Abstract
A female goat fetus was received by the Colorado State University–Veterinary Diagnostic Laboratory following an isolated abortion of twins by a reportedly healthy doe. Postmortem examination did not reveal any gross abnormalities. Histologic evaluation revealed pyogranulomatous and necrotizing bronchopneumonia with intracellular and extracellular gram-positive and non–acid-fast filamentous bacilli. Aerobic culture of the stomach contents and pooled lung and liver tissue yielded light growth of Nocardia sp., which was identified by MALDI-TOF MS and 16s rDNA sequencing as Nocardia farcinica.
A female goat fetus, of unspecified breed, from Southwest Colorado was submitted frozen for autopsy to the Western Slope branch of the Colorado State University–Veterinary Diagnostic Laboratory (CSU-VDL; Fort Collins, CO, USA). The goat fetus and its twin (not submitted) were aborted 3-wk premature, by a reportedly healthy doe. The doe was from a herd of 17 goats, and the abortion occurred as an isolated event.
There were no gross lesions identified on autopsy, and the placenta was not available for examination. A diagnostic workup was performed at CSU-VDL and initially included: bacterial aerobic culture of the stomach contents and pooled lung and liver; Chlamydia spp. and Coxiella burnetii PCR testing on pooled lung, liver, and kidney tissue; and Toxoplasma gondii PCR testing on heart tissue. For these various PCR amplifications, DNA was first extracted from the samples (QIAamp DNA mini kit; Qiagen), and RT-qPCR was performed according to CSU-VDL protocols. Tissue samples of the lung, liver, kidney, heart, and brain were fixed in 10% neutral-buffered formalin for a minimum of 24 h and processed routinely to produce 4-μm thick sections. Histologic sections were stained with H&E, Brown and Hopps Gram, and acid-fast (Ziehl–Neelsen [ZN] and Fite) stains.
Histologic examination of the lung revealed that ~90% of the terminal bronchioles and alveolar spaces were filled with abundant degenerate neutrophils, macrophages, multinucleate giant cells admixed with fibrin, and karyorrhectic debris. The terminal airway epithelium was frequently attenuated or lost and sloughed into the lumen. Bronchioles were occasionally surrounded by large aggregates of lymphocytes and plasma cells (Fig. 1). The bronchi were unaffected. There were no remarkable microscopic findings in the other tissues examined. Numerous intracellular and extracellular filamentous gram-positive bacilli were within macrophages and multinucleate giant cells in terminal bronchioles and alveoli (Fig. 1C). The acid-fast stains were negative.
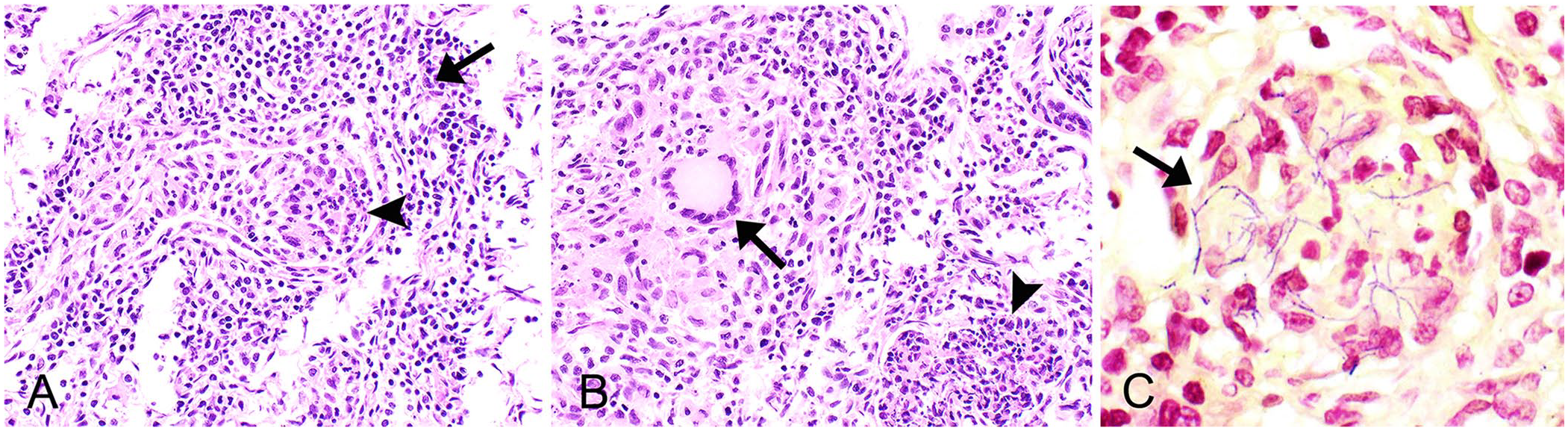
Pyogranulomatous pneumonia in an aborted female goat fetus lung.
Light growth of Nocardia sp. was obtained in the aerobic cultures of the stomach contents and pooled lung and liver tissues. This light growth was noted on trypticase soy agar with 5% sheep blood (Becton Dickenson) incubated in ambient air at 35°C, and Columbia agar with 5% sheep blood (Becton Dickenson) incubated in 5% CO2 at 35°C. Bacteria were identified as Nocardia farcinica by MALDI-TOF MS (Bruker) with a confidence score of 2.25. Aerobic cultures additionally revealed light mixed microbiota, consistent with surface contamination. Nocardia sp. was the predominant organism isolated.
