Abstract
Interest in causes of mortality of free-ranging, native North American lagomorphs has grown with the emergence of rabbit hemorrhagic disease virus 2 (RHDV2). Over the years 2013–2022, the Southeastern Cooperative Wildlife Disease Study received 119 Sylvilagus spp. case submissions from the central and eastern United States, comprising 147 rabbits. Most (86%) of these submissions occurred after detecting RHDV2 in the United States in 2020. Laboratory data from these rabbits were retrospectively evaluated for major causes, contributors to mortality, and pathogen detections. Gross and histologic examination was performed for 112 rabbits. Common primary causes of death included trauma (n = 49), bacterial disease (n = 31), emaciation (n = 6), and parasitism (n = 6). Among the 32 rabbits with bacterial disease, 12 were diagnosed with tularemia and 7 with pasteurellosis. Rabbits with pasteurellosis had disseminated abscessation, septicemia, and/or polyserositis. Less commonly, cutaneous fibroma (n = 2), notoedric mange (n = 2), encephalitozoonosis (n = 2), neoplasia (round-cell sarcoma; n = 1), and congenital abnormalities (n = 1) were diagnosed. RHDV2 was not detected in 123 rabbits tested. Although RHDV2 has not been detected in wild lagomorphs in the eastern United States, detections in domestic rabbits from the region emphasize the need for continued surveillance. Furthermore, continued surveillance for Francisella tularensis informs public health risk. Overall, increased knowledge of Sylvilagus spp. health furthers our understanding of diseases affecting these important prey and game species.
Keywords
Lagomorphs globally represent a diverse taxonomic group, including wild hares and rabbits in the family Leporidae. Specifically, New World rabbits (Sylvilagus spp.; cottontail rabbits; hereafter, cottontails) are comprised of 18 species of cottontail rabbits. 45 The eastern cottontail (Sylvilagus floridanus) is one of the most ubiquitous species in the United States, which has a diversified landscape and provides habitat for many other cottontail species, including the swamp and marsh rabbit (S. aquaticus and S. palustris, respectively) in the southeast, the New England cottontail (S. transitionalis) in the northeast, and the Appalachian rabbit (S. obscurus) in the Appalachian Mountains of the eastern United States. 14 Given their widespread distribution and high population densities in some areas, cottontails serve as important prey species for a variety of free-ranging carnivores and omnivores. 45 Opportunities for cottontail–human interactions are many, as made evident by their importance as game (fur and meat) species, their common presentation to wildlife rehabilitation centers, and efforts to control them as a pest species in some agricultural, commercial, and residential settings.23,41 Further, these and other settings may provide opportunities for wild rabbits to interface with domestic animals such as hunting dogs, pet rabbits housed outdoors, and livestock, furthering the importance of better understanding their natural diseases and causes of mortality.
Rabbit hemorrhagic disease virus 2 (RHDV2; Caliciviridae, Lagovirus europaeus) has emerged as an important disease of domestic and wild rabbits in North America. 34 RHDV2 was initially classified as a foreign animal pathogen in the United States and rapidly disseminated on a global scale following initial identification in France in 2010. 39 Its spread and broad host range were likely facilitated by human-mediated transport of rabbits and virus recombination events, respectively.30,39 The first North American detection of RHDV2 occurred in domestic rabbits from Canada in 2016, followed by detections in feral Oryctolagus cuniculus rabbits in 2018. 4 The first detection reported in the United States was in Ohio from domestic rabbits in 2018; the first detection in wild lagomorphs was in New Mexico in 2020.7,50 Subsequent spread prompted wildlife agencies to elevate surveillance efforts for RHDV2 in free-ranging rabbits, including increased submissions to veterinary and wildlife diagnostic services.
Wild rabbits infected with RHDV2 commonly are found dead without prior observation of clinical signs, but lethargy and epistaxis have been reported.7,27,54 Gross lesions also may include friable livers with an enhanced parenchymal reticular pattern or pinpoint capsular red foci, and pulmonary edema with variable pink to dark-red lung mottling. 27 Overlap in some of these lesions (e.g., epistaxis) with those seen with trauma or infection emphasizes the importance of ancillary molecular testing paired with histopathology. Histologic lesions associated with RHDV2 may include massive hepatocellular dissociation and necrosis or apoptosis, splenic congestion or necrosis, renal tubular hemorrhage or injury, and pulmonary congestion, hemorrhage, or edema.27,54 Research into RHDV2 transmission and environmental stability is needed to better understand population health risks to rabbits, but RHDV2 can persist in animal tissues for up to 3 mo in field settings, and scavenging birds and flies have been implicated as mechanical vectors.21,31,38
Prior to RHDV2 surveillance in North America, wild rabbit carcasses were most likely submitted for testing out of concern for tularemia, which is caused by Francisella tularensis. This bacterium is widespread throughout the Northern Hemisphere and can infect ≥65 wildlife species, as well as humans.1,47 F. tularensis is highly contagious with a low infectious dose, transmitted by various means (vector- and airborne, ingestion, via skin breaches) and is considered a potential bioweapon; hence, it is designated a Tier 1 select agent by the U.S. Centers for Disease Control and Prevention.13,51 Numbers of human tularemia cases in the United States increased in 2011–2019, with the central region considered endemic and accounting for >60% of cases.9,33 Common sources of human exposure have shifted from hunting and processing of infected lagomorphs to tick or other insect vector bites. 33 Subspecies F. t. tularensis (type A) has a terrestrial transmission cycle with rabbit, hares, and tick reservoirs; muskrats (Ondatra zibethicus) and beavers (Castor canadensis) serve as primary hosts in the aquatic transmission cycle of F. t. holarctica (type B).17,18 Experimentally induced infection with type A strains caused hepatic and splenic microabscesses in wild-caught cottontails; type B strains induced clinical signs from lethargy to mild fever and rarely were fatal. 11 The classic lesions for rabbit cases of septicemic tularemia include white foci in the liver, spleen, lungs, and lymph nodes.5,20,28
Although rabbit hemorrhagic disease, tularemia, and other zoonoses have historically motivated rabbit mortality investigations, complete diagnostic evaluations provide the opportunity to explore other causes and contributors to morbidity in wild lagomorphs that may not have otherwise been submitted. Much of the current research on rabbit disease focuses on laboratory or domestic rabbits of economic importance (e.g., O. cuniculus), with work on cottontails often more focused on zoonotic pathogens, a single disease process, or reviews of commonly detected parasites.3,6,16,28,48,55 Our objectives were to retrospectively evaluate a robust diagnostic dataset to determine common causes of morbidity and mortality and describe common and uncommon diseases in free-ranging cottontails in the central and eastern United States, over a 10-y period (Jan 1, 2013–Dec 31, 2022). Additionally, we evaluated cottontails for characteristic gross and histologic lesion patterns of tularemia along with any deviations from these patterns. To our knowledge, and after searching Google and PubMed for papers using keywords such as: “cottontail”, “cottontail rabbits”, “Sylvilagus”, and “eastern cottontail”, a comparable retrospective study evaluating the pathology and causes of morbidity and mortality in cottontails in this region of United States has not been published previously.
