Abstract
Brucella canis is a cause of canine infertility and abortion. Veterinarians and veterinary laboratorians screen for antibodies to B. canis with serologic tests including a rapid slide agglutination test (RSAT; D-Tec CB, Zoetis, San Diego, CA). False-positive results are possible because of cross-reactivity to antibodies to some gram-negative bacteria. Cross-reactivity has been reported between antibodies of Brucella abortus and Leptospira spp. with serologic tests for bovine brucellosis; however, this has not been documented with serologic tests for canine brucellosis, to the author’s knowledge. The RSAT was evaluated with the sera from dogs experimentally challenged with 1 of 4 serovars of Leptospira spp.: L. kirschneri serovar Grippotyphosa, or L. interrogans serovars Canicola, Icterohaemorrhagiae, or Pomona. Experimental infections were confirmed through results of microscopic agglutination testing and/or lateral flow immunochromatography testing. The sera of 32 dogs collected at day 0 and days 7, 10, and 14 yielded negative results with the RSAT. Antibodies produced through experimental infections to these 4 serovars of Leptospira spp. did not cross-react with Brucella antigen with the RSAT; therefore, cross-reactivity of anti-leptospiral antibodies may not be of concern for B. canis rapid slide agglutination testing of dogs.
Keywords
Brucella canis is a small gram-negative intracellular bacterium, and the dog is its reservoir host.2,12 B. canis may cause embryonic loss, abortion, epididymitis, orchitis, and prostatitis; however, many dogs are asymptomatic carriers of the bacteria and therefore are potential sources of infection to susceptible dogs.2,12 B. canis is antigenically most similar to B. ovis and differs from 3 other Brucella spp.—B. abortus, melitensis, and suis—that may rarely infect dogs.2,12 These 3 species grow in smooth colonies rather than rough or mucoid colonies, the latter of which are characteristic of B. canis and B. ovis (http://www.nasphv.org/Documents/BrucellaCanisInHumans.pdf). Colony appearance is attributable to smooth or rough lipopolysaccharide (LPS) of the respective cell walls of Brucella spp., which is the primary antigenic epitope of serologic tests for Brucella. 8
Gram-negative bacteria of genus Leptospira may also cause abortion in dogs, and therefore, testing for antibodies to both Leptospira spp. and Brucella spp. is performed.2,10,12 Concurrent testing is more common for cattle that have aborted or have other reproductive problems and are suspected to be infected with B. abortus or Leptospira spp.6,7 However, identifying cattle infected with B. abortus may be challenging because cross-reactivity of antibodies produced through immunization with Leptospira spp., 7 and other gram-negative bacteria including Yersinia enterocolitica 0:9 and Escherichia coli O157:H7,4,9 has been documented with some serologic tests for B. abortus and may yield false-positive antibody test results for B. abortus. Similarly, identifying dogs infected with B. canis may be challenging. The LPS of the cell walls of gram-negative bacteria Bordetella bronchiseptica and Pseudomonas aeruginosa are antigenically similar to those of B. canis, and antibodies to these bacteria have been reported to yield false-positive B. canis test results. 1 However, similar cross-reactivity with serologic tests for B. canis has not been reported for antibodies produced through natural infection with or vaccination against Leptospira spp. Nonetheless, some veterinarians have anecdotally extrapolated that antibody tests for B. canis behave in the same manner as antibody tests for B. abortus and therefore will yield false-positive results with sera containing anti-leptospiral antibodies.
Dogs that are suspected of being infected by B. canis or those that are healthy and within the breeding pool may be screened for antibodies to B. canis. To date, the only commercial test sold to veterinarians and veterinary laboratorians for canine brucellosis is D-Tec CB (Zoetis, San Diego, CA), a rapid slide agglutination test (RSAT). This RSAT uses a suspension of B. ovis, which cross-reacts with B. canis, stained with rose bengal. 12 B. canis antibodies within sera, if any, bind to the suspended antigen, leading to grossly observed agglutination. False-positive results are possible with this test; its specificity was reported at 83%. 3 The purpose of our study was to evaluate the performance of this commercial RSAT for antibodies to B. canis with canine sera containing antibodies to Leptospira spp.
