Abstract
Interstitial lung diseases of sheep and goats, which are caused by a range of infectious, parasitic, and toxic agents, have substantial negative health and welfare impacts globally. Within this category of pulmonary disease, entities such as peste des petits ruminants (PPR) can undermine the livelihood of farming communities in sub-Saharan Africa and Southeast Asia; enzootic pneumonia, maedi, and ovine pulmonary adenocarcinoma compromise the productivity of farm enterprises where sheep are housed for prolonged periods. I detail the pathogenesis and lesions caused by a range of viral, bacterial, parasitic, and toxic agents that target the pulmonary interstitium in small ruminants, ultimately resulting in parenchymal damage and clinical disease. These lesions range from the progressive distortion of alveolar walls by infiltrating lymphocytes and macrophages following small ruminant lentiviral infection, to the acute impact of alveolar septal thromboembolism in Bibersteinia trehalosi infection, and eosinophil-mediated necrosis of alveolar walls triggered during the migratory larval stages of parasitism by Dictyocaulus filaria. In addition, I review the pathologic impact of neoplastic type II pneumocytes extending over the interstitial scaffold in cases of sheep pulmonary adenocarcinoma (jaagsiekte) and the toxic injury induced by plants (Trema and Crotalaria sp.) and other toxins (carbolic dips, 3-methyl indole) on the pulmonary interstitium.
Interstitial lung disease (from inter “between” plus sistere “to stand” or “stand between”) describes the consequence of injury to the cells and structures supporting or partitioning the lung (i.e., the pulmonary interstitium). The term thus includes tissue injury affecting or arising from the alveolar and/or interlobular septa caused by a range of pathogens and injurious processes. The initiating injury impacts the interstitium via the hematogenous or aerogenous routes. The resulting inflammation then can lead to overlapping acute-exudative, organizing-subacute, and fibrotic-chronic phases.
18
Degeneration and/or necrosis of the constituent layers of the alveolar wall activates inflammation that varies in type (e.g., lymphoplasmacytic, granulomatous, eosinophilic) depending on etiology. The associated term bronchointerstitial pneumonia is variously used to describe diseases in which airway epithelial necrosis accompanies injury to alveolar walls or in which mononuclear leukocytes infiltrate alveolar septa and circumscribe airways as the bronchial- and bronchiolar-associated lymphoid tissue (
Interstitial lung disease of small ruminants has substantial economic impact: globally, peste des petits ruminants (
Viral causes
