Abstract
The vector-borne protozoan parasite Trypanosoma cruzi causes Chagas disease in humans, dogs, and many other mammalian hosts. Canine Chagas disease is increasingly diagnosed in dogs of the southern United States where triatomine insect vectors occur, and there are limited veterinary testing options; only the indirect fluorescent antibody (IFA) test is offered at a single accredited diagnostic laboratory. We evaluated a multiplex microsphere immunoassay (MIA) for the detection of antibodies against T. cruzi in dogs and compared it with existing serologic methods to establish cutoff values and relative sensitivity and specificity. We tested 135 canine sera that had been characterized using the IFA and off-label use of 2 commercial rapid assays with our multiplex MIA against 12 antigens: 9 T. cruzi antigens, a negative control recombinant protein (green fluorescent protein, GFP), a Leishmania antigen, and a canine parvovirus antigen (used as an antibody control given near-ubiquitous parvoviral vaccination). The median fluorescence intensity (MFI) ratio between each T. cruzi antigen and GFP was calculated for every sample. Samples with an antigen:GFP MFI ratio > 4 SDs above the mean of 25 known-negative sera were considered positive to that antigen. Samples testing positive to ≥ 2 antigens were considered positive for T. cruzi antibodies. Compared to the IFA, our multiplex MIA had a relative sensitivity of 100% and specificity of 97.0%. Given its precision, high-throughput format, potential for automation, and lack of subjective interpretation, our multiplex MIA should be considered a valid and improved assay for T. cruzi antibodies in dogs.
The vector-borne protozoan parasite Trypanosoma cruzi is endemic to the Americas and causes Chagas disease in a wide range of mammalian hosts, including humans and dogs. It is estimated that 288,000 people in the United States have Chagas disease, with origins of infection varying from travel-related to endemic spread. 13 Dogs are an important host of interest to veterinary public health because they are an effective sentinel species and experience similar disease manifestations to humans.9,19 Studies in Texas have estimated a prevalence of Chagas disease of 20.3–31.6% in domestic dogs, with even higher estimates in kennel or shelter environments (up to 57.6%).6,7,14 The average annual seropositivity in canine samples submitted to the Texas A&M Veterinary Medical Diagnostic Laboratory (TVMDL; College Station, TX, USA) is > 20%; additionally, demand for serologic testing at TVMDL has more than doubled in the last decade, from 1,129 test requests in 2014 to > 2,500 in 2022 (unpublished TVMDL data). Between 2014 and 2018, over 1,200 positive T. cruzi antibody tests were reported by TVMDL in samples submitted from Texas alone (unpublished TVMDL data).
Diagnosing Chagas disease can be challenging given that the parasite’s life cycle offers only brief windows of parasitemia during which T. cruzi organisms can be detected, primarily by PCR assay of blood or by hematologic methods. The parasite can be observed histologically in the heart and other affected tissues; however, this is primarily achieved postmortem. Serologic methods are the primary means of antemortem testing for Chagas disease. Because there is currently little evidence of patients self-clearing the parasite, seropositivity is considered to indicate current infection. In humans, the U.S. Centers for Disease Control and Prevention (CDC) considers a single test insufficient to definitively diagnose Chagas disease, and instead recommends using “two or more tests that use different techniques and detect antibodies to different antigens” 4 ; this recommendation is echoed by another publication on diagnosing Chagas disease in the United States. 10 For dogs, only the indirect fluorescent antibody (IFA) test is available as a validated option in accredited diagnostic laboratories; however, this test requires specialized equipment, is interpreted using subjective evaluation of fluorescence patterns, and is prone to cross-reaction with other closely related pathogens (e.g., Leishmania) given that it uses whole T. cruzi parasites as antigen. Commercial rapid tests designed and approved for human use have been used in research settings to screen dogs for T. cruzi antibodies, 17 but this is not an option for routine testing. Given these limitations, it is currently not practical to diagnose canine Chagas disease with the same level of certainty as the CDC’s recommendations for human diagnosis.
