Abstract
In females, the paramesonephric (syn: Müllerian) duct gives rise to the uterine tubes, uterus, cervix, and part of the vagina. Segmental uterine aplasia resulting from a paramesonephric duct abnormality has been reported in a range of species including bovids, canids, felids, equids, camelids, and lagomorphs. Here we document segmental aplasia of the left paramesonephric duct in a New Zealand white rabbit. The proximal 70 mm of the left uterine tube was present and terminated in adipose tissue. A 10 × 2 × 1-mm tag of cream tissue was present and was composed of sheets of adipose tissue and streams of smooth muscle, but otherwise, there was no evidence of the left uterine horn, supporting a diagnosis of unilateral uterine aplasia (uterus unicornis) analogous to a human class II (unicornuate uterus) lesion of the “no horn” subtype. In addition, our case had a concurrent uterine tube fimbrial cyst, minor cysts in the left kidney, and mammary gland hyperplasia with secretory activity. We suggest the adoption of a uniform classification system specifically for lagomorph uterine anomalies. Large-scale multi-center studies documenting prevalence of such lesions would facilitate identification of trends in laterality and other factors.
Uterine disorders, including endometrial hyperplasia and uterine neoplasia, arise in female lagomorphs with relatively high prevalence.2,13,25 In females, the paramesonephric duct arises as a longitudinal invagination of the craniolateral urogenital ridge epithelium and gives rise to the uterine tubes, uterus, cervix, and part of the vagina.9,20 Segmental uterine aplasia resulting from a paramesonephric duct abnormality has been reported in a range of species, including bovids, 16 canids, 15 felids,5,15 equids, 17 camelids, 8 and lagomorphs.3,22,23 Here we document segmental aplasia of the left paramesonephric duct in a New Zealand white rabbit with accompanying histology.
A 195-d-old female New Zealand white rabbit was euthanized at the end of a surgical study involving left forelimb surgery and subsequent imaging. The study followed local ethical committee and UK Home Office regulations. No abnormalities were detected during the study period. Immediately following euthanasia, the group of animals to which this rabbit belonged each underwent a routine postmortem examination, primarily for collection of mammary tissue for other studies, and to screen for underlying lesions that might influence mammary development. In our case, the fimbria of the left uterine tube had a 5-mm fluid-filled cavitation. The proximal 70 mm of the left uterine tube was present and terminated in adipose tissue. A 10 × 2 × 1-mm tag of cream tissue was present within the adipose tissue, but the left uterine horn and cervix were absent (Fig. 1). The left kidney had rare, ~0.5-mm, dark-gray, cavitated, cortical foci. No other abnormalities were detected macroscopically.
