Abstract
Over a 3.5-year period, 4 Rocky Mountain goats (Oreamnos americanus), housed at a single facility, developed clinical disease attributed to infection by Caprine arthritis encephalitis virus (CAEV). Ages ranged from 1 to 10 years. Three of the goats, a 1-year-old female, a 2-year-old male, and a 5-year-old male, had been fed raw domestic goat milk from a single source that was later found to have CAEV on the premises. The fourth animal, a 10-year-old male, had not ingested domestic goat milk but had been housed with the other 3 Rocky Mountain goats. All 4 animals had clinical signs of pneumonia prior to death. At necropsy, findings in lungs included marked diffuse interstitial pneumonia characterized histologically by severe lymphoplasmacytic infiltrates with massive alveolar proteinosis, interstitial fibrosis, and type II pneumocyte hyperplasia. One animal also developed left-sided hemiparesis, and locally extensive lymphoplasmacytic myeloencephalitis was present in the cranial cervical spinal cord. Two animals had joint effusions, as well as severe lymphoplasmacytic and ulcerative synovitis. Immunohistochemical staining of fixed sections of lung tissue from all 4 goats, as well as spinal cord in 1 affected animal, and synovium from 2 affected animals were positive for CAEV antigen. Serology testing for anti-CAEV antibodies was positive in the 2 goats tested. The cases suggest that Rocky Mountain goats are susceptible to naturally occurring CAEV infection, that CAEV from domestic goats can be transmitted to this species through infected milk and by horizontal transmission, and that viral infection can result in clinically severe multisystemic disease.
Introduction
Caprine arthritis encephalitis virus (CAEV; family Retroviridae, subfamily Orthoretrovirinae; genus Lentivirus) is a member of a subfamily of small ruminant lentiviruses (SRLVs).1-12 The SRLVs do not cause immunodeficiency but instead cause chronic multisystemic inflammatory disease in their host species, such as goats (family Bovidae, subfamily Caprinae, genus Capra) and sheep (family Bovidae, subfamily Caprinae, genus Ovis). The virus genome integrates into host DNA, causing persistent infection with a tropism for monocytes, macrophages, and dendritic cells.3,7 Mammary gland epithelial cell viral infection is common, and transmission is most often due to ingestion of milk from infected dams. Horizontal infection via direct contact occurs but is less common. 7 The present report involves an instance of CAEV-like infection in Rocky Mountain goats (Oreamnos americanus; family Bovidae, subfamily Caprinae), suspected to have been caused by ingestion of milk from infected domestic goats and by horizontal transmission from infected Rocky Mountain goats.
In early July 2004, an approximately 1-year-old female Rocky Mountain goat (goat 1) was referred to the Oregon State University College of Veterinary Medicine Teaching Hospital (Corvallis, Oregon) with a 3-week history of respiratory distress, exercise intolerance, and weight loss. The goat had been treated the previous November for pneumonia and was reported to be small for its age. The current respiratory condition had not responded to treatment with multiple antibiotics. Physical examination revealed tachypnea (R = 120 bpm), increased lung sounds bilaterally, and generalized muscle wasting. Arterial blood gas as compared to normal domestic goat values revealed respiratory acidosis (pH 7.245; normal range: 7.350–7.450) with metabolic compensation (pCO2 79.7, normal range: 35.0–45.0; HCO3 33.8, normal range: 24–26; tCO2 36.2, normal range: 25–28), hypoxia (pO2 23.4, normal range: 75–100), hyperkalemia (7.32, normal range: 3.5–6.5), and hyperglycemia (122, normal range: 50–75). The animal was treated with antibiotics and intranasal oxygen. Thoracic radiographs showed a marked generalized interstitial lung pattern that appeared worse dorsally, most consistent with viral proliferative or progressive pneumonia. Four days later, the arterial blood gas was repeated and revealed hypercapnia (pCO2 53.5, normal range: 35.0–45.0), hypoxia (pO2 29.9, normal range: 75–100), hyperglycemia (122, normal range: 50–75), and hyperlactemia (10.36, normal range: 0.5–2.0). A complete blood cell count was performed. A mild leukocytosis, compared to normal domestic goat values, was present (total white blood cell count 14,900/µl, normal range: 4,000–13,000/µl) with a mild neutrophilia (9,685 neutrophils/µl, normal range: 1,200–7,200/µl). Morphologically, 1+ reactive lymphocytes were reported. A final diagnosis of chronic pneumonia of unknown etiology was given. The patient was discharged on continued antibiotic therapy with added nonsteroidal anti-inflammatory medication and continued nasal insufflation of oxygen. The animal continued to deteriorate over the next 3 weeks and was euthanized and presented for necropsy at the Oregon State University Veterinary Diagnostic Laboratory.
