Abstract
In late summer/early fall of 2013, 2 South American camelids from central Washington were diagnosed with fatal bluetongue viral disease, an event which is rarely reported. A 9-year-old intact male llama (Lama glama), with a 1-day history of anorexia, recumbency, and dyspnea before death. Abundant foam discharged from the mouth and nostrils, and the lungs were severely edematous on postmortem examination. Histologically, there was abundant intra-alveolar edema with fibrin. Hemorrhage and edema disrupted several other organs. Bluetongue viral RNA was detected by reverse transcription polymerase chain reaction (RT-PCR), and serotype 11 was identified by sequencing a segment of the VP2 outer capsid gene. Approximately 1 month later, at a site 150 miles north of the index case, a 2-year-old female alpaca with similar, acutely progressive clinical signs was reported. A postmortem examination was performed, and histologic lesions from the alpaca were similar to those of the llama, and again serotype 11 was detected by PCR. The occurrence of bluetongue viral infection and disease is described in the context of seasonal Bluetongue virus activity within the northwestern United States and southwestern Canada.
Bluetongue is a vector-borne disease caused by Bluetongue virus (BTV; family Reoviridae, subfamily Sedoreovirinae, genus Orbivirus 21 ) that is transmitted mainly through biting midges belonging to the Culicoides genus.6,17,22 Transmission through other biting insects2,8 and mother-to-fetus transmission 4 have also been documented. Vector distribution plays an important role in the distribution of different serotypes of BTV, with 5 serotypes in North America, 4 vectored by C. sonorensis (BTV-10, -11, -13, and -17) and 1 by C. insignis (BTV-2).5,25 Given the predominance of vector-based transmission, bluetongue is a seasonal (late summer to early fall) disease in temperate climates, 9 such as the northwestern United States and southwestern Canada.3,5
Bluetongue virus has been documented to infect domestic ruminants, wild ruminants, and carnivores.12,14 Infection by BTV can result in very high morbidity and mortality rates in sheep and some wild ruminants, although the virus typically causing subclinical infections in other ruminants, such as cattle.14,17 Until recently, camelids were thought to be resistant to BTV. In 2007, an alpaca died from BTV-1 infection in Germany, 10 and in 2008, 2 outbreaks of BTV-1 infection occurred in 2 llama herds in France. 18 Seroprevalence of BTV within South American camelids in Germany was also examined in 2008–2009 and was found to be 14.3%. 24 In the United States, there is a report of an alpaca dying from bluetongue (unidentified serotype) in 2008 in California. 19 South American camelids experimentally inoculated with BTV-8 developed only mild clinical signs. 23 This report describes natural BTV-11 disease in 2 South American camelid facilities in the context of a seasonal bluetongue event in multiple species, and provides an initial investigation into the seroprevalence of BTV in camelids within the northwestern region of the United States.
In August 2013, a 9-year-old intact male llama (Lama glama) was referred to Washington State University Veterinary Teaching Hospital (WSU VTH) from the Pasco, Washington area. The animal had a 1-day history of anorexia and a several-hour history of reluctance to rise, coughing, foaming at the mouth, open-mouth breathing, and lung crackles. The llama died in transit and was submitted to the Washington Animal Disease Diagnostic Laboratory (WADDL) for postmortem examination. Cardiac blood was collected for postmortem serology. Gross postmortem findings included abundant foam discharging from the mouth and both nostrils. The lungs were severely edematous and congested. There was mild submucosal edema of the trachea, mild multifocal hemorrhage of the aortic tunica adventitia and pancreas, and mild serosanguineous peritoneal effusion. Representative sections were collected from organs and immersion fixed in neutral buffered 10% formalin. Fixed tissues were embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin for histology. Fresh samples from lung, liver, spleen, and kidney were collected and stored at −20°C.