Small ruminant lentiviruses
Closely related small-ruminant lentiviruses (
Small ruminant lentiviral infection of sheep: ovine progressive pneumonia
OPP, also known as maedi or lymphoid interstitial pneumonia, is a chronic, progressive respiratory disease of sheep that occurs worldwide, although many island states, such as Iceland, New Zealand, and Australia, are free of the causative virus. Natural transmission typically occurs among sheep >1-y-old through the inhalation of nasal secretions following prolonged close contact, as occurs during winter housing. 11 In addition, vertical transmission of infection occurs in lambs by ingestion of infected colostrum and milk.2,7 Epidemiologic modeling estimates that transmission rates amongst housed sheep are ~1,000 fold higher than among pastured sheep, in which transmission is considered negligible. 38 Ewes kept entirely at grass are unlikely to experience transmission frequently enough for infection to persist, and infection in flocks should dissipate as older ewes are replaced. In-utero infection is thought to be of minor epidemiologic significance.
Infection is characterized by both long immunologic and clinical latency resulting in the highest prevalence of clinical disease in animals >3-y-old. 38 Several weeks of viremia follow in sheep that fail to eliminate infection: >8 wk are required for seroconversion, which reflects ongoing slow build-up of viral antigen, rendering OPP both difficult to detect and control. There is a further delay before an animal becomes infectious: this suggests that transmission occurs when a particular threshold level of “free” virus is attained. This may also imply increasing infectiousness as the disease progresses. 38
Infected lungs fail to collapse, are heavy, diffusely pale-gray-to-tan, firm-to-rubbery, and frequently feature costal imprints. Pale-gray-to-tan mottling is also visible on sectioning, with bulging cut surfaces (
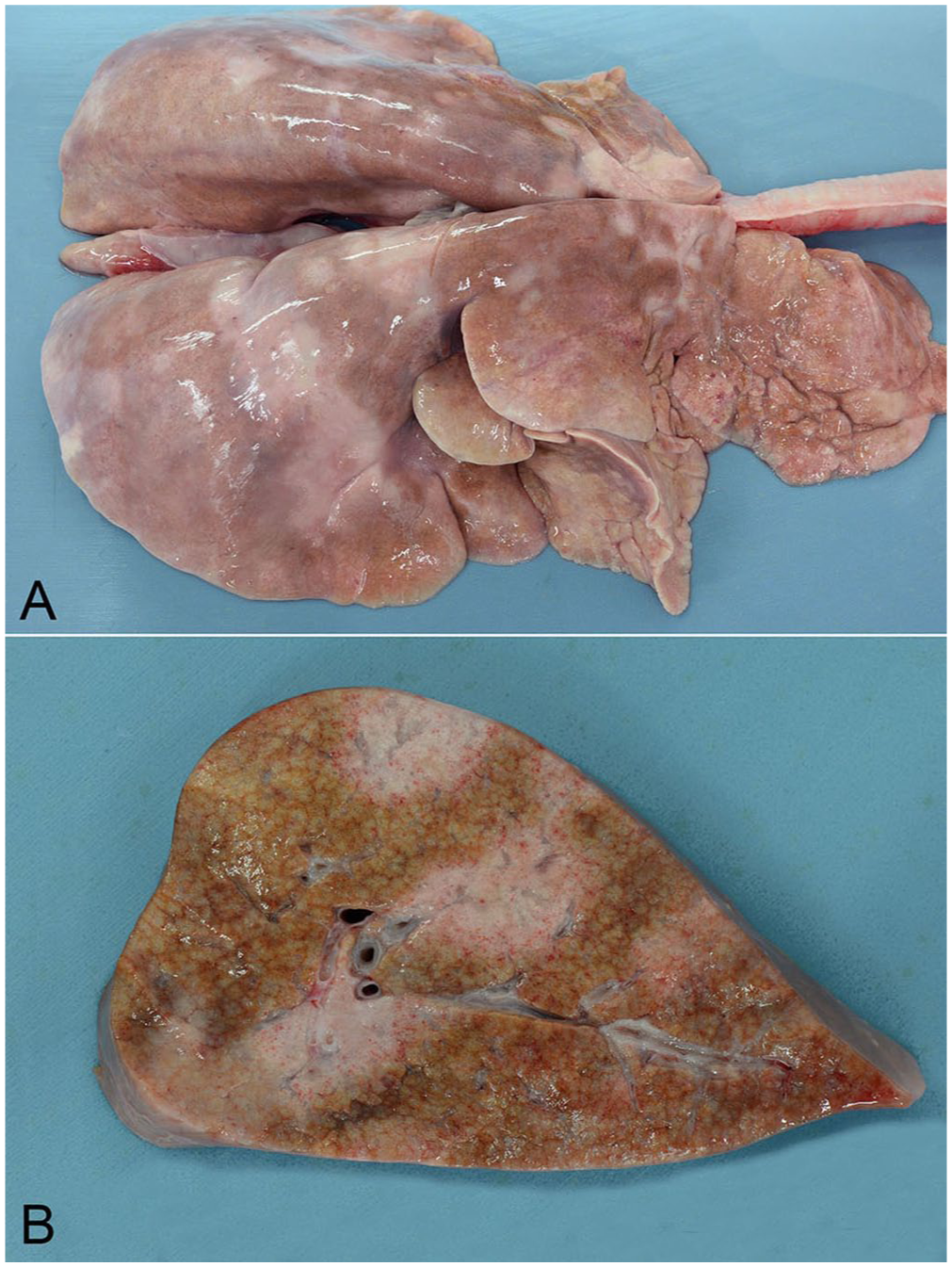
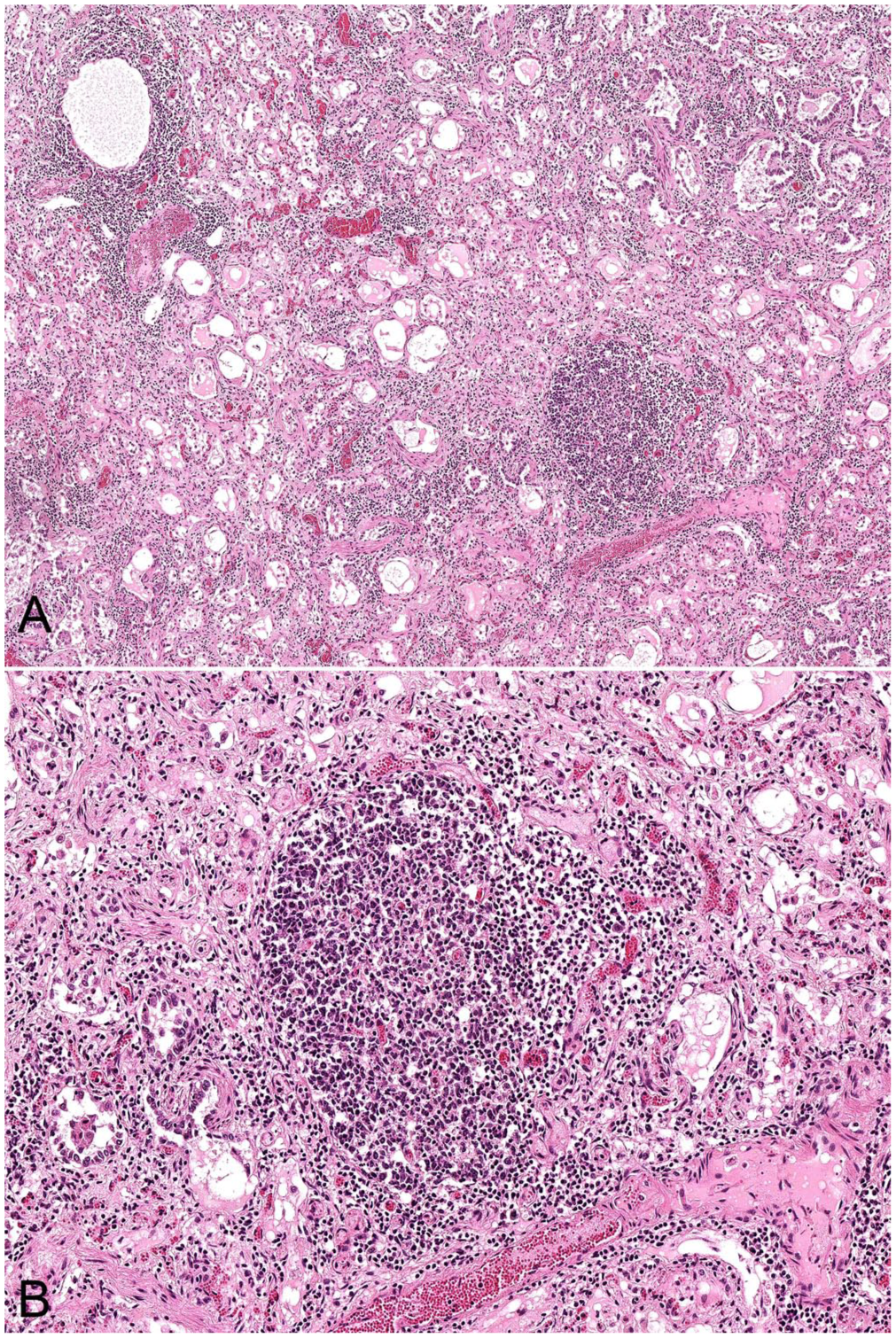
Microscopic appearance of the lungs of a ewe with ovine progressive pneumonia (maedi). H&E.
Once VMV enters a cell, single-stranded RNA is transformed into double-stranded DNA (provirus) using reverse transcriptase and is then inserted into the genome of the host cell, producing lifelong persistent infection. Viral RNA and proviral DNA can be detected in a broad spectrum of cells. However, complete viral replication and assembly are likely to occur only in mature macrophages in target organs such as the lung. Other cells may persistently harbor virus in a latent state in which viral antigens are not produced in sufficient quantities to be detected by the host immune system. Given that VMV is macrophage-tropic and not T lymphocyte–tropic, immunodeficiency does not result. 30
