Abstract
Viral nervous necrosis (viral encephalopathy and retinopathy) is caused by piscine nodavirus (Nodaviridae, Betanodavirus). Since 1986, this highly infectious virus has caused mass mortalities of up to 100% in farmed saltwater and freshwater fish around the world (with the exception of South America and Antarctica), affecting >60 species across 10 orders. The Atlantic blue marlin (Makaira nigricans Lacépède, 1802) is a top-level predator found throughout the tropical waters of the Atlantic and Indo-Pacific oceans. Despite their popularity as a sportfish, relatively little is known about the Atlantic blue marlin and other billfish. We describe here chronic betanodavirus infection in a juvenile Atlantic blue marlin, which is, to our knowledge, the first report of disease in M. nigricans.
Betanodaviruses are 25–30-nm diameter, icosahedral, non-enveloped, single-stranded, positive-sense RNA viruses with a genome consisting of 2 genes: RNA1 encoding for RNA-dependent RNA polymerase, and RNA2 encoding the coat protein. 2 First reported in Japan in 1986, the highly infectious piscine nodavirus has caused mass mortalities of up to 100% in farmed saltwater and freshwater fish around the world (with the exception of South America and Antarctica), affecting >60 species across 10 orders. Fish in aquaculture facilities have been shown to be infected subclinically. 1 Piscine nodavirus has also been detected subclinically in >120 species of wild marine fish across 19 orders, and implicated in mortality events of wild sea bass and grouper in the Mediterranean. 1 Additionally, piscine nodavirus has been detected in several invertebrate species, including bivalves, octopus, and squid (Korean nervous necrosis virus), none of which had clinical disease, and were considered carriers. 1
Four exemplar isolates of species of betanodavirus have been identified, named based on the fish species in which they were initially detected: Redspotted grouper nervous necrosis virus (RGNNV), Barfin flounder nervous necrosis virus, Striped jack nervous necrosis virus, and Tiger puffer nervous necrosis virus.1,2,5,7–9,12 A fifth species, turbot nodavirus, was proposed in 2004 and is now widely accepted as a fifth genotype. 8 Other proposed species are the Atlantic cod nervous necrosis virus 1 and Korean shellfish nervous necrosis virus. 1 The strains have differing geographic and optimal temperature ranges that play a major role in host susceptibility. RGNNV is the most common and widely distributed genotype, infecting fishes that inhabit tropical and temperate (25–30°C) waters.1,2 RGNNV is the only genotype able to cause disease in freshwater species and has been detected in persistent infections. 1
Acute disease and high mortalities are most common in larvae and juvenile fish; however, clinical disease with high mortality has been documented in adults of several species.1,9,12 Although gross lesions are not typical, clinical manifestations of acute disease include neurologic signs such as tremors and abnormal swimming behaviors (whirling, corkscrewing, vertical floating), as well as darkened skin pigmentation, inappetence, skin and fin erosions, eye lesions, and self-isolation.1,2,5,7,8,12 Hyperinflated swim bladders have also been reported. 4 Piscine nodavirus primarily targets the neurons of the CNS, especially in the retina and brain, resulting in clinical neurologic signs.1,2,5,7,8,12 In acutely affected fish, cytoplasmic vacuolation and necrosis, with diffuse granulocytic inflammation, are seen in the gray matter of the brain and the retina. Generally, in more chronic infections and in surviving fish, vacuolation and neuronal necrosis are replaced by granulomatous inflammation with nodules of concentrically arranged macrophages.1,2,5,7,8 Some species-specific variations in histologic lesion progression have been observed.1,2,5,7,8
The Atlantic blue marlin (Makaira nigricans Lacépède, 1802) is a top-level predator found throughout the tropical waters of the Atlantic and Indo-Pacific oceans. They are large solitary animals with few predators, including humans and shortfin mako (Isurus oxyrinchus) and great white (Carcharodon carcharias) sharks. Atlantic blue marlins are valued for their meat throughout the world, and the population is under intense pressure from longline fishing and bycatch from commercial tuna fisheries. Given these reasons, in addition to the reduction of prey species and climate change, along with their considerable generational time (9 y), age of maturity (4 y), and long lifespan (20–25 y), they are considered threatened by the International Union for Conservation of Nature. 3 Despite being a charismatic and valuable sportfish, relatively little is known about the Atlantic blue marlin and other billfish. We report here a chronic betanodavirus infection in a juvenile Atlantic blue marlin. To our knowledge, no reports of disease in M. nigricans have been published given that we found no results from multiple internet databases and search engines including Google, PubMed, and Web of Science, using the following search terms: marlin, billfish, and/or Makaira, in combination with pathology, disease, and/or betanodavirus.
