Abstract
A 17-y-old Arabian mare was presented to the Auburn Large Animal Veterinary Teaching Hospital with a long-term history of intermittent mild recurrent colic that responded to medical treatment. CBC revealed mild lymphopenia; serum biochemistry findings were of increased gamma-glutamyl transferase and creatine kinase activities, hyperferremia, hyperglycemia, hypomagnesemia, and hypokalemia. Abdominocentesis was compatible with low-protein transudate. Due to the progression and duration of clinical signs, the owner elected euthanasia. Postmortem examination and histopathology confirmed a cholangiocarcinoma. The neoplastic cells were arranged in large cysts containing lakes of mucin that comprised 90% of the tumor volume; thus, a mucinous variant was determined. The neoplastic cells had strong cytoplasmic immunolabeling for cytokeratin 19 and lacked immunolabeling for hepatocyte paraffin 1, supporting bile duct origin. Cholangiocarcinomas are infrequent tumors in horses with nonspecific and slow progressive clinical signs, including recurrent colic. Mucinous cholangiocarcinomas are seldom reported in veterinary medicine and, to our knowledge, have not been reported previously in horses.
A 17-y-old Arabian mare was presented to the Auburn Large Animal Veterinary Teaching Hospital (Auburn, AL, USA) with a 3-wk history of anorexia, gastric ulcers, and suspected right dorsal colitis. The horse had a long-term history of intermittent mild recurrent colic that responded to flunixin meglumine (1.1 mg/kg). On presentation, the patient was alert, with a temperature of 37.1°C (RI: 37.5–39.6°C), tachycardia (56 bpm; RI: 28–40 bpm), tachypnea (30 bpm; RI:10–14 breaths per minute), and pale-pink and moist mucous membranes. CBC revealed mild lymphopenia (1.39 × 109/L; RI: 1.5–5.0 × 109/L), and serum biochemistry showed increased gamma-glutamyl transferase (GGT; 0.53 µkat/L, RI: 0.03–0.48 µkat/L) and creatine kinase (CK; 16.7 µkat/L, RI: 1.7–6.7 µkat/L) activities, hyperferremia (40.3 µmol/L; RI: 23.1–31.0 µmol/L), hyperglycemia (13.6 mmol/L; RI: 4.5–7.0 mmol/L), hypomagnesemia (0.62 mmol/L; RI: 0.70–0.86 mmol/L), and hypokalemia (3 mmol/L; RI: 3.5–4.5 mmol/L).
Transrectal palpation revealed doughy contents in the large colon and no other significant abnormalities. Abdominal ultrasound examination indicated thickening of the right dorsal colon, and a sample of peritoneal fluid yielded a protein-poor transudate (7 g/L), with an estimated count of < 3,000 cells/µL. The differential cell count was 85–90% nondegenerate neutrophils, 5–10% large mononuclear cells (macrophages or mesothelial cells), and 2–5% small lymphocytes. The large mononuclear cells were sometimes lightly vacuolated and occasionally contained phagocytized cellular debris, consistent with macrophages. A fecal sample was submitted the same day for Salmonella culture, which was negative. Flunixin meglumine (1.1 mg/kg IV) and enteral water with electrolytes (4 L q3h) were administered to stabilize the patient and improve comfort. Additional treatment included sucralfate (20 mg/kg PO q8h) and omeprazole (4 mg/kg PO q24h) to treat the gastric ulcers. The patient had transient improvement but became anorexic again, with signs of colic during the following 5 d. Possible right dorsal colonic displacement or impaction were suspected, and exploratory surgery was offered. However, due to the progression and duration of the clinical signs, the owner elected euthanasia.
