Abstract
Most small ruminant farms in tropical climates are plagued by Haemonchus contortus, a hematophagous, abomasal parasite. Heavy burdens of this parasite can cause anemia, hypoproteinemia, weight loss, and mortality in susceptible animals. Haemonchus contortus is becoming a major health concern in New World camelids as well, namely llamas (Llama glama) and alpacas (Vicugna pacos), yet little research has been conducted regarding its prevalence or pathology in these species. Herein, we present a retrospective review of llamas and alpacas that were admitted to The University of Georgia Veterinary Teaching Hospital and Athens Diagnostic Laboratory between the years 2002 and 2013. Antemortem fecal egg count (FEC) estimates performed on 30 alpacas were negatively correlated with hematocrit, hemoglobin, and red blood cell count. Total protein was not significantly correlated with FEC. On postmortem examination, 55 of 198 camelids, including 2 from the aforementioned antemortem review, were infected with H. contortus, with llamas (42.6%) having a significantly higher infection rate than alpacas (22.2%). In 15.7% of the total cases, the parasite was the major cause of death. Common gross lesions included peritoneal, thoracic, and pericardial effusions, visceral pallor, subcutaneous edema, and serous atrophy of fat. Histologic lesions included centrilobular hepatic necrosis, hepatic atrophy, lymphoplasmacytic inflammation of the mucosa of the third gastric compartment (C3), extramedullary hematopoiesis in both the liver and spleen, and the presence of nematodes in C3. Our study emphasizes the importance of H. contortus diagnosis and herd monitoring in New World camelids, particularly llamas.
Introduction
Haemonchus contortus is a gastrointestinal nematode that attaches to the abomasum of small ruminants (sheep and goats) and feeds on whole blood.1,4,11,13 Additional blood is lost into the gastric lumen because of mucosal irritation, resulting in clinical anemia. Adult worms are 2–3 cm long and can easily be observed at autopsy. The female worm is identified by the twisting of the white reproductive tract with the red, blood-filled intestinal tract, giving this parasite the nickname “barber pole worm.”
Clinical signs of infection include weight loss, exercise intolerance, submandibular edema (“bottle jaw”), and mucous membrane pallor. 1 Severity of antemortem infection is primarily measured in 2 ways: fecal egg count (FEC) and the FAMACHA system (FAffa MAlan CHArts). In the former, the total number of eggs per gram (EPG) is calculated by counting trichostrongylid eggs, which are ellipsoidal with a thin, smooth wall and contain a morula. 1 The eggs of trichostrongyle nematodes (e.g., genera Haemonchus, Trichostrongylus, Teladorsagia, Ostertagia, Oesophagostomum) are all very similar in size and shape and cannot be accurately differentiated on FEC. A coproculture, polymerase chain reaction (PCR) assay, or lectin staining is required to confirm the presence of Haemonchus spp. antemortem. 3 FECs are typically used in a herd setting to monitor anthelmintic efficacy through the use of FEC reduction tests. They can also be used to diagnose trichostrongylids postmortem, if no worms are noted grossly at autopsy. The FAMACHA system is an inexpensive, rapid method used to categorize the level of antemortem anemia based on conjunctival mucous membrane color.13,18 The chart contains a color scale of 1–5, with 1 being red and 5 pale white. The chart is held next to the conjunctiva, and a score is assigned based on the closest matching color (Fig. 1).
