Abstract
We attempted to determine if Leptospira spp. antibodies induced by vaccination would cross-react with Borrelia burgdorferi antigens used in a commercial automated immunofluorescent assay (AccuPlex 4 BioCD; Antech). Staff- and student-owned dogs (n = 31) were recruited at a veterinary teaching hospital in a B. burgdorferi nonendemic area. The dogs were randomized and administered 1 of 4 commercial Leptospira spp. vaccines that contained serovars Canicola, Grippotyphosa, Icterohaemorrhagiae, and Pomona, then booster vaccinated 3 wk later. Blood was collected on weeks 0, 3, 4, 8, and 12. After confirming that maximal Leptospira spp. titers occurred on week 4, aliquots of sera from week 4 were shipped frozen for analysis of B. burgdorferi antibodies against OspA, OspC, OspF, P39, and SLP with the AccuPlex system. Week 4 sera from all 31 dogs had a titer of 1:100 for at least 1 Leptospira spp. serovar. Titers of 1:800 or greater were detected against multiple serovars in 27 dogs. None of the samples contained antibodies against the B. burgdorferi OspA, OspC, OspF, P39, and SLP peptides used in the commercial assay. The B. burgdorferi peptides used in the AccuPlex system do not recognize naturally occurring Leptospira spp. antibodies or those induced by the commercial Leptospira spp. vaccines administered in our study.
Keywords
In small animal veterinary medicine, 2 of the most common pathogenic spirochete genera are Borrelia spp. and Leptospira spp. There are a number of potentially pathogenic Borrelia species, including Borrelia burgdorferi and Borrelia miyamotoi, and multiple potential pathogens in the Leptospira group, including serovars within Leptospira interrogans and Leptospira kirschneri.1,5 Prevalence rates for the 2 genera vary geographically, based on environmental issues for Leptospira spp. and on the tick vector (Ixodes spp.) for B. burgdorferi. Leptospira spp. are found throughout the United States, whereas B. burgdorferi is found in areas with Ixodes spp. Commercial vaccines containing 2 or 4 Leptospira serovars are currently marketed and appear to effectively prevent disease and reduce shedding. 5 Therefore, these vaccines are used widely.
Although, within the United States, B. burgdorferi is most prevalent in the Northeast and Upper Midwest regions, seropositive dogs have been detected in many states, including Colorado, in which Ixodes spp. are not endemic. 3 Most B. burgdorferi seropositive dogs found in atypical locations merely reflect the fact that the owners moved from an endemic region. 3 However, although Leptospira spp. and Borrelia spp. genera are believed to be antigenically distinct, individual peptides are now commonly used in B. burgdorferi serologic tests.4,6,7 A number of multiple natural and artificial peptides—outer surface proteins (OspA, OspC, OspF), an outer membrane protein (P39), the C6 peptide, and a proprietary synthetic peptide (small Lyme peptide [SLP])—have been used in different assays. Although some commercial assays use the same peptides of outer surface proteins,4,6 the source of the peptides can vary, and sometimes the information about their production is proprietary. Thus, it is possible that cross-reactions between antibodies against Leptospira spp. and B. burgdorferi could exist at the peptide level.
In a previous study of Leptospira antibody responses to vaccination using client-owned dogs, most dogs had antibody titers >1:800 to several serovars for a period of time after vaccination. 2 Because Leptospira spp. vaccines are used widely and high titers can develop, there has been concern that cross-reactive antibodies may exist with some of the B. burgdorferi antigens currently used in veterinary laboratories.
