Abstract
Chlamydial abortion in small ruminants is usually associated with Chlamydia abortus infection. Although Chlamydia pecorum has been detected in aborted ruminants and epidemiological data suggests that C. pecorum is abortigenic in these species, published descriptions of lesions in fetuses are lacking. This work describes fetoplacental lesions in a caprine abortion with C. pecorum infection, and further supports the abortigenic role of C. pecorum in ruminants. A 16-month-old Boer goat aborted twin fetuses at ~130 days of gestation. Both fetuses (A and B) and the placenta of fetus A were submitted for postmortem examination and diagnostic workup. At autopsy, the fetuses had moderate anasarca, intermuscular edema in the hindquarters (A), and brachygnathia and palatoschisis (B). In the placenta, the cotyledons were covered by yellow fibrinosuppurative exudate that extended into the adjacent intercotyledonary areas. Histologically, there was severe suppurative and necrotizing placentitis with vasculitis (arteriolitis) and thrombosis, multifocal lymphohistiocytic and neutrophilic hepatitis (A), and fibrinosuppurative enteritis in both fetuses. Chlamydia antigen was detected in the placenta by the direct fluorescent antibody test and in fetal intestines by immunohistochemistry. Nested polymerase chain reaction of DNA extracted from formalin-fixed, paraffin-embedded sections of placenta and intestine amplified 400 bp of the Chlamydia 16S rRNA gene that was sequenced and found to be 99% identical to C. pecorum by BLAST analysis. Other known abortigenic infectious agents were ruled out by specific testing. It is concluded that C. pecorum infection is associated with fetoplacental lesions and sporadic abortion in goats.
Chlamydial infections caused by ubiquitous obligate intracellular Gram-negative bacteria in the family Chlamydiaceae are associated with a wide spectrum of diseases in animals and humans,8,22,32 with some chlamydial species causing zoonotic infections. 27 Diseases caused by these agents include abortion, pneumonia, gastroenteritis, encephalomyelitis, conjunctivitis, arthritis, and/or sexually transmitted diseases.18,27,34
The taxonomy of chlamydial microorganisms has been controversial. In the late 1990s, the only genus (Chlamydia) in the family was subdivided into 2 genera: Chlamydia and Chlamydophila. 9 In 2010, the “Subcommittee on the taxonomy of Chlamydiae” (International Committee on Systematics of Prokaryotes) proposed that a single genus (Chlamydia) should be considered within the family.12,13 This nomenclature will be followed in this article.
The first 2 chlamydial species identified based on morphological and phenotypical characteristics were C. trachomatis and C. psittaci. However, it was soon recognized that a number of isolates did not fit into either species group, and that members of the C. psittaci species represented a diverse group of microorganisms that differed serologically, genetically, and in host tropism, including pathogens of humans, nonhuman mammals, and birds. 22 With the advancement of gene sequencing and immunological analyses, new species were identified, including C. abortus, C. pecorum, C. pneumoniae, C. felis, C. muridarum, C. caviae, and C. suis.5,16
Two species are primarily implicated in ruminant diseases, C. abortus (formerly C. psittaci serotype 1) and C. pecorum (formerly C. psittaci serotype 2).1,10 C. abortus has long been recognized as a common cause of late-term abortion in sheep, being the etiologic agent of the disease known as ovine enzootic abortion, responsible for large economic losses in the livestock industry.14,24 C. abortus enzootic abortion in goats is clinically and pathologically similar to that occurring in sheep. 31 C. abortus is one of the recognized zoonotic chlamydial species. Although human cases are rare, C. abortus has the ability to colonize the human placenta and cause abortion in pregnant women. 27
Chlamydia pecorum was proposed as a separate species in 1992 based on DNA–DNA hybridization and immunologic analyses of strains that were associated with polyarthritis, encephalomyelitis, pneumonia, diarrhea, and inapparent enteric infection in sheep and cattle. 10 Chlamydia pecorum is commonly detected in the digestive and genital tracts of healthy ruminants; however, it has also been associated with reproductive and systemic disorders in sheep, goats, buffalo, and cattle.3,10,11,19,21,33
Although the involvement of C. pecorum in small ruminant abortion cases has been previously recognized,4,28 –30 the abortigenic and zoonotic impacts of C. pecorum are still unknown, 4 and there are no reports, to our knowledge, describing fetal lesions caused by this agent in ruminants. In the current work, we describe placental and fetal lesions caused by C. pecorum in a case of sporadic abortion in a goat.