A juvenile male Atlantic blue marlin in good body condition with mild postmortem autolysis was found dead on the beach at Grand Isle, Louisiana, in 2011. Because this was an extremely rare occurrence, and the animal was in sufficiently good condition to make a postmortem examination worthwhile, the fish was transported to the Louisiana Aquatic Diagnostic Laboratory at the Louisiana State University–School of Veterinary Medicine (Baton Rouge, LA, USA) for autopsy. On gross examination, the pericardial tissues were hyperemic and edematous with ~10 mL of watery, tan, turbid fluid in the pericardial sac. Three trematodes were present in the pyloric stomach and ceca. No other gross abnormalities were noted. Samples of all major organs, the eyes, and the whole brain were collected and fixed in 10% neutral-buffered formalin for 2 d, trimmed and processed routinely, and stained with H&E in preparation for histologic examination.
Histologic lesions were limited to the cerebrum and eye, in which severe granulomatous encephalomyelitis and mild choroiditis were observed, respectively. Large numbers of macrophages infiltrated the neuropil and often formed randomly distributed granulomas throughout white and gray matter (Fig. 1A, 1B). Granulomas were composed of epithelioid macrophages and fewer lymphocytes and glial cells arranged in concentric layers. Glial cells were abundant and scattered throughout the neuropil, occasionally surrounding degenerate and necrotic neurons (satellitosis). Immunohistochemistry (IHC) for betanodavirus was performed at the Istituto Zooprofilattico Sperimentale delle Venezie (IZSDV; Legnaro, Italy) on paraffin-embedded blocks of brain tissue. 13 Briefly, IHC detection of viral antigens was performed using rabbit polyclonal serum against nodavirus (PAb 283, 1:2,500; IZSDV). Slides were incubated in trypsin solution for antigen retrieval, then washed in Tris-buffered saline (TBS). Slides were incubated in antiperoxidase blocking solution to block endogenous peroxidase activity, then rinsed twice in TBS. Nonspecific antibody sites were blocked with normal horse serum, washed in TBS, and then incubated with PAb 283. Slides were washed 3 times with TBS, then incubated with biotinylated universal antibody anti-rabbit/mouse, washed again, and avidin–biotin peroxidase complex and horseradish peroxidase was then added. Slides were washed a final time, then incubated with 3-amino-9-ethylcarbazole (AEC) chromogen, and finally counterstained with Harris hematoxylin and mounted in glycerol–gelatin. Betanodavirus antigens were illuminated by red chromogen detected on light microscopy. Positive red immunohistostaining was seen within the cytoplasm of macrophages of glial nodules and a few neurons (Fig. 1C). Other infectious agents were not detected with special histochemical stains (Grocott methenamine silver [GMS], periodic acid–Schiff, Ziehl–Neelsen acid-fast, Gram stains).