We attempted to determine if Leptospira spp. antibodies in sera of client-owned dogs that were recently administered 1 of 4 commercial vaccines would cross-react with the B. burgdorferi antigens used in a commercial automated immunofluorescent assay (AccuPlex 4 BioCD). 4 The AccuPlex assay utilizes outer surface proteins (OspA, OspC, OspF), an outer membrane protein (P39), and a proprietary synthetic peptide (small Lyme peptide [SLP]) as antigens for the detection of B. burgdorferi. The assay also detects antibodies against Ehrlichia canis and Anaplasma phagocytophilum as well as Dirofilaria immitis antigen. 4
Healthy dogs were recruited from staff and students by announcing the study via email to first-, second-, and third-year veterinary students at the Colorado State University Veterinary Teaching Hospital (Fort Collins, CO). Inclusion criteria stipulated that dogs were healthy, weighed ≥10 kg, and had a known vaccination history that did not include Leptospira spp. or B. burgdorferi within the past year. Physical examinations were performed on each enrolled dog. The Colorado State University Institutional Animal Care and Use Committee approved the study, and all owners gave informed consent to participate.
A total of 31 dogs were enrolled in the study: 16 were female, and 15 were male. The median age of dogs enrolled in the study was 6 y (range: 1.5–11 y), and the median weight of dogs enrolled in the study was 23 kg (range: 11–38 kg). None of the dogs had been administered a Leptospira spp. vaccine or B. burgdorferi vaccine within the past year, and some dogs (n = 6) were known to never have been administered a Leptospira spp. vaccine.
The 31 dogs were administered 1 of 4 commercial vaccines that all contained the Leptospira spp. serovars Canicola, Grippotyphosa, Icterohaemorrhagiae, and Pomona (LeptoVax 4, Boehringer-Ingelheim, St. Joseph, MO; Nobivac Lepto4, Merck Animal Health, Whitehouse Station, NJ; RECOMBITEK 4 Lepto, Merial, Duluth, GA; Vanguard L4, Pfizer Animal Health, New York, NY). All available vaccines were used in case differences in cross-reacting antibodies varied by vaccine. The groups were assigned by random number generator, and then all dogs were subcutaneously administered a 1-mL dose of the respective vaccine on week 0 and booster vaccinated on week 3 per manufacturers’ instructions. The number of dogs administered vaccines 1 (LeptoVax 4), 2 (Nobivac Lepto4), 3 (RECOMBITEK 4 Lepto), or 4 (Vanguard L4) were 8, 8, 7, and 8, respectively.
Blood was collected via jugular venipuncture prior to initial vaccination on week 0 and prior to booster vaccination on week 3. Blood was sampled again on weeks 4, 8, and 12. Blood samples were placed in serum tubes, and after clot formation and centrifugation, the serum was separated and stored at −80°C until analysis. The samples were thawed only once.
The Leptospira spp. microagglutination test (MAT) is considered the test of choice for dogs with suspected leptospirosis. 5 Leptospira spp. MAT titers were determined by standard operating procedure of the Veterinary Diagnostic Laboratory, Colorado State University (Fort Collins, CO). For the purpose of our study, MAT titers ≥1:100 were considered positive. Low titers are defined as ≤1:400 and high titers at ≥1:800. After confirming that the maximal Leptospira spp. titers occurred on week 4, aliquots of the week 4 sera were shipped frozen for analysis of B. burgdorferi antibodies (AccuPlex 4 BioCD system, Antech Diagnostics, Lake Success, NY).
