Abstract
A validated second-generation SNAP 4Dx Plus (Idexx) incorporates new peptides for improved detection of antibodies against Anaplasma and Ehrlichia tick-borne pathogens in dogs. We compared the first- and second-generation SNAP 4Dx Plus using dogs naturally infected with Anaplasma or Ehrlichia species, or dogs seroreactive by an E. canis indirect fluorescent antibody test (IFAT). The second-generation immunoassay was more sensitive than the first-generation for dogs infected with A. phagocytophilum (51.1% and 29.2%, respectively), A. platys (63.6% and 35.3%, respectively), E. canis (96.2% and 88.3%, respectively), or E. ewingii (73.7% and 70.8%, respectively), and for dogs seroreactive by E. canis IFAT (87.3% and 83.9%, respectively). The second-generation immunoassay detected significantly more Anaplasma- or Ehrlichia-infected dogs that were Anaplasma (p < 0.001) or Ehrlichia (p = 0.031) seroreactive, respectively, than did the first-generation test. When Ehrlichia seroreactivity by E. canis IFAT and both immunoassays was compared, significantly more E. canis–infected dogs were seroreactive by E. canis IFAT than the first-generation (p = 0.006) but not the second-generation (p = 0.125) immunoassay. Significantly more E. ewingii–infected dogs were seroreactive by the first- (p = 0.011) and second-generation (p = 0.049) immunoassays than the E. canis IFAT. Medical records available for 7 dogs that were Anaplasma seroreactive by the second-generation but not the first-generation immunoassay revealed case management decisions that might have been different with an immediate anaplasmosis diagnosis, including earlier doxycycline therapy and less hospitalization. The second-generation SNAP 4Dx Plus test offered improved serologic detection of Anaplasma and Ehrlichia in naturally infected dogs.
Anaplasma and Ehrlichia spp. are gram-negative, intracellular, tick-borne pathogens (TBPs) that can infect a wide range of mammalian hosts, including dogs and people. 9 Common Anaplasma and Ehrlichia spp. in the United States include A. phagocytophilum (Aph), A. platys (Apl), E. canis (Ec), E. ewingii (Eew), and E. chaffeensis (Ech), transmitted by Ixodes scapularis (Aph), I. pacificus (Aph), Rhipicephalus sanguineus (Apl and Ec), and Amblyomma americanum (Eew and Ech).2,11,19 In dogs, Aph and Apl can cause canine granulocytic anaplasmosis and canine cyclic thrombocytopenia, respectively4,15; Ec and Eew can cause canine monocytic ehrlichiosis and canine granulocytic ehrlichiosis, respectively.11,24 E. chaffeensis is not commonly detected in dogs nor has it been experimentally linked to disease; however, it can cause severe clinical disease in humans.10,29 Clinicopathologic abnormalities and severity of Anaplasma and Ehrlichia infections in dogs can vary depending on the infecting species but commonly include fever, anemia, and thrombocytopenia.4,8,11,13,15,16,18,19,24
Tests used in the diagnosis of canine anaplasmosis and ehrlichiosis include microscopy to visualize intracellular morulae in stained blood smears, PCR to amplify pathogen DNA, and serology to detect antibodies in response to infection. In general, visualization of morulae or DNA amplification by PCR are most effective during the acute phase of infection; serologic tests are most useful days to weeks after the initial infection when IgM and IgG antibodies become detectable. Anaplasma and Ehrlichia serologic tests include indirect fluorescent antibody tests (IFATs) and immunoassays. IFATs utilize whole-cell antigen, potentially binding more cross-reactive antibodies; immunoassays utilize species-specific peptides and are more specific. 24 These differences were demonstrated in experimentally infected dogs in which species-specific immunoassays were highly specific against their respective TBPs; more cross-reactivity was measured with whole-cell IFATs. 25 Limitations of serologic tests include the lack of antibody detection during the acute phase of infection, and specifically for IFATs, the requirement for highly trained technicians and specialized laboratory equipment to generate test results. However, point-of-care (POC) immunoassays provide the benefit of rapid, qualitative results without the need for specialized equipment or training. POC testing can be moved out of the laboratory to the clinic, equipping the veterinarian with a valuable tool to assess tick-borne diseases (TBDs).6,12