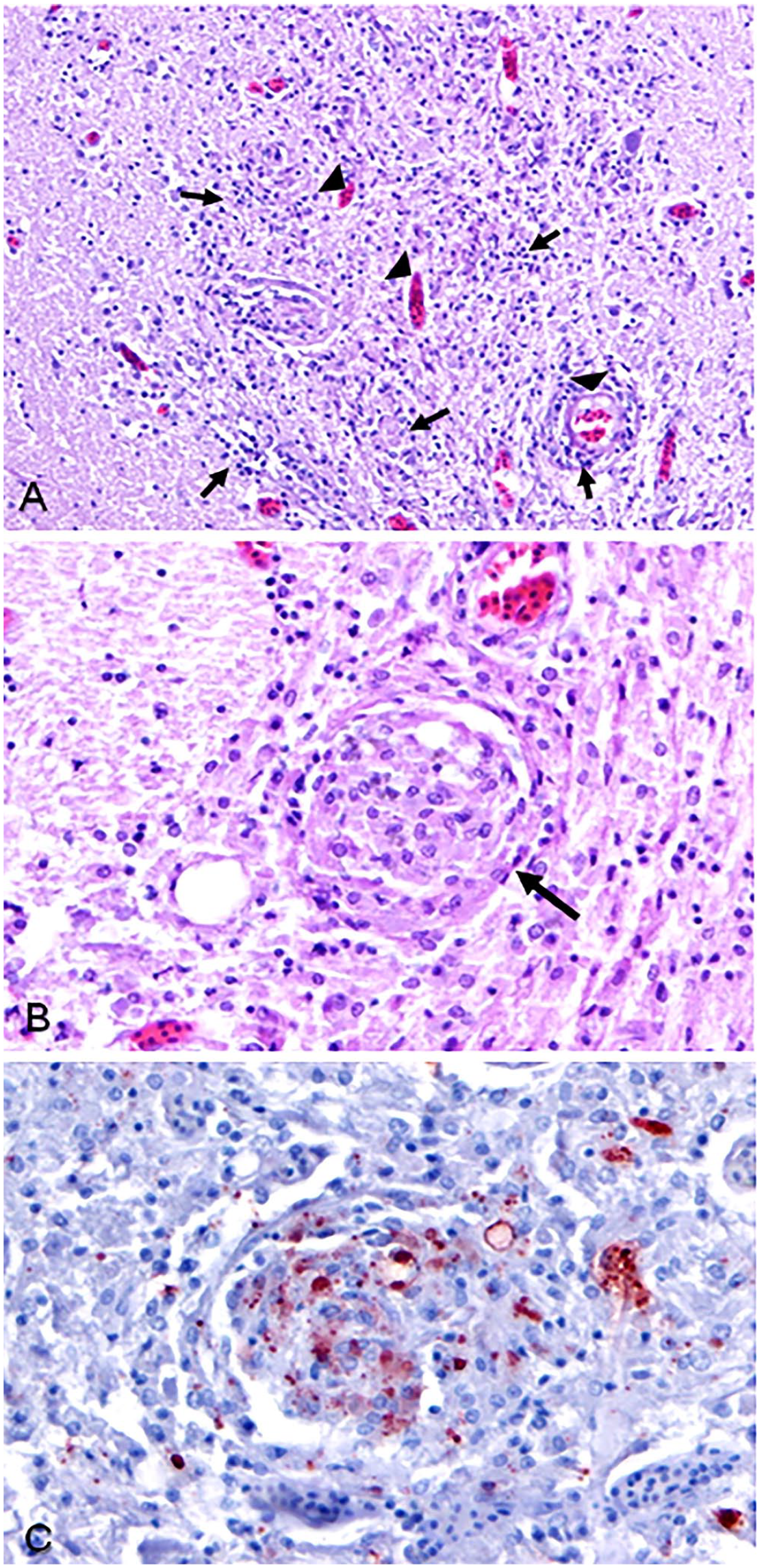
Betanodavirus meningoencephalitis in an Atlantic blue marlin.