Blood-borne infected macrophages can infect myeloid or stromal cells in the bone marrow, which facilitates ongoing production of infected monocytes, resulting in persistent life-long infection. 27 Immature macrophages thus act as “Trojan horses,” carrying the virus to target organs while allowing the virus to escape host immunity. Hematogenous dissemination of the virus occurs, mainly to the lungs within immature macrophages. Once these macrophages mature within the lung, they produce cytokines, such as interleukin (IL)8, in response to ongoing expression of env- and gag-encoded viral proteins. Production of type I and type II interferons by activated T lymphocytes stimulates ongoing lymphoid recruitment within the lung interstitium. 27
Breed differences in susceptibility to infection or genetic resistance are recognized: the seroprevalence in one study was 77%, 65%, and 15% in purebred Finn, Texel, and Suffolk sheep, respectively. A separate serologic study inferred that Basque dairy sheep had a particular genetic susceptibility. 36
Small ruminant lentiviral infection of goats
Chronic interstitial pneumonia in goats infected with SRLVs is much less common than its ovine counterpart. However, reports describe peribronchial and perivascular lymphoid cuffing, accumulations of homogeneous eosinophilic material in alveolar spaces, type II pneumocyte hyperplasia, and thickening of alveolar septa as a result of lymphocytic infiltration following infection.23,62 These animals were 5-mo to 3-y-old and had developed tachypnea, weight loss, and exercise intolerance. Mild chronic interstitial pneumonia has been reported in 58% of goats with SRLV antigen detected in affected lung tissue by immunohistochemistry. 47 A positive correlation was found between a diagnosis of chronic interstitial pneumonia and other pulmonary lesions, such as purulent bronchopneumonia and fibrinous pleuropneumonia. 47
Ovine pulmonary adenocarcinoma
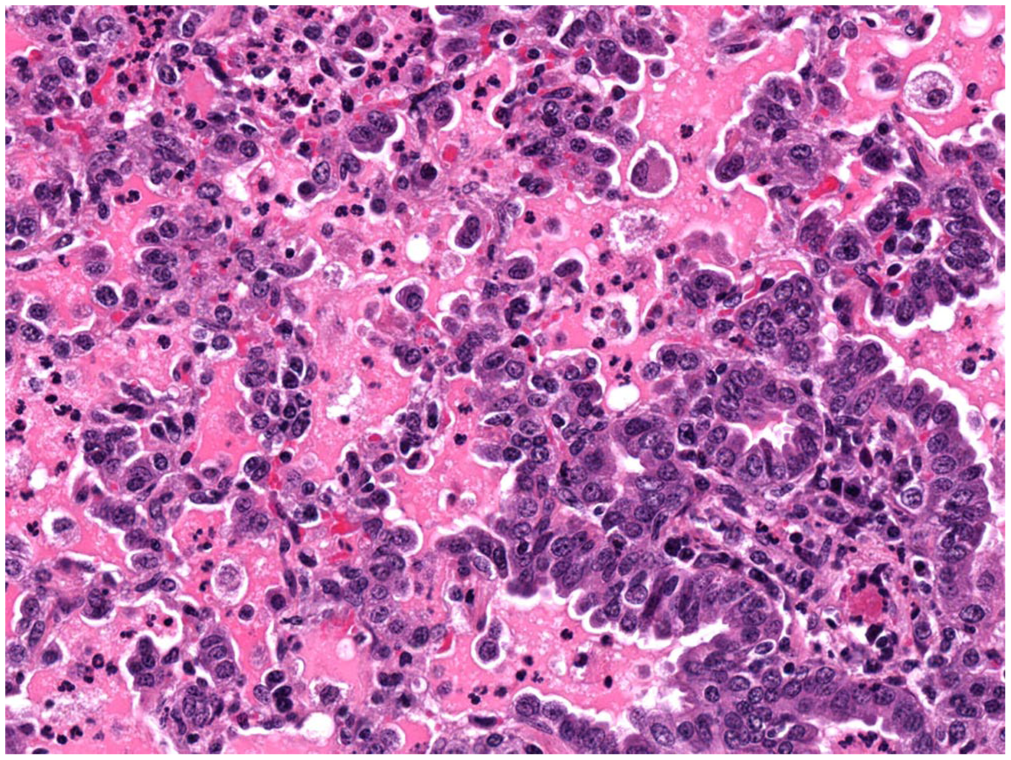
This invasive, well-differentiated adenocarcinoma of sheep (and rarely goats) is an interstitial lung disease in that cuboidal-to-columnar type II pneumocytes, which have undergone neoplastic transformation, extend along alveolar septa on a supporting fibrovascular scaffold infiltrated by leukocytes, with occasional nodules of loose mesenchymal tissue (also known as myxoid growths).32,64,71 This initial lepidic growth frequently progresses to acinar and/or papillary dominant patterns.
71
Grossly, the tumor is seen as locally extensive or firm gray nodules (
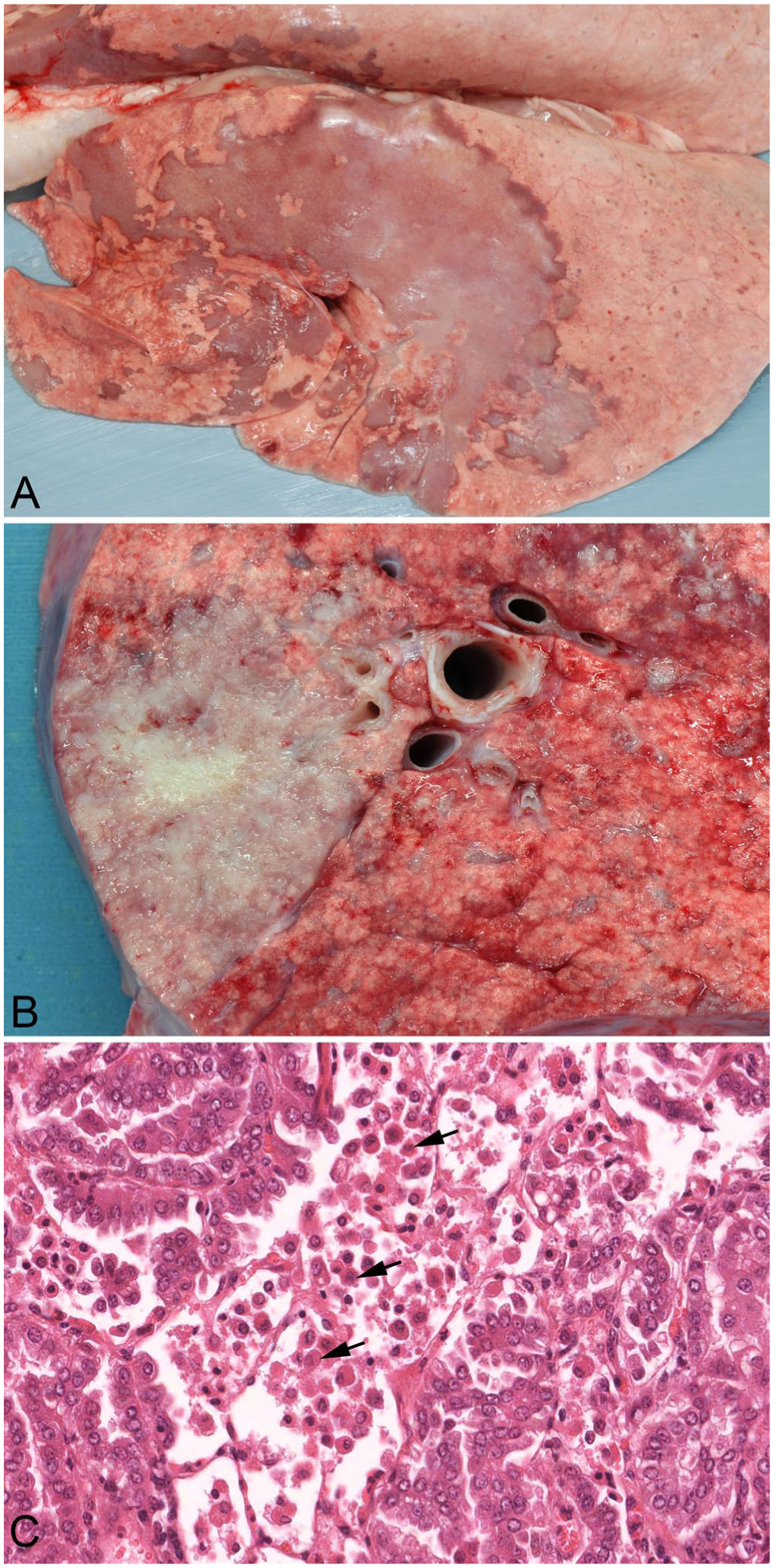
Ovine pulmonary adenocarcinoma in a 3-y-old ewe.