On postmortem examination, the animal was in good nutritional condition (body condition score 4 of 9), and its tissues had moderate autolysis (euthanasia to autopsy interval of ~12 h). The liver had white-tan, raised, 0.5–3-cm nodules on the hepatic lobes affecting < 10% of the hepatic parenchyma (Fig. 1A). On cut surface, the nodules were variably cavitated and filled with a dark green-brown mucoid material (Fig. 1B). The glandular portion of the gastric mucosa had 1–3-cm, irregular, chronic ulcers. Black cutaneous nodules, 0.5–1-cm diameter, were at the rostral margin (tragus) of the pinna, right temporal surface, perineum, below the external anal sphincter, and next to the vulvar labia. Similar masses were within the left parotid salivary gland. Displacement, torsion, entrapment, or impaction of the intestines was not observed during postmortem evaluation. Representative sections of the evaluated tissues were fixed in 10% neutral-buffered formalin and processed routinely to produce 4-µm, H&E-stained slides.
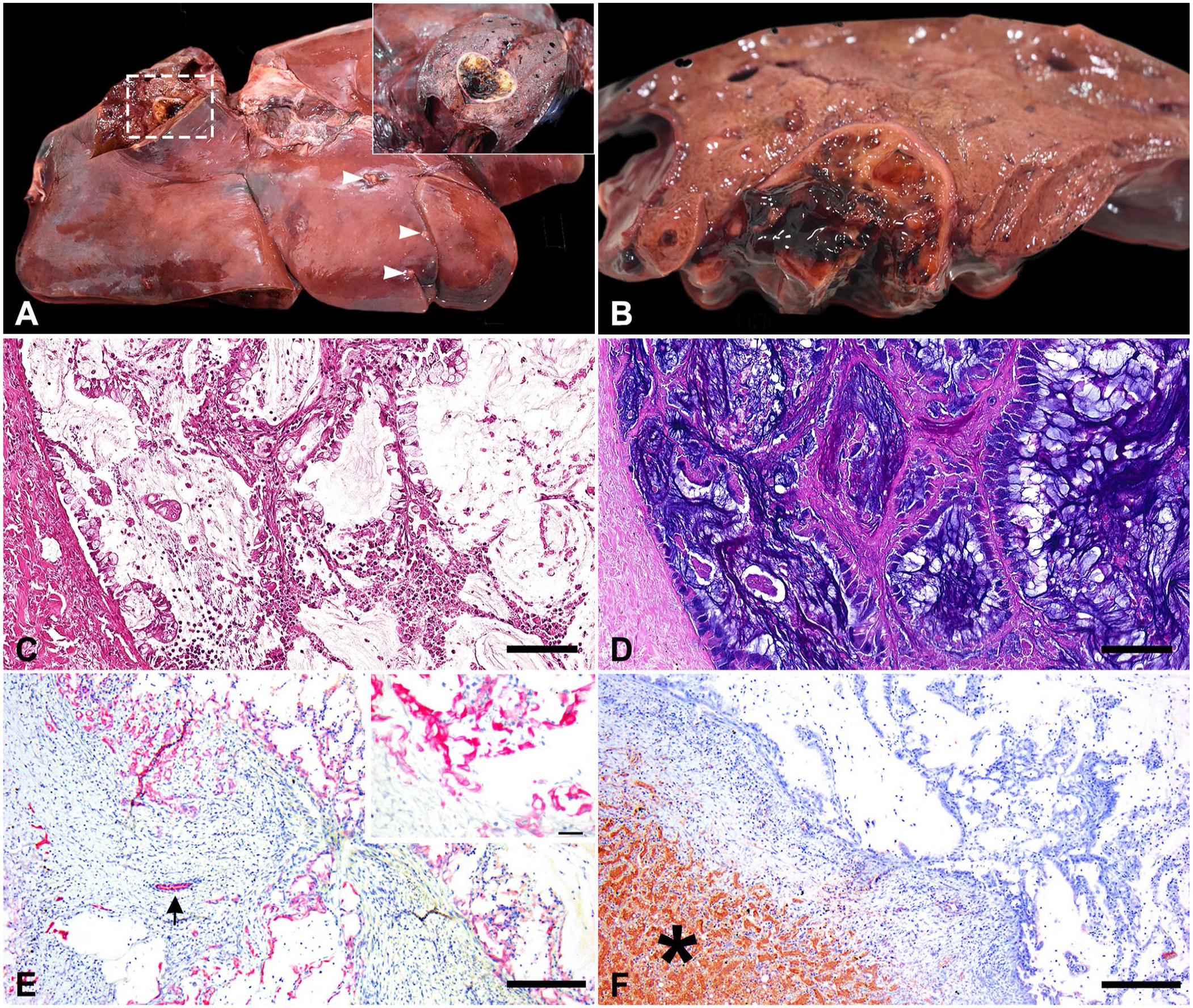
Intrahepatic mucinous cholangiocarcinoma in a 17-y-old Arabian mare.