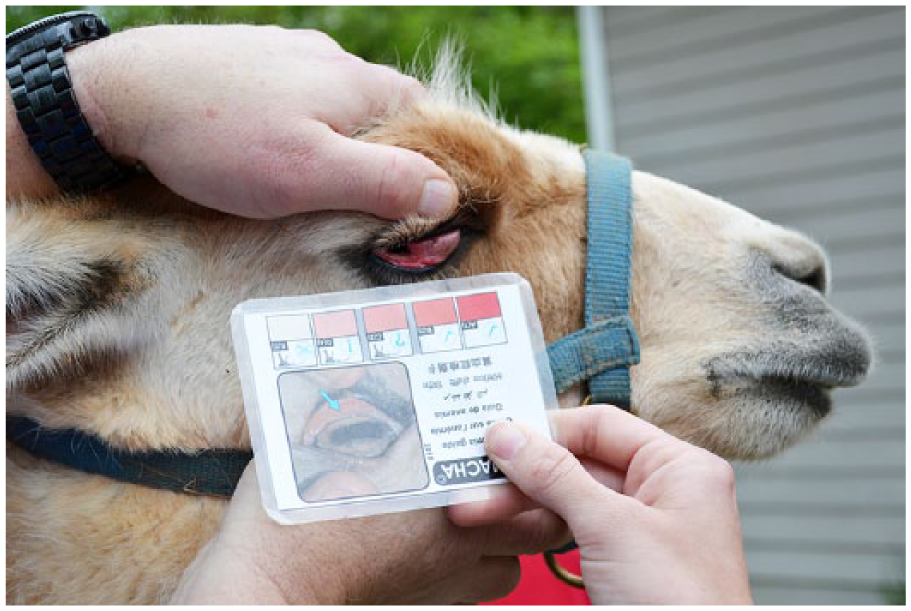
Demonstration of the FAMACHA (FAffa MAlan CHArts) technique in a llama (Llama glama).
The small ruminant industry is a major sector of livestock agriculture with ~2 billion sheep and goats worldwide. 5 Developing countries depend heavily on small ruminants for sustenance and income; therefore, H. contortus is considered a major economic problem for small ruminant farms worldwide, particularly those in warm, moist environments where the parasite thrives. Prevalence can reach as high as 80% in the summer, 4 and in severe infections, lambs can lose an estimated one-fifth of their circulating red blood cell mass volume in a single day.7,8
Like goats and sheep, camelid species (henceforth referring to llamas and alpacas only) are susceptible to H. contortus.9,12,15 Furthermore, North American camelids are commonly co-grazed with small ruminants, increasing the likelihood of infection.10,12,15,17 In camelids, H. contortus resides in the third gastric compartment (C3) and has become a major health issue for camelid farms in the southeastern United States. 9 H. contortus has been well studied in small ruminants, but primary literature on this infection in New World camelids remains sparse. As camelids are becoming more popular in the United States, parasite control should be emphasized. The objective of our review was to characterize the clinical and anatomic pathology of H. contortus infection in llamas and alpacas in the southeastern United States, in order to determine whether disease differed from that observed in small ruminants.
Materials and methods
Antemortem study selection
Medical records of adult camelids presented to The University of Georgia Veterinary Teaching Hospital (Athens, Georgia) from April 2003 to April 2013 were reviewed, representing 13 counties in northern Georgia, 4 in South Carolina, and 1 in North Carolina. To be selected for the study, the patient must have had fecal analysis and a complete blood cell (CBC) count, biochemical profile, or limited hematologic and biochemical assessment from a commercial analyzer a performed during their hospital stay (n = 30). CBC counts were performed using various commercial systems at 2 different time periods (2003–January 2007 b and January 2007–2013c,d). Biochemical profiles were analyzed using 2 commercial systems at 2 different time periods (2003–June 2010 e and June 2010–2013 f ). Fecal analysis was achieved using a modified McMaster test to search for parasite eggs.
Postmortem study selection
Final autopsy reports, including gross and histologic descriptions and associated parasitology reports, of all camelids >3 weeks old that were received at the Athens Diagnostic Laboratory (Athens, GA) for postmortem evaluation from January 2002 to April 2013 (n = 198) were reviewed, including 144 alpacas and 54 llamas, 2 of which were also included in the antemortem study. Cases had been received from 22 north Georgia counties, 4 South Carolina counties, and 1 county in North Carolina. Autopsy samples had been fixed in 10% neutral buffered formalin, paraffin embedded, and routinely stained with hematoxylin and eosin.