Alveoli adjacent to the tumor may become atelectatic, and development of the neoplasm may be compounded by SRLV or bacterial coinfections, necessitating careful histologic assessment of cases. Contemporaneous SRLV infection results in various degrees of interstitial lymphoplasmacytic infiltration, lymphoid hyperplasia, and fibromuscular hyperplasia in peritumoral regions.19,21,37 Coinfection rates of these retroviruses are estimated as 6–48%; the infection ultimately results in increased morbidity and mortality.29,37,58,67 This pathogenetic synergism between SRLVs and oncogenic retroviruses is hypothesized to result from: 1) increased lateral transmission of SRLV in coinfected animals resulting from the excessive production of lung fluid and thus expired infectious aerosol, 2) enhanced SRLV replication in the increased numbers of peritumoral intra-alveolar macrophages (
Peste des petits ruminants
PPR, also termed “goat plague” or “ovine rinderpest,” is a highly contagious and rapidly fatal virus-induced bronchointerstitial pneumonia of goats and sheep. In general, goats are more severely affected than sheep and morbidity and mortality rates can reach 90%.42,66 The disease has a major impact on the livelihood of farming communities in sub-Saharan Africa, the Arabian peninsula, and Indian subcontinent. 26 The PPR virus (PPRV; family Paramyxoviridae, taxon species Morbillivirus caprinae) is closely related to rinderpest virus, and the resulting lesions in sheep and goats are similar to those of rinderpest.
Aerosol transmission of this labile virus requires close contact, and outbreaks are commonly linked to movement of new animals into flocks. Infection via the oropharynx results in PPRV replication in draining lymph nodes and viremia resulting in the targeting of susceptible epithelia of the respiratory and alimentary tracts over 3–10 d.42,66 Experimental studies indicate that animals can shed virus via respiratory aerosol before the development of clinical signs.
24
In addition to the necrotizing oral and enteric lesions, which resemble those of rinderpest, PPR contrasts with rinderpest in causing fibrinonecrotic tracheitis, fibrinous pleuritis, and bronchointerstitial pneumonia. The pneumonia involves the cranioventral lobes and resembles the infection of dogs by canine distemper virus (another morbillivirus).
15
There is attenuation and necrosis of airway epithelia, type II pneumocyte hyperplasia, and the formation of epithelial syncytia (
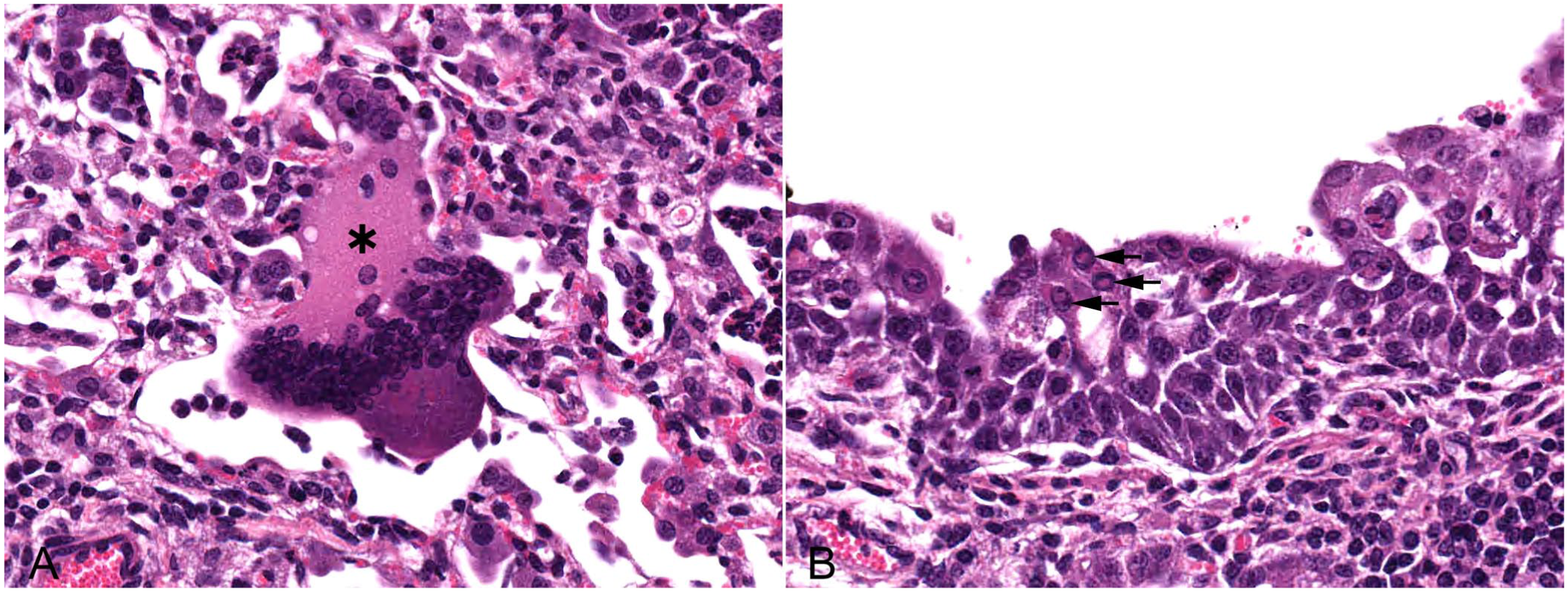
Microscopic appearance of peste des petits ruminants virus infection in the lungs of a 9-mo-old, mixed-breed goat. H&E.
The World Organisation for Animal Health and the Food and Agriculture Organization have set the ambitious target of eradicating PPR by 2030. 26 Factors favoring the achievement of this goal are the availability of efficient and safe live-attenuated vaccines to provide life-long immunity, and specific and highly sensitive detection assays. The absence of a long-term carrier state or of wildlife reservoirs along with lessons learnt from the successful completion of the Global Rinderpest Eradication Programme are further grounds for optimism.26,41
Bovine parainfluenza virus 3 and bovine respiratory syncytial virus infections
Bovine parainfluenza virus 3 (BPIV3; family Paramyxoviridae, taxon species Respirovirus bovis) causes bronchointerstitial pneumonia in lambs. BPIV3 is typically one component of polymicrobial respiratory infections that also include Mannheimia haemolytica and Mesomycoplasma (Mycoplasma) ovipneumoniae. 59 Uncomplicated infection does not appear to be an important cause of morbidity/mortality. 19 The virus replicates in alveolar macrophages, in the cytoplasm of type II pneumocytes, and in airway epithelia. These findings were confirmed by an immunohistochemical study of pneumonic sheep lungs collected from abattoirs in which BPIV3 antigen was found in 19% of lungs, particularly those with interstitial pneumonia. 25 The F-glycoprotein within the BPIV3 viral envelope facilitates pathogen entry through fusion with host cell membranes and the insertion of viral glycoproteins into the membranes of infected cells, which results in cell fusion and, on occasion, the formation of multinucleate cells. Initial inflammation of the bronchial and bronchiolar epithelium spreads to contiguous alveoli with attendant alveolar edema and neutrophilic infiltration. As time progresses, vacuolated and necrotic bronchiolar epithelium responds by proliferating and may feature acidophilic intracytoplasmic inclusions. 19 Atelectasis results from airway obstruction and subsequent changes include type II pneumocyte hyperplasia and accumulation of lymphocytes and plasma cells in alveolar septa and around blood vessels. A key pathogenetic attribute of BPIV3 is its capacity to undermine the bactericidal function of virus-infected alveolar macrophages. 19
When lambs were variously inoculated with bovine respiratory syncytial virus (BRSV; family Pneumoviridae, taxon species Orthopneumovirus bovis), ovine RSV, and human RSV, they did not have the severity of clinical signs or lesions observed in calves infected with BRSV. 4 Lesions following ovine RSV infection of lambs centered on airways (bronchitis and/or bronchiolitis and peribronchiolar lymphoid hyperplasia), but also featured an interstitial component (alveolar septal thickening). 16 Experimental inoculation of 1-wk- and 6-mo-old lambs with BRSV resulted in mild clinical respiratory signs and interstitial pneumonitis. 4 Thus the importance of RSVs as primary respiratory pathogens of sheep remains questionable. Notwithstanding this, lamb models of human RSV infection have been developed to elucidate the cellular and molecular mechanisms occurring in infected human infants. 1
Ovine gammaherpesvirus 2 infection
Ovine gammaherpesvirus 2 (OvGHV2; sheep-associated malignant catarrhal fever virus; family Orthoherpesviridae, taxon species Macavirus ovinegamma2), in addition to causing malignant catarrhal fever (MCF) in cattle, can cause sporadic MCF-like systemic necrotizing vasculitis in sheep, including development of chronic interstitial pneumonia.35,61 Given that sheep are a well-adapted or reservoir host for OvGHV2 and rarely develop disease, confirming a diagnosis of MCF-like disease can prove problematic, requiring the demonstration by qPCR of high viral loads in the tissues of affected animals and the use of in situ hybridization to co-locate viral antigen within lesions. 55 Cases of MCF-like disease occur in sheep from 2.5-mo to 3-y-old. Affected lungs have a diffuse rubbery consistency, costal impressions, and cut surfaces that do not exude fluid. Histologically, chronic interstitial, lymphohistiocytic pneumonia, including type II pneumocyte hyperplasia, is accompanied by vasculitis, vascular proliferation, and thrombosis. 55
Sheeppox
Sheeppox, caused by sheeppox virus (SPV; family Poxviridae, taxon species Capripoxvirus sheeppox), is endemic in the Middle East, Africa, and Asia. Infection affects all age groups of sheep. However, mortality is typically restricted to younger animals, in which rates of up to 50% are recorded.