On histologic examination, the liver parenchyma was effaced and replaced by a multifocal, cystic, moderately cellular, infiltrative, non-encapsulated neoplasm. Neoplastic cells formed papillary projections and fronds, delineated by columnar cells, with mucoid cellular differentiation supported by a fibrous stroma (Fig. 1C). The cysts and cells were filled with amorphous, granular, basophilic, laminated mucoid secretion, highlighted with Alcian blue mucicarmine stain, that comprised 90% of the neoplasm (Fig. 1D). The cells had moderate amounts of pale eosinophilic vacuolated cytoplasm. The nuclei were basal and round, with finely granular chromatin and 1 nucleolus. Anisokaryosis and anisocytosis were moderate. One mitotic figure was in 10 standardized (2.37 mm2) high-power fields (400×). Multifocally, neoplastic cells invaded the adjacent liver parenchyma and compressed hepatic cords. The surrounding hepatocytes had macrovesicular lipidic cytoplasmic vacuoles and eccentric nuclei. To better characterize the histogenesis of the liver neoplasm, immunohistochemistry (IHC) was performed for cytokeratin 19 (CK19; monoclonal mouse, 1:100, Leica) and hepatocyte paraffin 1 (HepPar1; monoclonal mouse, 1:100 dilution, Dako), as bile duct epithelium and hepatocyte markers, respectively. The neoplastic cells had strong cytoplasmic immunolabeling for CK19 (Fig. 1E) and lacked immunolabeling for HepPar1 (Fig. 1F). The cutaneous masses were malignant melanomas with metastasis to the left parotid gland. Histologic evaluation of the right dorsal colon was unremarkable. No significant lesions were evident in other tissues. Aerobic and anaerobic bacterial cultures from the liver yielded light growth of Escherichia fergusonii and Clostridium perfringens, respectively. No parasites were recovered in a Sheather flotation test.
Hepatic neoplasia includes primary epithelial neoplasms (hepatocellular, cholangiocellular, or mixed origin), embryonal hepatic tumors (hepatoblastomas), neuroendocrine hepatic tumors (hepatic carcinoids), and tumors derived from the hepatic mesenchymal components. 6 Malignant primary tumors are uncommon in young animals and humans, and most are classified as hepatoblastoma (HB) or hepatocellular carcinoma (HCC). 1 Cholangiocellular tumors include cholangiocellular adenoma (cholangioma, biliary adenoma), (benign) and cholangiocarcinoma (CC; cholangiocellular carcinoma, biliary carcinoma, bile duct carcinoma) as their malignant counterpart, and are anatomically classified according to the biliary system as intrahepatic, extrahepatic, or gallbladder.6,21 CC is reported in domestic animals, including dogs, cats, sheep, horses, and goats; a grading scheme does not exist for CC in these species.5,6 In humans and dogs, the incidence of CCs has been correlated with chronic infestation of flukes, such as Clonorchis sinensis.6,11 Other risk factors include intestinal parasites (Ancylostoma sp., Trichuris vulpis) and carcinogenic chemicals, such as furans in rodents, or nitrosamine, o-aminoazotoluine, and aramite in dogs.13,22,26,36 In our case, the light growth of E. fergusonii and C. perfringens was interpreted as postmortem bacterial growth without clinical significance or histologic correlation. In horses, predisposing factors for CC have not been described. However, a single case report correlated the presence of chronic cholangitis caused by gentamicin-resistant Klebsiella pneumoniae with concurrent CC. 7
Primary hepatic tumors are rare in horses, with metastatic tumors to the liver observed more commonly.1,4 HB and HCC are the most common hepatic tumors in horses, according to a retrospective study, and are mostly described in young 2–3-y-old animals.1,4 CC typically occurs in older animals, with a reported age of 11–23 y, and is only reported sporadically (Table 1).4,7,12,20,24,29,33–35,42 A combined or mixed HCC and CC have been reported in an 18-y-old Thoroughbred mare. A CC with a concurrent gastric squamous cell carcinoma was also described in an 11-y-old mixed-breed mare.20,29 Consistent clinicopathologic findings associated with CC in species other than cats and dogs are lacking due to insufficient published information. 5
Summary of signalment, and clinicopathologic and autopsy findings of cholangiocarcinoma cases reported in horses.