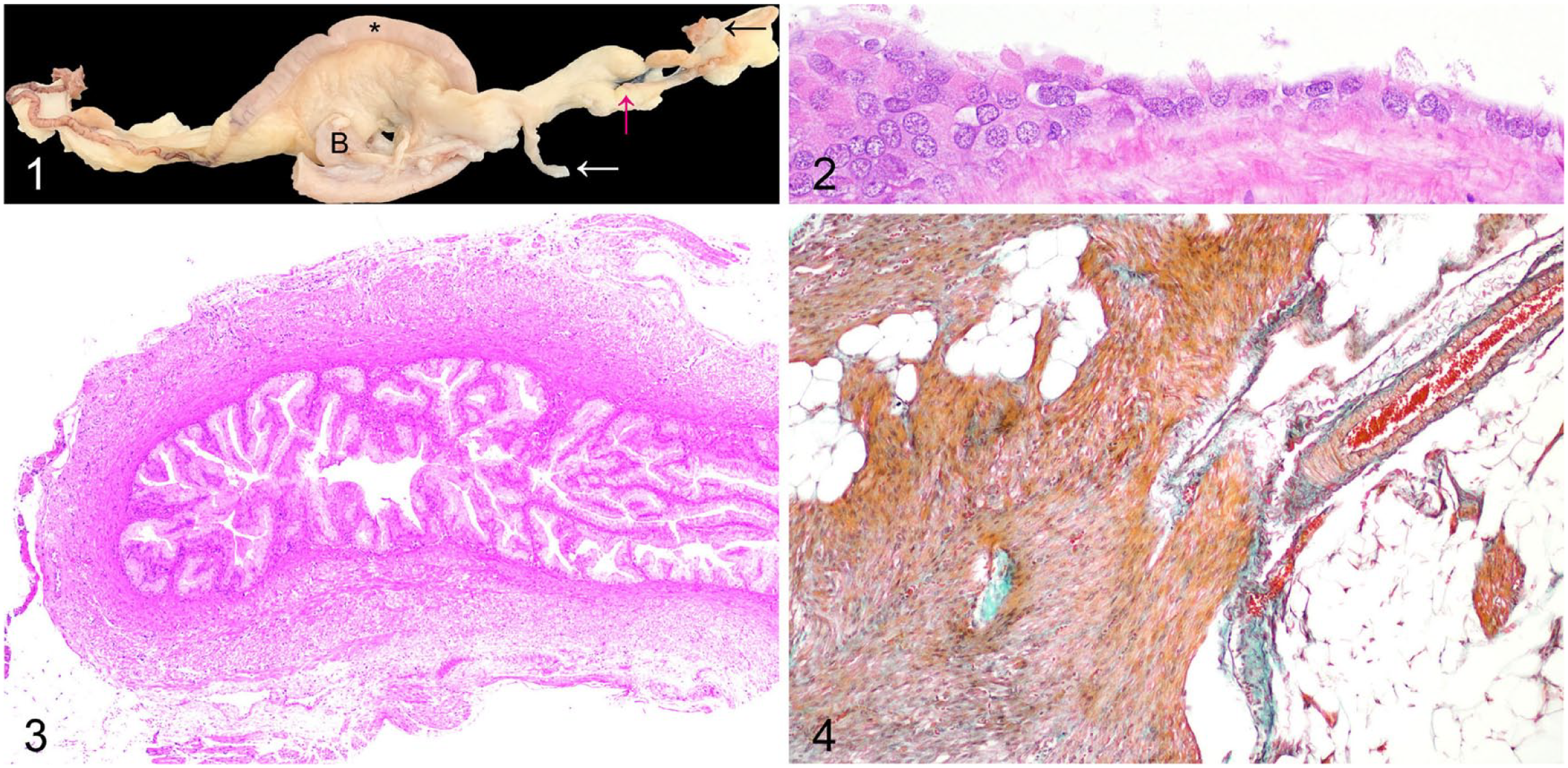
Segmental aplasia of the paramesonephric duct and fimbrial cyst in a New Zealand white rabbit. Figure 1. The fimbria of the left uterine tube has a 5-mm fluid-filled cavitation (black arrow). The proximal 70 mm of the left uterine tube terminates in adipose tissue (magenta arrow). A 10 × 2 × 1-mm tag of cream tissue is present within the adipose tissue (white arrow). The left uterine horn and cervix are absent. * = right uterine horn; B = urinary bladder. Figure 2. The fimbrial cyst is lined by ciliated pseudostratified columnar-to-attenuated cuboidal epithelium. H&E. Figure 3. The left uterine tube is blind ending. Section from level of the magenta arrow in Figure 1. H&E. Figure 4. The tag of tissue (white arrow in Fig. 1) is composed predominantly of sheets of adipose tissue and streams of smooth muscle cells. Masson trichrome.
Tissues were fixed for at least 24 h in 10% neutral-buffered formalin, processed routinely, sectioned at 4 µm, and stained with H&E. Histologic examination revealed that the fimbrial fluid-filled cavitation was lined by a ciliated pseudostratified columnar-to-attenuated cuboidal epithelium (Fig. 2). The blind-ending pouch of the left uterine tube was lined by ciliated columnar epithelium, with rare lymphocytes present in subepithelial foci. This uterine tube terminated in adipose tissue (Fig. 3). The tag of cream tissue in the expected location of the left uterine horn was composed of sheets of adipose tissue and streams of smooth muscle cells (Fig. 4) and expressed alpha–smooth muscle actin by immunohistochemistry (data not shown). The contralateral uterine horn was histologically normal. The ovaries had bilateral corpora lutea. The rare dark-gray cavitated foci in the cortex of the left kidney were histologically consistent with epithelial cysts. The mammary gland had numerous ectatic duct profiles that frequently contained large amounts of eosinophilic proteinaceous material together with multifocal lobuloalveolar hyperplasia with multifocal secretory activity (Fig. 5). 4 Our final diagnoses were segmental aplasia of the left paramesonephric duct leading to unilateral uterine aplasia (uterus unicornis) with co-occurrence of a uterine tube fimbrial cyst, minor cortical cysts of the left kidney, and mammary gland hyperplasia with secretory activity.