57
Infection with SPV is by the aerosol route or through skin abrasions and biting flies. Subsequent viremia results in viral localization in the skin and other organs, including the lungs in which variably sized pale-gray firm nodules develop throughout all lobes.9,20 Histologically, the lesion has an interstitial pattern with proliferation of type II pneumocytes and bronchiolar epithelium, along with perivascular and peri-airway lymphoid hyperplasia; infiltration of alveolar walls by neutrophils, macrophages, and lymphocytes; and foci of necrosis (
Proliferation of type II pneumocytes over alveolar walls, with fibrinocellular exudate within the alveolar lumens, of a sheep infected with sheeppox virus. H&E. (From: JPC Systemic Pathology Respiratory System, October 2023, P-V24, with permission; https://www.askjpc.org/vspo/show_page.php?id=TWdYeE9YalcvQXo0L09RTldpVUZZUT09)
Bacterial causes
Mesomycoplasma (Mycoplasma) ovipneumoniae infection
The term bronchointerstitial pneumonia describes diseases in which mononuclear leukocytes infiltrate alveolar septa and circumscribe airways. Mesomycoplasma (Mycoplasma) ovipneumoniae infection causes this pattern of pulmonary inflammation in sheep, resulting in disease that has been variously termed chronic enzootic pneumonia, atypical pneumonia, or chronic, non-progressive pneumonia. A somewhat similar disease presentation occurs in goats.28,54 M. ovipneumoniae can be recovered from the nasal chambers, trachea, and lungs of healthy sheep.
50
Subclinical or mild disease typically targets sheep 1–3-mo-old following the mixing of animals, the maintenance of high stocking densities, and/or overall poor air quality in indoor settings. Concurrent infection with BRSV, BPIV3, or M. haemolytica exacerbates clinical signs. Once infectious droplet nuclei are inhaled into the lower respiratory tract, M. ovipneumoniae adheres to ciliated epithelium, inducing ciliostasis
40
and inhibiting alveolar macrophage phagocytosis
48
dysfunction, as reflected in the accumulation of mucus and inflammatory exudate in alveoli and bronchioles. Infection results in bilateral cranioventral dark-red-to-plum consolidation (
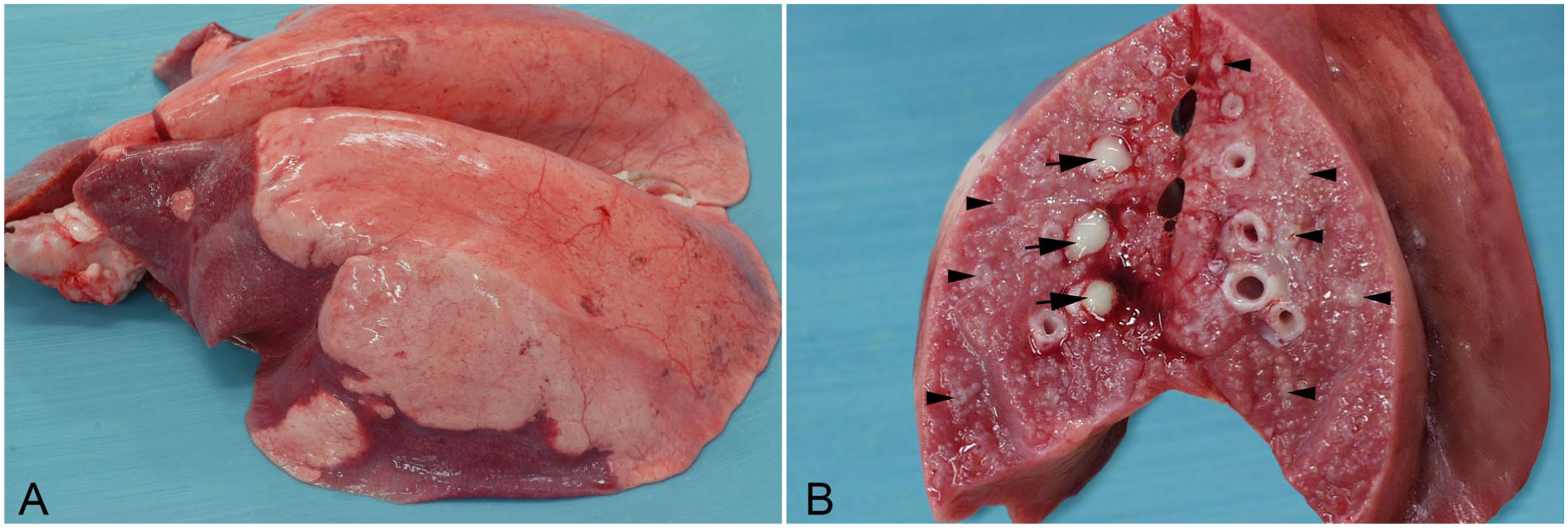
Mesomycoplasma (Mycoplasma) ovipneumoniae infection in a 15-mo-old sheep.
Microscopic appearance of chronic Mesomycoplasma (Mycoplasma) ovipneumoniae infection in the lungs of a 12-mo-old sheep. H&E.