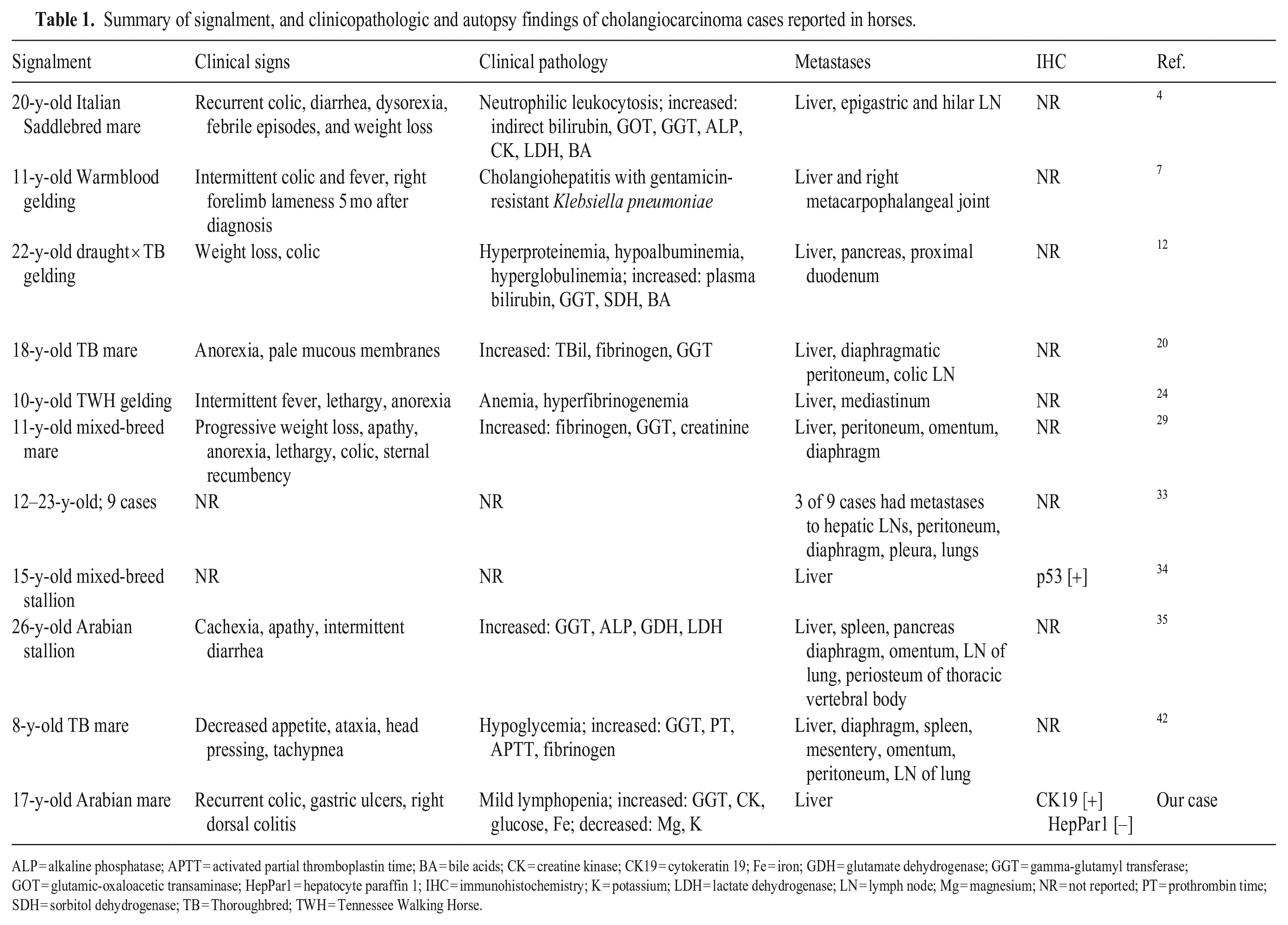
ALP = alkaline phosphatase; APTT = activated partial thromboplastin time; BA = bile acids; CK = creatine kinase; CK19 = cytokeratin 19; Fe = iron; GDH = glutamate dehydrogenase; GGT = gamma-glutamyl transferase; GOT = glutamic-oxaloacetic transaminase; HepPar1 = hepatocyte paraffin 1; IHC = immunohistochemistry; K = potassium; LDH = lactate dehydrogenase; LN = lymph node; Mg = magnesium; NR = not reported; PT = prothrombin time; SDH = sorbitol dehydrogenase; TB = Thoroughbred; TWH = Tennessee Walking Horse.
The clinical signs associated with CC in horses rely on scattered documented cases and are nonspecific and difficult to recognize due to their slow progression. 4 Reported signs include recurrent colic episodes without response to flunixin meglumine administration (0.5 mg/kg), diarrheic feces, dysorexia or anorexia, mild intermittent febrile episodes, apathy and lethargy, cachexia, sternal recumbency, ataxia, head pressing, seizures, and progressive weight loss (Table 1).4,7,12,20,24,29,35,42 The most relevant clinical sign in our case was recurrent colic. Displacement, volvulus, entrapment, torsion, or impaction was not seen during autopsy; the gross examination of the right dorsal colon was unremarkable. Postmortem resolution of intestinal displacements cannot be completely ruled out and the possible intestinal origin of the clinical signs of colic remains unknown. We speculate that the CC also contributed to the colic signs due to the metastases to all liver lobes; however, the patient had gastric ulcers that could have contributed to the recurrent colic clinical signs.