SNAP immunoassays (Idexx) have been available to detect antibodies against Anaplasma and Ehrlichia spp., and Borrelia burgdorferi, in dogs since 2001.15,23 Utilizing species-specific peptides, several generations of SNAP immunoassays have been developed, including SNAP 3Dx, SNAP 4Dx, and SNAP 4Dx Plus.6,28 With each new generation assay, species-specific peptides were added, expanding the number of detectable TBPs, which now include Aph and Apl (combined in one spot), Ec and Eew (combined in one spot), and B. burgdorferi on the first-generation SNAP 4Dx Plus immunoassay (SNAP 4Dx Plus [USDA PC 5P21.00], Idexx; hereafter, 1-Gen). 28 Released in summer 2022, a second-generation SNAP 4Dx Plus immunoassay (SNAP 4Dx Plus [USDA PC 5P23.01], Idexx; hereafter, 2-Gen) was validated in an effort to detect antibodies earlier in infection and to increase sensitivity. 1 Changes included the addition of species-specific peptides APH-4, APL-4, and Ec p16, which replaced the Ec p30-1 peptide. We have reported high sensitivity and specificity when detecting Anaplasma spp. (94.1% and 98.4%, respectively) and Ehrlichia spp. (93.4% and 96.8%, respectively) with the 2-Gen, along with significantly earlier Anaplasma antibody detection in dogs experimentally infected with Aph, compared with the 1-Gen. 1 High sensitivity and specificity have also been reported with the 1-Gen when detecting Aph (93.2% and 99.2%, respectively), Apl (89.2% and 99.2%, respectively), Ec (97.8% and 92.3%, respectively), and Eew (96.5% and 93.9%, respectively). 28
To better evaluate differences between 1-Gen and 2-Gen, we investigated the performance of the 1-Gen compared to the 2-Gen and the performance of both immunoassays compared to the Ec IFAT using clinical samples from dogs naturally infected with Anaplasma or Ehrlichia. We hypothesized that there would be a significant difference in Anaplasma and Ehrlichia seroreactivity between 1-Gen and 2-Gen and a significant difference in Ehrlichia seroreactivity between the immunoassays and Ec IFAT. We evaluated available medical records for dogs with discordant 1-Gen and 2-Gen results to identify potential changes in case management if veterinarians had obtained 2-Gen POC immunoassay results.
Materials and methods
Dogs
Between 2020 January 2 and 2021 September 24, results from canine vector-borne pathogen testing performed at the North Carolina State University (NCSU), College of Veterinary Medicine (CVM), Vector-borne Disease Diagnostic Laboratory (VBDDL; Raleigh, NC, USA) were reviewed retrospectively (2020 Jan 2–2021 Jun 16) or prospectively monitored (2021 Jun 17–2021 Sept 24) for dogs that tested PCR+ for Anaplasma and Ehrlichia. PCR+ results were reported for all dogs. Dogs PCR+ for Aph, Apl, Ec, or Eew were included in the serologic test comparisons based on available archived serum or blood for 2-Gen retrospective testing and available historical data from the 1-Gen (hereafter, 1-Gen-historical) or Ec IFAT for comparisons. If enough sample was available, dogs were also tested retrospectively by 1-Gen (hereafter, 1-Gen-retrospective) alongside the 2-Gen.
Retrospective serologic testing
Archived canine serum samples stored at 4°C, −20°C, or −80°C, or EDTA-anticoagulated whole blood samples stored at 4°C, were shipped from the NCSU-CVM-VBDDL on ice to Idexx (Westbrook, ME, USA) where they remained at −20°C or 4°C until tested by 1-Gen and 2-Gen. Samples were received in various shipments and were tested as received according to the manufacturer’s protocol. 28 Specific analyte differences between the 2 immunoassays were summarized (Table 1). At each testing time, all samples were anonymized and randomized before being tested, and results were interpreted visually by 3 operators, as described previously. 1
Anaplasma and Ehrlichia serologic tests used to compare seroreactivity in naturally infected dogs that were PCR+ for Anaplasma and Ehrlichia species.