Persistence of M. ovipneumoniae infection in the ovine respiratory tract may be linked to the delayed development of humoral immunity, resulting from frequent variation in the surface lipoprotein antigens of these organisms. Such variation is facilitated by a relatively small genome and propensity for genomic rearrangement. Expression of a polysaccharide capsule may also facilitate immune evasion.
49
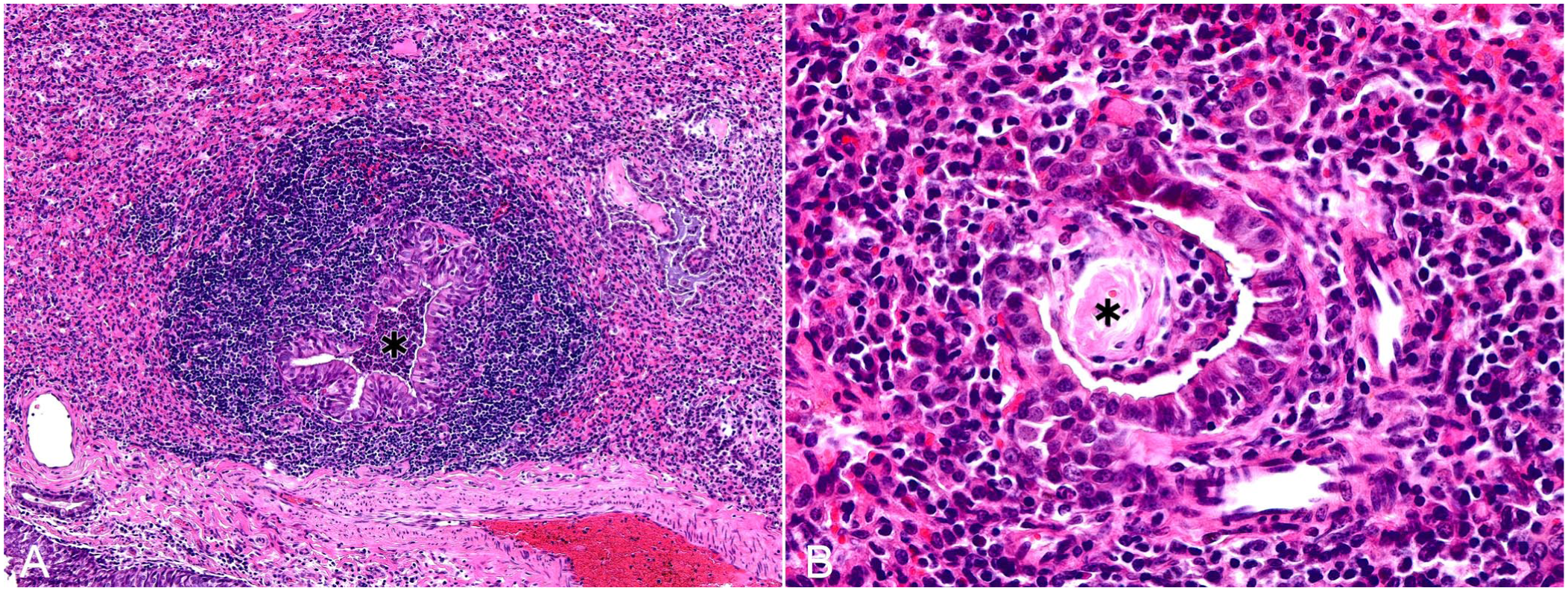
Microscopically, epithelial necrosis and inflammation occur in the airways, with marked circumscribing lymphoid hyperplasia that occasionally contains follicular aggregates. Bronchi and bronchioles are filled with a mixture of eosinophilic proteinaceous exudate, wispy basophilic mucus, and variable numbers of viable and degenerate neutrophils and sloughed epithelial cells. Surviving epithelium is frequently hyperplastic and infiltrated by neutrophils. Leukocytic infiltrates extend into the adjacent interstitium where alveoli are filled with admixtures of neutrophils, macrophages, and rarely multinucleate macrophage giant cells. Alveoli may feature type II pneumocyte hyperplasia. Variously sized nodular accumulations of eosinophilic collagenous material, previously termed hyaline scars, are inconsistently found within bronchiolar walls, resulting in distortion and stenosis of the lumen (
The clinical and economic impact of respiratory disease caused by M. ovipneumoniae is not restricted to domesticated small ruminants, given the population-limiting respiratory disease it causes in free-ranging Rocky Mountain bighorn sheep (Ovis canadensis canadensis) in the western United States. In this setting, ewes serve as a reservoir of infection for young animals.8,46
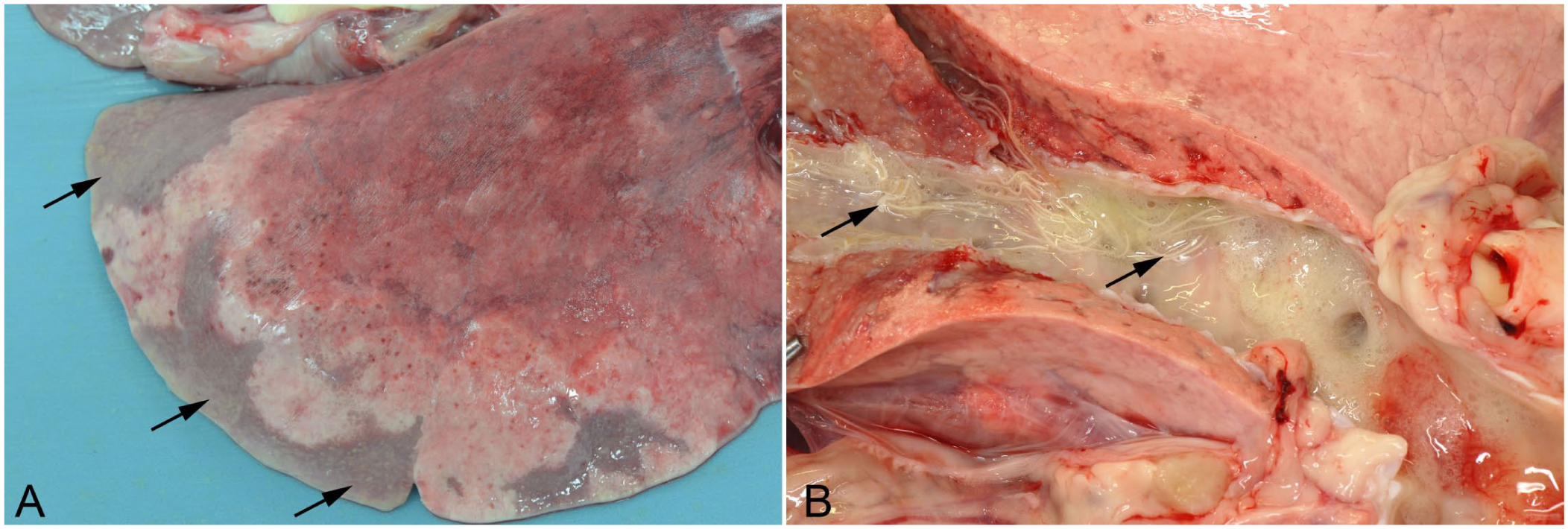
Bibersteinia trehalosi septicemia
Interstitial pulmonary inflammation and thrombosis result from systemic infection of typically weaned (6–10-mo-old) lambs with Bibersteinia trehalosi, although severe outbreaks of disease are described in adult sheep.19,22,63 This opportunistic pathogen resides in the tonsillar crypts and on the respiratory, gastrointestinal, and genital mucosae of sheep. Factors predisposing to the development of systemic infection include stressors, such as changes in pasture, overcrowding, inclement weather conditions, poor ventilation, high parasite burdens, and transportation.19,22,63Affected lambs are found dead unexpectedly, with subcutaneous hemorrhages over the neck and thorax, and ecchymoses over the pleural and epicardial surfaces and diaphragm. The lungs are bilaterally and diffusely congested and edematous with widespread focal hemorrhages and frothy blood-stained fluid in the airways. In addition to the pulmonary lesions, erosions and/or ulcers are found on the tongue, pharynx, and esophagus, with small pale-yellow lesions scattered throughout all hepatic lobes. Thromboemboli containing gram-negative coccobacilli form in the pulmonary blood vessels, resulting in acute necrosis of alveolar septa and fibrin, hemorrhage, and aggregates of degenerate leukocytes filling contiguous alveoli (
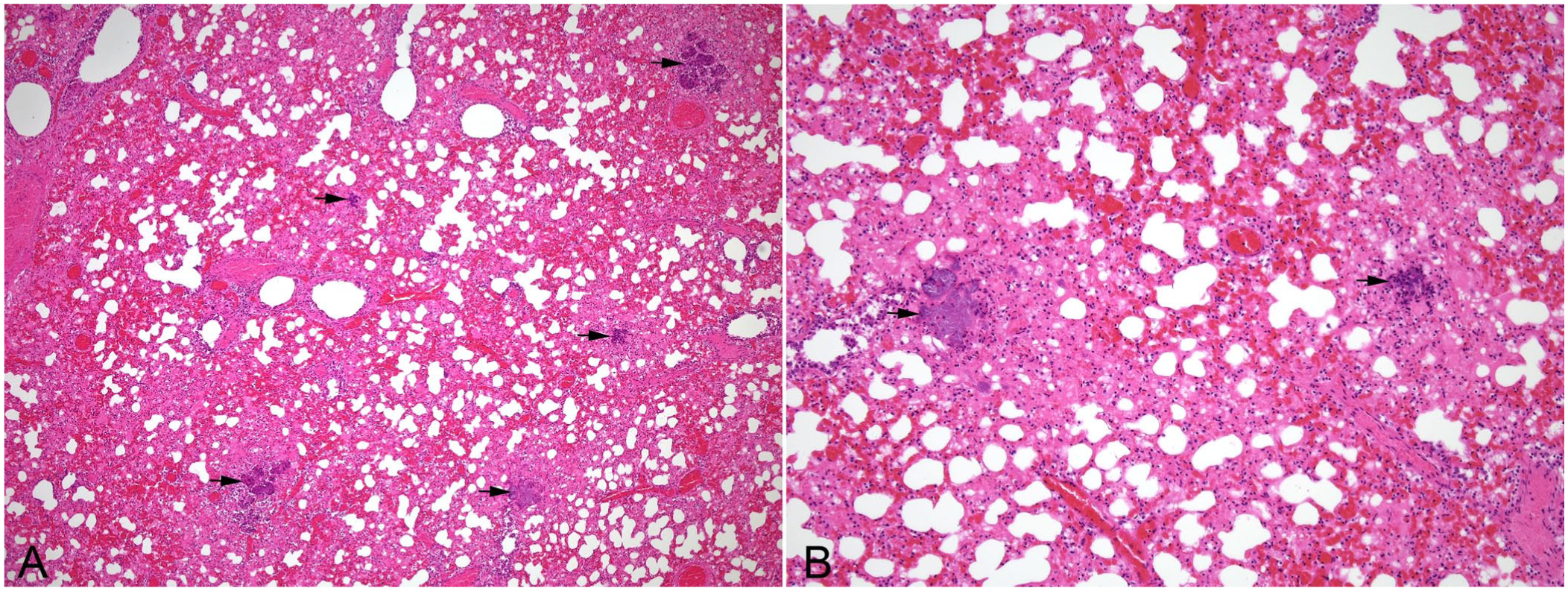
Microscopic appearance of Bibersteinia trehalosi infection in the lungs of a 7-mo-old lamb. H&E.
Parasitic causes