Human and veterinary diagnostic laboratories have developed and implemented multiplex microsphere immunoassays (MIAs) for a variety of infectious diseases using a commercial platform (xMAP Technology; Luminex).20,21 These assays use microscopic polystyrene beads (available in magnetic or non-magnetic forms) as the solid phase, to which proteins of interest can be bound. For indirect assays, antibodies in patient serum bind to antigens on the beads, and a secondary antibody conjugated with phycoerythrin (PE) serves as a marker for any bound antibodies. Each bead is uniquely labeled with an internal dye, allowing the instrument reader to determine which target is represented while measuring the signal from the PE conjugate (indicating the presence or absence of antibodies against that target in the patient serum).
Studies5,12 have described the use of a multiplex MIA to screen serum for T. cruzi antibodies in humans and dogs, tested with a panel of appropriate antigens of interest and demonstrating effective detection. Multiplex MIAs offer several improvements over classical T. cruzi serology (i.e., IFA): they eliminate the need for subjective interpretation by using an instrument reader; are performed using a 96-well plate; offer high throughput; increase precision via multiple readings per analyte and per sample; and offer high sensitivity and specificity by targeting multiple antigens. We aimed to characterize this multiplex MIA and determine its validity in detecting antibodies against various T. cruzi antigens in dogs, establishing cutoff values for seropositivity, and estimating sensitivity and specificity relative to existing methods. Additionally, we aimed to investigate the utility of our assay in monitoring seropositive dogs over time.
Materials and methods
As part of a longitudinal study of Chagas disease in south Texas kennels, 1 135 serum samples had been collected from hunting dogs. During that study, dogs were sampled at 0, 6, and 12 mo 1 ; our study used all 3 sampling times from 37 positive dogs, and 1 sampling point from each of 24 dogs that were T. cruzi negative at the 3 sampling times. Thus, 61 individual dogs were in our sample set, comprised of 11 different breeds, 1–12-y-old; 31 dogs were female, and 30 dogs were male. As part of the previous study, sera were characterized using the IFA test and 2 commercial rapid immunochromatographic tests (ICTs); an endpoint titer was determined for the IFA by serially diluting samples until a final dilution with positive fluorescence signal was reached. The rapid ICTs (Chagas StatPak, ChemBio; Chagas Detect Plus rapid test, InBios) were used off-label to test dog serum, given that both are FDA licensed and validated for human use only. To determine and validate cutoff criteria for the MIA, we used a subset of samples that had congruent positive (n = 27) or congruent negative (n = 31) results in all 3 of the serologic tests to which they were subjected in the original study. Validating the assay