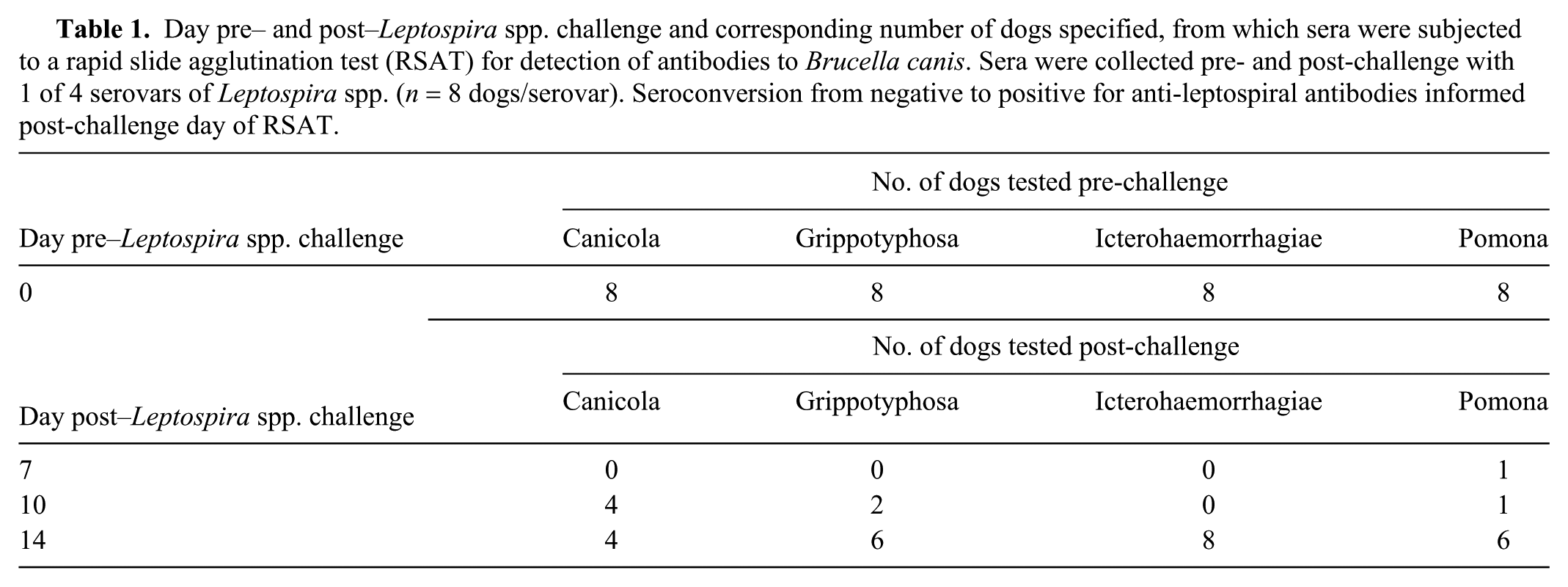
As described elsewhere, 5 32 male Beagle dogs 6–8-mo-old were inoculated with 1 of 4 leptospiral serovars—L. kirschneri serovar Grippotyphosa, or L. interrogans serovars Canicola, Icterohaemorrhagiae, or Pomona (8 dogs/serovar)—on 3 consecutive days (days 0, 1, and 2) through 3 routes (oral, conjunctival, and intranasal) on each day. Prior to the first inoculation, each dog was seronegative for leptospiral antibodies based on microscopic agglutination testing (MAT; titers < 1:100) and lateral flow immunochromatography testing (LFT; Witness Lepto, Zoetis, Lyon, France), the latter for the detection of canine IgM antibodies against these 4 serovars. Additionally, the 32 dogs had not received any vaccine containing leptospiral antigens. Sera were collected at days 0, 4, 7, 10, and 14 post-challenge and evaluated for a Leptospira-specific immune response, as determined with MAT and LFT. 5 Sera were stored at −20°C for 8 mo until tested by the RSAT. However, only sera collected at days 0, 7, 10, or 14 post-challenge were tested with the RSAT because the earliest detection of anti-leptospiral antibodies to any of the 4 serovars as determined with both MAT and LFT was day 7. 5 Experimental infections were confirmed through results of MAT and/or LFT. Day 7 or 10 was the selected post-challenge test day of the sera of some dogs based on MAT seroconversion (MAT ≥ 1:400) and positive LFT. Day 14 was the selected post-challenge test day of sera of the remaining dogs; by day 14 all had seroconverted based on MAT and/or LFT. Therefore, all 32 dogs were confirmed to have anti-leptospiral antibodies. 5
The RSAT was performed according to the manufacturer’s directions (https://www.zoetisus.com/contact/pages/product_information/msds_pi/pi/canine_brucellosis_antibody_test_kit_d-tec_cb.pdf). Briefly, one drop of positive control and one drop of canine serum are dispensed into the respective designated circles on the provided reaction card. Then, thoroughly mixed B. ovis antigen is dispensed into each of the circles. The 2 drops are mixed within each circle, and the resulting mixtures are observed for gross agglutination. The absence of agglutination within 2 min indicates the lack of B. canis antibodies within the canine serum. The presence of agglutination within 2 min indicates the need to perform the second part of the test (2-mercaptoethanol-rapid slide agglutination test [2ME-RSAT]). Consequently, 2 drops of canine serum are mixed with 2 drops of 2-mercaptoethanol (2-ME) in a separate tube. Next, one drop of this mixture is dispensed into a circle of a new reaction card. Then, one drop of B. ovis antigen is dispensed, and the drops mixed. If agglutination occurs within 2 min, antibodies to B. canis are likely present, and the dog is considered presumptively infected.
