations in rabbit feed or preferably, in target organs, is needed to confirm exposure. Several samples should be obtained from different areas of feed and after thorough mixing to ensure the homogeneity of mycotoxin concentration. However, by the time mycotoxicosis is suspected, most of the heavily affected portions of the feed may have already been consumed, leading to equivocal analytical results (i.e., low or false-negatives). Hence, a good standard practice is the collection and storage of representative feed samples before offering them for consumption. Dried or frozen feed samples in paper bags could be submitted to a laboratory and a broad screen for mycotoxins performed to investigate for combined effects. Masked mycotoxins pose inherent challenges in their identification by routine laboratory methods and might not be detectable; however, this is a research area under development.
Some research efforts have identified that serum and urine sphinganine:sphingosine ratio or OTA–acyl-glucuronide might show potential as markers for fumonisin and OTA exposure, respectively.25,120 Although exposure biomarkers hold promise as quick and cost-effective tools for mycotoxin exposure, there is no validated biomarker available for use in rabbits, to my knowledge.
A variety of compounds that can limit the effects of mycotoxins in rabbits and subsequently the risks associated with the presence of residues in their products have been tested in a research setting. These include binding compounds that can reduce mycotoxin bioavailability, such as glucomannan, 32 sodium bentonite, 68 coumarin, 68 antioxidants such as Vitamin E, 50 and selenomethionine,93,165as well as biological agents, such as Aspergillus awamori 1 and Bacillus subtilis, which can degrade mycotoxins. 126 However, a detailed discussion of these substances is beyond the scope of this review.
Knowledge gaps and areas for future research
Although a substantial body of research addresses mycotoxins in rabbits, certain aspects pose significant challenges and could benefit from additional research efforts. For instance, a prevalence study of mycotoxins in rabbit feed conducted across multiple countries would be useful as a starting point to determine the extent of the problem and indicate areas for future research. The evaluation of the results of up-to-date epidemiologic studies in conjunction with the constantly growing body of published experimental studies is important for the establishment of acceptable feed limits in rabbits, similar to other species, such as ruminants and swine, but also for the comprehension of natural mycotoxicoses. However, extrapolating from an experimental mycotoxin exposure study to a natural mycotoxicosis can be challenging and requires caution. First, experimental studies are conducted under controlled conditions both in terms of dosages and exposures, whereas natural mycotoxin exposures vary depending on environmental conditions, geography, etc. Moreover, the experimental dosages tested in rabbits are often higher than mycotoxin concentrations found in natural settings, or are occasionally administered through alternative routes (e.g., intraperitoneally). Although this approach has certain advantages (e.g., for studying specific organ effects), the extrapolation to natural mycotoxicoses is often challenging. For instance, parenteral routes of administration typically lead to higher bioavailability of substances given that first-pass hepatic metabolism is avoided, 144 or higher mycotoxin dosages could lead to more profound clinical effects.
Overall, there is an ongoing need for controlled feed studies investigating repeated oral exposures, particularly subchronic and chronic, at relevant dietary mycotoxin concentrations (as determined by recent regional or worldwide prevalence studies) that better replicate natural conditions. Furthermore, specific mycotoxins such as fusaric acid, fusarenon-X, or nivalenol, which are common co-occurrences with other better-studied mycotoxins, such as FB1 and DON, are yet to be studied in rabbits. Data are limited for other mycotoxins, such as ergot alkaloids. Understanding the metabolic pathways and toxicokinetics of each mycotoxin in rabbits is also crucial, as it can enhance knowledge in mycotoxin biomarker research, which has garnered significant interest in recent years. Finally, the publication of case reports or series of natural mycotoxicoses would be particularly useful for the establishment of a clinical description of mycotoxicoses in rabbits.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