The most significant histologic findings were pulmonary edema (Fig. 1) with rare, mild vasculitis (Fig. 1 inset) and hemorrhage. Edema and hemorrhage of the larynx, trachea, heart, aorta, and adrenal glands were also noted. Centrilobular hepatocytes were moderately disrupted by lipid-type degeneration.
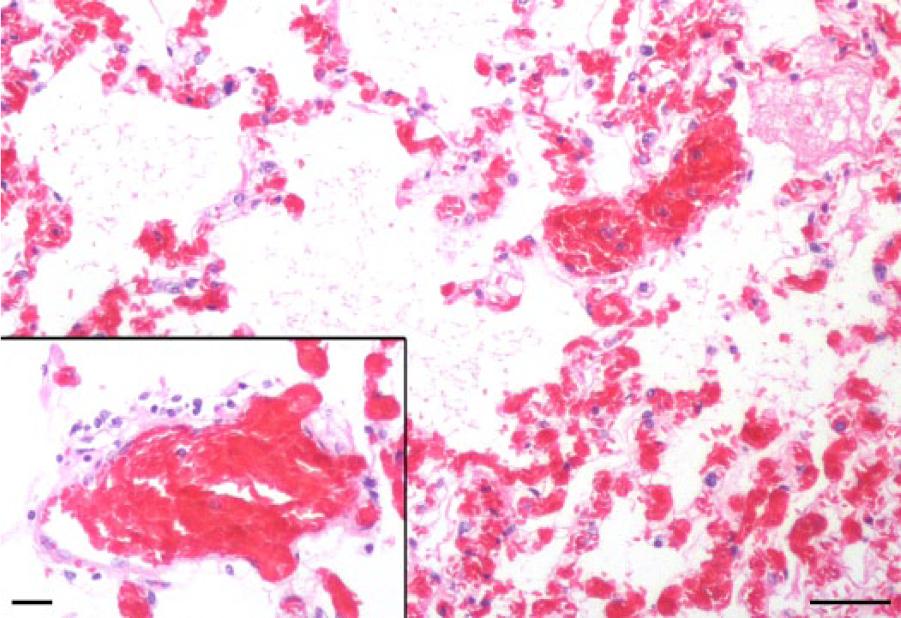
Llama (Lama glama); lung, with intra-alveolar edema and fibrin. Hematoxylin and eosin (HE). Bar = 200 µm. Inset: A few mononuclear inflammatory cells infiltrate the tunica adventitia of an artery. HE. Bar = 50 µm.
Sections of fresh-frozen lung were evaluated for BTV and West Nile virus (WNV) RNA by reverse transcription quantitative polymerase chain reaction (RT-qPCR) at WADDL. Small sections (approximately 2–3 mm3) of tissue were placed into 1.5-mL microcentrifuge tubes containing five 2.5-mm glass beads and 200 µL of Dulbecco modified Eagle medium. Tissues were homogenized for 30 cycles at speed setting 25. a RNA was isolated from homogenized splenic tissue using magnetic beads per manufacturer’s directions. b BTV RNA was assessed by using previously published primers and probe directed against the M6 gene, which codes for the NS1 protein. 26 WNV RNA was assessed by using previously published primers and probe directed against the envelope glycoprotein gene. 11 PCR testing for BTV RNA was positive; the WNV PCR was negative. Undiluted serum was negative for BTV antibodies by competitive enzyme-linked immunosorbent assay (cELISA; Fig. 2). c Samples resulting in optical densities (ODs) <50% of the mean of the negative controls were considered seropositive. Fresh-frozen lung samples were submitted for serotype identification to the National Veterinary Services Laboratories (NVSL) in Ames, Iowa. BTV-11 was identified with RT-PCR of the VP2 outer capsid protein gene followed by routine amplicon sequencing (standard operating procedure, personal communication, D. Johnson, NVSL, Ames, Iowa, 2014).