An approximately 2-year-old male Rocky Mountain goat (goat 2) developed signs of pneumonia, including weight loss and rapid breathing and a temperature of 39.6°C. The animal was placed on antibiotics with no improvement. One month after initial presentation to the referring veterinarian, this goat developed right-sided hemiparesis. Neither the chronic pneumonia nor the paralysis responded to antibiotic therapy; thus the goat was euthanized and presented for necropsy in September 2004. Both goats 1 and 2 had been acquired as kids and raised on raw domestic dairy goat milk from a local goat dairy. This source domestic goat flock was thought to be CAEV-free at the time these Rocky Mountain goat kids were being bottle-fed, but CAEV was confirmed in the flock by enzyme-linked immunosorbent assay (ELISA) of serum during a 2004 investigation.
A 5-year-old male Rocky Mountain goat (goat 3) had also received milk from the same domestic goatherd. The goat was presented for necropsy in 2007 following several weeks of dyspnea that was unresponsive to antibiotic therapy. In January 2008, a 10-year-old male Rocky Mountain goat (goat 4) from the same facility was presented for necropsy after being recumbent for 1 week with signs of pneumonia. This goat had been treated the previous July and August for pneumonia. This buck had not received milk from the CAEV-positive domestic goatherd but had been housed with goats 1–3.
Gross necropsy findings of goats 1–3 included poor body condition, with decreased body fat stores or serous atrophy of fat. Body condition of goat 4 was thin but with adequate body fat stores. The lungs of goats 1, 3, and 4 failed to collapse as indicated by rib impressions on the pleural surfaces and were diffusely mottled red and purple, firm, and meaty (Fig. 1). Gross pulmonary lesions were not detected in goat 2. In goats 3 and 4, red-tinged thoracic fluid was present (600 ml to 1 liter), and multiple, prominent firm, meaty, pale tan to white, 1–2 cm in diameter nodules were present in the caudal lung lobes. Some of the nodules had a slightly depressed center and were variably mineralized. The pleural surfaces of the lungs had multiple white plaques up to 4 cm in diameter that extended 1 mm into the underlying parenchyma (pleural fibrosis). The cranioventral portions of the lungs were consolidated, meaty, and red to purple. The tracheobronchial lymph nodes were enlarged.
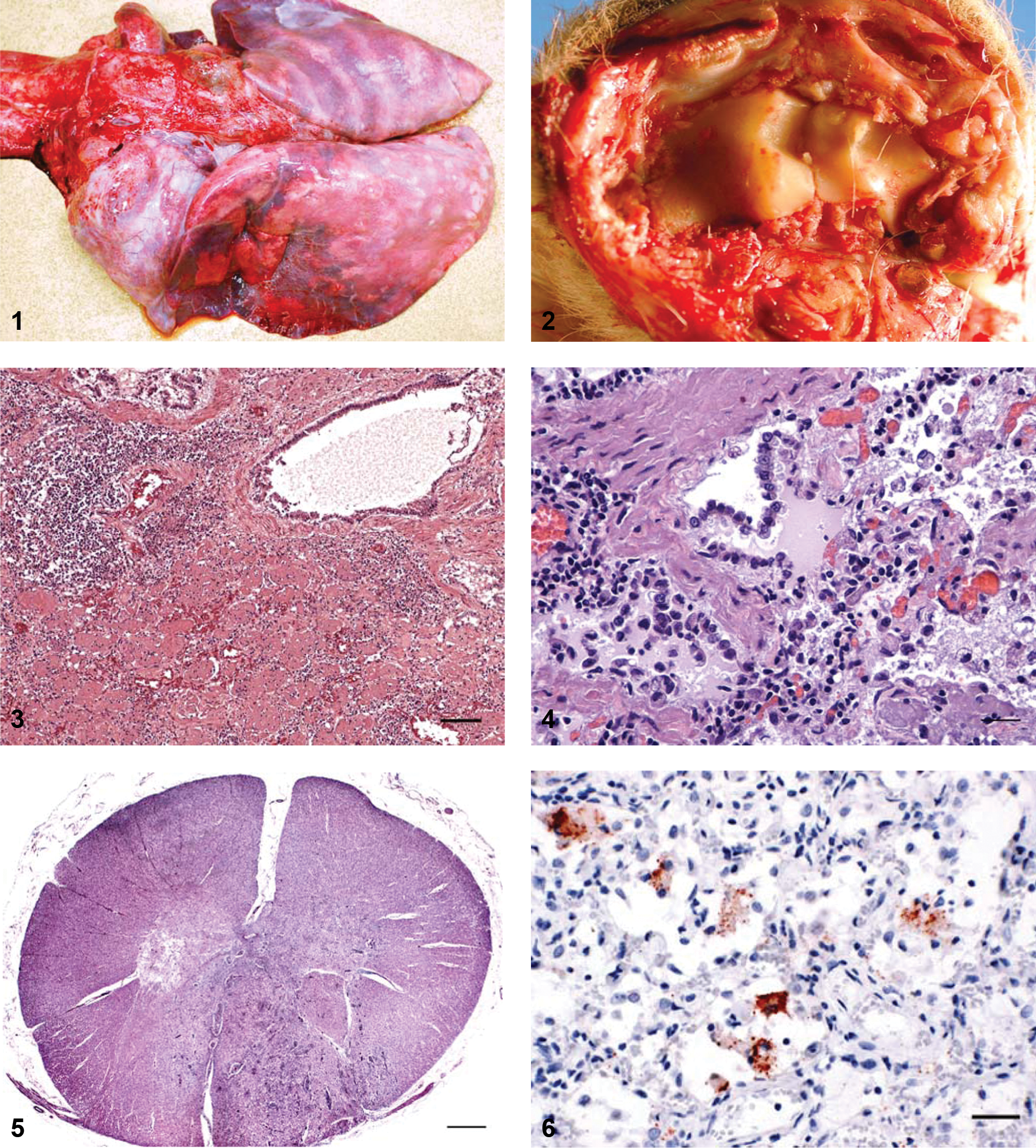
Rocky Mountain goat (Oreamnos americanus), goat 3. Gross image of lungs with diffuse, severe, interstitial pneumonia; cranioventral consolidation; and rib impressions.