APH-4 = A. phagocytophilum–specific peptide; APL = A. platys–specific peptide; Ec–p30, Ec-p30/p30–1, Ec–p16 (gp19) = E. canis–specific peptides; EENZ1 = Anaplasma genus–peptide; Eew-p28 = E. ewingii–specific peptide.
Historical serologic data
Historical data from dogs tested by 1-Gen or Ec IFAT at the time of sample submission or sample collection were obtained from the VBDDL database or by submitting veterinarians. For samples tested previously through the VBDDL, 1-Gen was performed according to the manufacturer’s protocol, with results verified visually by 2 technologists, and Ec IFATs were performed as reported previously. 25
Statistical analysis
Proportions for Anaplasma and Ehrlichia spp. PCR+ dogs were calculated as the total number of positive dogs over the total dogs tested. Sensitivity was calculated for the Anaplasma and Ehrlichia analytes as the number of seroreactive samples divided by the total number of Anaplasma spp. PCR+ and Ehrlichia spp. PCR+ dogs, respectively, or as the number of Ehrlichia analyte–seroreactive dogs divided by the number of Ec IFAT–seroreactive dogs. Associated 95% exact CIs were calculated by the Wilson–Brown hybrid method (Prism v.9.4.1 for Windows; GraphPad). Comparisons between the serologic tests were analyzed in R (v.4.2.1, https://www.R-project.org) using a mid-p-value McNemar test for matched pairs, 95% CIs, and 2-tailed p-values for the following paired samples: 1-Gen-retrospective and 1-Gen-historical; 1-Gen (combined data from 1-Gen-retrospective and 1-Gen-historical) and 2-Gen; Ec IFAT and 1-Gen; and Ec IFAT and 2-Gen. Comparisons were made for the Anaplasma and Ehrlichia analytes and by groups of PCR+ dogs, including all infected dogs, genus infections, and species infections.
Medical records
We requested medical data and client consent for use of the data in our study by contacting attending veterinarians of PCR+ dogs via email, a notification attached to the reported results, or both. Differences in presentations, clinical abnormalities, diagnostic workups, hospitalization time, and treatment were evaluated for dogs with discordant 1-Gen and 2-Gen results, focusing on potential changes in case management or outcome if veterinarians had POC results from the 2-Gen. Medical records included combinations of historical findings, physical examination findings, laboratory tests, and diagnostic imaging results. Laboratory test values for CBC, serum biochemistry profile (SBP), and urinalysis were considered abnormal if they were above or below RIs for the respective laboratory, which included Idexx, Antech, university teaching hospitals, or in-house testing equipment.
Results
Anaplasma- and Ehrlichia-infected dogs
Between 2020 January 2 and 2021 September 23, 14,155 dogs were tested for Anaplasma and Ehrlichia infections by PCR through the NCSU-CVM-VBDDL. Dogs PCR+ for Anaplasma or Ehrlichia spp. (n = 324; 2.3%; 95% CI: 2.1, 2.6) included Aph (n = 109; 0.77%; 95% CI: 0.64, 0.93), Apl (n = 18; 0.13%; 95% CI: 0.08, 0.2), Ec (n = 120; 0.85%; 95% CI: 0.71, 1), Ech (n = 3; 0.02%; 95% CI: 0.006, 0.06), and Eew (n = 76; 0.54%; 95% CI: 0.43, 0.67). Two dogs were coinfected with Apl and Ec.