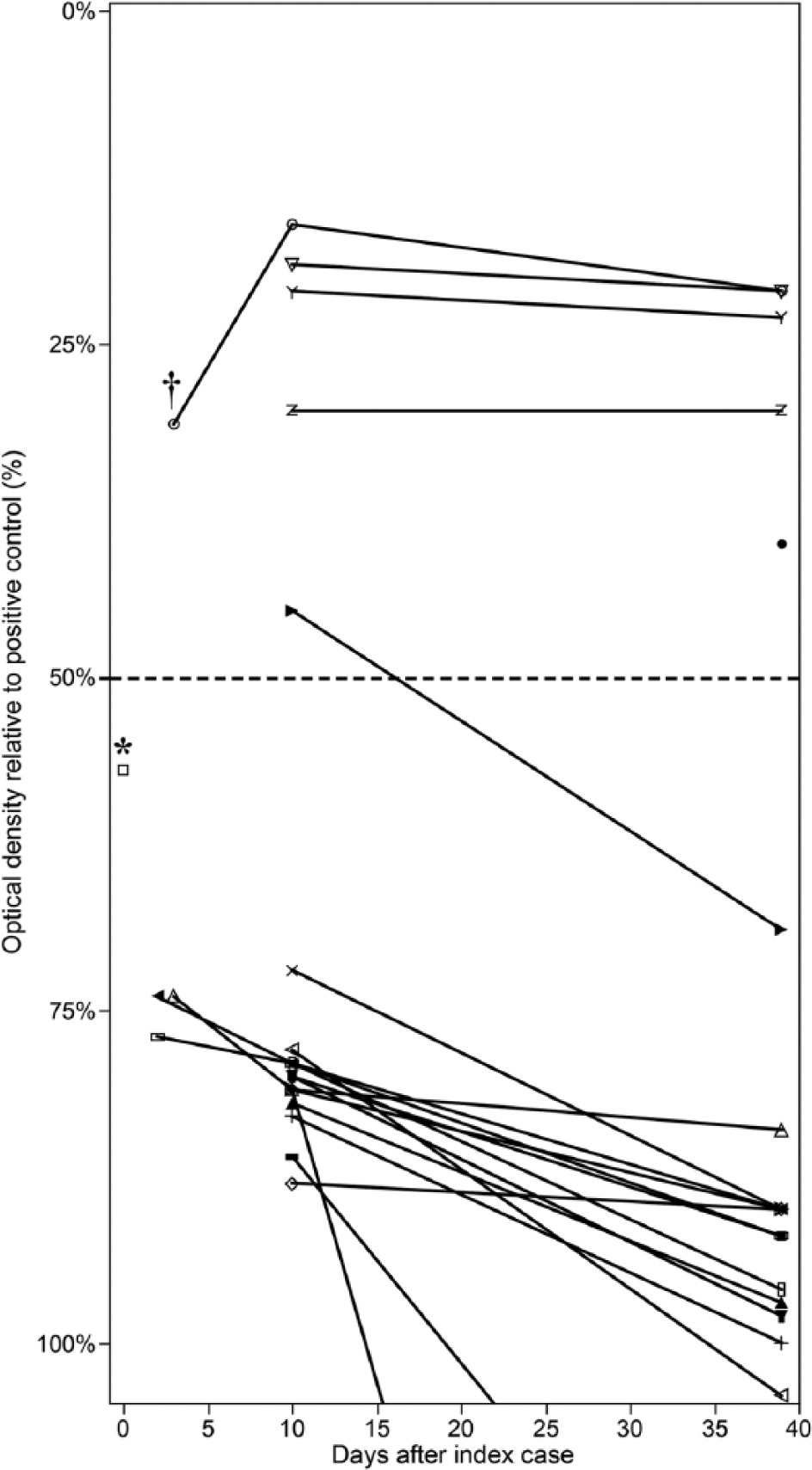
Competitive enzyme-linked immunosorbent assay results from the llama ranch (individual animals signified by a different symbol). Samples resulting in optical densities (ODs) <50% of the mean of the negative controls (dotted line) were considered seropositive. The index case (*) was seronegative at death. Five animals tested positive for Bluetongue virus antibodies over the course of the analysis. One animal seroconverted. Only 1 animal (†) had any detectable rise in antibody OD levels.