Dictyocaulus filaria
Dictyocaulus filaria is the most pathogenic lungworm of sheep and goats, with goats being more susceptible to infection and often more severely affected clinically than sheep. 69 Severe infections occur in animals <1-y-old following grazing pasture contaminated with infective third‑stage larvae (L3). Lungworm infection in small ruminants is usually less severe than that in cattle, with the clinical signs of coughing, dyspnea, and loss of body condition developing 2–3 wk after infection and largely attributable to the obstruction of bronchi by adults. L3 larvae can overwinter on pasture, and spring pastures contaminated with larvae from the previous year, along with larvae from adults developing from arrested larvae in ewes, are sources of infection for young, susceptible animals. Acquisition of larvae typically results in disease in lambs or kids during the second half of their first grazing season. The development of strong immunity mitigates reinfection of older animals. 53
Ingested L3 larvae ex-sheath in the small intestine, penetrate the mucosa, and migrate to the mesenteric lymph nodes via lymphatic channels. Here they mature to the L4 stage before migrating to the lungs via lymph and blood vessels. Once they reach the alveolar capillaries within the pulmonary interstitium some 7 d after infection, larvae breakout of the vasculature and enter the airways. They undergo a final molt to the L5 stage within bronchioles, and adults begin laying eggs (patency) ~4 wk after ingestion. Patency usually lasts 1–3 mo, with peak larval output occurring 39–57 d after infection. 53 The interstitial nature of this infection is to the fore during the prepatent phase at 7–25 d post-infection, during which time L4 larvae exit pulmonary capillaries and enter alveoli and bronchioles. This disruption and necrosis of alveolar walls activates adjacent pulmonary epithelial cells to secrete alarmins, leading to the production of cytokines and chemokines, such as eotaxin, which in turn attract eosinophils, neutrophils, and macrophages. 18 Alveolar septa are then thickened by hyaline membranes, fibrin, and the above leukocytes. Prominent infiltrates of admixed intact and degranulated eosinophils extend from alveolar walls to fill contiguous alveoli and bronchioles with dense eosinophilic granulomas, which retard the migration of some larvae. As time progresses, surviving larvae translocate to bronchioles and bronchi where they mature into adults, surrounded by eosinophil-rich aggregates (eosinophilic bronchitis and bronchiolitis), sloughed and hyperplastic epithelia, circumscribing lymphoid hyperplasia, and smooth muscle hypertrophy. Chitin, shed during molting, acts as a pathogen-associated molecular pattern triggering a host immune response; proteases, glycolytic enzymes, and lectins produced by the parasites directly contribute to tissue injury. 18 Granulomas surrounding nematode eggs and dead larvae, as well as atelectasis and alveolar overinflation/microemphysema, are features of the surrounding parenchyma.
Grossly visible wedge‑shaped, red-to-gray, consolidated lesions occur with patent infection in the caudal aspects of the caudal lobes (
Dictyocaulus filaria infection in a ewe.