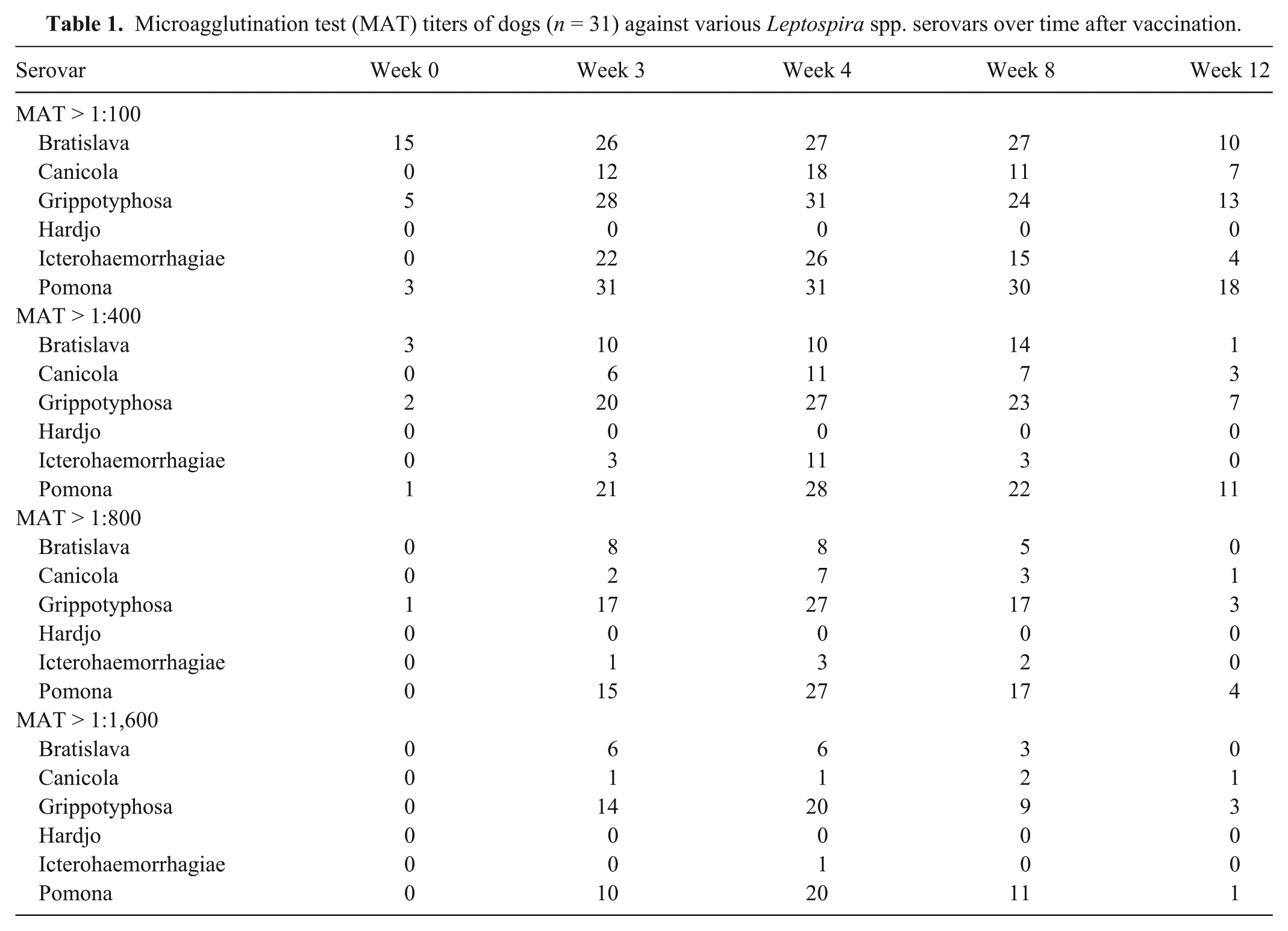
Prior to vaccination, titers to Leptospira spp. were detected in some dogs (Table 1). Of the 5 dogs with serovar Grippotyphosa antibodies at 1:100 on week 0, 3 dogs were vaccinated more than a year prior. Of the 3 dogs with serovar Pomona antibodies at 1:100 on week 0, 2 dogs were vaccinated previously. After vaccination, dogs developed positive Leptospira spp. MAT titers of different magnitudes that varied by serovar and by vaccine over time (Table 1). As seen with another study using client-owned dogs, 2 maximum antibody titers were seen on week 4, when most dogs had titers >1:800 to at least 1 serovar (Fig. 1). High titers were most commonly against Leptospira serovars Grippotyphosa and Pomona.