Sera of all 32 dogs collected at day 0 and at days 7, 10, or 14 post-Leptospira challenge (Table 1) yielded negative RSAT results. Testing with the addition of 2-ME was therefore unnecessary. The positive control and antigen concurrently yielded positive results for all tests.
Day pre– and post–Leptospira spp. challenge and corresponding number of dogs specified, from which sera were subjected to a rapid slide agglutination test (RSAT) for detection of antibodies to Brucella canis. Sera were collected pre- and post-challenge with 1 of 4 serovars of Leptospira spp. (n = 8 dogs/serovar). Seroconversion from negative to positive for anti-leptospiral antibodies informed post-challenge day of RSAT.
The dogs experimentally challenged by 1 of 4 serovars of Leptospira spp. produced anti-leptospiral antibodies that did not cross-react and yield positive results with this RSAT for B. canis. These findings are different than those reported for B. abortus antibody tests with bovine sera containing vaccine-induced anti-leptospiral antibodies; various test methods yielded false-positive results post–Leptospira spp. vaccination. 7 Findings between these 2 studies may differ because of the species of Brucella for which an immune response was being detected, the different serovars of Leptospira to which the animals were subjected, the manner in which anti-leptospiral antibodies were induced in vivo, the days of Brucella testing after immunization, and the types of tests used. 7
The cell wall of B. canis has rough or mucoid LPS, and the cell wall of B. abortus has smooth LPS (http://www.nasphv.org/Documents/BrucellaCanisInHumans.pdf). 8 Possibly, the smooth LPS of the cell wall of B. abortus more closely resembles the LPS of Leptospira spp. such that antibodies directed to them are cross-reactive. 7 The rough LPS of B. canis and B. ovis may be antigenically different from the LPS of Leptospira spp., and therefore, antibodies to the latter did not induce agglutination with this RSAT, which uses B. ovis antigen. The serovars of Leptospira to which these dogs were challenged for our study were 4 of the 6 serovars for which cattle in the previous study were vaccinated. 7 The remaining 2 serovars may be antigenically different from the other 4 serovars such that the antibodies they induce yield false-positive test results for antibody to B. abortus. However, this possibility is unlikely because all serovars belong to the species L. kirschneri or L. interrogans. 7 The methods by which antibodies were induced in vivo also differed between these 2 studies. In the previous study, 7 anti-leptospiral antibodies were induced through vaccination, and in our study, anti-leptospiral antibodies were induced through experimental infection. These antibodies should be similar, however, because both bovine and canine leptospiral vaccines are efficacious at preventing leptospirosis. The day of testing for B. abortus antibodies was beyond 14 d in the previous study 7 and ended by day 14 in our study. In the previous study, 7 the greatest number of vaccinated cattle (8 of 48) tested positive for antibodies to B. abortus and Leptospira spp. on day 28 post-vaccination. Therefore, if RSAT of canine sera collected beyond 14 d post-challenge was performed, testing may have yielded false-positive results. Also, the types of tests used in the previous 7 and current studies differed. An equivalent test used in the previous study to the RSAT used in our study is the rose bengal test. The procedure and interpretation of this rapid agglutination test is similar to the RSAT, but the antigen for the rose bengal test is B. abortus instead of B. ovis. As noted above, the LPS of these species differ and therefore may have caused the different test performances with sera containing anti-leptospiral antibodies.
Limitations of our study include the small number of dogs enrolled, use of an experimental infection model for leptospirosis, single time-point testing post–Leptospira spp. challenge for anti–B. canis antibodies, and not testing the sera of control dogs not experimentally infected with Leptospira spp. or the sera of dogs with confirmed B. canis infection. The most significant limitation is the lack of testing beyond day 14 for these experimentally infected dogs. The sera of these dogs may have yielded positive RSAT results beyond day 14 as the leptospiral infection progressed. This could be clinically important because, for some dogs, clinical signs of leptospirosis may not appear until after day 14 of natural exposure 11 ; therefore, these dogs may not be tested for antibodies to either Leptospira spp. or B. canis until day 14 or later. Likewise, bacteremia and antibody production as well as clinical signs may not occur until 3 wk post natural exposure to B. canis. 2
Footnotes
Acknowledgements
The author thanks Jillian Brooks and Josh Lizer for technical assistance. A portion of this research was presented as an abstract at the 2016 Society for Theriogenology Conference.
Declaration of conflicting interests
The author was an employee of Zoetis, the manufacturer of D-Tec CB and Witness Lepto, at the time of this study.
Funding
Zoetis employed the author during the period of study and provided the D-Tec CB tests.
