Abstract
Here we report the isolation of Streptococcus hillyeri from a thoracic sample from a horse. A 17-y-old Hungarian Sport Horse mare was referred to the equine clinic of the University of Veterinary Medicine Budapest, Hungary, with suspected pleuritis. Upon arrival, the horse was febrile and had tachycardia, severe inspiratory dyspnea, and tachypnea. Thoracic ultrasonography revealed severe bilateral pleural effusion, and a large area of lung consolidation. After sampling of both hemithoraces, 66 L of turbid exudate were drained. Based on these findings, a tentative diagnosis of septic pleuritis was made, and the horse was immediately started on a course of broad-spectrum antibiotics, a NSAID, an anticoagulant, and intravenous fluids. Despite intensive care, the clinical parameters deteriorated, and the horse was euthanized 6 d later. Cytology confirmed septic pleuritis, with short chains or groups of coccoid bacteria. Anaerobic culture yielded gram-positive cocci from both hemithoraces in almost pure culture, which we identified as S. hillyeri by 16S rDNA and whole-genome analysis. Additionally, we identified 4 previously unassigned Streptococcus sp. sequences as S. hillyeri. Of these, 3 were obtained from aborted equine fetuses and a fourth from a donkey mastitis case, supporting the pathogenic nature of S. hillyeri in these host species.
Pleuritis is rarely an independent condition in horses and is usually the sequel to severe bacterial bronchopneumonia with extension of inflammation to the pleura, when the condition is termed pleuropneumonia. 12 Predisposing factors, most often long-term transportation, prolonged head elevation, strenuous exercise, general anesthesia, or primary viral infection, are needed to compromise local defense mechanisms and allow colonizing facultative pathogenic or commensal bacteria to gain access to and induce inflammation in the lower airways. 1
The bacterial genera involved in pleuritis are most often natural inhabitants of the horse’s mucous membranes and are aerobes (Actinobacillus spp., Bordetella spp., Escherichia coli, Klebsiella spp., Pasteurella spp., and β-hemolytic streptococci) or anaerobes (Bacteroides spp., Clostridium spp., Fusobacterium spp., Peptostreptococcus spp., and Prevotella spp.).1,13,14 The prognosis is usually poorer if anaerobes are involved in the process. 10 Although several species can be isolated from pleuropneumonia, and mixed infections are likely, Streptococcus equi subsp. zooepidemicus is considered the main causative agent.1,10
Other streptococci are rarely associated with pneumonia or pleuropneumonia, with β-hemolytic Streptococcus dysgalactiae subsp. equisimilis and the primarily human-associated S. pneumoniae identified from lower airway infection in horses.4,16 Here we report the isolation of another, recently recognized equine Streptococcus, S. hillyeri, and describe the associated clinical manifestation and laboratory investigation of septic pleuritis in a horse.
A 17-y-old Hungarian Sport Horse mare was referred to the Department and Clinic of Equine Medicine, University of Veterinary Medicine Budapest (Üllő, Hungary) with suspected pleuritis. According to the owner, the horse started losing weight ~1 wk before admission. On the day of presentation, the referring veterinarian reported dyspnea, elevated heart rate, cyanotic mucous membranes, and prolonged capillary refill time. The veterinarian performed thoracic ultrasonography, where he found a large amount of fluid in the thoracic cavity and subsequently referred the horse to the clinic for drainage and further treatment.
Upon arrival, the horse was in poor body condition (body condition score: 3 of 9, body weight: 510 kg), and the body surface was wet in places. The horse was febrile; rectal body temperature was 38.8°C (RI: 37.0–38.0°C). Heart rate was 80 bpm (RI: 28–42 bpm), corresponding to tachycardia. The oral mucosa was cyanotic; capillary refill time was 6 s (RI: <2 s). The horse had severe inspiratory dyspnea accompanied with tachypnea (respiratory rate: 45 breaths/min; RI: 10–18 breaths/min). Thoracic ultrasonography revealed severe bilateral pleural effusion. The pleural fluid was anechoic to hypoechoic, and a large amount of proteinaceous material was detectable. The maximum depth of the fluid was ~25 cm. Several comet-tail artefacts were seen, and a large area of lung consolidation was also found. Abdominal ultrasonography revealed a large amount of anechoic peritoneal fluid. No other abnormalities were seen. CBC revealed a stress leukogram; elevated alkaline phosphatase and lactate dehydrogenase activities, increased triglyceride, glucose, and lactate concentrations, and hypoalbuminemia were also detected (Suppl. Table 1). Both hemithoraces and the abdomen were sampled for cytologic and microbiologic examination.