Joint lesions were present in goats 3 and 4. In goat 3, the right hock was swollen, and the periarticular tissues were hemorrhagic and contained clots of fibrin. The synovium was thickened, and the joint fluid was blood-tinged. The carpal joints of goat 3 were grossly normal, but both carpi of goat 4 were swollen, and the articular cartilage of the right carpus was diffusely, dull, roughened, and tan (Fig. 2). The medial side of the joint contained brown–green friable casts of material that expanded the joint space and extended along the bursa of the tendon sheaths. The joint capsule was severely thickened by firm, white, fibrous tissue rimming prominent papillary projections of synovium into the joint. The left carpus, stifles, hocks, and fetlock joints had similar lesions. Joints examined in goats 1 and 2 were grossly normal.
Histopathological lesions in the lungs of all animals were variable with the most typical changes consisting of thickened alveolar septa, mainly due to fibrosis, and deposition of intra-alveolar amphophilic to eosinophilic fibrin-like material with a few entrapped macrophages (Figs. 3, 4). Perib ronchiolar lymphoplasmacytic infiltrates or interstitial nodular aggregates of lymphocytes (Fig. 3) were present in some sections from all 4 animals. Foci of type II pneumocyte hyperplasia were also visible, mainly in areas in which the alveoli were not occluded by the fibrin-like material (Fig. 4). Foamy intra-alveolar macrophages were often present, but neutrophils were uncommon.
Multiple sections of spinal cord from different levels of goat 2 were examined microscopically. An area within the cranial cervical spinal cord contained prominent lymphoplasmacytic inflammation with necrosis and reactive astrocytes (gemistocytic astrocytes), most prominent within the white matter of the ventral tracts, with some extension into the ventral horn gray matter (Fig. 5). In other areas, there were irregular zones of severe parenchymal rarefaction with numerous gemistocytic astrocytes. Neurons within affected gray matter were generally intact. The most cranial cervical spinal cord section had no significant lesions, and there was no inflammation or necrosis in sections from lower levels of the spinal cord. There were no lesions in the nerve roots or ganglia, nor were there lesions in sections of axillary nerve, sciatic nerve, brainstem at the level of the obex, cerebrum, or midbrain through the rostral colliculi of this animal. Sections of the central nervous system from goats 3 and 4 lacked microscopic lesions. Central nervous system tissue from goat 1 was not examined microscopically.
Histopathologic findings in synovium from goats 3 and 4 were similar. The joint capsule was severely thickened by loose connective tissue containing proliferative, branching blood vessels with extravascular fibrin and foci of necrosis, some of which were lightly mineralized. A leukocytic vasculitis (mostly mononuclear cells) affected some arteries, and there was multifocal thrombosis of several arterioles and capillaries. The synovial membrane contained loosely packed plasma cells and lymphocytes and was variably ulcerated and replaced by a thick mat of hypereosinophilic material admixed with cellular debris.