Materials and methods
Diagnostic submissions and case data
The Southeastern Cooperative Wildlife Disease Study (SCWDS) Research and Diagnostic Service, located at the University of Georgia (Athens, GA, USA), assists 17 state wildlife agencies throughout the central and eastern United States with wildlife mortality investigations. We evaluated retrospectively the SCWDS Research and Diagnostic Service database for all lagomorph cases submitted from January 1, 2013–December 31, 2022. During this time, SCWDS received 122 lagomorph submissions, comprising 164 individual animals; however, we excluded from analysis feral domestic rabbits (O. cuniculus domesticus) and wild lagomorphs other than cottontail species. Similarly, we excluded submissions from the western United States (i.e., California, Arizona) because they were from outside our geographic area of interest (i.e., central and eastern United States), and generally comprised rare submissions that were limited to liver samples for RHDV2 testing. Species identification generally was provided by the submitting wildlife biologists, rehabilitators, and/or veterinarians and confirmed upon evaluation, when sex (gross gonad identification) and age (juvenile or adult, based on morphologic characteristics 41 ) were determined.
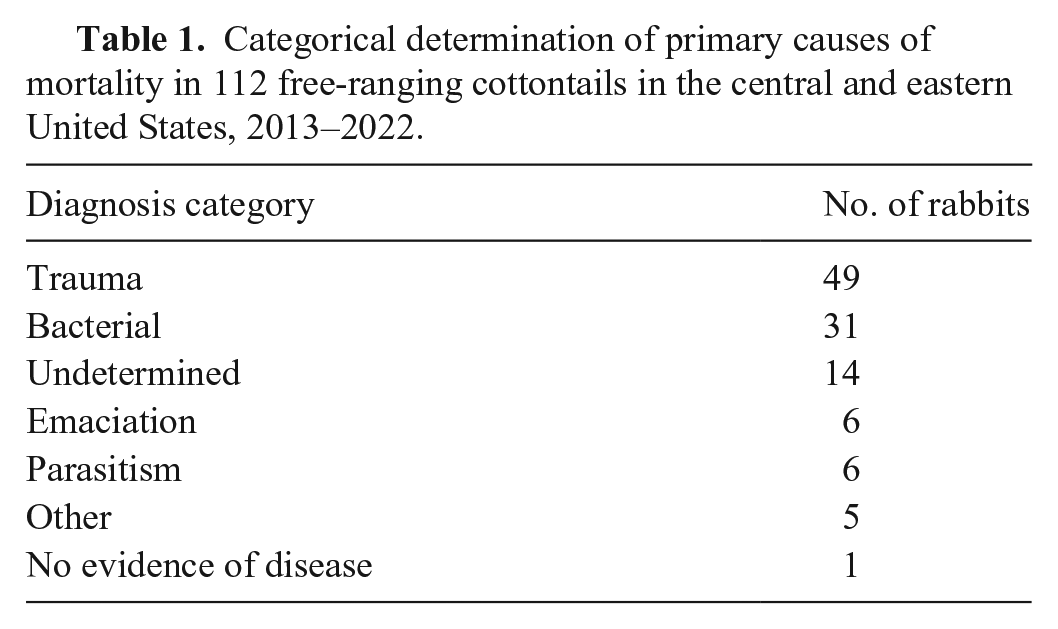
Case data included age, sex, date of death and/or date of carcass collection (also categorized by year and season), clinical signs and method of dispatch (e.g., killed, hunter-harvested, etc.) or field findings related to carcass discovery (when relevant), geographic location (county, state), nutritional condition, gross and histologic lesions, and ancillary test results. The season was according to meteorologic start dates in the Northern Hemisphere (i.e., winter starts Dec 1, spring starts Mar 1, summer starts Jun 1, and fall starts Sep 1). Nutritional condition was subjectively determined based on skeletal musculature and visceral and subcutaneous adipose stores, and assessed as poor (moderately reduced or absent subcutaneous and visceral fat stores, and variable muscle atrophy), fair (mildly reduced fat stores and appropriate musculature), or good (appropriate fat stores and musculature). Major causes and contributors to morbidity and/or mortality were organized into general categories (i.e., trauma, bacterial disease, poor nutritional condition; Table 1).
Categorical determination of primary causes of mortality in 112 free-ranging cottontails in the central and eastern United States, 2013–2022.
Laboratory testing
Routine screening of liver and/or spleen samples for F. tularensis with a PCR test 44 was performed at the Athens Veterinary Diagnostic Laboratory (AVDL; Athens, GA, USA), an AAVLD-accredited facility. Aerobic culture on select tissues based on gross and/or histologic lesions also was performed by the AVDL. Samples from all rabbits underwent F. tularensis PCR assay except when insufficient fresh tissue was available and/or a liver sample was submitted solely for RHDV2 testing.
From 2020 to 2022, a liver sample from each rabbit was routinely tested for RHDV2 by PCR at SCWDS. 15 Briefly, liver samples were mechanically homogenized in Dulbecco phosphate-buffered saline with RNase-free zirconium oxide beads (Bullet blender tissue homogenizer; Next Advance). The homogenate was centrifuged at 600 × g for 5 min, and RNA was extracted (MagMax pathogen DNA/RNA kit; Applied Biosystems) on a magnetic particle processor (KingFisher; Thermo Fisher) per the manufacturer’s instructions. Viral RNA extracts underwent reverse-transcription quantitative real-time PCR (RT-qPCR) with primers and probe targeting the vp60 gene of RHDV2 (7500 FAST real-time system, Applied Biosystems; TaqMan fast virus 1-step master mix, Thermo Fisher). 15
Results
Case submissions, including demographic, geographic, and temporal data
We received 119 cottontail case submissions at SCWDS from the central and eastern United States from 2013 to 2022. Most cases (n = 102; 86%) were submitted in 2020–2022 during heightened concern and surveillance efforts for RHDV2 in the region. Submissions originated from 17 states, including Florida (n = 29), North Carolina (n = 16), Georgia (n = 15), Kansas (n = 11), Kentucky (n = 9), Missouri (n = 8), South Carolina (n = 5), Arkansas, Tennessee, Virginia, and West Virginia (n = 4 each), and Louisiana and Nebraska (n = 3 each). One submission each came from Indiana, Maryland, Pennsylvania, and Alabama (Fig. 1).