Clinicopathologic findings are usually nonsignificant or nonspecific and include neutrophilic leukocytosis, hypoalbuminemia, hyperglobulinemia, hyperbilirubinemia, hyperfibrinogenemia, hypoglycemia, increased prothrombin time, activated partial thromboplastin time, bile acids, and indirect reacting bilirubin, and serum activities of aspartate aminotransferase (AST), GGT, alkaline phosphatase (ALP), CK, lactate dehydrogenase (LDH), and sorbitol dehydrogenase (SDH; Table 1).4,12,20,24,29,35,42 Only GGT and CK increases were recorded in our case. GGT is an enzyme bound to the membrane of biliary epithelium and its increase is related to biliary hyperplasia and cholestasis. 15 While the GGT activity was mildly increased in our case, ALP remained within the RI. GGT in horses is considered more sensitive than ALP, with increased reported responsiveness in experimental cholestasis compared to ALP; GGT increased 9-fold whereas the ALP increase was 2-fold, explaining the difference between values in our case. 14 In cholestatic disorders, GGT is increased, and the hepatocellular enzymes (SDH, AST) remain in low elevations, as in our case. 15 The increased GGT serum activity in our patient was mild. In fact, GGT serum activity was mildly increased in the 3 recorded serum biochemistry panels performed in our patient over 10 d, which might represent a standard deviation from the RIs without clinical significance. This contrasts with the previously reported cases of a CC in horses, in which GGT was markedly increased. The difference between these cases and our case is the extensive metastatic disease evidenced on autopsy in other published cases.4,12,20,29,35,42 CK activity was elevated in our case. CK is a cytoplasmic enzyme from skeletal and cardiac muscle and is increased after muscle injury and intravenous and intramuscular injections. 38 Our case had signs of colic (kicking, rolling) and an abdominocentesis, which together elicited muscle damage and subsequent CK serum activity increase.