A 16-month-old Boer doe grouped with 34 other goats in a herd of 140 animals in California aborted twin fetuses at ~130 days of gestation. There was no recent history of abortions in the herd, and the herd had no history of vaccination against C. abortus or other abortifacients. Both fetuses (A and B) and a piece of placenta expelled with fetus A were submitted to the California Animal Health and Food Safety (CAHFS) laboratory of the University of California, Davis. Postmortem examination was conducted, and samples of placenta and tissues from both fetuses, including liver, brain, thymus, heart, pancreas, kidneys, lungs, spleen, skeletal muscle, tongue, trachea, thyroid gland, abomasum, esophagus, salivary gland, skin, conjunctiva, urinary bladder, and small and large intestines were immersion-fixed in 10% neutral buffered formalin for 24 hr, and processed routinely for the production of 4-µm-thick sections that were stained with hematoxylin and eosin for histological examination. Formalin-fixed, paraffin-embedded (FFPE) sections of placenta and small intestines from both fetuses were processed for immunohistochemistry (IHC) for detection of Chlamydia spp. antigen using a mouse anti-Chlamydia lipopolysaccharide (LPS) monoclonal antibody, a at a dilution of 1:50 as a primary antibody. Antigen unmasking was accomplished by 15-min incubation in acidulated pepsin. Sequential incubations with horseradish peroxidase–labeled anti-mouse polymer, b followed by 3-amino-9-ethylcarbazol (AEC) substrate chromogen, c enabled visualization of the bound antibody. Similarly, FFPE sections of placenta were processed for the detection of Coxiella burnetii antigen by IHC using a mouse monoclonal antibody. d Antigen unmasking was not necessary, and antibody binding was detected as previously described. Appropriate positive and negative controls were used for each of the IHCs. An impression smear from the placenta was processed for Chlamydia spp. antigen detection by direct fluorescent antibody test (FAT) using a fluorescein-labeled mouse monoclonal antibody directed against Chlamydia LPS. e
Samples of placenta and abomasal fluid, lung, and liver from both fetuses were processed routinely for aerobic and microaerophilic bacterial cultures on sheep blood agar, and incubated at 35–39°C for 48 hr. Additionally, samples of placenta, lung, and abomasal fluid from both fetuses were processed for Campylobacter spp. and Brucella spp. cultures. Smears of kidney from both fetuses were analyzed by FAT for the detection of Leptospira spp. antigen using a multivalent conjugant of rabbit origin. f
Fetal thoracic fluid samples from both fetuses were processed for the detection of antibodies against pestiviruses (Bovine viral diarrhea virus [BVDV] 1 and 2 and Border disease virus [BDV]) by serum virus neutralization test, 7 Bluetongue virus (BTV) by enzyme-linked immunosorbent assay (ELISA), g Coxiella burnetii by ELISA, h Brucella spp. by Rose Bengal test, 35 Toxoplasma gondii by latex agglutination test, i and several Leptospira interrogans serovars (including L. Icterohaemorrhagiae, L. Canicola, L. Grippotyphosa, L. Hardjo, and L. Pomona) by microscopic agglutination test, as previously described. 2 All of the aforementioned testing was performed according to CAHFS standard operating procedures and following the manufacturers’ recommendations. Additionally, a frozen brain sample from fetus B was submitted to the National Veterinary Services Laboratory (NVSL; Ames, Iowa) for the detection of genomic RNA of Schmallenberg virus and bunyaviruses within the California and Bunyamwera serogroups by polymerase chain reaction (PCR).