Serologic assay comparisons
Excluding Ech, of the 321 Anaplasma and Ehrlichia PCR+ dogs, 240 (74.8%) had available serum (n = 237) or blood (n = 3) for 2-Gen testing. PCR+ results for these dogs included Aph (n = 94), Apl (n = 11), Ec (n = 79), including 1 Apl/Ec coinfection, and Eew (n = 57). Of the 240 dogs tested by 2-Gen, 225 had sufficient sample volume to also test by 1-Gen. We found no significant differences in Anaplasma or Ehrlichia seroreactivity in the 1-Gen-retrospective and 1-Gen-historical paired samples (n = 211) in any of the PCR+ groups (Suppl. Table 1); thus, for subsequent 1-Gen comparisons, data from 1-Gen-retrospective (n = 225) were combined with data from 1-Gen-historical (n = 15) for dogs not tested retrospectively. Using PCR positivity or Ec IFAT seroreactivity as indicators of true-positives, the 2-Gen detected more Anaplasma- and Ehrlichia-seroreactive dogs than the 1-Gen (Table 2). In comparisons between 1-Gen and 2-Gen using paired samples (n = 240), the 2-Gen test detected significantly more Anaplasma-seroreactive dogs when grouped by all PCR+ dogs (p < 0.001), Anaplasma genus PCR+ dogs (p < 0.001), Aph PCR+ dogs (p < 0.001), and Ehrlichia genus PCR+ dogs (p = 0.013); and 2-Gen detected significantly more Ehrlichia-seroreactive dogs when grouped by Ehrlichia genus PCR+ dogs (p = 0.031; Table 3; Suppl. Table 1). The Apl/Ec coinfected dog was seropositive for both Anaplasma and Ehrlichia by both immunoassays.
Sensitivity of 1-Gen and 2-Gen immunoassays using samples from naturally infected Anaplasma or Ehrlichia spp. PCR+ dogs or dogs seroreactive by E. canis indirect fluorescent assay test (IFAT) as true-positives.
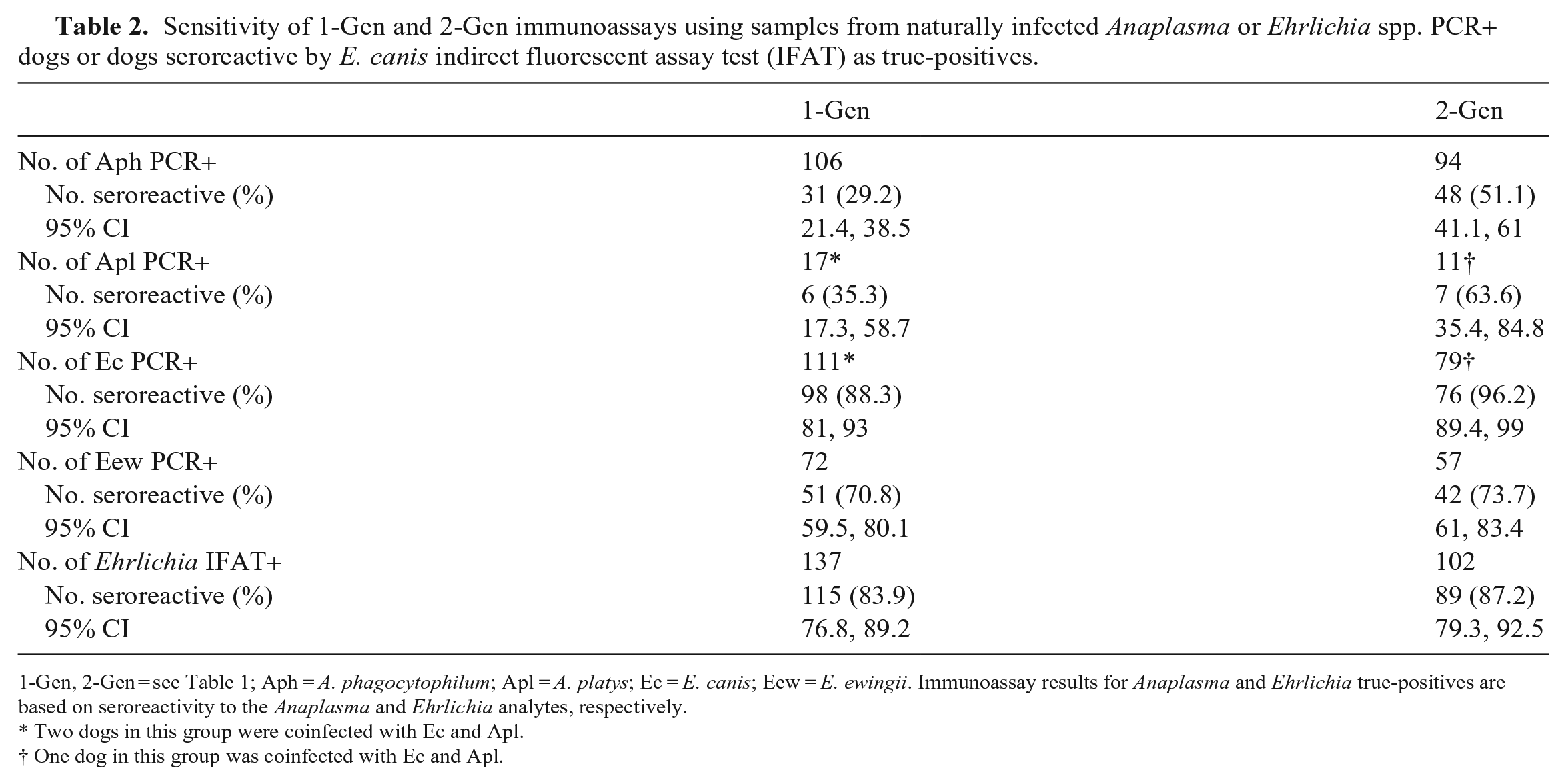
1-Gen, 2-Gen = see Table 1; Aph = A. phagocytophilum; Apl = A. platys; Ec = E. canis; Eew = E. ewingii. Immunoassay results for Anaplasma and Ehrlichia true-positives are based on seroreactivity to the Anaplasma and Ehrlichia analytes, respectively.