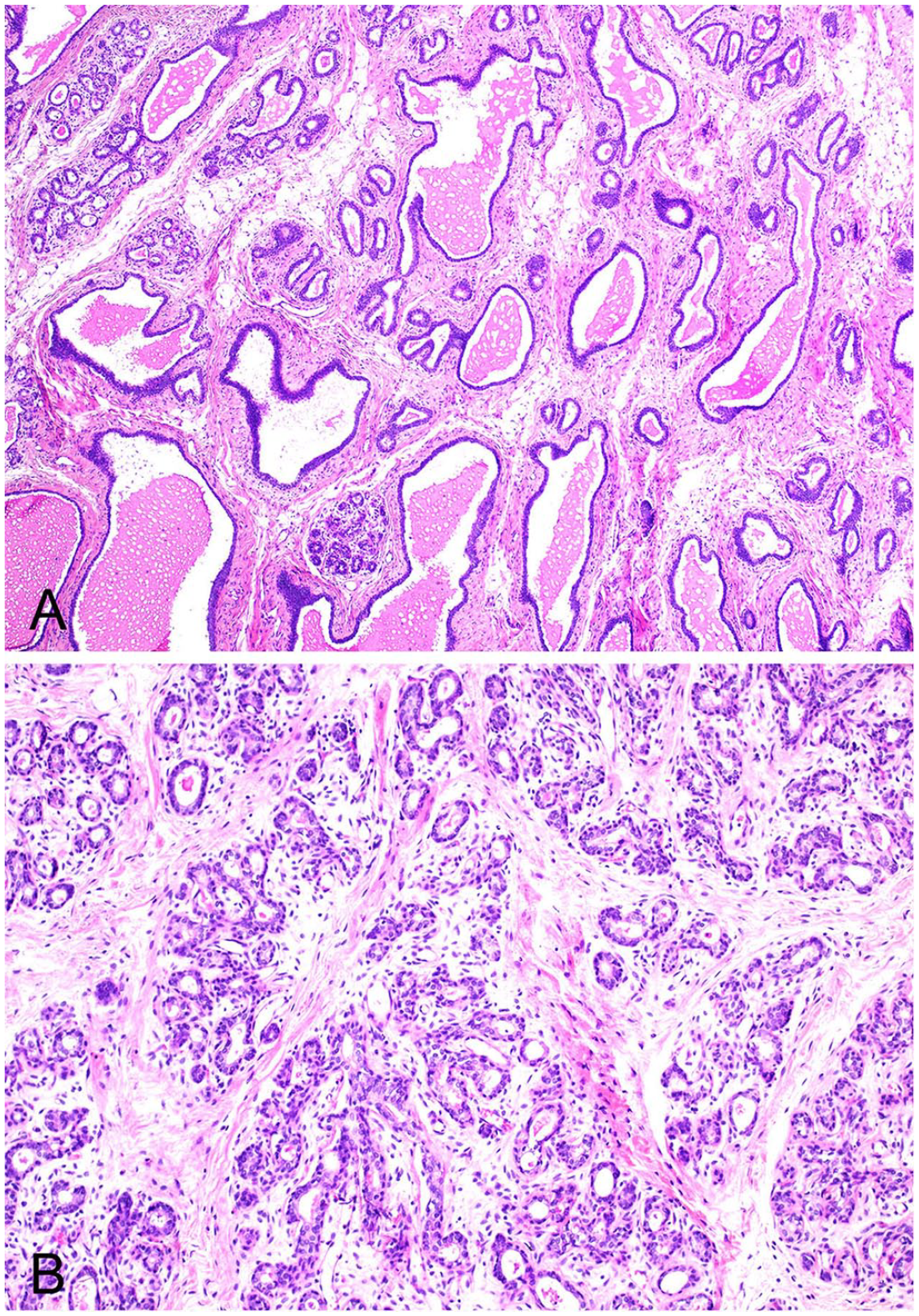
Mammary gland in a New Zealand white rabbit.
A 2022 review article emphasized the need to apply consistent terminology to failures of development of the reproductive tract. Using that diagnostic framework, our case was inferred to exhibit an XX ovarian disorder of sexual development and a female phenotype.9,21 In the medical literature, disorders of sexual development are now described as differences of sexual development. 11
During embryonic development, the paramesonephric (syn: Müllerian) ducts, derived from intermediate mesoderm, originate from coelomic epithelial invaginations of the urogenital ridges lateral to the mesonephros. The paramesonephric ducts run in a cranial-to-caudal direction and give rise to uterine tubes, uterus, cervix, and the cranial vagina through 3 processes: organogenesis, unification (fusion), and canalization (septal resorption).8,9,20,24
In humans, classification systems have been developed to describe anomalies of Müllerian duct development. In 1988, the American Fertility Society produced an essentially pictorial 7 criterion classification system that was adopted widely.1,7,24 In 2021, the American Society for Reproductive Medicine presented a new classification system, partly in response to concerns that the previous system did not adequately categorize vaginal and cervical anomalies, and was not easily applicable to complex anomalies. 19 In the case of partial uterine agenesis, this classification system includes consideration of whether functional endometrium is present. It is important to note that rabbits have a duplex uterus, 10 and therefore neither of the human systems are fully applicable to rabbits given their divergent anatomy.