with these samples of congruent serostatus was done because there remains uncertainty about the true infection status for animals that have discordant test results, and there is no accepted gold standard serologic test for T. cruzi exposure in human or veterinary medicine.8,10,15,16 Once we had established cutoff criteria for the MIA with this smaller validation sample set, we then used the full sample set from the 37 dogs sampled at multiple times to monitor changes in serostatus over time.
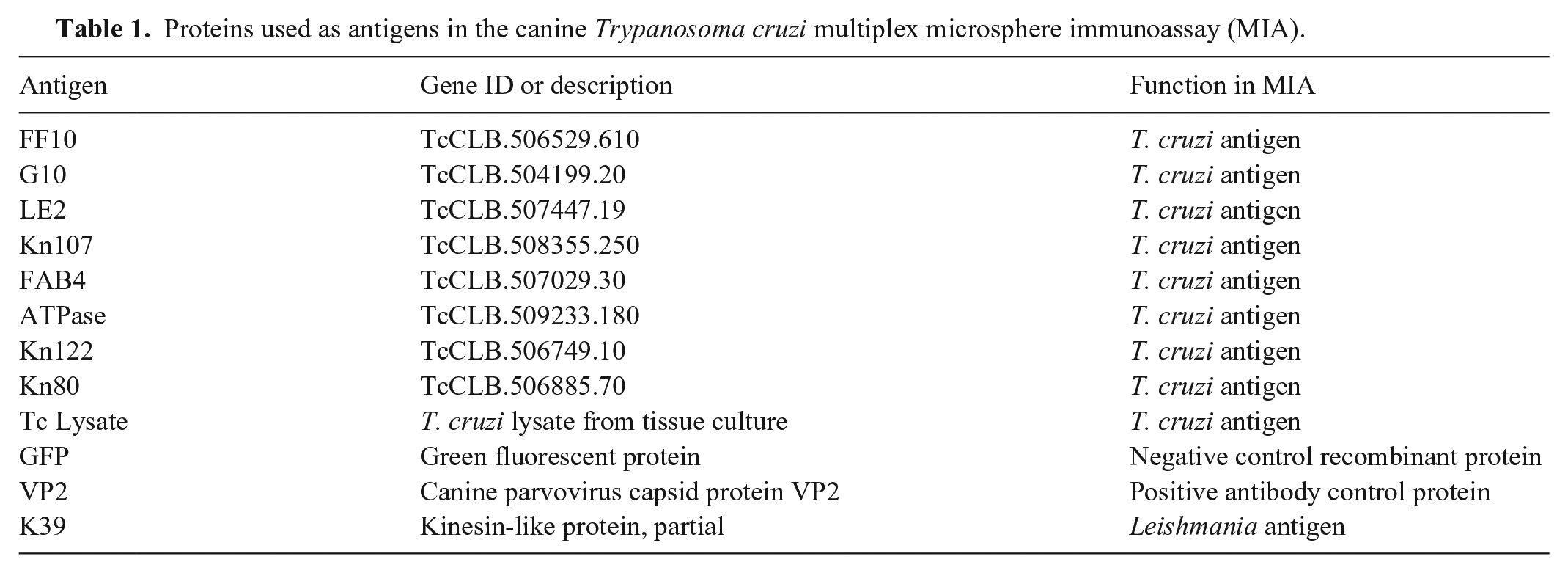
We evaluated 12 antigens in our multiplex assay (Table 1); 9 antigens from T. cruzi were prepared and coupled to the xMAP beads as described previously 5 : FF10, G10, LE2, Kn107, FAB4, ATPase, Kn122, Kn80, and a whole-organism lysate. Green fluorescent protein (GFP) was used as a negative recombinant antigen. A canine parvovirus protein (VP2; MyBioSource) was used as an antibody control; because all dogs in our study were assumed to have been vaccinated, samples failing to react to this protein would reflect inadequate antibody production or sample integrity issues. We included a Leishmania protein (K39; MyBioSource) to check for cross-reactivity. 18 In addition to the study samples, 3 control sera were also tested: negative and positive Leishmania controls, and a negative parvovirus control (VMRD).
Proteins used as antigens in the canine Trypanosoma cruzi multiplex microsphere immunoassay (MIA).
Samples were tested as described previously for dog sera. 12 A mixture containing all 12 antigen-coupled beads (Luminex) in assay buffer (PBS with casein) was prepared and added to 96-well, flat-bottom plates. The plates were affixed to magnetic holders, and the beads were allowed to settle to the bottom of the wells. Excess assay buffer was removed from the plates, leaving the beads behind, and 100 µL of each serum sample (diluted 1:500) was added in duplicate wells. One set of wells had only plain assay buffer added as a blank sample. The plates were sealed with foil and incubated at room temperature on a compact orbital microplate shaker (Thermo Fisher) set to 600 rpm for 1 h. After incubation, the plates were washed 3 times; for each wash, the magnetic holder was used to keep the beads in place, and 200 µL of assay buffer was added and then removed from the wells. Anti-canine IgG conjugated with PE (Rockland) was used as a secondary antibody at a working dilution of 1:100; 150 µL of the PE conjugate was added to each well, the plates were again sealed with foil, and incubated at room temperature on a shaker for 90 min. After the second incubation, the plates were washed 3 times. The beads were then resuspended with 150 µL of assay buffer and read (MAGPIX instrument, xPONENT software; Luminex); the median fluorescence intensity (MFI) for all 12 antigens in each well was measured. The average net MFI (average MFI of both sample wells minus the MFI of the blank wells) for each antigen on each sample was used for subsequent analysis.