Macroscopically, equine CCs are described as spherical to pear-shaped, raised, umbilicated, encapsulated, multifocal-to-coalescing, white-to-yellow masses in the liver that are firm due to fibrous tissue or mineralization.4,12,20,29 Mass diameters are variable and range from 0.2–25 cm.12,20,42 In most of the equine published cases, widespread carcinomatosis is reported, including in the diaphragmatic peritoneum, omentum, mesentery, pancreas, proximal duodenum, right metacarpophalangeal joint, colic lymph nodes, spleen, periosteum of thoracic vertebra, lungs, tracheobronchial lymph nodes, and mediastinum.7,12,20,24,29,35,42 Extrahepatic metastases were not detected in our case.
In horses, the reported histologic features of CC include the formation of tubules, acini, ducts, papillae, and solid lobules, with stromal fibrosis, central areas of necrosis, and mineralization with frequent mitotic figures.12,20,29,35,42 Mitotic activity is one of the diagnostic criteria used to differentiate CC from benign biliary lesions. However, in our case, the mitotic count was only 1 in 10 standardized (2.37 mm2) high-power fields (400×). We used the criteria of local invasion and intrahepatic metastases to define our case as a malignant tumor. In well-differentiated tumors, neoplastic cells and ducts contain mucin. 6 The presence of mucin is more typical of biliary origin than metastatic tumors of pancreatic or mammary origins, usually considered important differential diagnoses.6,27,30,39 Occasional accumulation of mucin within the tumor forms large aggregates or “lakes.” 30 Mucin stains weakly with H&E and is better highlighted with Alcian blue stain. 5 A cholangiocellular cystadenocarcinoma is a histologic variant that has cysts with papillary projections, with single or several layers of neoplastic biliary epithelial cells. 5 The cysts contain abundant mucinous secretion; however, the relevance of this histologic subtype is unclear because they exhibit behavior similar to CC. 5 Mucin production was observed in the cholangiocellular areas of a mixed HCC and CC reported in a mare. 20
Mucinous CC in animals has been reported in cats and a captive sloth bear (Melursus ursinus).16,17,19 Although mucin production is a common feature reported in CC, the description of a mucinous variant in these case reports is vague and not detailed. In human cases, mucinous adenocarcinomas are identified arising from mammary gland, pancreas, colon, and gallbladder, and by convention, at least 50% of the tumor volume must be composed of large extracellular lakes of mucin containing floating carcinoma cells.3,37 In animals, intestinal mucinous adenocarcinomas are defined by the same criterion. 25 Because 90% of the mucinous extracellular material formed the neoplasm volume in our case, we propose the “mucinous” descriptor in our morphologic diagnosis based on an extrapolation of the defined human histologic criterion.