Sections from FFPE placenta (A) and small intestine (B) were submitted to the Wyoming Veterinary Diagnostic Laboratory (Laramie, Wyoming) for detection of Chlamydia spp. by nested PCR and sequencing. DNA was isolated using a commercial kit j protocol for paraffin-embedded tissue. Briefly, 25 mg of the paraffin sections were dewaxed in 1.2 mL of xylene followed by 2 × 1.2 mL ethanol washes. The tissue was then pelleted and DNA extracted according to the manufacturer’s directions. Samples were tested for Chlamydia spp. using a previously described nested PCR assay. 23 Briefly, genus-specific, first-round primers were used to amplify 436 bp of Chlamydia 16S rRNA, followed by a second round of PCR using semi–species-specific inner primers. The PCR amplicon from the first-round reaction was sequenced in both directions using the forward and reverse primers by Sanger sequencing k to obtain 400 bp of sequence. Sequences were compared to genetic sequences in GenBank using BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
At postmortem examination, fetus A, a 3.2-kg male with a crown-to-rump length (CRL) of 41 cm in a good state of postmortem preservation, had moderate anasarca with scrotal and intermuscular edema in the hind quarters. Fetus B, a 2.4-kg female with a CRL of 39 cm in a moderate state of postmortem decomposition, had moderate anasarca, brachygnathia superior and inferior, and palatoschisis affecting the hard palate. Grossly, the cotyledons of a piece of placenta expelled with fetus A were covered by loose, yellow, friable exudate that extended to the adjacent intercotyledonary areas. These were variably red to white gray, moderately thickened, and had lost normal translucency.
Microscopically, there was marked diffuse suppurative and necrotizing placentitis characterized by expansion of the chorionic surface and stroma by abundant degenerate neutrophils admixed with necrotic cellular debris (Fig. 1), mineral, and abundant extracellular indistinct basophilic bacteria. Some deep chorionic arterioles showed mural fibrinoid necrosis, and their walls were infiltrated by inflammatory cells, including degenerate neutrophils and lytic cellular debris (arteriolitis; Fig. 1). Occasional medium-sized blood vessels were occluded by fibrin thrombi. Scattered sloughed mono- and binucleate trophoblastic cells contained abundant intracytoplasmic indistinct granular basophilic bacteria consistent with chlamydial reticulate bodies (Fig. 2). Incidentally, there was a focal area of cartilaginous metaplasia in the chorioallantoic stroma.
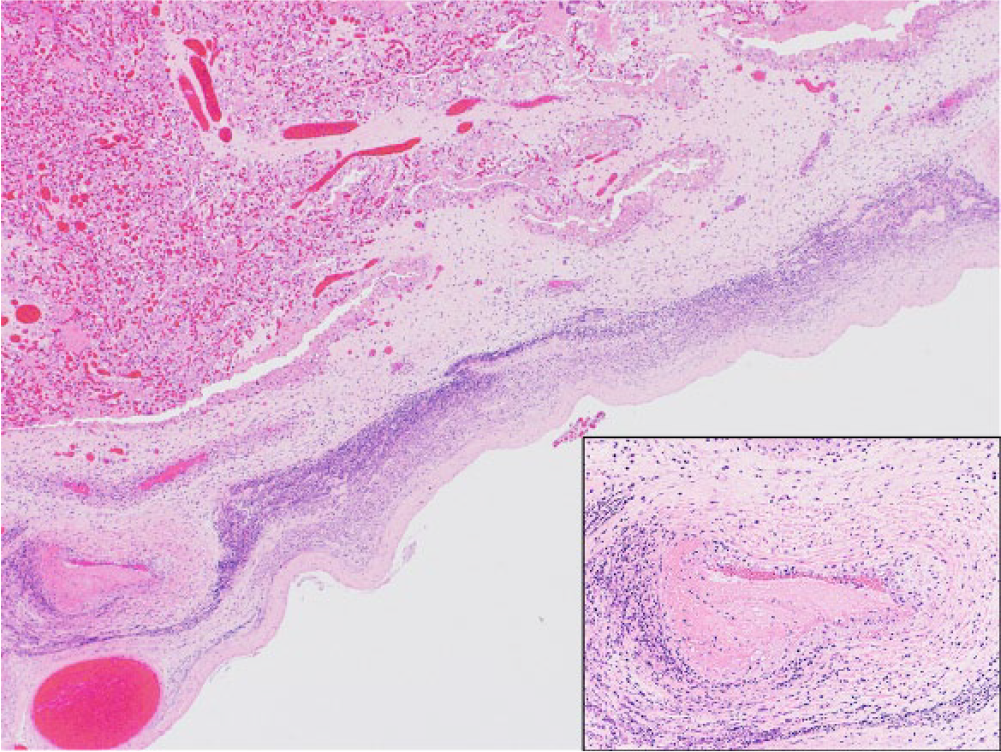
Caprine placenta. Suppurative placentitis characterized by severe diffuse expansion of the chorionic stroma by infiltrating neutrophils and necrotic debris, with associated necrotizing mural arteritis (lower left corner and inset). Hematoxylin and eosin.