For all samples, the average net MFI of each antigen was divided by that of GFP (referred to as the antigen:GFP ratio), which allowed analysis to be performed on a normalized value as well as corrected for any samples that had high reactivity against GFP (i.e., background or nonspecific reactions). 22 A subset of 25 samples that were previously negative for T. cruzi antibodies by all 3 serologic methods were designated as a “negative pool,” and the mean antigen:GFP ratio for each antigen was calculated for these samples. As described previously,5,12 study samples with antigen:GFP ratios > 4 SDs from the mean of the negative pool were considered seroreactive to that antigen.
Statistical analyses were performed (Stata software; StataCorp), and graphical representations of results via heatmaps were generated (Prism 9 software; GraphPad) to compare results between groups and visualize seroconversion over time. A receiver operating characteristic (ROC) using IFA as the reference test was used to determine the optimal number of antigens required to designate a sample as positive for antibodies against T. cruzi, along with assay sensitivity and specificity relative to the IFA. Correlation analysis was performed to investigate the relationship between IFA endpoint titer and the number of positive MIA antigens.
Results
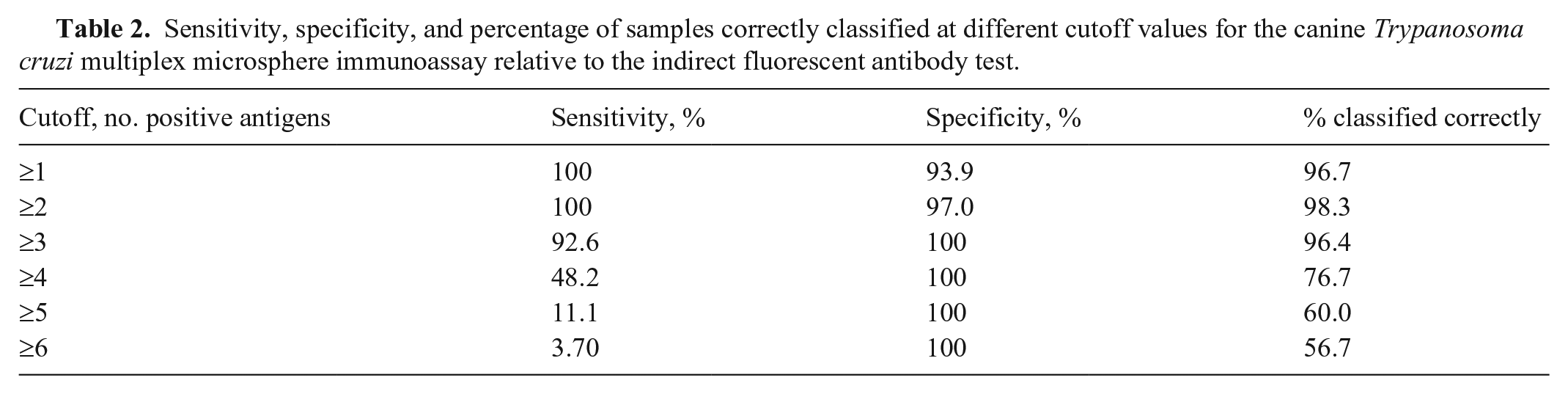
The area under the curve (AUC) for the ROC was 0.999 (Fig. 1), indicating that our multiplex assay is highly comparable to the IFA reference test. The ROC analysis indicated an optimal cutoff of reaction to ≥ 2 antigens to classify a sample as seropositive (Table 2). Using this criterion, the multiplex assay had relative sensitivity of 100% and specificity of 97.0%, with only a single result disagreeing with the reference test. The group of samples that were positive on IFA had notably higher antigen: GFP ratios than the IFA-negative group (Fig. 2). The number of antigens for seropositive samples was highly correlated with the previous IFA titer (r = 0.868, p < 0.0001). All study samples were reactive on the parvovirus protein, indicating no sample quality issues and that all dogs produced detectable IgG antibodies. The Leishmania positive control had high reactivity against the Leishmania antigen; the negative control had none (Fig. 2). Notably, the Leishmania positive control was also seroreactive to 2 T. cruzi antigens (whole-organism lysate and Kn107). A single T. cruzi–negative sample was considered seroreactive for the Leishmania antigen using the diagnostic criterion established for T. cruzi (4 SD above the T. cruzi–negative pool).