Two days after the first llama was presented to WSU VTH, 4 additional llamas from the same farm were presented to WSU because of dyspnea. All 4 were examined, and venous blood was collected for serum chemistry panels, complete blood cell counts, BTV serology, and BTV RNA testing. After several days of observation, all of the llamas appeared healthy, were breathing normally, and were returned to their farm. One of the 4 llamas was seropositive for BTV by cELISA (Fig. 2) with a decreasing OD value between days 3 and 10 post–index case mortality. Experimental inoculation of camelids with BTV has demonstrated peak clinical signs around 5 days postinfection (dpi). 23 Extrapolating this data to our serology results, the rising antibody values between days 3 and 10 postmortality would correspond to approximately 8–18 dpi. Rising antibody levels days 8–18 dpi is consistent with the time period for rising antibody levels in sheep and cattle (7–14 dpi) 7 and South American camelids (6–13 dpi). 23 All 4 of the llamas were negative for BTV RNA on whole blood. The negative RT-qPCR result from the blood of the animal with BTV antibody is consistent with previous observations that viremia is transient, and falls below detectable limits, coinciding with seroconversion. 23 Complete blood cell counts and serum chemistry panels revealed various abnormalities such as hyperglycemia, elevated creatine kinase, neutrophilia, and lymphopenia, in 2 of the 4 animals (data not shown); however, there were no consistent blood parameter abnormalities among the 4 affected llamas. On further epidemiologic investigation, 2 herd mates had died 1 week prior with similar signs of recumbency, anorexia, and dyspnea. Postmortem examinations were not performed, and cause of death is unknown.
An on-farm investigation was performed 10 days after the index case. The farm held 20 llamas ranging in age from 1.5 to 18 years; there were 2 intact males, 5 castrated males, and 13 females. No abnormal findings were found on physical exams of any of the llamas. The llamas were living on 5 acres of sprinkler-irrigated grass pasture with multiple outbuildings for shade and cover. There was also a large pond used as a reservoir for irrigation water. Wet areas were present throughout the pasture; however, there were no large areas of mud and only a minimal amount of mud was present around the edges of the pond. The pond was tested and sprayed for mosquitoes through the summer prior to the animals exhibiting clinical signs. At the time of the investigation, the owners were applying a pyrethrin- and permethrin-based fly spray to every llama daily and had set out several insect traps to reduce the numbers of midges. Blood was drawn from the jugular vein from all 20 llamas for BTV serology at WADDL. Five of the 20 llamas were BTV seropositive. Convalescent sera from 19 of 20 animals were assayed 39 days after the original visit. Seroconversion was compared between the different time points of collection. To further compare antibody levels, the ODs from the cELISA were normalized as a percentage of the positive control (Fig. 2). 7 The results showed that 4 animals remained seropositive, 1 llama seroreverted or possibly had an initial false positive, and no llamas had evidence of decreasing OD values (rising antibody levels) between days 10 and 39. An additional llama, not tested by ELISA in the first round, was tested and was BTV seropositive. The lack of decreasing OD values later in the time course suggests a single infection event within the herd.
In mid-September, after the initial on-farm investigation was completed, we were informed of 3 sheep flocks 70 miles north of the llama operation suspected of having bluetongue. One flock had 3 of 58 total sheep showing clinical signs consistent with bluetongue (swollen ears and or muzzle, sore feet, anorexia) with 1 death loss. A second flock had 3 of 4 sheep with bluetongue signs (sore feet), and a third flock with 1 of 2 sheep showing anorexia and respiratory signs. Whole blood was collected from 4 sheep from the 3 farms and was tested for BTV RNA at WADDL to determine if the llama bluetongue case was of the same serotype as local sheep bluetongue outbreaks. Two of the 4 sheep tested for BTV RNA were positive by RT-qPCR at WADDL. One of these positive samples was submitted for serotyping to NVSL, which revealed serotype 11, matching what was found earlier in the llama case. Sera were collected from the 4 sheep and 12 other sheep and evaluated for BTV antibodies to compare seropositive rates between sheep and camelids. Eight of the 16 (50%) sheep were BTV seropositive, as compared with approximately 25% of the tested camelids being seropositive (Table 1).