Differential diagnoses for CC include HCC. IHC is a valuable tool in reaching a definitive diagnosis. HepPar1 is a marker for hepatocellular differentiation, and it has been used for diagnosis of HCC in canine and human patients.9,31 Normal biliary epithelium and neoplastic cells of biliary origin do not immunolabel for HepPar1, thus we used this marker to differentiate CC from HCC, although mucin production is not a common feature described in HCC. However, it is important to emphasize that HepPar1 immunolabeling is not exclusive of hepatocellular neoplasms; in fact, normal intestinal epithelium will immunolabel for HepPar1. 32 Canine non-hepatic neoplasms with reported HepPar1 immunolabeling include metastatic intestinal tumors to the liver, large and small intestinal carcinomas, mucinous intestinal carcinomas, adrenocortical carcinoma, testicular interstitial cell tumors, melanoma, salivary carcinomas, and leiomyosarcomas.31,32 HepPar1 is useful to distinguish hepatocellular tumors from cholangiocellular tumors; however, it cannot differentiate metastatic carcinomas.
CK7 has been used to identify bile ducts in equine tissues to differentiate cholangiocarcinoma from HCC and HB 1 ; however, a case report of a cancer of unknown primary in a mare yielded a negative result with lack of immunolabeling of normal bile ducts using CK7. 2 Thus, we favor the use of CK19 in equine liver. The biliary epithelium is composed of cholangiocytes that represent ~5% of the liver cell population and typically express intermediate filaments as CK7 and CK19. 5 However CK7 and CK19 are not exclusive markers for biliary neoplasms. CK7 immunolabeling has been reported in intestinal, hepatocellular, pancreatic, pulmonary, urothelial, endometrial, and mammary gland carcinomas in cats; and in urothelial, prostatic, ovarian, endometrial, apocrine anal sac, ceruminous gland, and mammary gland carcinomas in dogs.8,28 CK19 in humans is reported in intestinal and pancreatic carcinomas, whereas in veterinary medicine this marker immunolabels HCC, mammary carcinomas of dogs, and pancreatic carcinomas in cats.10,18,23 CK19 immunolabeling is associated with poor histologic differentiation and highly aggressive biologic behavior in hepatocellular tumors in dogs.40,41 There are no large studies assessing the use of this marker in horses. The increased amount of p53 protein was demonstrated (based on IHC) in a 15-y-old male horse with a CC. 34 In humans, mucinous cholangiocarcinoma has immunolabeling for mucin IHC markers MUC1, MUC2, MUC5AC, MUC6, CK7, CDX2, p53, Smad4, and epidermal growth factor receptor (EGFR), and lacks immunolabeling for CK20. 3 Metastatic carcinomas represent a higher challenge when distinguishing them from CC. When large masses are present within the pancreas, intestine, mammary gland, or uterus, a metastatic carcinoma is favored over a CC. However, in our case, a primary tumor arising from another anatomic location was not detected at postmortem examination, and the lack of immunolabeling for HepPar1 reinforced the biliary origin over hepatocellular origin because CK19 is not an exclusive marker of biliary epithelium. In humans, distinctions rely on the use of IHC markers when the primary origin of the mucinous CC is dubious.
CCs are uncommon tumors in old horses but should be considered a differential diagnosis in animals > 11-y-old with nonspecific clinical signs, including recurrent colic episodes and elevated GGT activity. We obtained no cases of intrahepatic mucinous CC diagnosed in horses in a comprehensive search of Google, PubMed, CAB Direct, Web of Science, and Scopus, using search terms “mucinous cholangiocarcinoma,” “mucinous cholangiocellular carcinoma,” “horse,” and “equine,” suggesting that this condition has not been reported previously in horses. The use of biliary IHC markers and an exhaustive postmortem evaluation is critical when possible metastatic carcinomas arising from the intestine, pancreas, or mammary gland are suspected and cannot be ruled out.
Footnotes
Acknowledgements
We thank Lisa Parsons (Histology Laboratory, Pathobiology, College of Veterinary Medicine, Auburn University) for technical assistance and slide preparation, and Dr. Andrew Miller (Cornell University) for providing the technical protocols for CK19 and HepPar1 immunohistochemical stains.
Declaration of conflicting interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The authors did not receive any specific grant from the public, commercial, or not-for-profit funding agencies.