Muellerius capillaris
Muellerius capillaris is the most common lungworm of small ruminants. Heavy infections by migrating larvae and adults result in chronic interstitial pneumonia with diffuse infiltration of alveolar septa by leukocytes and intra-alveolar fibrinous exudation.5,51 More typically, rather than causing diffuse interstitial inflammation, M. capillaris infection results in the formation of gray, <1-mm to several cm, subpleural nodules within the dorsal aspects of the caudal lobes (
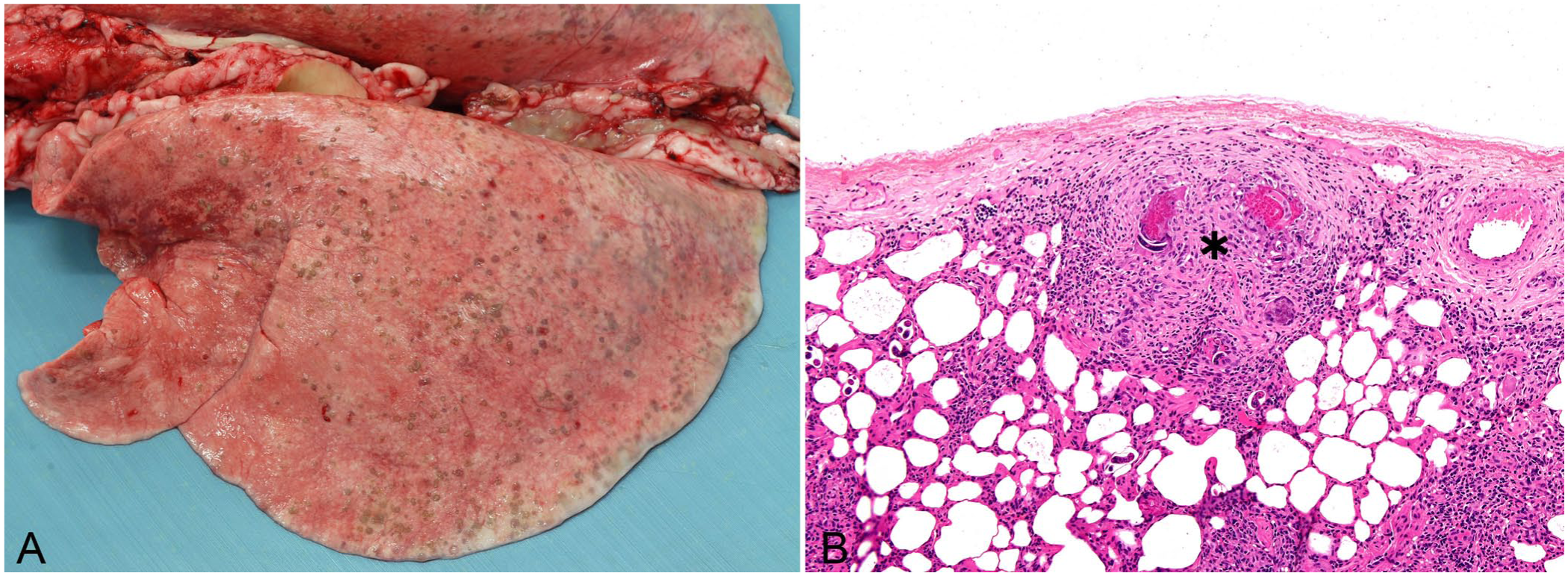
Muellerius capillaris infection in a 4-y-old ewe.
Both sheep and goats become infected most commonly in warmer, more moist geographic regions. Light infection rarely causes clinical disease; reduced weight gain and breeding performance are reported. 6 Rather than having a direct life cycle like D. filaria, M. capillaris requires a gastropod intermediate host in which infective larvae develop. Infection of small ruminants follows inadvertent ingestion of mollusks containing infective larvae or the ingestion of infective larvae that have emerged from the gastropod host. Once they reach the small intestine, larvae penetrate the mucosa, transit to the mesenteric lymph nodes, and migrate via the thoracic duct to the heart and pulmonary arterioles. Larvae and adults are found in nodules in the lungs. Adults are present 25–38 d after infection and may survive and shed larvae for >4 y. 53
Toxic causes
Trema micrantha
Ingestion of the leaves of the tree Trema micrantha (family Ulmaceae) causes pneumopathy in sheep and goats. 48 This tree, also known as the Jamaican nettle tree, is native to tropical South America, Central America, Jamaica, Cuba, Puerto Rico, and southern Florida. The leaves of the plant are palatable to livestock, especially when forage is scarce. Poisoning by this plant typically causes hepatic necrosis in sheep, goats, and cattle. However, a pulmonary form has been described in sheep, resulting in dyspnea, edema of the face, eyelids, and vulva, and subcutaneous emphysema. On postmortem examination, the lungs do not collapse, are diffusely congested and heavy, and feature costal imprints.56,70 Microscopically, injury is focused on the pulmonary interstitium with hyaline membrane formation, along with type II pneumocyte hyperplasia. Mild-to-moderate infiltrates of neutrophils, macrophages, lymphocytes, and plasma cells are noted in alveolar, bronchial, and bronchiolar lumens. 56
It is hypothesized from studies of related plant species that the toxic effect of T. micrantha is attributed to the metabolite(s) of a constituent glycoside generated by either the rumen microbiota and/or hepatic detoxification. These compounds target type I pneumocytes and endothelium, resulting in increased vascular permeability and hyaline membrane formation.56,70 Doses of as little as 20 g/kg liveweight over an interval of 30 d can cause poisoning. 70
Crotalaria pallida
The green leaves of Crotalaria pallida, a species of flowering plant of the family Fabaceae, found in Florida, Puerto Rico, Brazil, and India, cause acute pneumotoxicosis in sheep at a dose of 2.5 g/kg bodyweight. 12 When experimentally administered, sheep develop dyspnea and die within 13–22 h. The main gross lesions are hydrothorax and severe pulmonary and subpleural edema, with alveolar, interstitial, and perivascular edema observed microscopically, along with mild multifocal intra-alveolar infiltrates of neutrophils. This family of plants (which also includes C. dura and C. globifera) contain pyrrolizidine alkaloids, the metabolites of which cause injury to the pulmonary interstitium in other species. 19
Carbolic dip
In the early 1980s, outbreaks of interstitial lung injury were reported in sheep in Scotland 1–5 d after dipping in proprietary carbolic dips. 45 Although a toxic constituent was never elucidated, it was presumed that pulmonary injury resulted from skin absorption of an unknown pneumotoxin. Young, well-conditioned sheep of the Suffolk and Border Leicester breeds were affected most commonly, and grossly lungs were hyperemic and edematous. Damage to alveolar septa along with regeneration in the form of type II pneumocyte hyperplasia were key features. Septal injury included interstitial edema and filling of alveoli with proteinaceous fluid, cell debris, erythrocytes, neutrophils, and macrophages. 3
3-methylindole
Three-methylindole (
Conclusions
Inflammation of the pulmonary interstitium is a well-documented response in small ruminants to a range of pathogens and injurious processes that target alveolar septal epithelium and/or endothelium as well as other supporting pulmonary structures. The attendant lesion, diffuse in distribution, progresses from acute and exudative to chronic and fibrotic in surviving animals. Secondary bacterial infections may exacerbate their clinical impact and obscure their histomorphologic features.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