Summary of laboratory data for Bluetongue virus in camelids and sheep.*
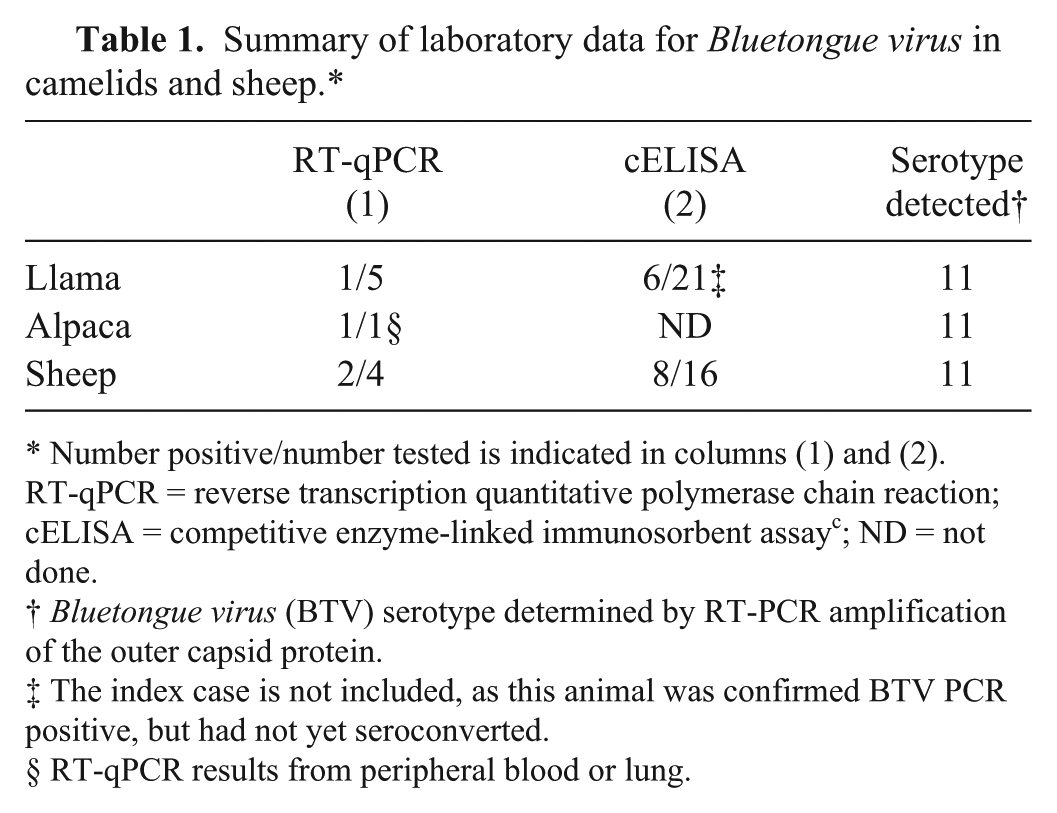
Number positive/number tested is indicated in columns (1) and (2). RT-qPCR = reverse transcription quantitative polymerase chain reaction; cELISA = competitive enzyme-linked immunosorbent assay c ; ND = not done.
Bluetongue virus (BTV) serotype determined by RT-PCR amplification of the outer capsid protein.
The index case is not included, as this animal was confirmed BTV PCR positive, but had not yet seroconverted.
RT-qPCR results from peripheral blood or lung.