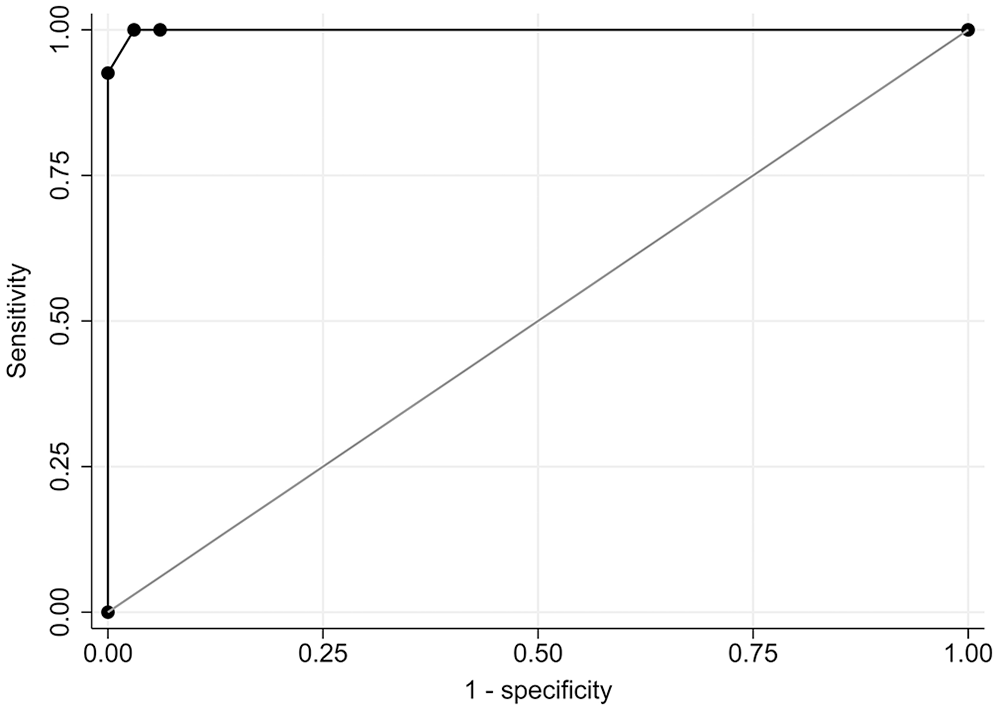
Receiver operating characteristic (ROC) curve for our canine Trypanosoma cruzi antibody multiplex microsphere immunoassay using the indirect fluorescent antibody as a reference standard. Area under the ROC curve = 0.999.
Sensitivity, specificity, and percentage of samples correctly classified at different cutoff values for the canine Trypanosoma cruzi multiplex microsphere immunoassay relative to the indirect fluorescent antibody test.
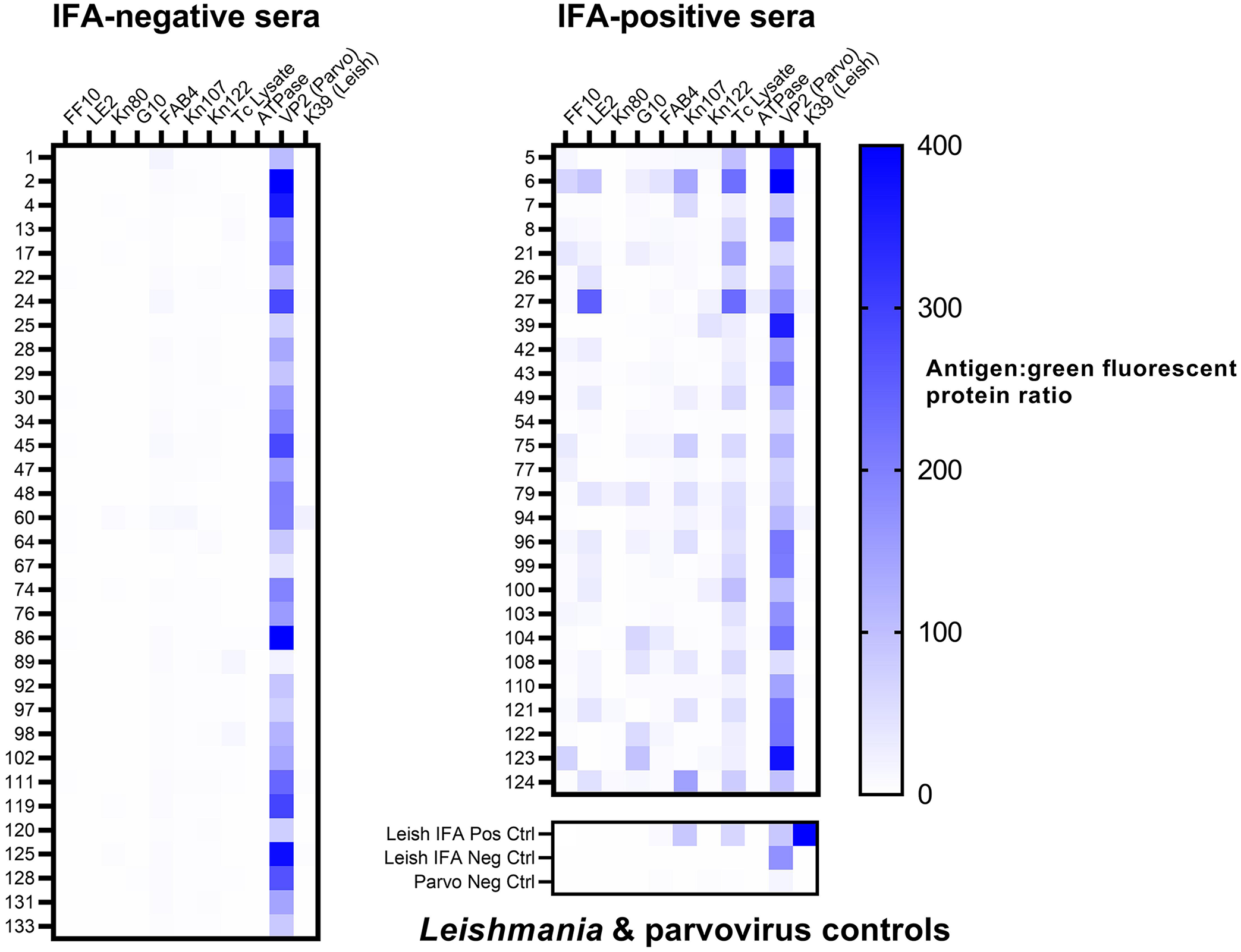
Heatmap of multiplex microsphere immunoassay reactivity of 60 canine serum previously tested for Trypanosoma cruzi antibodies by an indirect fluorescent antibody (IFA) test (grouped by previous IFA result), and reactivity of Leishmania and canine parvovirus controls.
Of the 37 dogs for which all 3 longitudinal samples were tested (Fig. 3), 8 were considered in the original study to have seroconverted between sampling times based on a consensus result of at least 2 positive serologic tests (dogs 3, 13, 22, 25, 28, 97, 131, 133). 1 Using the MIA, 3 (38%) of these dogs (25, 28, 133) had apparent seroconversion using our optimized diagnostic cutoff values (positive to ≥ 2 antigens); 2 more dogs (97, 131) were below the cutoff criterion but were seropositive against a single antigen. Once seropositive, the number of antigens remained relatively stable over time, fluctuating on no more than 2 antigens for most dogs (and with no dogs reverting from seropositive to seronegative over time). All samples that were IFA seropositive were positive on the multiplex assay; the patterns and magnitude of antigen reactions were unique to individual dogs.