Statistical analyses
Camelids with clinical pathology data was divided into anemic (n = 21) and nonanemic (n = 9) categories. The anemic category was composed of patients with a red blood cell (RBC) mass below the established alpaca reference interval for the laboratory analyzer c (<10.2 × 106 cells/μl, unpublished data) or below previously published llama reference intervals (<10.1 × 106 cells/μl). 6 FECs of anemic and nonanemic animals were compared using Student t-tests. Pearson correlation analysis was used to test for correlations of FEC and the following hematologic parameters: hematocrit (HCT), RBC count, hemoglobin (HGB), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and total protein (TP). Antemortem statistical analyses were completed using commercial software. g A binomial proportion test was run to compare the number of llamas versus alpacas that were diagnosed as infected on postmortem examination. Additionally, Pearson chi-square, Fisher exact, and binomial proportion tests were used to look for an association between sex and postmortem diagnosis of haemonchosis among camelids and also within alpaca and llama species groups. Postmortem statistical analyses were executed in the program R (version 2.15.1, https://CRAN.R-project.org). The level of significance was set at p ≤ 0.05.
Results
Antemortem clinicopathology
Average erythron parameters, TP, and FECs are reported in Table 1. Student t-tests revealed that FEC estimates were significantly higher in anemic animals than nonanemic animals ( p = 0.0143). Anemic animals had an average FEC estimate of 441 ± 591.1, whereas nonanemic animals had an average FEC estimate of 85 ± 113.9. Pearson correlation analysis revealed that FEC estimates were negatively correlated with HCT (r = −0.75, p < 0.0001), RBC (r = −0.67, p = 0.0002), and HGB (r = −0.76, p < 0.0001). FEC was not significantly correlated with MCV, MCH, MCHC, or TP ( p = 0.1552, 0.5319, 0.7909, and 0.9418, respectively).