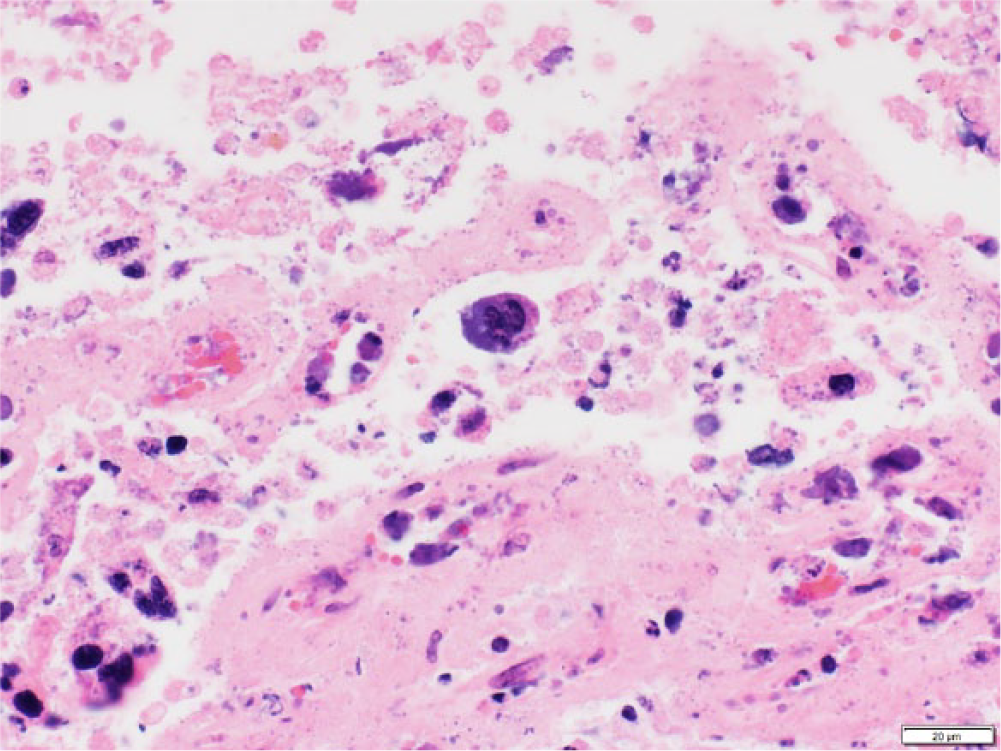
Caprine placenta. A binucleate trophoblastic cell (center) contains abundant intracytoplasmic basophilic bacteria consistent with chlamydial reticulate bodies. Bar = 20 μm.
In the tissues of fetus A, there was multifocal moderate histiocytic and neutrophilic hepatitis, with a random but predominantly portal and periportal distribution, and sinusoidal leukocytosis. In the small intestine, there was moderate fibrinosuppurative exudate admixed with necrotic debris filling the lumen (Fig. 3). Occasionally, sloughed enterocytes contained abundant intracytoplasmic bacteria morphologically resembling chlamydial reticulate bodies (Fig. 3). In the mucosa, intestinal villi were shortened, and some crypts were mildly distended, lined by flattened epithelial cells, and filled with neutrophils (cryptitis; Fig. 4A). In fetus B, there was also severe fibrinosuppurative and necrotizing enteritis with pseudomembrane formation and neutrophilic cryptitis. No microscopic lesions were observed in the other examined tissues in both fetuses.