Two dogs in this group were coinfected with Ec and Apl.
One dog in this group was coinfected with Ec and Apl.
Analysis of 1-Gen and 2-Gen immunoassays using samples from naturally infected Anaplasma or Ehrlichia PCR+ dogs.
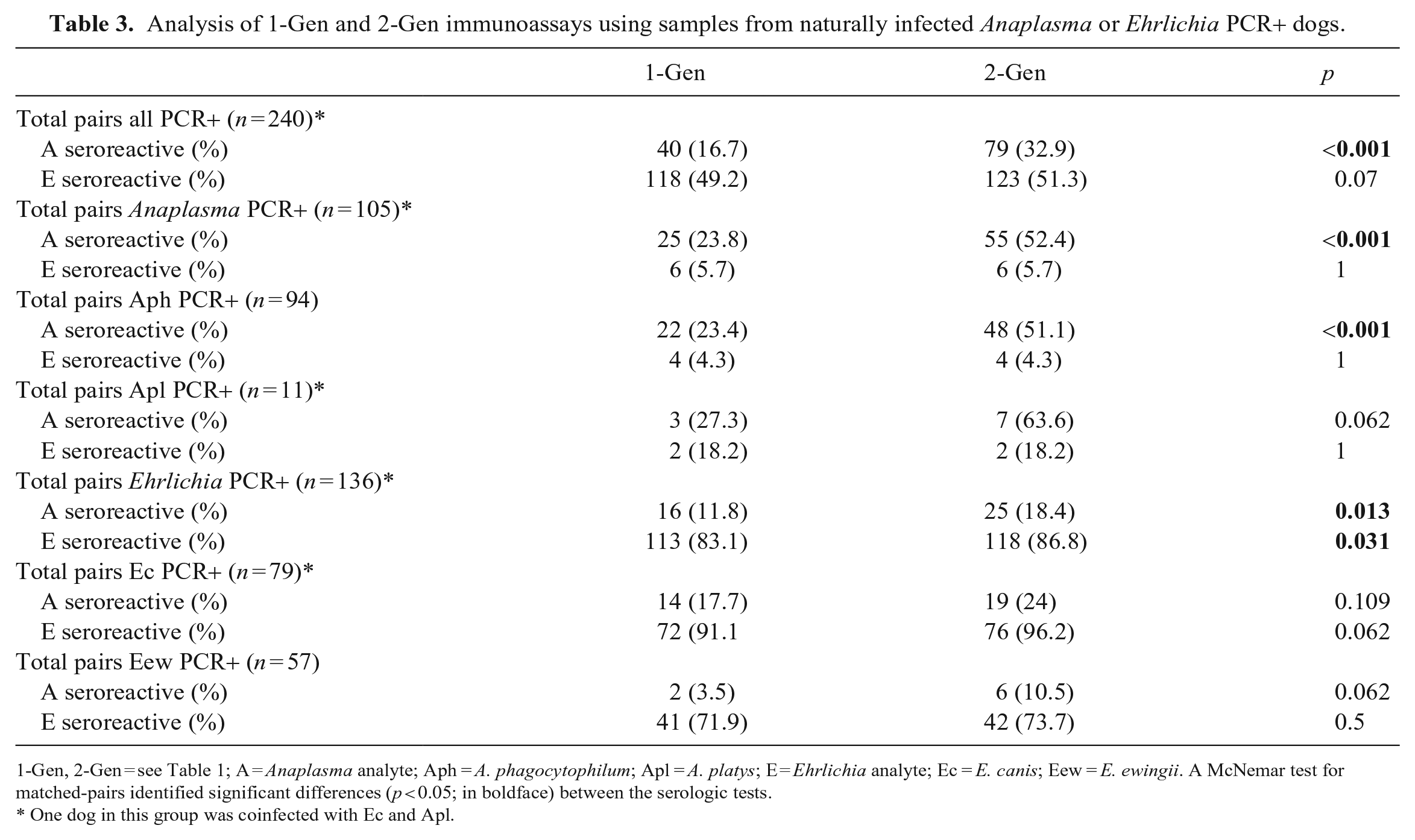
1-Gen, 2-Gen = see Table 1; A = Anaplasma analyte; Aph = A. phagocytophilum; Apl = A. platys; E = Ehrlichia analyte; Ec = E. canis; Eew = E. ewingii. A McNemar test for matched-pairs identified significant differences (p < 0.05; in boldface) between the serologic tests.
One dog in this group was coinfected with Ec and Apl.
Of the 196 Ec or Eew PCR+ dogs, 160 (81.6%) were tested by Ec IFAT, with some also tested by 2-Gen (n = 112) and 1-Gen (n = 159). When comparing Ehrlichia seroreactivity, significant differences between the tests were only observed when the dogs were separated by Ec and Eew. In the Ec PCR+ group, we measured significantly more seroreactive dogs by the Ec IFAT compared with 1-Gen (p = 0.006) but not 2-Gen (p = 0.125). Within the Eew PCR+ group, there were significantly more seroreactive dogs by 1-Gen (p = 0.011) and 2-Gen (p = 0.049) than the Ec IFAT (Suppl. Table 2).
Medical records
Records from veterinary institutions (university teaching hospitals, private practices, and diagnostic laboratories) were provided for 50 of 324 (15.4%) dogs, of which 45 were tested by both the 1-Gen and 2-Gen. Of the 45 dogs, 