Clinicopathologic values for camelids at The University of Georgia Veterinary Teaching Hospital (Athens, Georgia) with Haemonchus contortus infections between the years 2002–2013.*
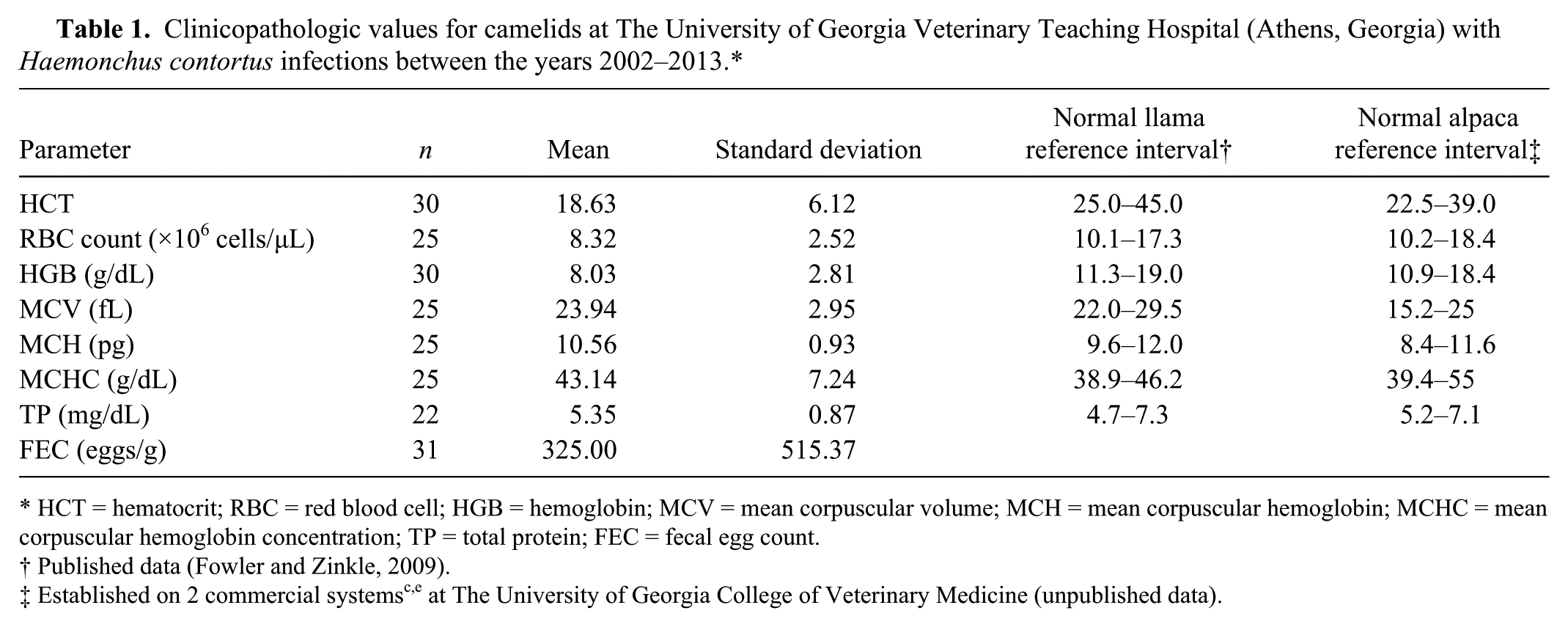
HCT = hematocrit; RBC = red blood cell; HGB = hemoglobin; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; TP = total protein; FEC = fecal egg count.
Published data (Fowler and Zinkle, 2009).
Postmortem study
Of the 198 camelids that were autopsied (144 alpacas, 54 llamas), 55 showed evidence of H. contortus infections (32 alpacas, 23 llamas), either through the presence of nematodes within C3 (Fig. 2) or trichostrongylid eggs in the feces. The rate of infection was significantly higher in llamas (42.6%) than alpacas (22.2%; p = 0.004). Of all camelids, 28.4% of females and 26.7% of males were infected. There was no association between sex and infection among camelids as a group ( p = 0.913), nor among individual alpaca ( p = 0.391) and llama groups ( p = 0.695). Infected animals ranged from 2.5 months to 17 years old. H. contortus was considered the major cause of death in 31 cases (17 alpacas, 14 llamas) as a result of a significant worm burden, FEC, and the lack of other diseases. These animals were categorized as “severely infected” (Table 2). In 8 of the H. contortus–infected cases (14.5%), H. contortus nematodes were identified; however, another major cause of death was designated at autopsy. In these cases, H. contortus was often considered a key secondary player, and these cases were categorized as “moderate.” The final 16 cases of H. contortus infections (29.1%) were incidental findings characterized by the presence of low numbers of eggs on fecal flotation. These cases were categorized as “mildly infected.” Other frequent causes of death in adult camelids included infection with Parelaphostrongylus tenuis, hepatic lipidosis, trauma, and gastrointestinal perforation secondary to ulceration.