After sampling, both hemithoraces were drained using 32-F drains, through which 48 L of red-orange, slightly turbid, odorless fluid was drained from the left and 18 L from the right cavity. While awaiting microbiologic and cytologic results, the horse was immediately started on a course of broad-spectrum antibiotics (gentamicin, amoxicillin–clavulanic acid, and metronidazole), an NSAID (flunixin meglumine), an anticoagulant (enoxaparin), and intravenous fluids (lactated Ringer, Gelofusine; B. Braun). Thoracic fluid (~20 L) was drained daily during the following 3 d when the drains were removed. During this time, the dyspnea was significantly milder. Two days later, bilateral pleural effusion was detected again, and the severe dyspnea reappeared. Also, severe ventral edema developed. Despite intensive care, the horse’s condition deteriorated from both the clinical and laboratory perspectives. Given the poor prognosis and lack of response to treatment, the horse was euthanized with the owner’s consent. The carcass was disposed of without postmortem examination at the owner’s request.
The total cell counts of EDTA-anticoagulated bodily fluid samples were measured (Advia 120; Siemens). Then the samples were centrifuged for 10 min at 360 × g, and monolayer smears were made from the sediments for cytologic evaluation (Reag-Quick Panoptic stain; Reagens).
The sample taken from the left and right hemithoraces had a slightly elevated leukocyte count (left: 8.6 × 109/L, right: 9.9 × 109/L; RI: <5 × 109/L) and increased total protein levels (left: 34.1 g/L, right: 31.2 g/L; RI: 10–25 g/L). Cytologically, the sediment smears contained a small proportion of erythrocytes and an abundance of nucleated cells, mostly intact and degenerate neutrophils, some with karyorrhexis. Several reactive macrophages and occasional lymphocytes could also be seen (Fig. 1). A monopopulation of coccoid bacteria, both intracellular and free, appeared in abundance in chains or in small groups (Fig. 2), together identifying the sample as exudate of a septic pleuritis. No malignant, atypical cell forms were observed.
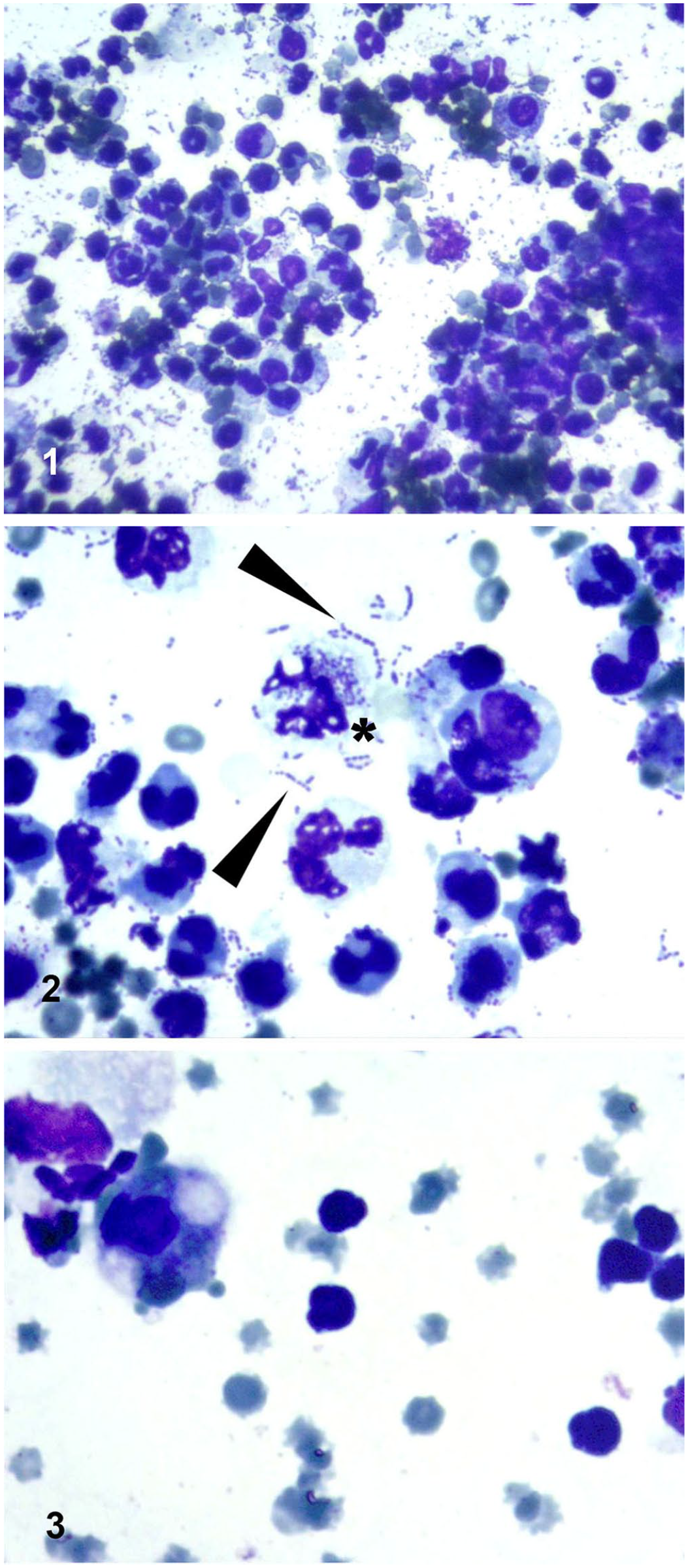
Cytologic preparation of thoracic and abdominal fluid samples taken at admission.