18 (40%) were Ehrlichia seroreactive by both 1-Gen and 2-Gen, with no discordance between the tests; however, 16 (35.6%) dogs were Anaplasma seroreactive by the 2-Gen compared to 9 (20%) by 1-Gen (p = 0.008). Of the 7 discordant cases, 6 dogs were Aph PCR+ and 1 dog was Ec PCR+ (Table 4). The Ec PCR+ dog was Ehrlichia seroreactive by both the 1-Gen and 2-Gen, but the 2-Gen detected seroreactivity to Anaplasma, as well. Case details for each dog are outlined, showing differences in presentations, clinical abnormalities, diagnostic workups, hospitalization time, and when dogs were treated with doxycycline (Table 4). Medical records revealed that POC 1-Gen testing was performed for 6 dogs within 1–2 d of presentation with results that matched our 1-Gen data; dog 1 was not tested at the POC. Similarities between all dogs included CBC and SBP testing within 1 d of presentation and detection of thrombocytopenia (Table 4). All dogs were eventually treated with doxycycline; however, treatment was not started until ≥ 4 d after presentation for 4 dogs, including 1 dog that was not treated with doxycycline for 13 d. Four dogs were hospitalized for ≥ 2 d. Follow-up information was available for 5 dogs, all of which were reported to be either in full recovery or had clinical improvements after doxycycline treatment was started.
Medical record summary for dogs with discordant 1-Gen and 2-Gen results.