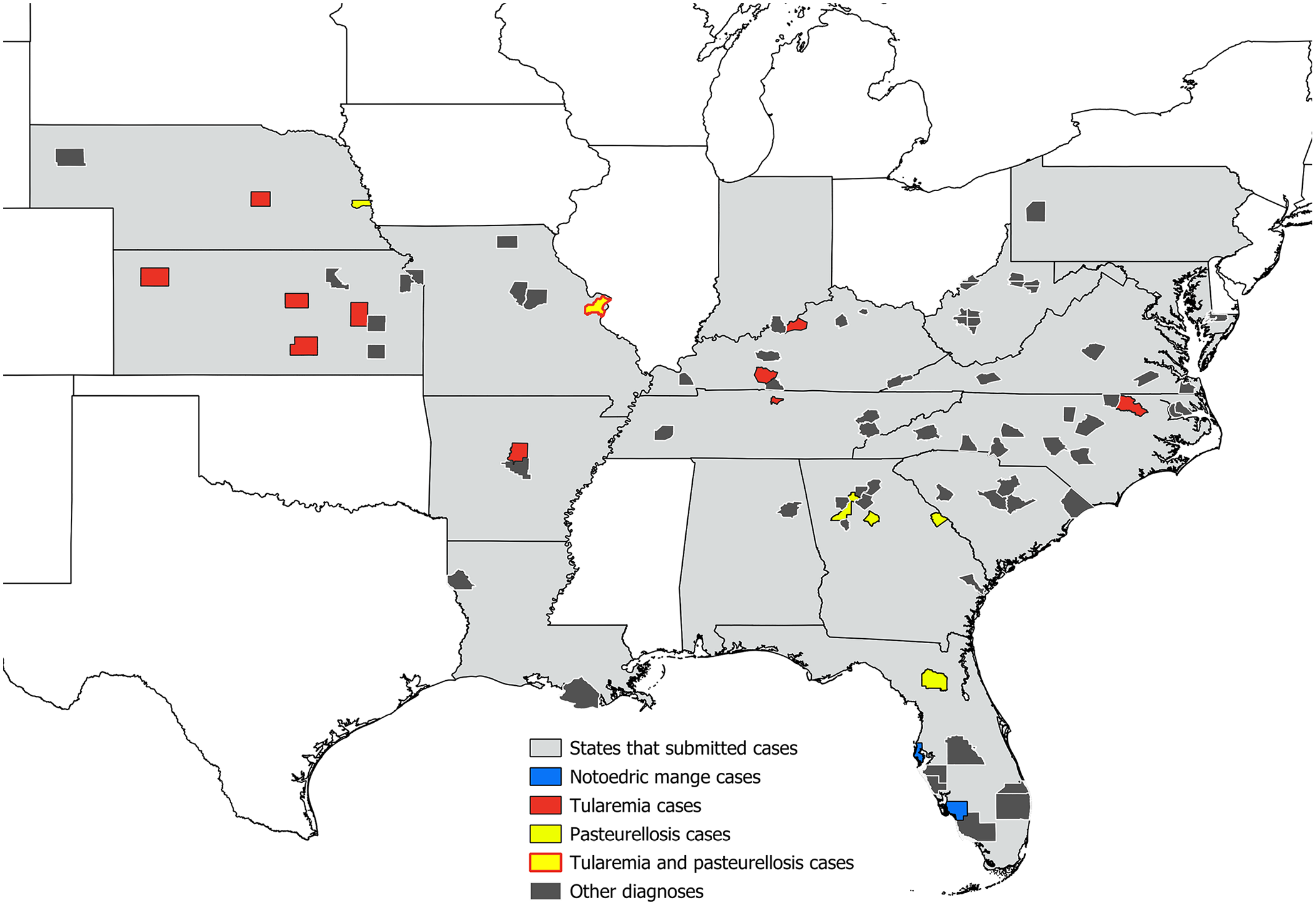
County location data from 112 free-ranging cottontails in the central and eastern United States that underwent diagnostic evaluation at the Southeastern Cooperative Wildlife Disease Study, 2013–2022. Diseases of interest are highlighted; 2 tularemia were from Sedgwick County, Kansas, for a total of 12 cases from 11 counties.
These 119 submissions included 147 individual rabbits, from 1 to 21 individuals per submission (median of 1; mean of 1.24 rabbits per submission). Most (n = 112; 76%) were eastern cottontails, with fewer marsh rabbits (n = 6; 4%) and Sylvilagus spp. (identified to genus only (n = 29; 20%). Most were females (n = 68; 46%), and 33% were males (n = 48). Sex was not recorded for 31 (21%) rabbits, including one submission of 21 rabbits for which age was not recorded. Of the 147 individual rabbits, 89 (61%) were adults and 34 (23%) were juveniles; age estimates were not available for 24 rabbits. Of the 147 individual rabbits, 82 (56%) were found dead; 42 (29%) were dispatched or euthanized; 16 (11%) died under observation; 2 (1%) were hunter-harvested; method of death was unknown in 5 (3%).
For 112 rabbits, cause(s) of morbidity and/or mortality were determined based on the evaluation of either full carcasses or representative tissue sets from field autopsy. Among these diagnostic cases, submissions were greatest from 2020 to 2022 (n = 94). By year, 51 were from 2021; 25 were from 2022; 18 were from 2020; 4 were from 2019; and 3 were from 2014. Five years of the study (2013, 2015–2018) had only 2 rabbit submissions each, and only 1 rabbit was submitted in 2012. Submitted rabbits died and/or were collected in the following seasons: summer (n = 34), fall (n = 29), spring (n = 27), and winter (n = 22; Fig. 2).