The Nocardia sp. isolate was further characterized by DNA sequencing. DNA was extracted by placing the isolate in a pathogen lysis tube (Qiagen) followed by a 16S ribosomal RNA conventional PCR assay using forward and reverse primers (16S F: 5′-TTGGAGAGTTTGATCCTGGCTC-3′; 16S R: 5′-ACGTCATCCCCACCTTCCTC-3′) and the following cycling conditions: 94°C for 5 min; 35 cycles of 94°C for 30 s, 60°C for 30 s and 72°C for 2 min; and a final extension step at 72°C for 2 min. Results were screened on an agarose gel, and bands were extracted and bidirectionally sequenced. Sanger sequencing was performed at Quintara Biosciences (San Francisco, CA, USA), and a consensus sequence was based upon both the forward and reverse sequences. A BLAST search (https://blast.ncbi.nlm.nih.gov/Blast.cgi) of the 1,094-bp amplicon showed that the isolate was 98.6% similar to N. farcinica (MW563834.1). The sequence from this isolate was deposited into GenBank (OQ781153). PCR testing for Chlamydia spp., Coxiella burnetii, and Toxoplasma gondii was negative.
Nocardia spp. are ubiquitous saprophytic bacteria in the class Actinomycetes, order Mycobacteriales, along with Mycobacterium and Corynebacterium spp. There are > 100 characterized species of Nocardia found in the soil, water, and decaying organic matter worldwide. 20 Approximately half of these species have been associated with clinical disease in predominantly immunocompromised animals and/or humans. Although opportunistic infections are more common, occasionally patients without an underlying predisposing condition are affected. 20 Pathogenic Nocardia spp. are facultative intracellular, gram-positive, partially acid-fast bacteria that evade host immunity by inhibiting phagosome-lysosome fusion, neutralizing phagosome acidification, secreting superoxide dismutase, and altering lysosomal enzymes in neutrophils and macrophages. 19 Identification and diagnoses of nocardial infections are often difficult, given that Nocardia spp. grow more slowly in culture than frequently contaminating microbiota. Treatment of nocardial infections is often also difficult given the extended antimicrobial use and surgical excision or debridement of cutaneous lesions. Despite the occurrence of infections with the same Nocardia spp. in animals and humans, zoonotic nocardiosis has not been reported.
Nocardiosis predominantly occurs as granulomatous or pyogranulomatous inflammation in cutaneous and subcutaneous tissues from traumatic inoculation or as pneumonia from inhalation. 20 Both routes of infection can lead to CNS involvement and lead to life-threatening systemic disease. 14 In humans, ocular infections and osteomyelitis have been reported as unusual primary infections from direct inoculation. 20 Nocardia infections have been identified in a multitude of animal species, including cattle, sheep, goats, dogs, cats, pinnipeds, cetaceans, fish, birds, horses, pigs, bivalves, a llama, reindeer, and Alpine chamois.2–5,8,9,11–13,15,18, 21 ,22 In cattle, nocardiosis is seen most commonly as environmentally derived mastitis that results from poor hygienic conditions in dairy herds. The most common Nocardia species implicated in bovine mastitis are N. asteroides, N. farcinica, N. nova, and N. brasiliensis. 6 In goats, environmentally derived mastitis from N. farcinica is considered a significant cause of mastitis in Sudan. 12 Only one other case report of goat mastitis caused by Nocardia (N. asteroides) has been reported, similarly in Sudan. 7 A report from 1973, identified N. caviae (current name N. otitidiscaviarum) as the cause of granulomatous pneumonia in an adult goat in Malawi. 10 The pathogenic species implicated in disease of cats and dogs vary geographically and include N. nova, N. otitidiscaviarum, N. asteroides, N. farcinica, and N. brasiliensis.13,15,19 In dogs, coinfection with canine distemper virus (CDV; Morbillivirus canis) is commonly reported as a result of CDV-induced immunosuppression. Nocardiosis is a major cause of mortality in intensively cultured fish in Japan leading to large economic losses. Infected fish can exhibit anorexia, emaciation, and erratic swimming. 11 Most infections in marine mammals are disseminated, and the most frequently affected organs are the lungs and thoracic lymph nodes. 18
Sporadic abortions caused by Nocardia have been reported in cattle, pigs, and horses.1,2,5,16 These abortions are primarily caused by N. asteroides; however, abortion and stillbirth caused by N. farcinica in cattle has been reported twice.1,17 One case of suspected nocardial placentitis from an ovine abortion was reported. 22 Histologically, all of the aborted bovine and equine fetuses had pyogranulomatous or suppurative pneumonia with systemic involvement and placentitis. The dams were reported as systemically healthy, and the pathogenesis is poorly understood. It has been proposed that uterine infection by environmental contamination leads to placentitis and subsequent fetal infection. The bronchopneumonia of the fetus in our case may suggest aspiration of amniotic fluid secondary to placentitis. However, the placenta was not submitted for evaluation in our case.
The most common infectious causes of abortion in goats include Chlamydia abortus, Listeria monocytogenes, Toxoplasma gondii, Coxiella burnetii, Leptospira spp., and Brucella melitensis. These infectious organisms generally cause necrotizing and/or suppurative fetal lesions or minimal le-sions with marked autolysis. Pyogranulomatous pneumonia is an uncommon finding in aborted fetuses of all species. In aborted goats with pyogranulomatous pneumonia, nocardiosis should be considered as a primary differential diagnosis.
Footnotes
Acknowledgements
We thank Gene Niles for performing the postmortem examination, the Colorado State University–Veterinary Diagnostic Laboratory technicians for performing the tissue processing and staining, and Michael Russel for performing ancillary tests. Helena Vogel is a Molecular Pathology Fellow in the National Institutes of Health (NIH) Comparative Biomedical Scientist Training Program supported by the National Cancer Institute in partnership with Colorado State University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no outside financial support for the research, authorship, and/or publication of this article.