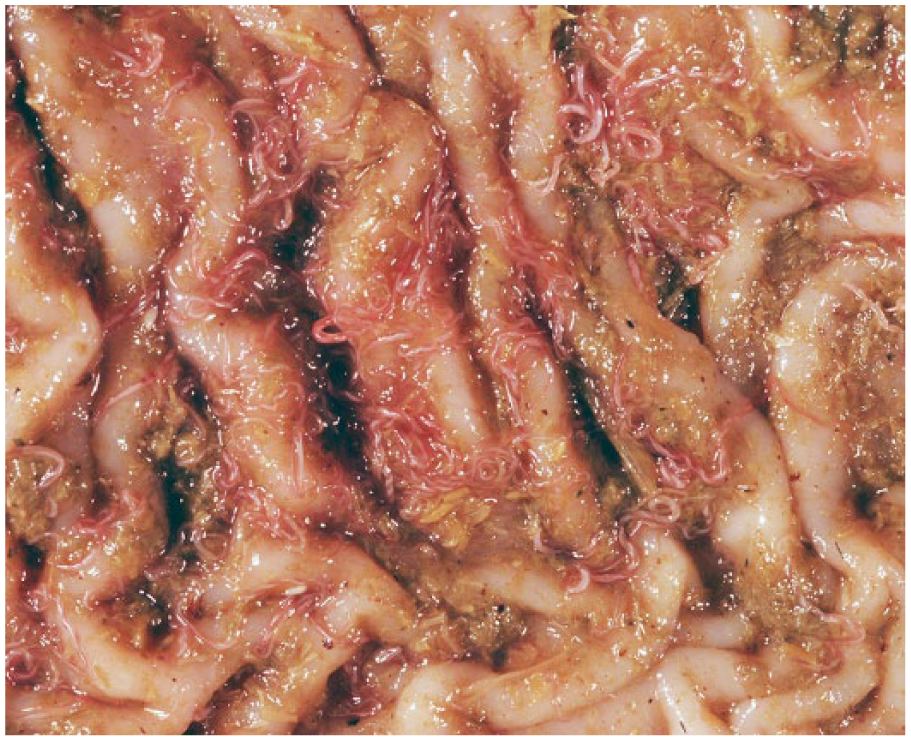
Haemonchus spp. nematodes in the third gastric compartment of a llama (Llama glama). Females are identified by the twisted “barber pole” appearance. Photo by Dr. Corrie Brown.
Prevalence of Haemonchus contortus infections on postmortem examination of camelid species.*
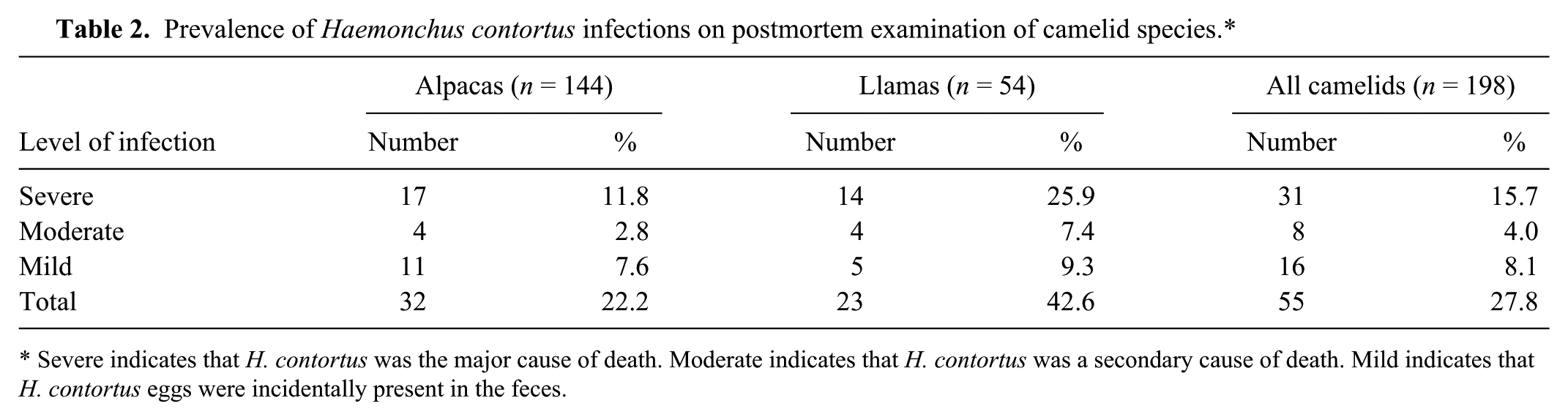
Severe indicates that H. contortus was the major cause of death. Moderate indicates that H. contortus was a secondary cause of death. Mild indicates that H. contortus eggs were incidentally present in the feces.