WBC counts in the abdominal fluid were within the RI (2.5 × 109/L; RI: <5 × 109/L), and the protein level was mildly increased (27.7 g/L; RI: <25 g/L). The smear of the abdominal fluid sediment contained RBCs predominantly, and slightly increased numbers of nucleated cells, with lymphocytes being the most abundant, followed by neutrophils, and macrophages or mesothelial cells. No bacteria or malignant cell forms were observed (Fig. 3). The findings suggested the presence of a transudate in the abdomen due to chronic stasis.
Bacteriologic culture of the bodily fluids was carried out by spreading the samples onto 5% Columbia sheep blood agar and McConkey agar plates (Biolab). Cultures were incubated both aerobically and anaerobically (AnaeroGen; Thermo) at 37 ± 1°C for 24–48 h. The Gram stain and catalase test were performed on pure cultures. For further identification, the 16S rDNA of isolate S23-3001-2 was Sanger-sequenced, and both isolates underwent long-read whole-genome sequencing and phylogenetic analysis (Suppl. Method 1).
No growth was observed on the plates cultured aerobically, and no pathogen was cultured from the abdominal fluid. The anaerobe culture of the pleural fluid samples from both sides yielded catalase-negative, gram-positive cocci in almost pure culture, forming 1–2-mm, non-hemolytic, gray-translucent, glistening colonies on Columbia sheep blood agar.
The partial 16S rRNA gene sequence of the selected study isolate (S23-3001-2, PQ085642) had the closest match with that of the type strain of S. hillyeri (100% pairwise nucleotide identity [PNI]), followed by S. caledonicus (96.5%), 5 as the next closest species match. Interestingly, sequences of a few other unidentified Streptococcus sp. isolates had close hits (98.0–99.4% PNI), 3 of them well above the ≥98.7% threshold suggested for species delineation. 6
Whole-genome sequencing resulted in a sequencing coverage of 318.05 × (S23-3001-1) and 338.83 × (S23-3001-2) after quality-filtering the sequencing data. Flye (https://github.com/mikolmogorov/Flye) assembled a single contig from each isolate, which appeared to be circular and had a size of 2,227,889 bp (S23-3001-1) and 2,227,898 bp (S23-3001-2). Both could be classified as Streptococcus sp. and had a completeness of 99% without any contamination identified. The core genome consisted of 291 genes (genes present in >99% of both isolates) and IQ-TREE (http://www.iqtree.org/) unequivocally clustered both isolates to the reference of S. hillyeri with high statistical support (Fig. 4), confirming the result of the species identification using 16S rDNA. As well, our isolates and the reference S. hillyeri isolate clustered with the isolate from donkey mastitis (isolate 20-37394), with S. caledonicus being found at the lowest genetic distance. We could separate all distinct species with high support values in the phylogenetic tree (Fig. 4). Both our isolates had 99.7% average nucleotide identity (ANI) with the S. hillyeri reference genome. Again, the whole genome of the Streptococcus sp. isolated from the donkey mastitis case was clustered to the same branch (Fig. 4) and had 97.0–97.1% ANI compared to the S. hillyeri genomes.
