Abstract
We report meconium aspiration in 2 sibling goat kids, and characterize the expected lesions of aspiration pneumonia in conjunction with the rare lesion of otitis media. Grossly, the lungs were multifocally consolidated, and there was yellow-green exudate within the middle ear. Histologically, the lung was characterized by pyogranulomatous pneumonia and foreign-body reaction around aspirated debris. Within the lumen of the middle ear, aspirated squamous cells, keratin, meconium debris, and neutrophils, without evidence of bacteria, were accompanied by a subepithelial accumulation of lymphocytes, plasma cells, and fewer macrophages. This is an especially rare phenomenon, which is thought to result from transport of meconium from the oropharynx through the auditory tube (Eustachian tube) to the middle ear.
Fetal meconium release and subsequent aspiration of amniotic fluid is not considered to be a part of normal parturition, based on studies in the dog, sheep, rabbit, human, and non-human primate. 9 When it occurs in these species, and is reported anecdotally in case reports in other species, there are frequently serious neonatal disease consequences. Meconium release into amniotic fluid is often detected initially by a yellow hue or staining of the exterior of the animal. Meconium staining of the skin or haircoat is often without consequence. When particulate meconium matter within the amniotic fluid is inhaled or ingested, additional tissue lesions, most consistently pneumonia, may be noted at autopsy. Despite being sterile, meconium exerts its effects on visceral organs such as the lung by several interconnected mechanisms including obstruction, irritation, toxic insult, chemotaxis, and surfactant inhibition. 2 Meconium also acts as an enriched medium for bacterial growth, and bacteremia or sepsis may be seen concurrently in these cases.8,9
Depending on the extent and degree of involvement, animals may clear the material and recover, or death from respiratory distress may ensue. Meconium aspiration syndrome (MAS), coined for a similar scenario in human infants, to characterize the serious often fatal disease presentation and its sequelae, has been reported in foals as a cause of death at <5-d-old, in calves <2-wk-old, and in piglets.1,4,8 To our knowledge, MAS has not been reported previously in small ruminants.
Two sibling Nigerian goat kids were submitted for autopsy from a farm that raises ~30 Nigerian goats. The diet included hay, sweet feed (10% protein), and loose minerals. Injectable ivermectin was administered semi-annually for parasite control. Copper boluses were given to the does 1 mo prior to breeding. A 3-y-old Nigerian dwarf doe was bred to an ~6-mo-old buck. Gestation was considered normal. At the owner-attended birth of triplets, 2 of the 3 kids struggled to breathe. There was no reported history of dystocia. The owner attempted mouth-to-mouth resuscitation but, within 5 min of birth, both kids died; 1 buck kid weighed 1.2 kg, the doeling weighed 1.5 kg. The kids were presented for autopsy to the anatomic pathology laboratory at the University of Tennessee College of Veterinary Medicine (UT-CVM; Knoxville, TN). Placental membranes were not submitted for examination. The third kid, a doeling, died of respiratory distress at 1.5 wk, and was not autopsied.
The kids were determined to be term based on breeding history and fully formed anatomic structures such as teeth, haircoat, and crown-rump lengths. The female kid had meconium around the perineum and generalized yellow discoloration of the skin and haircoat. Both kids had partially aerated lungs that were pink and soft, and there were regions that were dark red, most prominently in the cranioventral aspect. Additionally, the inner ears, especially the tympanic bullae, contained thick green-to-yellow exudate (Fig. 1A). In the female kid, there was 7 mL of free blood in the abdomen, and a blood clot (estimated at 5 mL of blood volume) adhered to the edge of the fractured liver lobe, accounting for a 13% blood volume loss.