On external examination, H. contortus–infected animals were frequently in poor body condition and had pale mucous membranes. Gross anatomic lesions were generally more striking in the severely infected group. At autopsy, on opening the carcasses, prominent findings included subcutaneous edema and decreased viscosity of blood. The edema was most commonly located in the intermandibular area, with fewer cases having ventral edema. Abdominal, thoracic, and/or pericardial effusions were common. Overall, individuals had serous atrophy of fat and muscular and visceral pallor, particularly in the liver. If present, H. contortus nematodes were identified on the C3 mucosa and also within the C3 ingesta. The C3 mucosa was sometimes reddened, but generally there were no other gross mucosal lesions.
Samples were evaluated histologically for severely and moderately infected cases, excluding those tissues that were too autolyzed for histologic evaluation. Common histologic findings in the liver included hepatocellular atrophy, centrilobular necrosis, and extramedullary hematopoiesis (commonly present in the spleen). There was often lymphoplasmacytic inflammation of the lamina propria in C3. H. contortus larvae were deep within the C3 mucosa of 6 severely infected cases and were characterized by a thin-walled digestive tract and longitudinal cuticular ridges (Fig. 3).
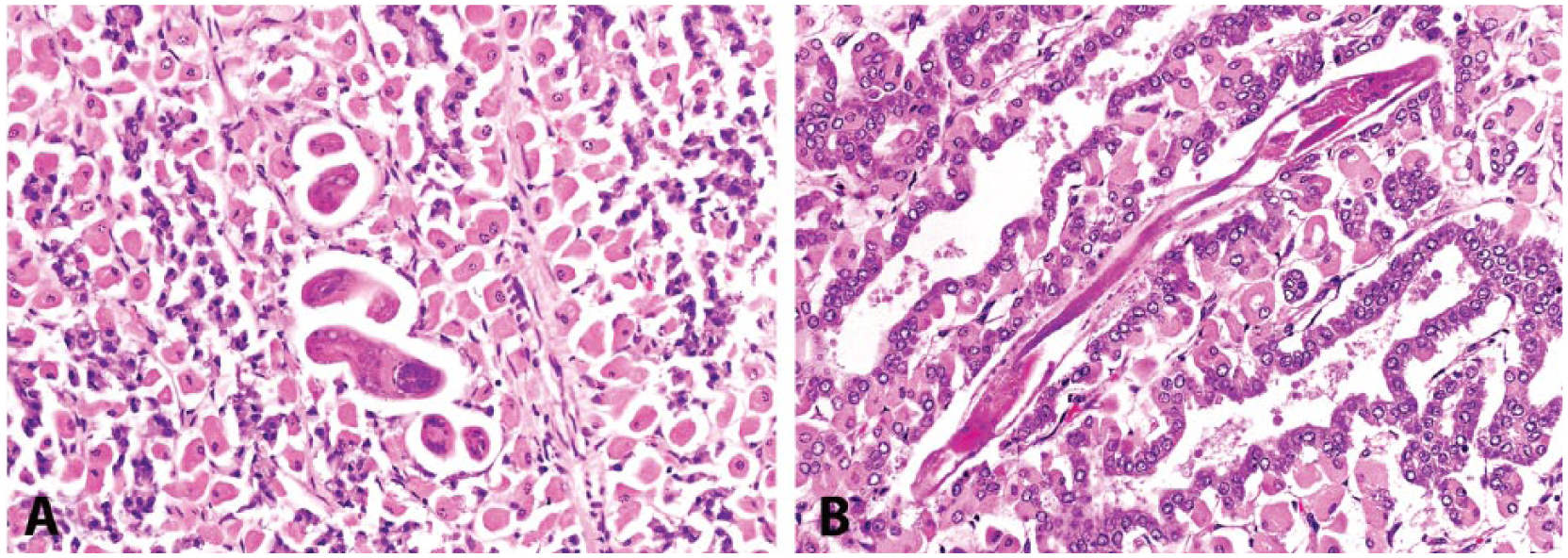
Cross-sectional (
Discussion
Our review confirms the high prevalence of H. contortus infections in New World camelids. The occurrence of infection, however, is likely higher than this review represents because of the rapid decomposition of nematodes postmortem. Autopsies should ideally be performed as quickly as possible after death, yet many camelids are not brought to the diagnostic laboratory in a timely manner, either because of delays in discovery of the carcass or in transport. Additionally, as a result of the retrospective nature of this review, a FEC was not collected on every autopsied animal, again lowering the level of H. contortus detection.