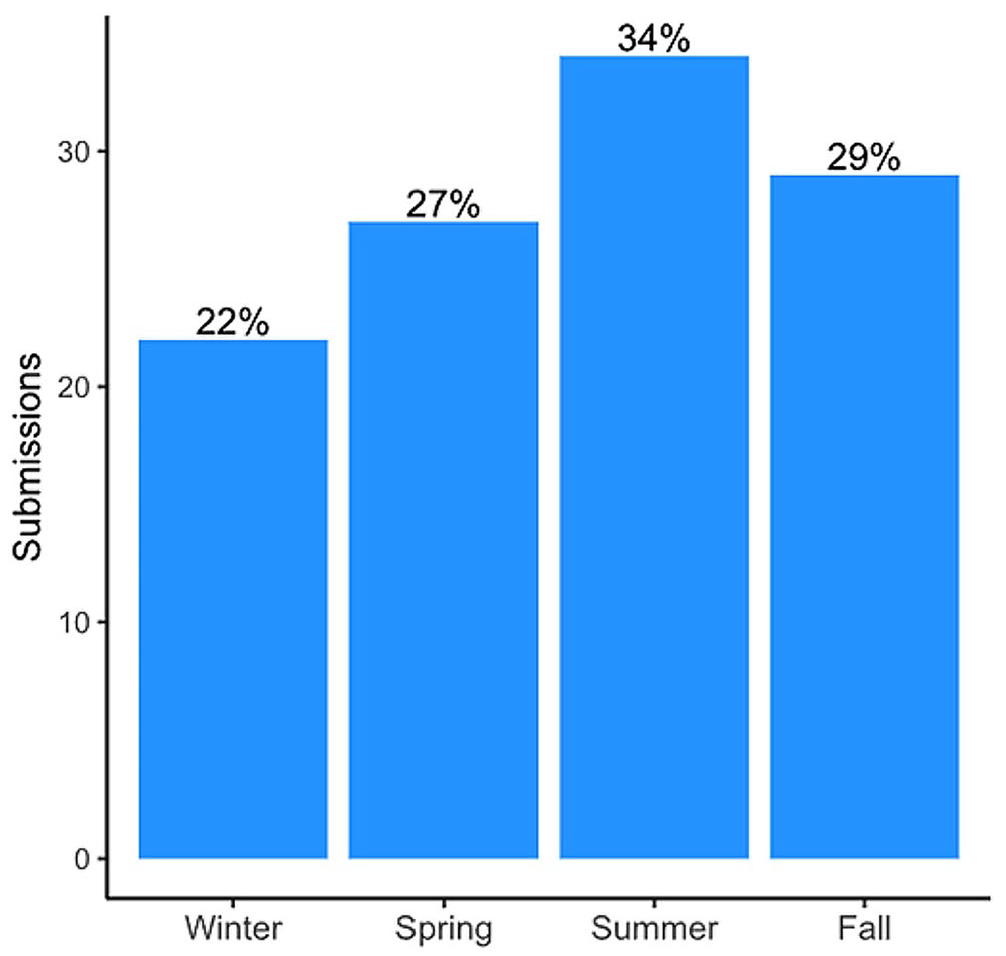
Histogram of seasonality data for cottontails in the central and eastern United States that underwent diagnostic evaluation at the Southeastern Cooperative Wildlife Disease Study, 2013–2022.
RHDV2 surveillance results
From 2020 to 2022, all rabbits without pathology or laboratory-based evidence of tularemia and with available fresh tissue samples (n = 123) were tested for RHDV2 via RT-qPCR. Of the 119 case submissions, 7 consisted of only liver samples for RHDV2 testing via histology and RT-qPCR. These 7 cases included tissue samples from 27 individual rabbits originating from Florida and Louisiana with submission histories of trauma and/or unexplained hemorrhages. RHDV2 nucleic acid was not detected in any of the 123 samples.
General causes and contributors to morbidity and/or mortality
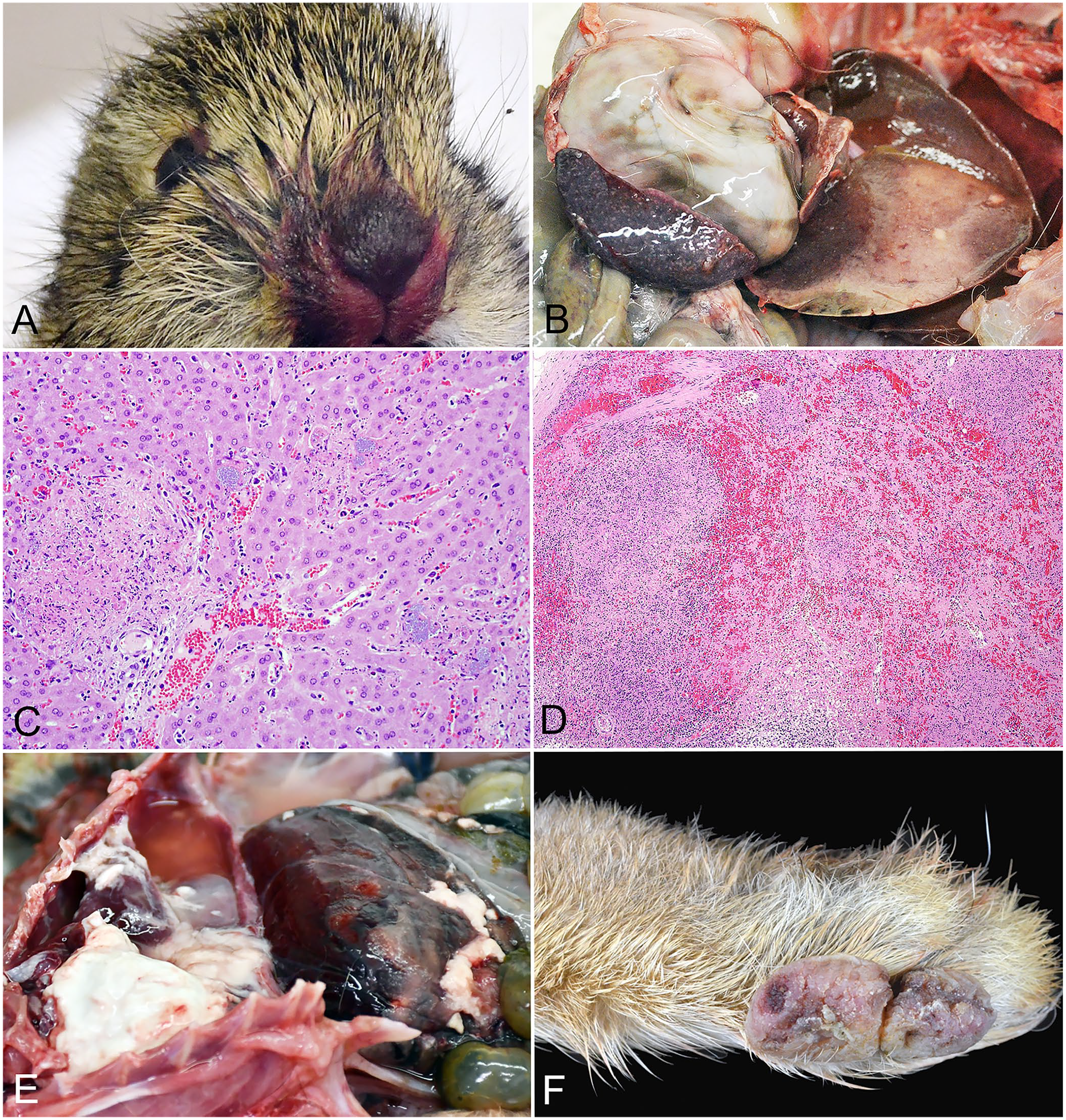
Trauma was the most common cause of death and was deemed the primary cause for 49 rabbits. The most common associated gross finding was extensive subcutaneous, intramuscular, and/or clotted cavitary hemorrhages (n = 48). This was followed by skull and/or vertebral fractures (n = 16), rib, pelvic, and/or long bone fractures (n = 12), and variably deep skin lacerations (n = 8). Fewer than 10% of rabbits each had reported gastrointestinal or diaphragmatic lacerations, epistaxis (Fig. 3A), ocular rupture, traumatic exophthalmos, vertebral subluxation or subluxations, or liver fractures. Rabbits often had at least 2 of these findings; however, the degree of detail and specificity in gross reports was highly variable.
Gross and histologic lesions of select diseases of free-ranging cottontails from the central and eastern United States.
Next most common was bacterial disease, the primary cause of morbidity and/or mortality in 31 rabbits. Other important causes included emaciation and parasitism, and primary causes of mortality (either cause of natural death or euthanasia; Table 1). However, there was frequent overlap between contributing causes of morbidity and mortality, such as lesions suggestive of both trauma and bacterial infection, or systemic bacterial infection in a rabbit in severely decreased nutritional condition. Most rabbits were in fair-to-good nutritional condition (82), and the remainder were in poor-to-emaciated nutritional condition or had no nutritional condition reported in gross findings.
Tularemia—demographic data, case histories, and gross and histopathologic findings