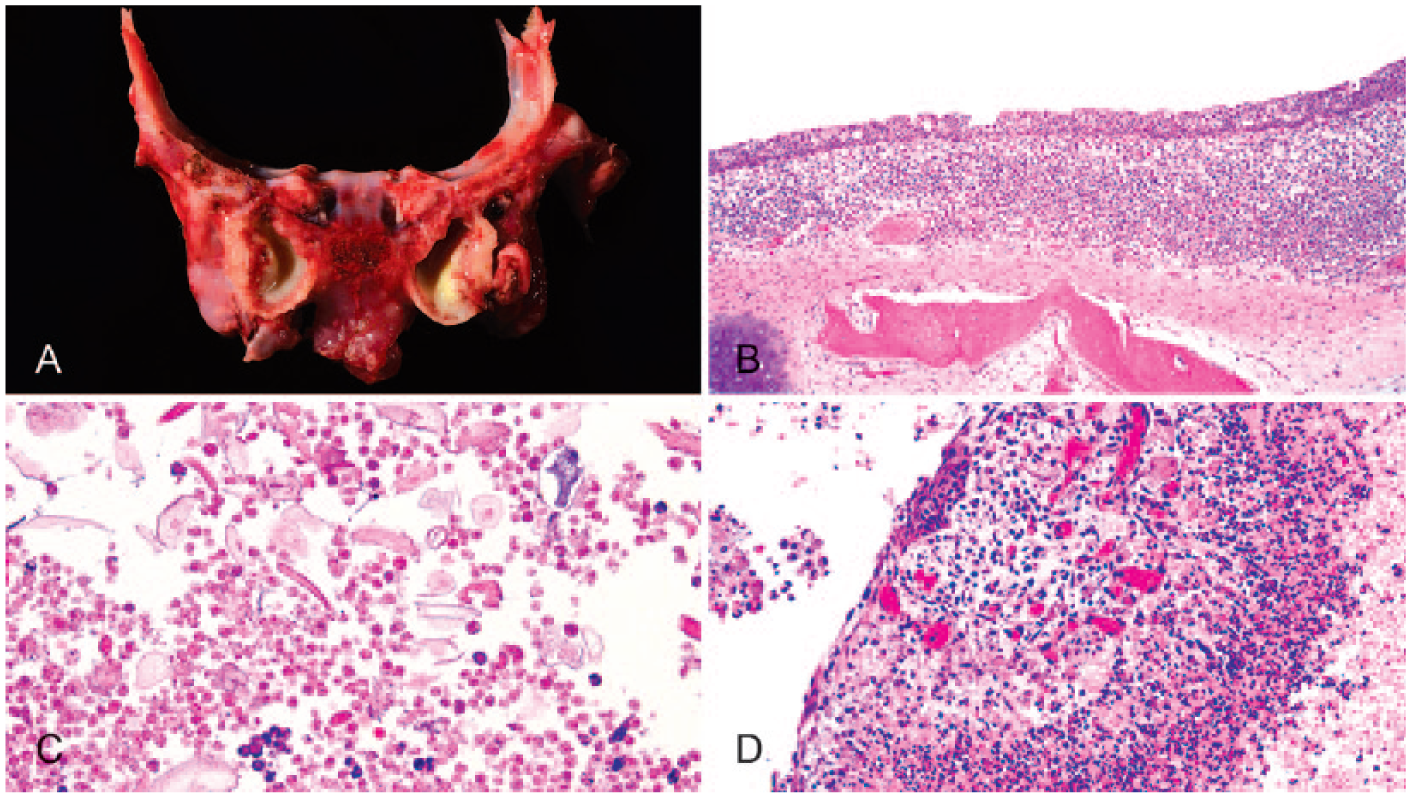
Middle ears of a neonatal goat kid with otitis media.
Tissues were collected into 10% buffered formalin, processed routinely, sectioned at 5 µm, and stained with hematoxylin and eosin. Histologic lesions were limited to the lung and middle ears (Fig. 1). The pulmonary inflammatory infiltrate was remarkable in extent, diffuse in its involvement, pyogranulomatous, and consistently expanded the alveolar lumens. Large numbers of alveolar macrophages and multinucleate giant cells were seen engulfing foreign material. Pulmonary sections consistently revealed large amounts of intra-alveolar and intra-bronchiolar material characterized as scrolls of lightly eosinophilic cytoplasm and rare nuclei, or as flakes of slightly refractile linear material, interpreted as keratin. Smaller numbers of neutrophils were present in alveoli and airways. Tissue Gram stains and acid-fast stains were negative for infectious agents. Special light microscopic stains for keratin (Ayoub–Shklar) confirmed small amounts of free keratin, and immunohistochemistry for cytokeratin (AE1/AE3) confirmed the presence of epithelial cells lining airways as expected, but not free in lumens. The likely reason for this absence of detection is that AE1/AE3 does not recognize all cytokeratins, including several layers of the epidermis. Additionally, amorphous yellow-to-brown granular material was present in air spaces and was interpreted as meconium. Hall bile stains were negative. Our diagnosis was moderate, subacute, pyogranulomatous, cranioventral bronchopneumonia with intralesional squamous epithelium, keratin, and meconium.