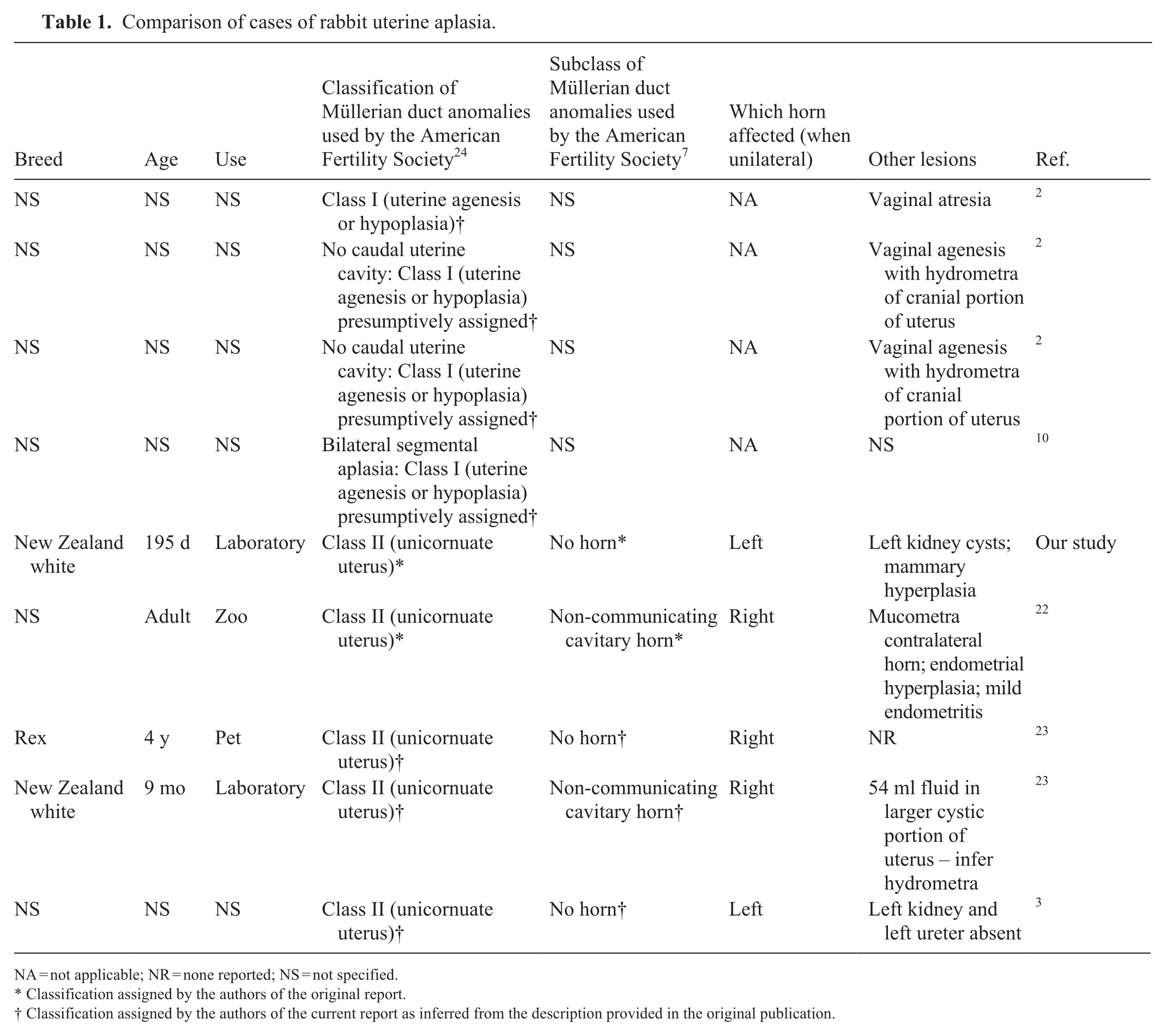
Based on the data available regarding published cases of lagomorph segmental uterine aplasia, we found it easier to attempt to apply the 1988 system to rabbit cases of uterine aplasia. 1 Based on this scheme, our case would appear to be most synonymous with a human class II unicornuate uterus anomaly. Class II lesions can be further subdivided into 4 groups: “communicating cavitary horn,” “non-communicating cavitary horn,” “non-communicating non-cavitary horn,” and “no horn.” 1 Our case would therefore be most fully classified as a class II (unicornuate uterus) “no horn” lesion. The presence of a very small remnant of smooth muscle raised the possibility of diagnosis of a “non-communicating, non-cavitary horn,” but given that the smooth muscle remnant was a small tag of tissue only, a diagnosis of “no horn” more accurately described the macroscopic lesion. Previous cases of class II segmental uterine aplasia in rabbits have either been classified as the “no horn” subtype or the “non-communicating cavitary horn” subtype (Table 1). It is of note that hydrometra or mucometra may be a sequela of the latter,22,23 and that this associated lesion has been recorded in other species with a unicornuate uterus.8,15 Interestingly, literature documenting segmental uterine aplasia in the cow, mare, and queen suggests that, depending on the extent of the lesions, viable pregnancies may be maintained in the contralateral uterine horn.5,6,16
Comparison of cases of rabbit uterine aplasia.