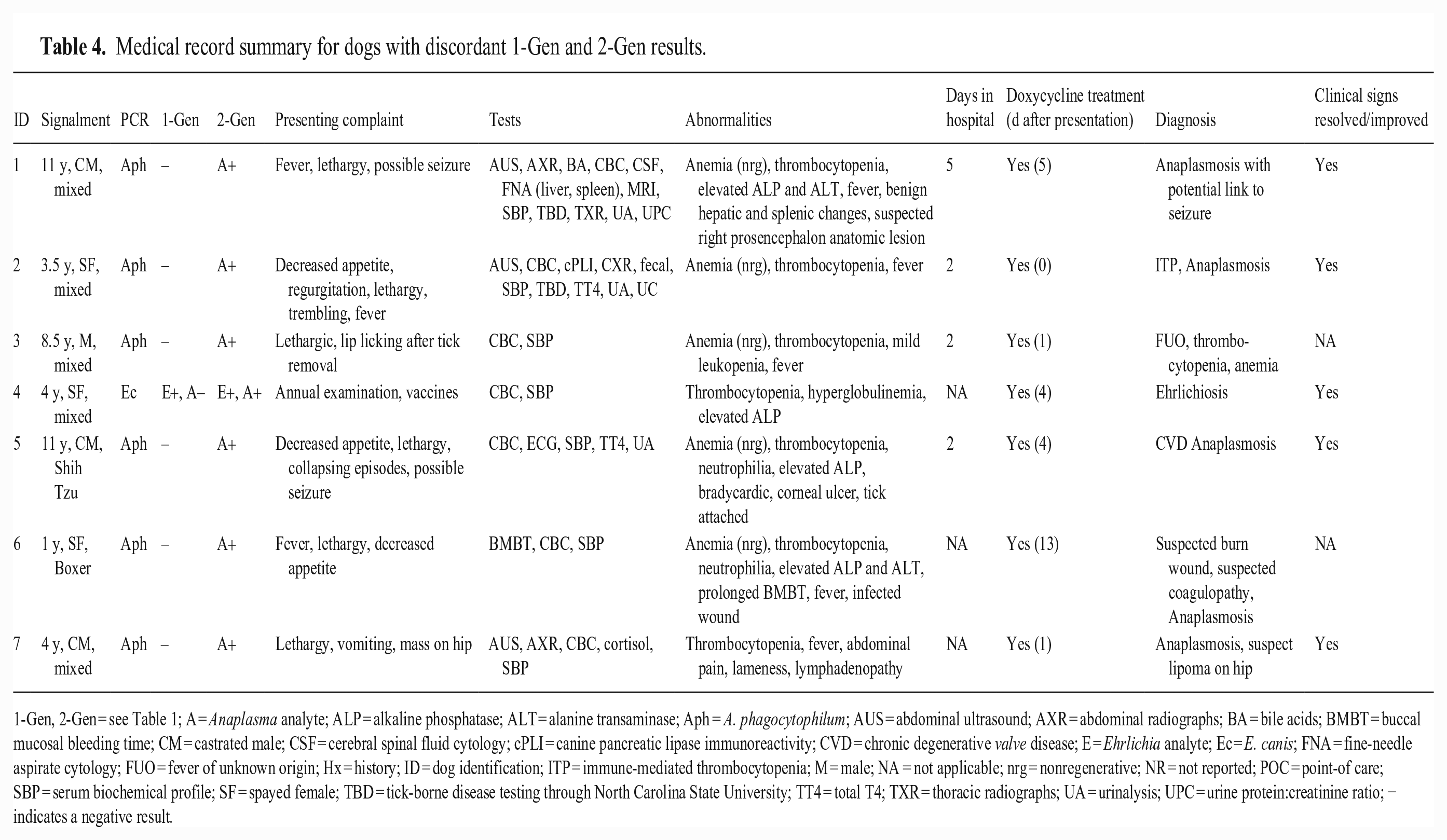
1-Gen, 2-Gen = see Table 1; A = Anaplasma analyte; ALP = alkaline phosphatase; ALT = alanine transaminase; Aph = A. phagocytophilum; AUS = abdominal ultrasound; AXR = abdominal radiographs; BA = bile acids; BMBT = buccal mucosal bleeding time; CM = castrated male; CSF = cerebral spinal fluid cytology; cPLI = canine pancreatic lipase immunoreactivity; CVD = chronic degenerative valve disease; E = Ehrlichia analyte; Ec = E. canis; FNA = fine-needle aspirate cytology; FUO = fever of unknown origin; Hx = history; ID = dog identification; ITP = immune-mediated thrombocytopenia; M = male; NA = not applicable; nrg = nonregenerative; NR = not reported; POC = point-of care; SBP = serum biochemical profile; SF = spayed female; TBD = tick-borne disease testing through North Carolina State University; TT4 = total T4; TXR = thoracic radiographs; UA = urinalysis; UPC = urine protein:creatinine ratio; − indicates a negative result.