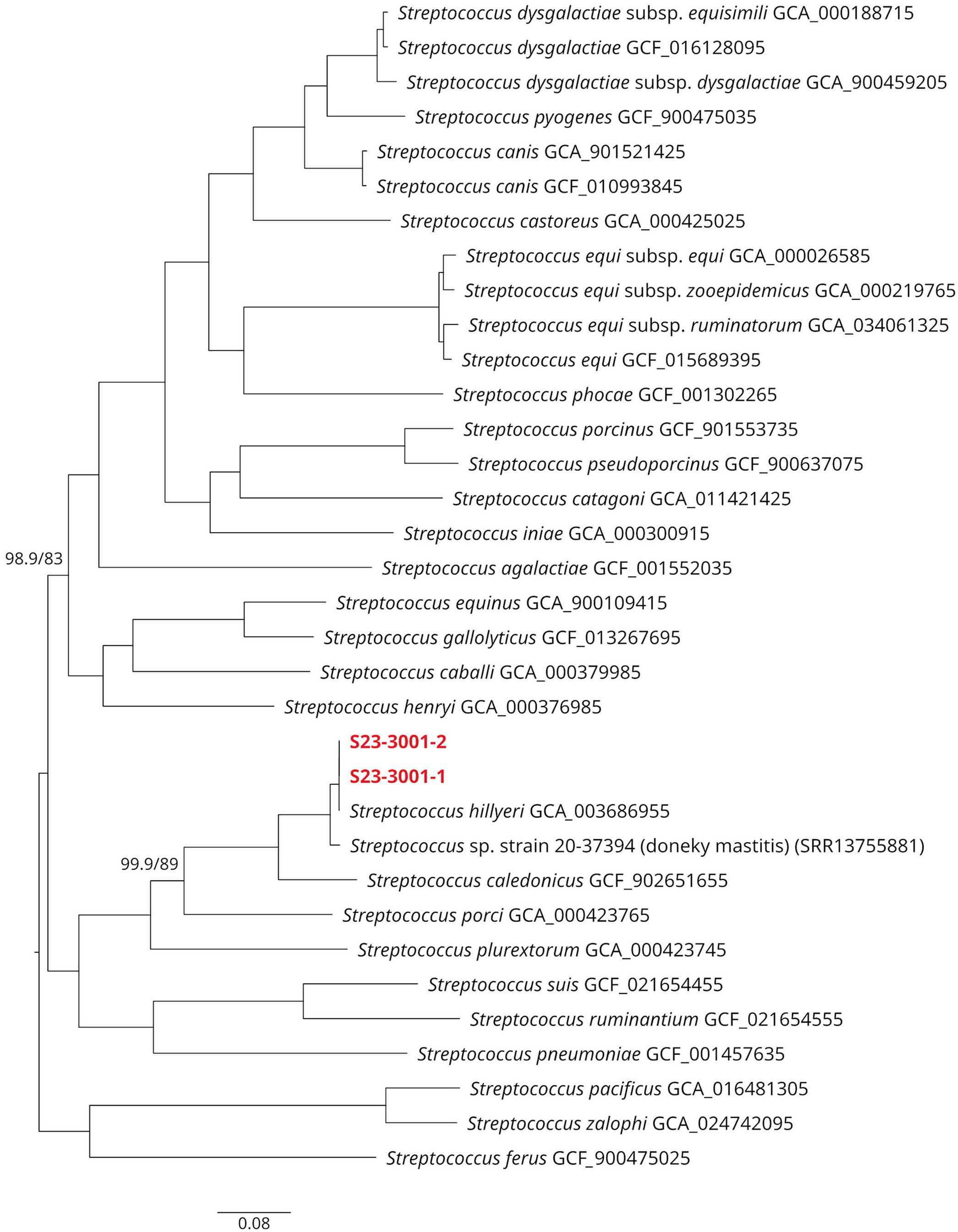
Core genome–based phylogenetic reconstruction of Streptococcus hillyeri depicted as a phylogram, including other closely related or relevant pathogenic Streptococcus species. All branches received maximum aLRT/rapid bootstrap statistical support (100/100), except where indicated. Our isolates are shown in red.
We identified 6 copies of the 16S rRNA genes in each de novo assembled genome, corresponding to 2 allelic variants. In each of the isolates, 1 allele was identified in 4 copies with 100% similarity to the S. hillyeri reference strain, consistent with the results of Sanger sequencing of the partial 16S rRNA gene. The other copy of the 16S rRNA gene had 3 point mutations and was present twice in both isolates. In the phylogenetic reconstruction of the 16S rRNA gene sequences, both copies clustered with the S. hillyeri reference strain. Highly similar accessions of a few other unidentified Streptococcus sp. were also clustered with the S. hillyeri reference sequence in the phylogenetic analysis of the 16S rRNA gene (Suppl. Fig. 1). Three (EU483249.1; EU_483250.1; EU_483251.1) were cultured from aborted equine fetus samples (unpublished data), and another one from mastitis of a donkey (MW_662649.1, isolate 20-37394). Whole-genome sequence data from our study were deposited in the NCBI Sequence Read Archive database under BioProject accession PRJNA1140127. The core genome alignment with related statistics, the alignment of 16S rRNA gene copies, and the unedited phylogenetic trees can be obtained from the Zenodo repository (doi: 10.5281/zenodo.12820377).