NA = not applicable; NR = none reported; NS = not specified.
Classification assigned by the authors of the original report.
Classification assigned by the authors of the current report as inferred from the description provided in the original publication.
It has been suggested by authors working with other species that there may be laterality in the prevalence of segmental uterine aplasia in some species, 8 but it is important to interpret datasets suggesting such associations with caution, particularly when case numbers are modest. In the case of lagomorphs, there are insufficient data to draw any conclusions as to whether laterality of segmental uterine aplasia exists (Table 1).
A historic case of left uterine horn segmental aplasia in a rabbit documented concurrent absence of the left kidney and left ureter. 3 In our case, the left kidney exhibited minor renal cysts, a common finding in laboratory rabbits that was not considered associated with the uterine lesion. 4
In our case, the fimbrial cyst of the uterine tube was also not necessarily considered to be related to the uterine lesion. Oviduct cysts are common in laboratory rabbits, 4 and paraovarian cysts located in the mesosalpinx have also been described in pet rabbits. 2
Our case had a notable degree of mammary gland hyperplasia with secretory activity. This was not considered likely to be related directly to the experimental protocol given that other rabbits in the same cohort did not have this lesion. Mammary gland hyperplasia with secretory activity has been attributed to pseudopregnancy or elevated serum prolactin levels resulting from either prolactin-producing pituitary adenomas or cyclosporine A administration.4,14,18 Rabbits are induced ovulators, with pseudopregnancy potentially resulting from stimuli other than breeding. 12 Pseudopregnancy seems a likely pathogenesis for the mammary hyperplasia in this individual. Furthermore, bilateral ovarian corpora lutea were detected histologically.
Our case of segmental aplasia of the paramesonephric duct resulting in a unicornuate uterus with no horn, with small remnants of presumed uterine smooth muscle remaining, also illustrates the co-occurrence of a fimbrial cyst in the proximal portion of the uterine tube. The current “International Harmonization of Nomenclature and Diagnostic Criteria (INHAND): Nonproliferative and Proliferative Lesions of the Rabbit” 4 is based on lesions that have been described or observed in laboratory rabbits, and congenital hypoplasia of the uterus and cervix is documented as “not observed but potentially relevant.” Our report confirms the occurrence of segmental aplasia of the left paramesonephric duct leading to unilateral uterine aplasia in a New Zealand white laboratory rabbit. We suggest that it would be beneficial to adopt a uniform classification system specifically for lagomorph uterine anomalies, and that adopting the elements of the 1988 American Fertility Society framework 1 is appropriate for this purpose, as demonstrated in Table 1. Large-scale multi-center studies documenting prevalence of such lesions would facilitate interrogation of the possibility of laterality in the prevalence of segmental uterine aplasia in rabbits, and identification of other trends such as the overrepresentation of specific breeds, which may exist but are currently unrecognized.
Footnotes
Acknowledgements
We thank Prof. Matthew Allen who provided this animal for postmortem examination and tissue collection at the end of the experimental protocol. We also acknowledge the expertise and care of the University of Cambridge Biomedical Services staff who are responsible for the husbandry of research animals. We thank Paul Crompton, Yvonne Pratt, and Emma Ward of the Department of Veterinary Medicine, University of Cambridge, for excellent technical expertise in the preparation of tissue sections.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Benjamin P. Davies is funded by an Anatomical Society PhD Studentship awarded to Katherine Hughes. Sara Hassouna Elsayed is funded by the Newton Mosharafa Fund from the British Council in liaison with the Egyptian Ministry of Higher Education and Scientific Research, the Cambridge Commonwealth, European and International Trust (MOHE Egypt Cambridge Scholarship), and the Cambridge University Postgraduate Research Student Covid-19 Assistance Scheme.