Of the 147 total rabbits, 111 (76%) were screened for F. tularensis via PCR assay. Samples were submitted predominantly based on the availability of fresh liver and/or spleen, as well as the presence of suspicious gross lesions of random, pale foci on capsular and cut surfaces of liver and spleen (Fig. 3B). Twelve rabbits (11% of tested rabbits) had F. tularensis nucleic acid detected. Among rabbits from which F. tularensis was detected, 7 had only liver tested, 3 only spleen, and 2 pooled liver and spleen. In cases without a detection, only liver was tested for 87 rabbits, pooled liver and spleen for 10, and only spleen for 1 rabbit. The remaining rabbit submission was limited to the head (submitted for rabies testing amid a local outbreak in wildlife), and thus, pooled tonsil and submandibular lymph nodes were tested.
All 12 rabbits diagnosed with tularemia were eastern cottontails. These were from 7 states, with 5 from Kansas, 2 from Kentucky, and 1 each from Missouri, Tennessee, Arkansas, North Carolina, and Nebraska (Fig. 1). Nine were adults, and 3 were juveniles. Seven were female, and 3 were males; in 2 cases, sex was listed as unknown. Eleven were found dead, including 2 previously observed as lethargic and unafraid of humans. The remaining rabbit was euthanized after it was found recumbent with anal bleeding. Four rabbits had a history of being found near other dead or sick rabbits.
Of the 12 rabbits diagnosed with tularemia, 11 underwent either abbreviated (because of biosafety and biosecurity concerns) or full autopsy. Nutritional condition was assessed in 10; all of these were in fair-to-good nutritional condition. Ten rabbits had gross lesions, including widely disseminated, random, pinpoint, splenic, and/or hepatic necrosis in 9 (5 in both liver and spleen; 4 in one or the other; Fig. 3B, Table 2). Of the 2 remaining cases, one had no compatible gross or histologic lesions but had advanced autolysis (which likely obscured lesion patterns), and the other had moderate autolysis with no apparent gross lesions but did have compatible histologic lesions. Seven rabbits had mottled red to dark-red lungs that variably oozed blood on cut section. Less commonly associated gross lesions included cecal and vermiform appendix serosal hemorrhages (n = 1) and serosanguinous thoracic effusion (n = 1).
Summary of gross and histologic lesions in cottontail rabbits with tularemia from the central and eastern United States, 2013–2022. Unless otherwise specified, gross and histologic lesions in the spleen and liver included generalized organ enlargement and myriad, pinpoint foci of necrosis. In some cases, limited autopsies were performed for biosafety reasons, and thus, all organs were not fully evaluated in all cases.