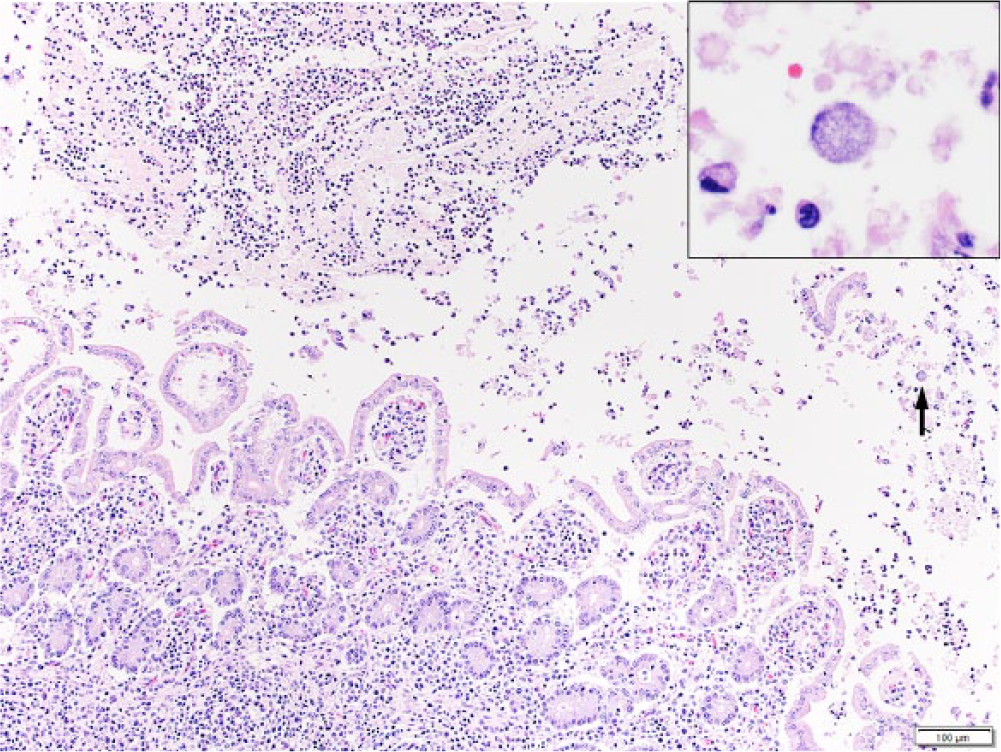
Caprine fetus A; small intestine. Fibrinosuppurative exudate in the lumen of the small intestine (upper left). Sloughed enterocytes contain abundant intracytoplasmic basophilic bacteria resembling chlamydial reticulate bodies (arrow and inset). Hematoxylin and eosin. Bar = 100 μm.
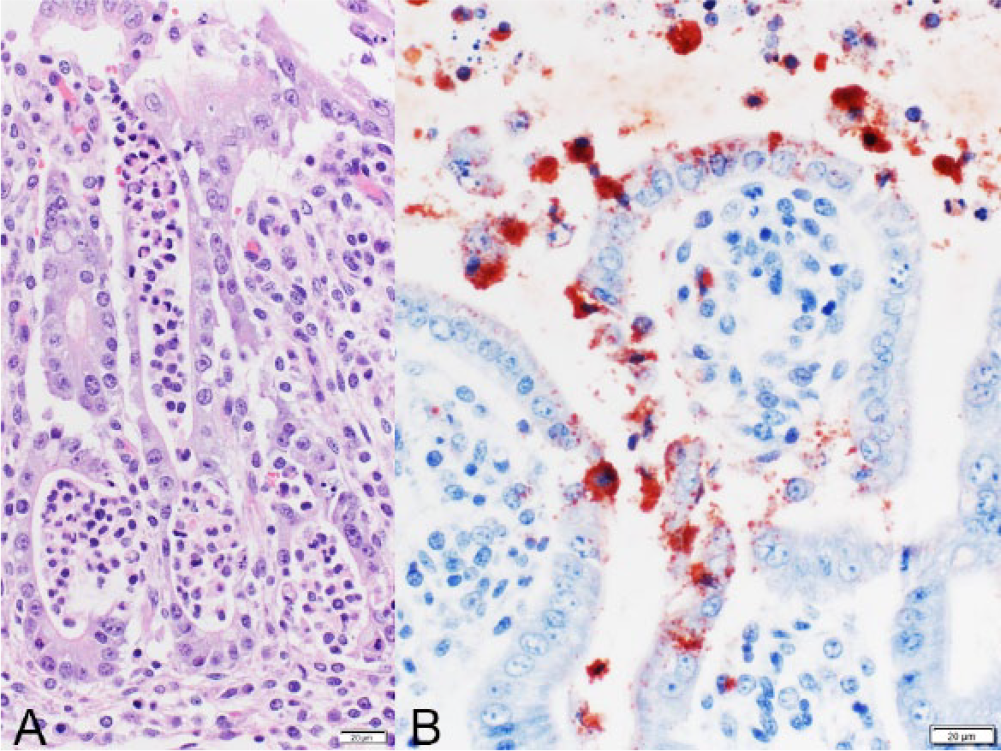
Caprine fetus A; small intestine.