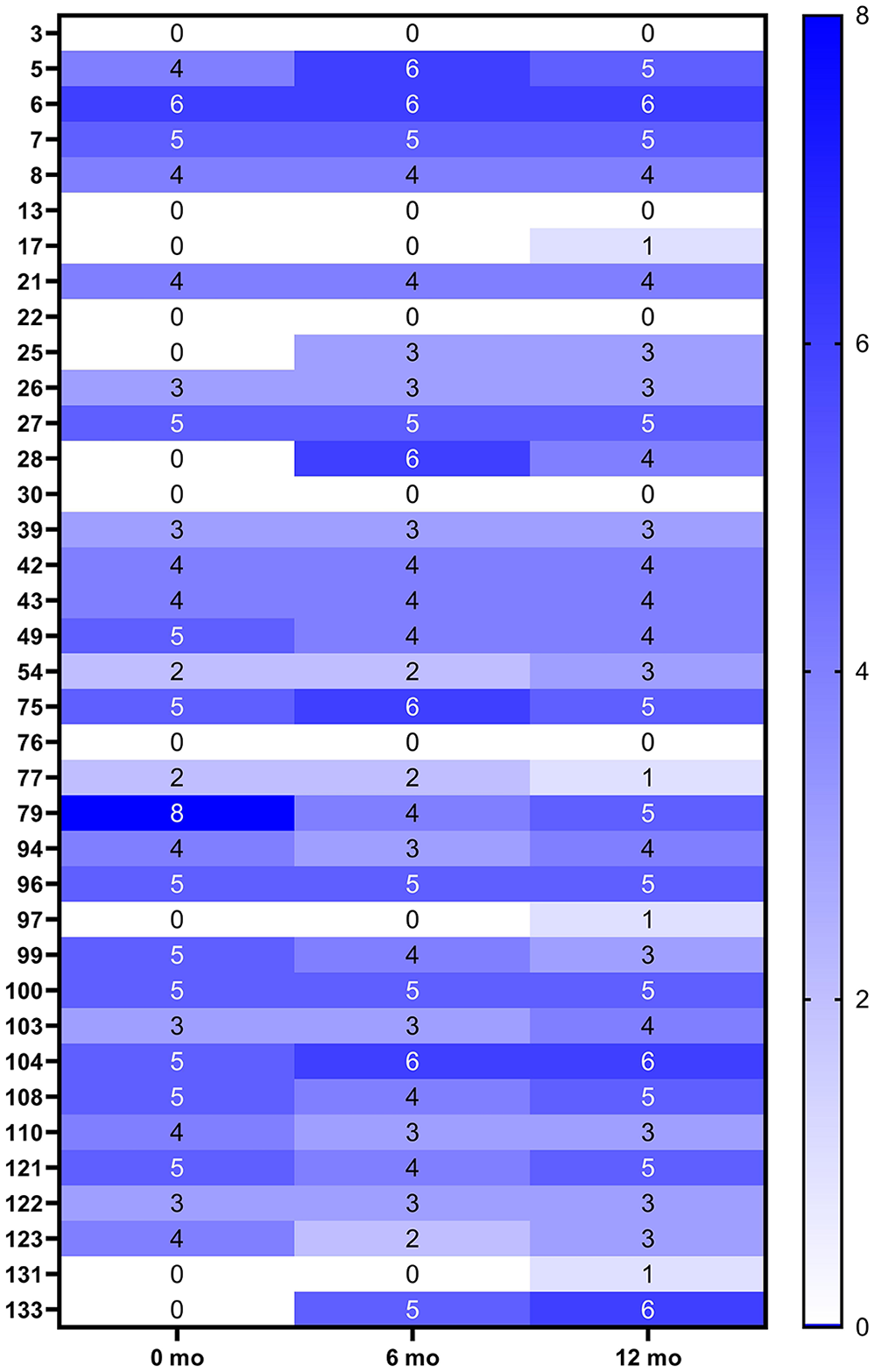
Heatmap of the seropositivity to the number of Trypanosoma cruzi antigens in the multiplex microsphere immunoassay at 0-, 6-, and 12-mo times for 37 dogs presumed to be positive based on previous testing.