A whole, frozen eye was submitted to the Mississippi State University–College of Veterinary Medicine (Mississippi State, MS, USA) for virus isolation, reverse-transcription PCR (RT-PCR), and sequencing. For cell culture evaluations, retina and optic nerve tissues were homogenized, diluted 1:5 on serum-free medium, and filtered through a 0.2-µm filter. In a 24-well plate, triplicate wells of E11 cells were inoculated with 100 µL or 50 µL of homogenate, incubated at 20°C and 28°C, and evaluated for 10 d; no cytopathic effect was evident. Cells were blind-passed and evaluated for another 10 d; no cytopathic effect developed. RNA was extracted from optic nerve and retina tissue using TRIzol reagent (Thermo Fisher), according to the manufacturer’s protocol. One-step hydrolysis probe real-time RT-PCR was performed on 100 ng of total RNA with primers and probes (QuantiTect virus qPCR kit; Qiagen) using methods described previously. 6 Both tissues were positive, with mean Ct values of 35.9 and 32.2 for retina and optic nerve, respectively, indicating that the concentration of viral RNA in the portion of the optic nerve that was sampled was ~13-fold higher than the retina sample. The RNA was subsequently subjected to nested PCR by first running a one-step RT-PCR using F2 and R3 primers 11 (Superscript III one-step RT-PCR kit; Thermo Fisher) in a 50-µL reaction. Cycling parameters were: 1 incubation cycle at 42°C for 45 min, and 95°C for 4 min; followed by 30 cycles at 95°C for 40 s, 55°C for 40 s, and 72°C for 40 s. This was followed by PCR using 2 µL of the product and amplifying the product with primers VNNF2-280 (ACCTGAGGAGACTCACCGCTC) and VNNR3 (CAGCGAAACCAGCCTGCAGG) and taq polymerase (Takara Bio) in 25-μL reactions using 1 cycle at 95°C for 4 min; then 30 cycles at 95°C for 30 s, 60°C for 30 s, and 72°C for 30 s; and 1 cycle at 72°C for 7 min. PCR yielded a 280-bp band that was purified and cloned into the TOPO TA plasmid (Thermo Fisher). The inserts of 3 separate cloned purified plasmids were sequenced by Big-Dye terminator Sanger sequencing reactions (Thermo Fisher) using T7 and T3 primers (Prizm 310 genetic analyzer; Applied Biosystems). The 212 bp between the primers of the 280-bp amplicon had 99% identity (209 of 212) to Lates calcarifer encephalitis virus (GenBank AY284974.1) and 98% identity (208 of 212) to several NNV isolates, including RGNNV (GenBank AY744705.1).
This is, to our knowledge, the first report of disease in an Atlantic blue marlin. Little is known of the susceptibility of the Atlantic blue marlin to disease, given the perception of its incompatibility with captivity, based on previous attempts by aquaria with other fast-moving pelagic species (thus preventing captive populations and study), and the general difficulty of acquiring ill wild specimens. Most specimens of this species are caught for sport, or as bycatch for other fisheries, and are presumed to be healthy animals that are rarely examined for lesions. In our case, the specimen was a rare gift, a stranding mortality that provided insight into the susceptibility of this species to a highly contagious, commonly fatal, piscine virus. In fish culture settings, horizontal transmission of NNV has been demonstrated by the oral route, bath, cohabitation, intramuscular injection, and intraperitoneal injection. 1 Vertical transmission has been reported in striped jack, Japanese flounder, barfin flounder, Atlantic halibut, European and Asian sea bass, and several grouper species. 1 However, the mechanisms of cellular entry have not yet been elucidated. In addition to cultured fish species, this virus has also been found in wild species spanning 19 orders, the majority associated with RGNNV.
In our case, the betanodavirus was most similar to Lates calcarifer encephalitis virus, which is within the RGNNV genotype. 10 This is cogent, considering the overlap of the geographic ranges of the Atlantic blue marlin and RGNNV. As a highly migratory piscivorous hunter, we presume that this marlin became infected by ingesting an infected fish, or potentially via water transmission by passing near high-density sea aquaculture cages, leading to disorientation and stranding. The lesions that we observed are consistent with those reported for chronic adult cases in other species, despite our case being aged as a juvenile. Considering the long maturation time of marlins, the target neurons of the CNS may have been fully developed at the time of infection, thus causing the difference in disease manifestation.
Other common causes of granulomatous encephalitis with granuloma formation in fish include local and systemic fungal and mycobacterial infections; these entities were ruled out via GMS and acid-fast stains on formalin-fixed, paraffin-embedded brain sections. Betanodavirus encephalitis was considered as the primary cause of death in our case given the effects of this virus in other fish species and the lack of evidence for metabolic or traumatic causes determined via gross and histologic examination of major organs. Given that Atlantic blue marlins are highly valued as meat and game animals, more consideration should be given to the study of these species and their susceptibility to contagious disease in addition to their response to other conservation threats.
Footnotes
Acknowledgements
We thank the late Cheryl Crowder for her contributions to this case report.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