Microagglutination test (MAT) titers of dogs (n = 31) against various Leptospira spp. serovars over time after vaccination.
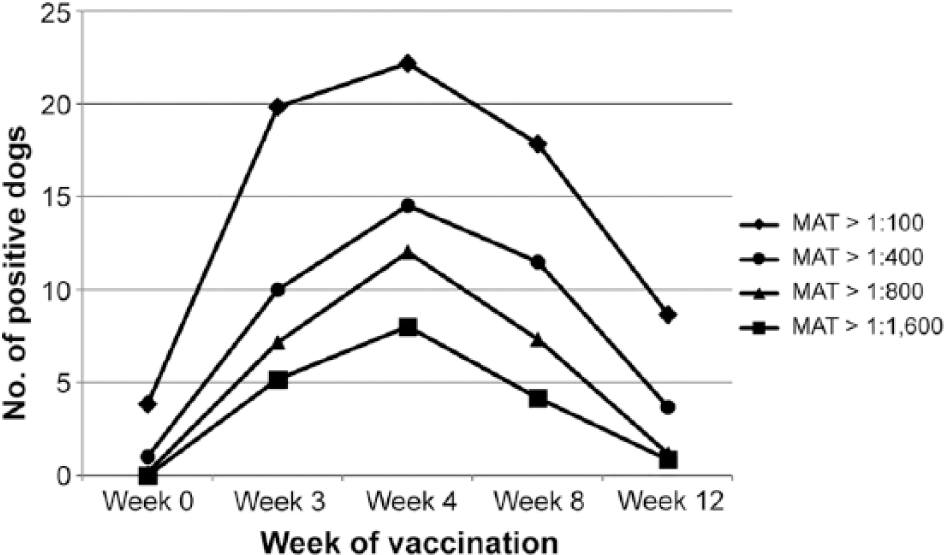
Number of dogs (n = 31) with different magnitudes of detectable Leptospira serovar microagglutination test (MAT) titers over time, before (week 0) and after vaccines were administered on week 0 and week 3.
None of the positive Leptospira spp. sera, regardless of titer or serovar with maximal titer, reacted with the B. burgdorferi antigens in the AccuPlex assay. Several dogs in this study had titers against several Leptospira spp. serovars at the beginning of the study. As none of the dogs were vaccinated within the past year, these positive results were most likely remnant Leptospira spp. antibodies from prior exposure, vaccination, or represent false-positive agglutination. Previous studies show that high titers can be documented at 56 wk post-vaccination. 2 It has been reported previously that antibodies against serovars Grippotyphosa and Pomona can cross-react with serovar Bratislava. 5 However, of the 15 dogs with 1:100 serovar Bratislava titers on week 0, only some had titers against serovars Grippotyphosa (2 dogs) or Pomona (2 dogs). To our knowledge, infection of a dog with serovar Bratislava has not been reported in Colorado. Thus, the serovar Bratislava positive results likely represent false-positive agglutination in most of these 15 dogs.
Our results support previous work that Leptospira spp. antibody titers >1:800 are commonly induced by administration of commercial Leptospira spp. vaccines. 2 Antibodies induced by the commercially available Leptospira spp. vaccines used in our study did not react with OspA, OspC, OspF, P39, or SLP peptides utilized in the AccuPlex assay. Whether that is true for other B. burgdorferi peptides and similar peptides synthesized by other laboratories or point-of-care manufacturers has not been published to our knowledge.
Footnotes
Acknowledgements
We thank the research associates, veterinarians, and students of the Center for Companion Animal Studies for their assistance with this project.
Declaration of conflicting interests
Dr. MR Lappin and Ms. AL Caress performed the study at Colorado State University, and the MAT titers were performed at CSU. Dr. S Moroff is employed by the sponsor, but was blinded to the MAT results until after the AccuPlex results had returned.
Funding
This study was funded by Antech Diagnostics.