Chlamydia spp. antigen was detected by FAT in direct smears of placenta of fetus A, and intralesionally by IHC in the placenta (A) and small intestine of both fetuses (Fig. 4B), but not in the liver of fetus A. Chlamydia spp. DNA was detected by nested PCR on FFPE sections of placenta (A) and small intestine (B). The sequence of a 400-bp region of the 16S rRNA gene in both samples was found to be 99% identical to C. pecorum by BLAST analysis (94–95% homology with C. abortus and C. psittaci). Other abortigenic pathogens were ruled out in the placental and fetal samples using direct (Leptospira spp., Brucella spp., C. burnetii, Campylobacter spp., and other aerobic and microaerophilic bacteria, and Schmallenberg virus and bunyaviruses of the California and Bunyamwera serogroups) and indirect (Leptospira spp., C. burnetii, T. gondii, BVDV, BDV, and BTV) detection methods.
Chlamydial abortion in small ruminants is usually associated with C. abortus infection. Although there is epidemiological data that suggests that C. pecorum is abortigenic in ruminants, to date there were no descriptions of microscopic fetal and placental lesions caused by the agent in these species, to our knowledge. Our report describes fetal and placental lesions in an aborted goat naturally infected with C. pecorum, and further supports the abortigenic role of C. pecorum in ruminants.
A few studies report on the detection of C. pecorum in aborted fetuses of various ruminant species. Detection of C. pecorum strains from cases of abortion in sheep and goats has been reported in France, 30 Tunisia (Tlatli A, et al. Isolation and typing of a local strain of Chlamydia pecorum of caprine origin. Proceedings of the 7th international conference on goats; 1999; Tours, France. In French), and Morocco. 28 Experimentally, intravenous inoculation of C. pecorum in pregnant ewes resulted in placental infection and abortion in some of the ewes. 26 In a study performed in Italy, C. pecorum was detected in the abomasal content of 3 out of 164 water buffalo (Bubalus bubalis) aborted fetuses from 2 of 80 herds, confirming sporadic naturally occurring fetal infections in this species. 3 Although all of these studies suggest that C. pecorum is abortigenic in ruminants, the studies lack histologic examinations of the fetuses and placentas. It is worth mentioning that the sole detection of the agent in placentas or fetuses does not necessarily confirm it as the cause of the abortions or its ability to cause fetoplacental lesions. Furthermore, in these studies, other common causes of abortion were not ruled out, and this is important in attributing causality, given the multifactorial etiology of the syndrome and the possible occurrence of coinfections with different reproductive pathogens, and even with different Chlamydia spp., as has been described in aborted buffalo fetuses coinfected with C. abortus and C. pecorum. 11
Without fulfilling Koch’s postulates, attributing causality to a potentially new reproductive pathogen in any animal species is challenging. Typically, this begins with elimination of usual pathogens and is followed by epidemiologic, clinical, pathological, and microbiological investigations that include a wide variety of ancillary laboratory tests that together support this pathogen-disease association. In the fetuses examined in our study, tests for most known abortigenic infectious agents of goats were all negative. Concurrently, several testing methods produced results indicative of chlamydial infection. Intralesional Chlamydia spp. was detected immunologically using antibody-based methods (direct FAT and IHC) in the placenta and fetal intestines. Finally, Chlamydia spp. DNA was detected by PCR, and subsequent sequencing allowed for identification of C. pecorum to a species level. Altogether, these results indicate an association between C. pecorum and the pathological process that led to the lesions and subsequent abortion in this goat.
Placental and liver lesions in our case were similar to those caused by C. abortus in ruminants. However, to our knowledge, intestinal lesions have not been described in C. abortus–infected fetuses, 31 and this may represent a distinct pathological feature of C. pecorum infection. Interestingly, C. pecorum is a well-recognized cause of enteritis in the postnatal life in ruminants. 33 Furthermore, the epidemiology of this case differed from that caused by C. abortus in that it was a sporadic abortion, rather than epizootic, as is usually the case with C. abortus.