Mycoplasma cultures performed on the lung of goats 1, 3, and 4 and on the bronchial lymph node of goat 1 were negative. Fluorescent antibody testing for Bovine herpesvirus 1 (formerly known as infectious bovine rhinotracheitis virus) and Bovine viral diarrhea virus was performed on lung and lymph node of goats 1 and 2, and results were negative. Thoracic fluid collected at necropsy from goats 3 and 4 was positive for CAEV antibodies by monoclonal antibody gp135 SU competitive ELISA (cELISA), a as previously described. 4 Paraffin-embedded tissue blocks were submitted to the California Animal Health and Food Safety Laboratory (Davis, California) for immunohistochemistry (IHC) for CAEV. Sections of lung tissue were submitted from all 4 goats. Sections of the lesion within the cranial cervical spinal cord were submitted from goat 2. Sections of synovium were submitted from goats 3 and 4. All samples were sectioned at 3–4 µm, mounted on charged slides, and autoclaved at pH 6.1 in a citrate-based antigen retrieval procedure. b Nonspecific protein binding was blocked by applying 1% casein to the slides prior to application of the primary monoclonal CAEV antibody a CAEP10A1, which reacts to CAEV-60, CAEV-Co, and Visna/maedi virus but not ovine progressive pneumonia virus, followed by biotinylated goat anti-mouse immunoglobulin c and avidin–biotin complex with horseradish peroxidase and 3-amino-9-ethylcarbazole chromogen. c Controls included nonspecific monoclonal mouse antibody d as a negative control and CAEV-infected tissues as positive control. In all 4 goats, IHC was positive in all tissues submitted (Fig. 6). For additional validation, a section of lung from a Rocky Mountain goat from this herd with Arcanobacterium pyogenes pneumonia was tested and was negative for CAEV by IHC. This goat had not received milk from the domestic dairy goat milk source, nor had this goat been housed with any of the other Rocky Mountain goats in the present study.
Lentiviruses, including all SRLVs, have previously been thought to form 2 distinct groups evolving independently in sheep or goats; however, studies suggest that field isolates from various geographical regions exist as a genetic continuum with viruses that do not simply cluster according to the host species from which they were isolated. 6 There is, in fact, phylogenetic reconstruction evidence that supports the existence of SRLV cross-species transmission in domestic and wild ruminants.6,11 This premise is supported by experimental infection of Mouflon sheep, viral recovery from wild ibexes,3,6 and in the current report of disease compatible with CAEV infection, confirmed by CAEV cELISA and CAEV IHC, in Rocky Mountain goats. Infection with Visna/maedi virus cannot be completely ruled out in the current case given that the CAEV IHC antibody used is cross-reactive with Visna/maedi virus, and thus further cases should be analyzed with more specific polymerase chain reaction testing protocols.
The main routes of transmission of the SRLVs are through ingestion of infected colostrum and milk and through the inhalation of respiratory secretions.1,6,9,10 Epidemiologic features in the current study support both horizontal transmission of CAEV between domestic dairy goats and Rocky Mountain goats via consumption of infected domestic goat milk and horizontal transmission between cohabitating animals. However, the possibility of horizontal transmission from goat 4 to the other 3 goats without transmission via the milk cannot be completely eliminated. Most CAEV infections are subclinical; thus, similar to most other retroviruses, animals may develop clinical signs after many years of incubation and may shed low levels of the virus undetected.
Clinical disease due to CAEV is caused by chronic debilitating inflammatory disease due to persistent infection of mainly cells of the monocyte–macrophage lineage.6,7 Clinically, infection is most often associated with chronic progressive arthritis in adult goats and leukoencephalomyelitis in young kids 2–4 months of age.2,5,8 Spastic hemiparesis due to unilateral cervical spinal cord lesions, as seen in goat 2, are a common presentation of CAEV infection in goats. 13 Infection can also cause interstitial pneumonia and mastitis.2,5 No mammary gland involvement was detected in the only doe examined in the present study, but she was a nulliparous animal, and microscopic examination of the mammary gland was not performed. All other typically reported manifestations of CAEV infection were exhibited by the goats in the current tudy.2,5,7,8 All 4 goats presented for clinical signs of chronic pneumonia; 1 young animal (goat 2) manifested with typical lesions of CAEV-induced leukoencephalomyelitis, and the oldest animals (goats 3 and 4) presented with concurrent arthritis.
The suspicion that CAEV can infect Rocky Mountain goats has implications for both wildlife resource management and for captive ungulate husbandry. Resource managers must strive to prevent direct contact between domestic and sylvatic goat species to prevent horizontal transmission. Similarly, protocols for the transfer of captive O. americanus between different facilities should include testing for CAEV in addition to routine quarantine procedures.
Footnotes
Acknowledgements
The authors thank Rocky Baker for expertise in CAEV cELISA and fluorescent antibody techniques, as well as Mr. Baker and Peggy Dearing for performing ancillary testing for other pathogens in these cases. The authors also thank the Oregon Wildlife Foundation for historical and clinical information on these animals.
a.
VMRD Inc., Pullman, WA.
b.
BioGenex Laboratories Inc., San Ramon, CA.
c.
Dako North America, Carpinteria, CA.
d.
Vector Laboratories, Burlingame, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