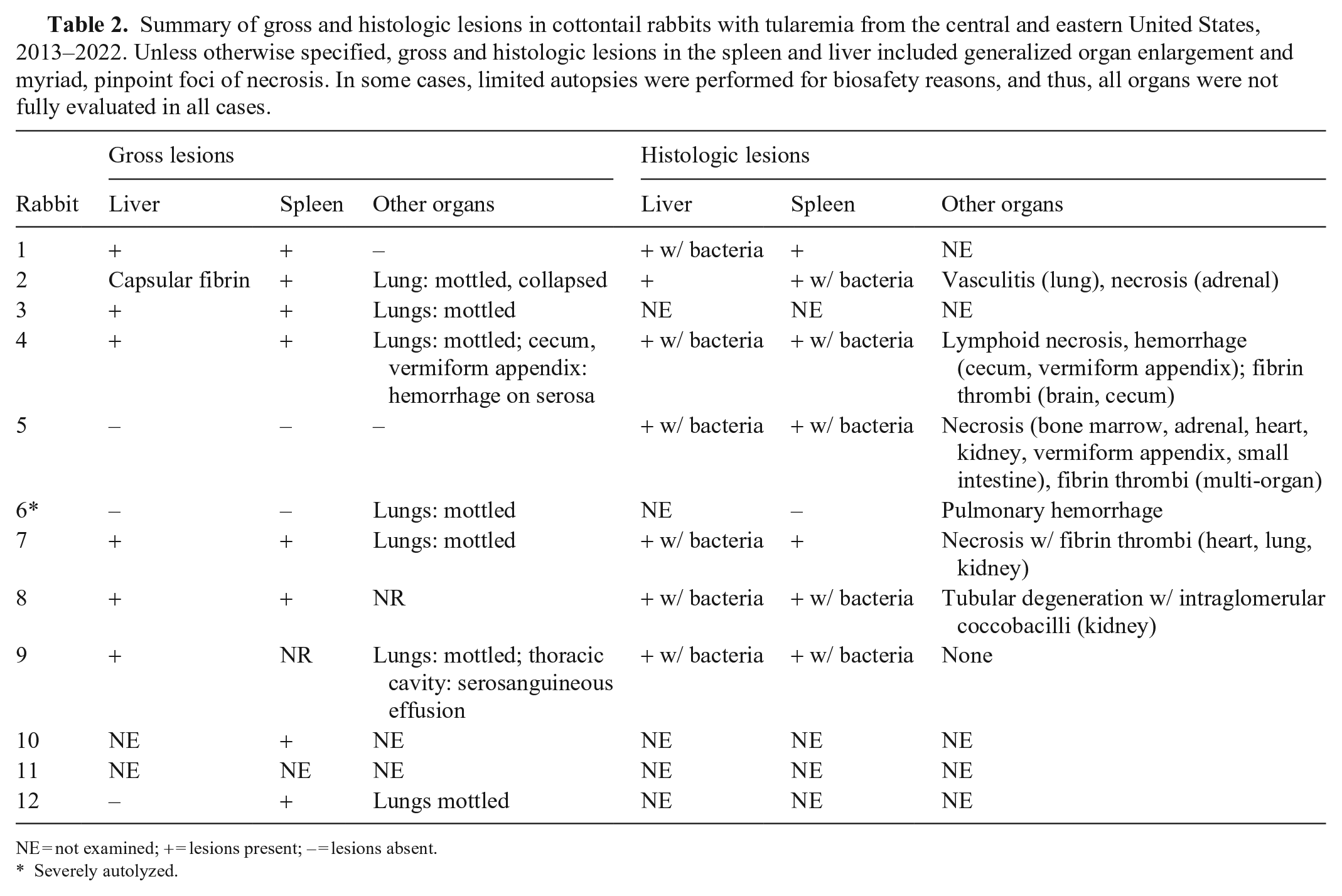
NE = not examined; + = lesions present; – = lesions absent.
Severely autolyzed.
Eight rabbits with tularemia had histologic examination of at least the liver or spleen, and all but one of these had histologic lesions characteristic of tularemia (Fig. 3C, 3D). 27 Gram-negative coccobacilli (morphologically compatible with F. tularensis) were around necrotic foci in the liver and/or spleen in 7 rabbits. Other associated histologic lesions included: multi-organ fibrin thrombi (n = 3), renal tubular degeneration and/or necrosis (n = 3), adrenal necrosis (n = 2), myocardial necrosis (n = 2), cecal and vermiform appendix necrosis and/or hemorrhage (n = 2), bone marrow necrosis (n = 1), small intestinal mucosal necrosis (n = 1), pulmonary vasculitis (n = 1), pulmonary hemorrhage (n = 1), and pulmonary necrosis (n = 1).
Three rabbits with tularemia had concurrent gross and histologic lesions compatible with trauma, considered likely secondary to tularemia but contributing to morbidity and/or mortality.
Other bacterial diseases
Other bacterial diseases were considered the primary cause of death in 19 rabbits (Suppl. Table 1). Pasteurella multocida was the most common isolate, arising in 6 of these rabbits as well as in another rabbit in which the primary cause of death was trauma. Of these 19 rabbits, other important isolates were Yersinia spp. in 4, Escherichia coli in 3, Streptococcus spp. in 2, Mammaliicoccus sciuri in 2, with 1 rabbit each with Enterococcus and Fusobacterium necrophorum. Five rabbits had 2 or 3 bacterial species isolated. Three rabbits did not have a predominant bacterial species isolated via aerobic culture, which instead resulted in the overgrowth of myriad suspected contaminants. Two rabbits did not have aerobic culture performed due to limitations in available fresh tissues.
In descending order, organs or tissues most often with bacterial infection (not including F. tularensis) and associated lesions were the liver (n = 15), lung (n = 10), brain (n = 10), kidney (n = 9), spleen (n = 8), skeletal muscle (n = 8), subcutis (n = 6), and heart (n = 5). Grossly, subcutaneous, intramuscular, and/or multisystemic abscesses were present in 5 rabbits. Four rabbits had wet, heavy, rubbery, red to dark-red lungs that failed to collapse. Multifocal, small, tan-to-red foci scattered throughout the hepatic and splenic parenchyma were seen in 3. Three rabbits had abdominal and/or thoracic serosanguineous effusion unrelated to the dispatch artifact. Extensive fibrinous thoracic and/or abdominal adhesions were present in 2, and 2 rabbits had splenomegaly.
Three cottontails had histologic lesions of inflammation and/or necrosis in the bone marrow and small intestines. Two had histologic lesions in the large intestines, uterus, and adrenal gland. Each had lesions in the testis, stomach, and urinary bladder. Fourteen rabbits had heterophilic inflammation in ≥1 organ, and 9 of these had associated necrosis. Six rabbits had distinct abscesses involving the subcutis, skeletal muscle, and/or viscera. Four of the 6 rabbits with pasteurellosis had significant fibrin exudation associated with heterophilic inflammation (Fig. 3E), and a similar fibrinous component was only seen in 1 non-pasteurellosis case, in which bacterial etiology was not evident via aerobic culture. Abscesses, pneumonia, pleuritis, and peri- and epicarditis often occurred in rabbits with pasteurellosis. Upper respiratory tract infection, conjunctivitis, and otitis media were not observed in any of the cottontails with pasteurellosis. All yersiniosis cases had characteristic, intralesional microcolonies of gram-negative bacilli, and 5 of the 6 pasteurellosis cases had compatible, intralesional, gram-negative coccobacilli.