Bilaterally, the middle ears were inflamed. Moderate numbers of lymphocytes and plasma cells were in the lamina propria, and the epithelium was multifocally metaplastic (Fig. 1B). The lumen contained amorphous yellow material resembling meconium, and occasional flakes and scrolls of keratin (Fig. 1C), accompanied by foamy macrophages. Additionally, focal areas of loose connective tissue contained large numbers of linear small vessels and mild edema, interpreted as granulation tissue (Fig. 1D).Tissue Gram stains and acid-fast stains were negative. The keratin stain showed scattered positive luminal content. Bile stains for meconium were negative. Our diagnosis was moderate, subacute, diffuse, granulomatous otitis media with intraluminal meconium (rare), squamous epithelium, and keratin. Similar gross and histologic lesions were identified in the second kid.
Our report is an example of meconium aspiration in 2 term goat kid siblings. The degree of pneumonia in utero resulted in respiratory failure as a cause of death in both kids, with one kid having the added complication of blood loss from presumed hepatic trauma. The cause was not determined, but the owner admitted to performing resuscitation efforts, and there is a remote possibility that the liver fractured during parturition, albeit that no history of dystocia was documented.
Meconium aspiration from contaminated amniotic fluid, and its consequences, are well documented in the human literature but less so in the veterinary literature. In humans, amniotic contamination occurs in 10–15% of deliveries, with 3% of live births having meconium in the lungs. 7 This condition has been reported to cause yellow staining of the haircoat or skin in many species. Under certain circumstances, related to its duration, extent, and pro-inflammatory properties, amniotic contamination can produce foreign-body pneumonia, which has been documented in veterinary species including neonatal foals, calves, piglets, and a dolphin.1,4,8,10
Hypoxic events lead to intestinal peristalsis and release of meconium into amniotic fluid in utero (Kane E. Meconium aspiration syndrome in foals. DVM360 2017;1. Available from: http://veterinarynews.dvm360.com/meconium-aspiration-syndrome-foals). Reported hypoxic events include compression of the umbilical blood vessels, placentitis, twisting or knotting of the umbilical cord, rupture of the umbilical cord, and/or placental insufficiency. 1 Hypoxia also predisposes the fetus to forcefully gasp and subsequently aspirate amniotic fluid contaminated with meconium deep into the lungs (Kane E, 2017). Alternatively, the fetus can inhale accumulated meconium from the oropharynx at the time of the first breath of air, although this is likely to result in less severe signs (Kane E, 2017). The oropharynx is also the source of meconium-contaminated amniotic fluid that reaches the middle ear. Meconium can contribute to lesion formation in many ways. When aspirated, meconium acts as a direct toxin or local irritant to the pulmonary epithelium. Meconium contains substances that are pro-inflammatory, are chemotactic to neutrophils, and activate complement. 2 Meconium causes dose-dependent inhibition of surfactant, which reduces surface tension, and results in grossly recognizable atelectasis and variable degrees of ventilation-perfusion mismatch. 2 Additionally, meconium acts like other large inhaled particulate matter, in that it partially occludes airways, and traps air during expiration. 9
Calves, piglets, and foals are the veterinary species studied most commonly in meconium aspiration trials. In one study of 52 calves that died within 2 wk of birth of either infectious or noninfectious causes, 42% had evidence of meconium, squamous cells, and/or keratin in the lungs. 8 Gross appearance varied from normal, to mild suppurative bronchopneumonia, to multifocal lobular atelectasis. 8 Microscopic changes were most consistent with mild diffuse alveolitis with exudation of a few neutrophils and fibrin. In cases considered to be of longer duration, macrophages and multinucleate giant cells predominated around meconium particles,1,8 as in our case. Older calves that died in the study 8 had lesser amounts of meconium visible in the lungs, suggesting that, with time and no new exposure, they may be able to clear meconium through mucociliary clearance and/or phagocytosis. 8 Additionally, aspiration of squamous cells and keratin alone produces at most a limited suppurative-to-granulomatous inflammatory reaction, because the sloughed cells are recognized as originating from self, are less irritating, and do not contain substantial amounts of pro-inflammatory cytokines. 8