The finding that llamas have higher infection rates than alpacas is interesting. One possible reason for this difference could be differences in immunity and stress levels between the 2 species. It is possible that the llamas in our study were either exposed to more stressful scenarios, such as show events or other travel, or that they had a lower capacity to handle these stressful scenarios; either scenarios could lead to immunosuppression and exacerbation of infection. Additionally, alpacas are generally more highly regarded as pets and better cared for than llamas. As a result, owner care and dewormer administration could be a reason for variations in the degree of parasitic infection. A clinical comparison study is indicated to determine if there are differing rates of infection in living camelids under the same living and treatment conditions.
As expected, anemic animals were more likely to have a higher FEC, and FEC was negatively correlated with anemia parameters including HCT, RBC, and HGB. Interestingly, however, TP was not negatively correlated with FEC. H. contortus adults feed on whole blood, so TP and HCT would both be expected to decrease. Although clinical dehydration may elevate both HCT and TP, additional factors may have affected TP and prevented a clear correlation with FEC. Inflammation, protein-losing enteropathies and nephropathies, as well as liver disease, could all have altered the TP concentration. Comparison studies of TP in H. contortus–infected small ruminants would be useful. Although studies have shown a decreasing TP associated with H. contortus infection in sheep and goats, values have not been compared to corresponding FECs.2,14,16 Even though TP was not significantly correlated with FEC, infected individuals were debilitated by both anemia and hypoproteinemia, to which the majority of the gross and histological findings could be attributed.
Because of its retrospective design, our study presented many challenges. Sample size was limited due to the requirement of having a CBC count, chemistry, and FEC performed during the same hospital visit. Future prospective studies would allow for greater consistency and sample size. Regardless, the highlights of this review include how important hematologic and pathologic effects of H. contortus in camelid species can be. H. contortus should be a top differential for recumbent camelids. Finally, clinicians and producers should monitor camelids showing poor body condition or exhibiting signs of weakness, pale mucous membranes, or dependent edema. Routinely monitoring camelids with FAMACHA checks and FEC should aid in diagnosing and preventing loss caused by parasitism.
Footnotes
Acknowledgements
We thank Drs. Deborah Keys and Nicole Gottdenker for their assistance with statistical analyses and Dr. Corrie Brown for providing the photograph of H. contortus in situ. Thank you to Dr. Wilson Yau for technical assistance with the photomicrographs.
Authors’ note
Information regarding research materials can be acquired by contacting the corresponding author.
Authors’ contributions
EE Edwards contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. BC Garner contributed to analysis and interpretation of data, and critically revised the manuscript. LH Williamson contributed to acquisition of data, and critically revised the manuscript. BE Storey contributed to acquisition and interpretation of data, and critically revised the manuscript. K Sakamoto contributed to conception and design of the study; contributed to analysis and interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
NOVA CCX stat analyzer, Nova Biomedical, Waltham, MA.
b.
Cell-Dyn 3500, Abbott Diagnostics, Lake Forest, IL.
c.
ADVIA 120 hematology system, Siemens Medical Solutions USA, Malvern, PA.
d.
HemaTrue analyzer, Heska, Loveland, CO.
e.
Hitachi 912, Roche Diagnostics, Indianapolis, IN.
f.
Roche P module, Roche Diagnostics, Indianapolis, IN.
g.
SAS version 9, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