Clinically relevant parasitism
Noteworthy parasitic diagnoses included 2 rabbits with notoedric mange, 19 2 with hepatic cestodiasis, and 1 rabbit with heavy, concurrent tick infestation, intestinal coccidiosis, and cestodiasis. The rabbits with notoedric mange were previously reported. 19 One rabbit had gram-positive microsporidia in the brain consistent with encephalitozoonosis. Two rabbits submitted together from the same location and date both had brain lesions compatible with encephalitozoonosis, although only one had gram-positive microsporidia detected in the brain histologically. Presumed incidental ecto- and endoparasitic infections were not tallied.
Neoplasia
Other findings of interest included fibromas in 2 rabbits (Fig. 3F) and multi-organ round-cell sarcoma in an additional rabbit in which mortality was attributed to bacterial disease. Based on characteristic histologic findings, including cytoplasmic inclusions, the cause of the fibromas was suspected to be caused by rabbit (Shope) fibroma virus infection. The sarcoma was noted microscopically in sections of the liver, spleen, kidney, adrenal gland, lung, ileum, trachea, meninges, and uterus. The liver from this rabbit was tested by a pan-herpesviral PCR assay, as a herpesvirus-associated lymphoproliferative disease has been reported in cottontails, but no viral nucleic acid was detected. 22
Discussion
Based on the increased submissions of rabbits for diagnostic evaluation throughout much of the United States after RHDV2 incursion, many state wildlife agencies submitted higher numbers of rabbits to SCWDS than ever before, as supported by our data on yearly case numbers in the eastern United States from 2020 to 2022.2,46 This provided a unique opportunity to investigate wild cottontail mortality more thoroughly. Although cottontails are an important game species, historically, they have not composed a major portion of the SCWDS diagnostic caseload. In our study, as with many wildlife diagnostic cases, most rabbits were found dead with incomplete histories (e.g., lack of knowledge of clinical signs or premonitory circumstances of death), supporting the utility of diagnostic evaluation to determine the cause of disease and death. Rabbit submissions (and death dates) spanned all 4 (temperate) seasons with no obvious seasonality trend. Females outnumbered males in submissions, and most were adults.
As for many wildlife species, we found that trauma, emaciation, and bacterial disease were the most common diagnoses in cottontails. Most died in fair-to-good nutritional condition, consistent with most deaths caused by the acute processes of trauma or bacterial sepsis. This may underrepresent the true causes of mortality, as successful predation of rabbits cannot be accounted for in our study. Additionally, young rabbits are small and may die in hidden areas, such as burrows, where they are not found. Our study also included primarily eastern cottontails, ubiquitous throughout the study area, as opposed to other cottontail species in the central and eastern United States, which have more limited distributions. Evidence of some diseases previously reported or experimentally induced in wild cottontails was not detected in our study via gross or histologic examination of tissues. These include reports of natural and experimental infections including cryptosporidiosis, herpesviral infection, cerebrospinal nematodiasis, leptospirosis, and West Nile virus infection, among others.22,24,40,43,49 Some of these may represent rare diseases in free-ranging cottontails or may be uncommon in our study region.
Despite increased surveillance efforts and detections of RHDV2 in domestic rabbits in some parts of the eastern United States at the time of submission, RHDV2 has not yet been detected in free-ranging cottontails in this region. 37 In public outreach materials, clinical and/or gross findings of nasal or other orifice bleeding often are used as possible indicators of RHDV2 infection and may serve as criteria for testing in some cases. 36 In wild rabbits, the most commonly reported presentation is death without signs of trauma. 36 However, it should be recognized that trauma is a common cause of death in wild rabbits, as recorded in our study, and nasal and orifice bleeding are nonspecific findings also seen in trauma cases. Additionally, one of the cases in our study was a rabbit with rectal bleeding in which the cause of death was attributed to tularemia. This underlies the importance of complete postmortem evaluation in cases for which determining the cause of death is deemed necessary as opposed to a targeted RHDV2 surveillance–based approach, and the overlap of this clinical sign with other conditions. Tularemia is generally considered a public health risk for any wild rabbit autopsy in the United States, and thus, high biosafety standards during postmortem evaluations should be practiced.
Bacterial disease was a common diagnosis in wild rabbits in our study. As in domestic rabbits, subcutaneous and intramuscular abscesses were a common manifestation of bacterial disease. 25 One of the most common isolates from these abscesses was P. multocida, although Yersinia enterocolitica, E. coli, F. necrophorum, M. sciuri, S. suis, and Pseudomonas aeruginosa also were isolated from abscesses. Similar to some of those bacteria isolated from wild rabbits in our study, bacteria associated with skin abscesses in domestic rabbits include Streptococcus spp., Staphylococcus aureus, P. multocida, and some anaerobes. 25 Another common manifestation of bacterial infections in wild rabbits in our study was septicemia, with both F. tularensis and P. multocida as major detected pathogens.