Piglets, both stillborn and live-born, have been reported to have meconium staining of the skin and/or haircoat. 1 One study suggested that the degree of meconium skin/haircoat staining was directly related to prior hypoxia. 1 Umbilical cord rupture was identified more commonly in piglets with meconium staining (33%) than those without (10%), and hence it was considered the most likely species-specific cause of periparturient hypoxia. 1 Chronicity of meconium aspiration was based on tissue reaction in that there was typically no meconium found in piglets surviving 1–3 d, free meconium in alveoli in piglets surviving 4–7 d, and engulfed meconium in macrophages and multinucleate giant cells, as in our case, in piglets surviving 8–15 d. 1 If one can extrapolate from the piglet study, this may suggest that meconium aspiration subsequent to hypoxia occurred at least a week before birth in our cases. As no other obvious periparturient lesions were identified in our goat kids, it would have been very interesting to evaluate the umbilical cords and placental membranes in these cases.
Movement of meconium-contaminated amniotic fluid via cilia to the level of the middle ear to induce otitis media has not been reported in the veterinary literature, to our knowledge. There are a few reports in human infants with MAS.3,5–7,9 The link between meconium contamination at birth on development of perinatal and juvenile recurrent middle ear infections in humans is controversial. Conflicting reports suggest that human infants born through meconium-contaminated fluids have increased risk and incidence of ear infections in the first 2 y of life, 7 whereas other authors suggest that there is no increased incidence. 6 In one study of human infants, 55 of 130 had meconium aspiration pneumonia and 17 of 130 had otitis media, suggesting that the latter is a much more rare manifestation of MAS, and unlike the pulmonary lesions, much less likely to result in mortality. 3
Two forms of otitis media are reported in human infants: those with suppurative inflammation alone and those with suppurative inflammation in addition to luminal meconium debris. 3 The goat kids of our study had aspirated squamous cells and/or keratin within the middle ear, rare meconium debris, and an inflammatory response. Of interest, all cases of otitis media associated with aspiration of meconium-contaminated amniotic fluid in infants, showed evidence of infection elsewhere, mainly pneumonia or meningitis. 3 In those affected tissues, the cultured organisms were gram-negative bacteria. Although the presence of culture-positive organisms might support bacteremic spread to the middle ear, it would be an unusual location for ultimate colonization. Additionally, the presence of inflammatory exudate and meconium debris within the middle ear may support the growth of bacteria as part of either a concurrent bacteremia, or from an infection ascending the auditory tube. Aseptic collection of a bacterial culture sample from the middle ear may enhance chances of culturing causative bacteria for either local or systemic disease or both, given that the site is remote and can be approached with sterile technique. Unfortunately, our cases were not cultured and the possibility of concurrent bacteremia cannot be ruled out.
Application of special light microscopic stains for bacteria and mycobacteria were negative, and hence bacteria were not strongly considered as contributing to the inflammatory tissue response in our cases. Intact auditory membranes do not hinder ascent of bacteria into the middle ear, and hence migration through the auditory tube may act as another possible means of bacterial spread and colonization. 3 Once in the middle ear, the irritating properties of meconium may account for metaplastic epithelial changes noted along the lining epithelium 3 as in our case, and in rare instances to auditory loss. 5
Footnotes
Acknowledgements
We thank the histologic lab at UT-CVM for the processing of tissues, special stains, and immunohistochemistry. Additionally, Joy Chambers provided excellent graphic arts support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