Marked pleural effusion in horses can be most likely of primarily septic, traumatic (hemorrhage, chylothorax), or neoplastic origin, or a result of congestive heart failure. 11 In our case, the septic nature of the process was suspected based on the fever and the presence of fibrin. The cytologic findings and isolation of bacteria in almost pure culture confirmed the diagnosis of septic pleuritis. Pleuropneumonia is considered a devastating condition in equine medicine, with survival rates of 43.3–87.6%.12,15 Fibrinous pleuritis further worsens the prognosis, as well as the general condition of the animal at admission, and results in decreased survival rates. 15 The relapse of our case after drainage and its subsequent deterioration indicated a poor prognosis, and euthanasia was justified.
Septic pleuritis in horses is most often a sequel to bacterial bronchopneumonia rather than a primary disease. 12 The clinical and cytologic findings in our case suggested bronchopneumonia, hence secondary septic pleuritis is plausible, although we could not identify any of the frequent predisposing factors associated with bronchopneumonia.8,12 A primary viral infection was not investigated, so cannot be ruled out. Notably, the amount of the pleural exudate was exceptionally large in our case. Forty-eight and 18 L could be drained at the first attempt and a further 20 + 20 L in each of the following 3 d. In the case of such massive pleural effusion and resistance to intensive therapy, the exclusive role of the pleuropneumonia should be questioned, and an underlying process sought. 11 Thoracic mesothelioma and lymphoma are 2 conditions that can result in extreme amounts of pleural effusion, hence must be included in the differentials. 3 Although no malignant cell forms were identified during cytologic examination, neoplasms can lack a diagnostic cytologic picture, and only biopsy or postmortem histopathology can confirm their presence. 3 Unfortunately, however, no pathology studies were performed that would have allowed either the identification of a neoplastic background or definitive confirmation of lung involvement.
In both pleuritis and pleuropneumonia of any origin, involved bacteria are often the facultative pathogens or commensals colonizing the upper respiratory tract or the oropharynx of the horse. 12 According to microbiome studies, streptococci are the most frequent colonizers of such niches. 2 In addition to the most frequent facultative pathogen, S. equi subsp. zooepidemicus,1,10 other streptococci may also be involved in airway infections, such as S. dysgalactiae subsp. equisimilis or S. pneumoniae.4,16 Although other species of the genus have been detected in samples of healthy horses, such as S. pluranimalium, S. minor, and S. hillyeri,2,7 there are no reports suggesting their pathogenic nature. The only, to our knowledge, report on S. hillyeri described the isolation of the organism from a routine tracheal sample of a horse, without discussing involvement in any disease. 7 The S. hillyeri strain that we obtained was very likely the causative agent of the pleuritis, and thus provides a disease report unambiguously related to this species.
Interestingly, the 16S rDNA sequence of the study strains clustered with other sequences originating from Streptococcus sp. isolates of pathologic samples of Equidae, in addition to the S. hillyeri type strain. Three of these have been isolated from aborted equine fetuses, another from a donkey mastitis sample. 9 In the latter case, the whole-genome sequence was available, and both whole-genome and 16S rDNA analysis confirmed the donkey isolate to be S. hillyeri (Fig. 4). These observations suggest that our case may not be unique and make it plausible that S. hillyeri is part of the normal mucosal microbiota of horses and donkeys and can occasionally act as a facultative pathogen in both host species.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241306724 – Supplemental material for Streptococcus hillyeri isolated from septic pleuritis in a horse
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241306724 for Streptococcus hillyeri isolated from septic pleuritis in a horse by Ervin Albert, Imre Biksi, Levente Laczkó, László Miló, Karolina Cseri, Renáta Bőkényné Tóth, Dalma Papp, Dóra Halmay, Emese Bódai and Zoltán Bakos in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Ms. Edit Csuka and Ms. Ágnes Vu for their excellent technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the intramural strategic research fund of the University of Veterinary Medicine Budapest (grant SRF-001).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