Among bacterial diseases of rabbits, tularemia is important due to both human and animal health concerns. In cottontails from the central and eastern United States, tularemia was a relatively common diagnosis, especially in endemic areas. Nearly half of the tularemia cases in our study were from Kansas, the state with the fourth largest number of reported human cases in the entire United States from 2011 through 2019. 9 We also documented cottontails with tularemia in other states with higher numbers of reported human cases nationwide, such as Arkansas and Missouri. 9 Although lesions characteristic of tularemia generally were visible upon opening rabbit carcasses, F. tularensis was detected by PCR in one rabbit with severe decomposition that precluded detection of gross or histologic lesions, and another less autolyzed rabbit that lacked gross lesions but had characteristic histologic lesions. In squirrels, F. tularensis was detected in 6 of 15 multiple squirrel species submitted without apparent gross or histologic lesions at a veterinary diagnostic laboratory in Washington, United States. 33 In our study, the most consistent gross lesion pattern was multifocal, randomly and/or widely distributed, small, white-to-tan foci in the liver and spleen. Histologically, these areas corresponded to necrosis and usually, but not always, were associated with gram-negative coccobacilli. Lungs often were grossly mottled pink to dark-red, and in some cases, areas of hemorrhage and necrosis were microscopically evident; however, it was unclear if these were directly related to F. tularensis infection or related to peri- or postmortem changes. Similar lesions can be seen in rabbits infected with other pathogens, including Yersinia pestis, Y. pseudotuberculosis, Y. enterocolitica, and C. piliforme, highlighting the importance of complete diagnostic evaluation in these suspect cases. 55 In our study, lesions in tularemia cases tended to be more necrotizing than yersinosis cases; however, gross and histologic lesions sometimes overlapped, and both diseases tended to have intralesional aggregates of gram-negative bacteria. Both pathogens could cause hepatosplenomegaly and pinpoint foci of random necrosis, characteristic of a septicemic disease process. Thus, good biosafety practices and careful ancillary testing, often first ruling out tularemia, are necessary when working up wild rabbit diagnostic cases, particularly in areas where tularemia is relatively common. Routine aerobic culture of affected viscera, without cold enrichment, was able to detect Yersinia spp. in all cases with compatible histologic lesions. A recent retrospective study in 46 of 87 (53%) hunted rabbits from the United Kingdom attributed white-spotted liver lesions to coccidiosis in most cases, specifically Eimeria stiedai, which could be grossly misdiagnosed as tularemia. 10 Coinfections are also possible, as reported in a rabbit submitted from Missouri that was coinfected with F. tularensis and E. stiedai. 26
Pasteurellosis is one of the major diseases described in domestic and laboratory rabbits and also was diagnosed in cottontails in our study 35 ; systemic pasteurellosis lesions generally resembled those described in domestic and laboratory rabbits, and included pneumonia, pleuritis, peri- and epicarditis, metritis, and lesions consistent with septicemia, often including abundant fibrinous exudate. 3 In domestic rabbits, pasteurellosis with abscessation often is a sequela to traumatic wounds, especially from intraspecific fighting. In wildlife, similar fighting could be a potential cause, as could unsuccessful attempts at predation or other traumatic injuries. One wild eastern cottontail with pasteurellosis in our study had a history of cat attack, and abscesses were adjacent to partially healed puncture wounds. P. multocida is the most common isolate from cat bite wounds in humans and is part of the normal nasopharyngeal and upper respiratory tract flora in cats. 29 While P. multocida may be transmitted via cat bites, other means of potential spread include contact with other rabbits, which can develop subclinical infections and shed the bacteria, potentially infecting other rabbits. 35 Other manifestations of pasteurellosis in domestic and laboratory rabbits, including otitis media and interna, rhinitis, and conjunctivitis, were not seen in our wildlife cases. 35
Most cottontails in our study underwent aerobic, but not anaerobic, culture. More thorough microbiologic characterization to better understand bacterial disease in these species is a worthy future direction of study. Rabbits in our study were often found dead, and acute, fatal septicemia without previous clinical signs is reported in Oryctolagus and Sylvilagus spp. with septicemic diseases such as pasteurellosis and tularemia. 35 Further study of serotypes of P. multocida affecting wild cottontails could reveal interesting comparisons to those in their domestic counterparts. With previous reports of antimicrobial-resistant bacterial strains in cottontails, further microbiologic study may also improve understanding of important strains of bacteria carried by these rabbits. 52
Neoplasia and tumors were rare findings in our study, likely partly due to the generally short average lifespan of many wild cottontails and low annual survival rates (i.e., 5–40%) of adults. 45 Few (<25%) of wild eastern cottontails reach 2-y-old, and they rarely survive beyond 3-y-old. 53 In domestic rabbits, neoplasia increases in prevalence with age, with nearly half of the cases occurring in rabbits >6-y-old. 8 In eastern cottontails, fibromas, lipomas, and malignancies such as renal adenocarcinoma, lymphoma, nephroblastoma, and unidentified malignant neoplasia have been reported.12,32 The round-cell neoplasm diagnosed in our study was histomorphologically consistent with a myeloid neoplasm; however, limitations in available immunohistochemistry in rabbits (i.e., that do not use anti-rabbit antibodies) precluded further workup of this case, and samples were not submitted or archived for electron microscopy at the time of diagnosis. While virologic testing was not performed for our 2 fibroma cases, histologic lesions resembled those reported in leporipoxvirus-associated fibromas in cottontails. 42
As with most retrospective diagnostic studies of free-ranging wildlife, our study results were biased by their opportunistic collection. Interest in and criteria for reporting of sick or dead wild rabbits and submissions for postmortem diagnostic evaluation vary over time. Poor carcass condition due to prolonged postmortem intervals affected gross and histopathology interpretation, and may also affect ancillary test sensitivity. State wildlife agencies have different management and submission priorities and sometimes use other diagnostic laboratories or services, so the results of our study may not provide an accurate picture of reported cottontail mortalities in a particular state or region. Similarly, many wildlife professionals are aware of major diseases of wildlife and may not submit all carcasses for evaluation unless there is a specific regional, host taxonomic, or pathogen concern, or there are potential human or domestic animal health implications, such as for tularemia screening or RHDV2 surveillance. Previous studies cover parasitic diseases of cottontail species from the southeastern United States in more depth, and thus, a comprehensive evaluation of all incidental parasites was not a focus of our research.6,28,48
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241259000 – Supplemental material for Causes of morbidity and mortality in wild cottontail rabbits in the eastern United States, 2013–2022
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241259000 for Causes of morbidity and mortality in wild cottontail rabbits in the eastern United States, 2013–2022 by Alisia A. W. Weyna, Victoria A. Andreasen, Caitlin E. Burrell, Melanie R. Kunkel, Rebecca Radisic, Chloe C. Goodwin, Heather Fenton, Brian S. Dugovich, Rebecca L. Poulson, Mark G. Ruder, Michael J. Yabsley, Susan Sanchez and Nicole M. Nemeth in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank SCWDS member state wildlife management agencies in Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, South Carolina, Tennessee, Virginia, and West Virginia for their continued long-term support and submission of diagnostic cases. Further, we thank SCWDS partnering federal agencies, including the U.S. Fish and Wildlife Service and the U.S. Geological Survey Ecosystems Mission Area. We thank current and former SCWDS diagnosticians and staff (including Xuan Hui Teo, Aidan O’Reilly, Martha Frances Dalton, Kevin Niedringhaus, Brandon Munk, Lisa Last, John A. Bryan II, and Natalie Stillwell) for case workup and ancillary testing and Paul Oesterle (SCWDS) for the map figure. We also thank University of Georgia Histology Laboratory and Athens Veterinary Diagnostic Laboratory staff for their technical expertise, including the preparation of histology slides, and conducting ancillary tests (bacterial culture, PCR), respectively.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study and the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. Support from the states to SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