Finally, in early October (approximately 1 month after the first confirmed llama BTV infection), postmortem examination was performed by a referring veterinarian on a 2-year-old female alpaca (Vicugna pacos) located approximately 150 miles north of the llama farm. The alpaca was 1 of 2 on the premises, and it had acute clinical signs similar to the index case: recumbency, dyspnea, and death within 12 hr. Representative sections of lungs, tongue, heart, liver, kidney, and diaphragm were immersion fixed, sent to WADDL, and processed for histology as indicated above. Histology was similar to the llama index case, with pulmonary edema and vascular degeneration being the primary lesions. Lung from this animal was positive for BTV by RT-PCR at WADDL, and determined to be BTV-11 by PCR at NVSL.
Later in the season (mid-October), BTV-11 was reported even farther north in 2 sentinel cattle herds in British Columbia, Canada (http://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?page_refer=MapFullEventReport&reportid=14418. Accessed on June 27, 2014). In these 2 herds (total of 451 susceptible animals), no clinical signs were demonstrated. BTV has only been reported 6 times in this area of Canada since originally being detected in 1975.3,5 This suggests that the late summer and early fall of 2013 was relatively active for BTV infection in this area. There is speculation that climate change is associated with a change in epidemiology and distribution of the disease. 16 A 2014 study reports that BTV can overwinter in C. sonorensis 15 suggesting that factors that increase survival of these midges during milder winters could contribute to an increasingly northern distribution of the disease. Continued monitoring in British Columbia is suggested to identify increased rates of northward BTV incursions.
In the cases presented herein, the same serotype that was causing bluetongue outbreaks in sheep was also causing the disease in the camelid herds. BTV-11 has been reported from outbreaks of bluetongue in sheep in the Pacific Northwest in the past,1,20 and was responsible for several of the infections in Canada. 3 It has been speculated that different variants of BTV of the same serotype can be more virulent than others. 13 It is unknown whether this is a different variant of BTV-11 from previous outbreaks.
In the 5 camelid bluetongue cases that reported clinical signs, including 2 from this report, all reported weakness, reluctance to rise or recumbency, open-mouth breathing or respiratory distress, and death within 24 hr of the first observed clinical signs. Three of the 5 cases reported anorexia; 2 of the 5 reported coughing and abnormal lung sounds (stertor or crackles). One of the 5 cases reported abortion, paresis, disorientation, and foaming at the mouth.10,18,19 Other commonly reported signs in sheep such as transient fever and edema of the lips, muzzle, and ears were not reported in these camelid cases.
It is unknown whether bluetongue will become a significant disease in camelids. The disease incidence and sero-prevalence suggest that the BTV disease susceptibility of camelids is more similar to cattle than to sheep. It is also unknown whether the clinical signs reported herein are the result of a novel disease process, or if camelids have had morbidity and mortality from BTV in this area previously and it was not diagnosed or reported. Collectively, this report reminds veterinarians and ranchers that BTV should be included as a differential diagnosis in South American camelids displaying acute dyspnea during BTV season in endemic areas. Diagnosis is often difficult given the nonspecific clinical signs and lack of pathognomonic histologic findings; thus, confirmatory laboratory testing is recommended when there is sufficient suspicion of BTV infection. Further investigation into BTV pathogenesis in camelids is required to answer the questions discussed above.
Footnotes
Acknowledgements
We thank Dr. R. Ross for referring the second BTV case, the sheep ranchers for allowing sampling of their animals, and D. Bradway and the Immunodiagnostic Section at WADDL for performing the molecular diagnostics and serology for BTV and WNV. We are grateful to the National Veterinary Services Laboratories, Ames, Iowa, for confirming and serotyping the Bluetongue virus.
a.
Mini-Beadbeater, Bio Spec Products Inc., Bartlesville, OK.
b.
Ambion MagMax-96 viral RNA isolation kit, Thermo Fisher Scientific, Grand Island, NY.
c.
Bluetongue virus antibody test kit (cELISA), VMRD Inc., Pullman, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