Discussion
We successfully demonstrated the utility of a multiplex MIA in the detection of antibodies against T. cruzi in dogs compared to existing test methods. Although other serologic methods, such as IFA and rapid ICTs, have been used extensively in diagnostic and research settings and are considered validated for those purposes based on human literature and their correlation with clinical signs, few previous studies have been conducted that quantify their sensitivity and specificity relative to a gold standard. The multiplex MIA has been used in studies to test dogs, humans, non-human primates, and mice3,5,12; however, sensitivity and specificity had not been estimated for diagnostic purposes. Although we determined an optimal single cutoff point of 2 positive antigens to balance the inverse relationship between sensitivity and specificity, when implementing our assay in a true diagnostic setting, it may be most useful to utilize a 3-tiered approach bracketing cutoff values to maximize both values (i.e., classifying samples as negative below the cutoff that gives 100% sensitivity, positive at the cutoff that gives 100% specificity, and “suspect” in between). Such an approach would maximize the sensitivity of the assay by detecting dogs that are in the very early stages of seroconversion because classifying them as “suspect” would trigger follow-up testing that could lead to earlier diagnosis and clinical intervention.
Our evaluation of the performance of our multiplex MIA was dependent on several assumptions. First, although there is no perfect “gold standard” for T. cruzi testing, we used the IFA as the reference test in the ROC analysis and determination of sensitivity and specificity for the MIA. Second, antibody-positive dogs were assumed to be infected because of the lack of evidence of self-clearing once an animal has been exposed to the T. cruzi parasite. The sample size in our study was relatively small given the decision to use well-characterized samples. Additionally, the samples that we selected for validation were those that had the clearest seropositive or -negative reactions based on previous testing, and we did not explore MIA performance in classifying discordant or “borderline” results in animals that are truly infected or not infected; hence, we may have positively biased our estimates of sensitivity and specificity. 11 Estimates of sensitivity and specificity could be improved by using larger sample sets and statistical analyses that account for the lack of a gold standard (e.g., latent class analysis). Regardless of these limitations, our multiplex assay demonstrated high agreement with the IFA.
Existing T. cruzi antibody assays are well known to cross-react with Leishmania antibodies, confounding detection of animals that could have been exposed to both pathogens. In addition to a whole-organism lysate, the inclusion of individual T. cruzi proteins in our assay (including FF10, which is unique to this organism) is intended to increase specificity. Given that the Leishmania-positive serum was positive on the T. cruzi assay, but none of the T. cruzi–positive samples were significantly reactive with the Leishmania antigen, we cannot conclude whether cross-reactivity will carry over to the MIA testing platform. Additional studies with known Leishmania-positive and -negative samples, from areas in which T. cruzi exposure is unlikely, could ultimately provide more definitive answers to this question. Other studies2,3 have used different sets of T. cruzi proteins in the multiplex assay, and one of the advantages of the multiplex platform is the ability to expand or modify antigens to optimize the test. If implementing our assay in settings in which more definitive discrimination from Leishmania positivity is crucial (e.g., where infection with either pathogen is possible), additional proteins unique to T. cruzi may be added to increase specificity.
Compared to the longitudinal results of another study, 1 our MIA had perfect agreement with the IFA results but did not detect seroconversion against any antigens in 3 of the 8 dogs that were seropositive in 2 rapid ICTs (dogs 3, 13, 22). Given that these 3 dogs were consistently negative by both IFA and MIA, the rapid ICTs may be more sensitive (and thus less specific) than either IFA or MIA. Seroconversion was documented in dogs 97 and 131 in the previous study; however, these dogs were seroreactive to only 1 antigen on our MIA at only the 18-mo sampling time (and thus classified as negative); these dogs were PCR positive at 18 mo in the previous study, indicating they were truly infected at that sampling time. 1 These results may indicate that positivity to even 1 antigen can detect early infection and should be considered as a suspicious result in the clinical and epidemiologic context. Our multiplex assay may be useful in monitoring at-risk populations of dogs (e.g., in kennel settings) over time, in which a change in seropositivity between sampling times—even against a single antigen—could trigger immediate follow-up testing by PCR, allowing diagnosis and possible intervention for acute cases sooner than conventional serology alone.
Footnotes
Acknowledgements
We thank Lisa Auckland for assistance with sample preparation. We thank Dr. Pam Ferro and TVMDL for support throughout this project. Negative canine parvovirus serum was kindly provided by Ed Felt and VMRD. We also thank Heather Darby and Luminex for on-site training and support in setting up the MAGPIX instrument.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
American Kennel Club Canine Health Foundation grant 02448 provided funding for collection of the dog samples used in our study.