Lesions caused by chlamydiae are not pathognomonic; therefore, an etiologic diagnosis based on pathologic findings only is not possible unless chlamydial elementary bodies are stained in tissue sections. Unfortunately, these histochemical methods are poorly sensitive and not species specific. 21 Etiologic confirmation requires isolation of the agent, still regarded as the standard method for diagnosis; however, various chlamydial strains require special facilities and expertise that are not widely available in veterinary diagnostic laboratories. Molecular methods such as PCR followed by sequencing are more sensitive, specific, and less time-consuming methods for the detection and identification of these bacteria.3,4,6,14,20,29
Some of the pathological findings in the fetuses examined in our study, such as cartilaginous metaplasia of the placenta in fetus A, and brachygnathia and palatoschisis in fetus B, cannot be explained by C. pecorum infection alone. Because brachygnathia has been described in aborted ruminants with orthobunyavirus infection, 15 we tested fetus B for the detection of bunyaviruses by PCR, with negative results. It is possible that other unidentified teratogens may have induced these lesions.
The mechanisms by which C. pecorum reaches the placenta and fetus are unknown. C. pecorum subclinical intestinal infections have been reported in flocks with and without a history of abortion, with animals acting as asymptomatic carriers.6,17,19,25,29 The enteric subtypes of C. pecorum are unlikely to be invasive and reach the placenta in pregnant animals when a well-balanced host–bacteria relationship is occurring.26,32 Exceptionally, some carrier animals may develop severe disease as a result of stress, lapsing into clinical disease. 32 Accordingly, a previous study has suggested that C. pecorum might spread from the intestine through the blood circulation as a result of unknown physiologic or pathologic events, and reach the placenta, thus inducing abortion. 4 No such stressful factors were identified in our case.
Interestingly, in our case, a newly introduced male was hypothesized to be a potential source of the infection. The male had served several does prior to introduction into the case herd, and, retrospectively, the former owner reported infertility and apparent abortion in several of these does, suggesting possible venereal transmission from this buck to the doe in the case herd. Unfortunately, no diagnostic testing was done on those animals, so the significance of this history is not known. The buck and the remaining pregnant does in the case herd that were served by him were treated with oxytetracycline; the buck remained in the herd and was used in subsequent seasons. No further abortions attributable to C. pecorum have occurred since the reported case.
Under natural conditions, C. pecorum infection was associated with placental and fetal lesions and spontaneous abortion in goats, with gross and microscopic lesions similar to those caused by C. abortus in small ruminants, with the exception of necrotizing enteritis. Because there is antigenic cross-reactivity between C. abortus and C. pecorum, 29 distinction cannot be made based on immunologic diagnostic tests (FAT or IHC), and etiologic confirmation requires molecular methods. The prevalence of C. pecorum as a cause of ruminant abortion should be further investigated.
Footnotes
Acknowledgements
We thank Jacqueline Cavender (Wyoming State Veterinary Laboratory) for technical assistance.
Authors’ contributions
F Giannitti contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. M Anderson contributed to conception and design of the study; critically revised the manuscript; and gave final approval. M Miller contributed to acquisition, analysis, and interpretation of data, and critically revised the manuscript. J Rowe contributed to acquisition and interpretation of data; critically revised the manuscript; and gave final approval. K Sverlow and M Vasquez contributed to acquisition of data, and critically revised the manuscript. G Cantón contributed to design of the study; contributed to interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval.
a.
Catalog no. 1681, Virostat Inc., Portland, ME.
b.
EnVision+ K4001, Dako North America Inc., Carpinteria, CA.
c.
K3464, Dako North America Inc., Carpinteria, CA.
d.
AB-COX-MAB, U.S. Department of Defense, Critical Reagents Program.
e.
D3 DFA culture confirmation kit, Diagnostic Hybrids Inc., Athens, OH.
f.
National Veterinary Services Laboratory, Ames, IA.
g.
Bluetongue virus antibody test kit, VMRD Inc., Pullman, WA.
h.
Q-Fever (Coxiella burnetii) antibody test kit, IDEXX, Hoofddorp, The Netherlands.
i.
Toxotest-MT test kit, Eiken Chemical Co., Tokyo, Japan.
j.
QIAamp, Qiagen Inc., Valencia, CA.
k.
Clemson University Genomics Institute, Clemson, SC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