Discussion
Compared to 1-Gen, the 2-Gen detected significantly more Anaplasma-seroreactive dogs naturally infected with Anaplasma and significantly more Ehrlichia-seroreactive dogs naturally infected with Ehrlichia, demonstrating improved serologic detection of canine anaplasmosis and ehrlichiosis. Two Anaplasma peptides (APH-4 and APL-4) added to the 2-Gen, released in June 2022, had been shown to improve the detection of antibodies against Anaplasma in dogs.1,5,7 In dogs experimentally infected with Aph, the additional 2-Gen Anaplasma peptides were significantly more likely to detect antibodies against Aph (p = 0.0156) earlier than the 1-Gen Anaplasma peptide (EENZ1). Seroconversion was also detected earlier in 4 of 6 dogs experimentally infected with Apl when tested by the 2-Gen; however, the difference was not significant (p = 0.375). 1
In our study, in which we evaluated naturally infected dogs, significantly more dogs were Anaplasma seroreactive by the 2-Gen than 1-Gen in all Anaplasma PCR+ groups except Apl PCR+. One explanation could be that Aph frequently causes acute, self-limiting infections, whereas Apl infections are more persistent and may have longer-lasting antibodies.8,11,13,15,16,18,20 Interestingly, significantly more dogs were Anaplasma seroreactive by the 2-Gen than 1-Gen in Ehrlichia PCR+ dogs, which may represent detection of antibodies from previous or current Anaplasma infections. A marked increase in Anaplasma antibodies has been reported in dogs experimentally and sequentially infected with Apl, followed by Ec. 11 This effect was also seen in dogs that had become PCR negative for Apl and even one dog that had become Anaplasma seronegative, but when infected with Ec, became Anaplasma seroreactive again.
A new Ehrlichia peptide (Ec p16), derived from immunoreactive glycoprotein 19, was added to the 2-Gen, which also contains the original 1-Gen peptides, Ec-p30 and Eew p28. Using the Ec p16 peptide, several studies have demonstrated improved detection of antibodies against Ec in dogs.1,3,21,27 Earlier Ec antibody detection was reported in 4 of 6 dogs experimentally infected with Ec; however, the difference was not significant (p = 0.125) when compared to the 1-Gen. 1 In our study, compared to 1-Gen, the increase in Ehrlichia seroreactivity detected by the 2-Gen was only significant in the Ehrlichia genus PCR+ group (p = 0.031). The difference between the 1-Gen and 2-Gen, with respect to earlier Ehrlichia antibody detection, could be difficult to quantify in dogs naturally infected with Ec given the chronicity of Ehrlichia infections and persistence of antibodies.11,22,26 Chronic canine ehrlichiosis is frequently documented as a waxing and waning illness.11,14,17,19 This is reflected in our study by the high percentage of seroreactive (96.2%) Ec PCR+ dogs, compared to the low percentage of seroreactive (51.1%) Aph PCR+ dogs.
We found that the 2-Gen was more like the Ec IFAT than the 1-Gen with respect to detecting Ec antibodies. The Ec IFAT detected significantly more Ec antibodies than the 1-Gen but not the 2-Gen, likely signifying recognition of the Ec p16 peptide present in both the whole-cell Ec IFAT and the 2-Gen but not the 1-Gen. In Eew PCR+ dogs, both the 2-Gen and 1-Gen detected significantly more seroreactive dogs than the Ec IFAT, likely reflecting the Eew-p28 peptide, an Eew-specific peptide not found in Ec but present in both immunoassays.
We evaluated medical records from 7 dogs that were seronegative by 1-Gen but Anaplasma seroreactive by 2-Gen and found that veterinarians for 6 of those dogs made case management decisions based on a seronegative POC 1-Gen result. TBP POC immunoassays are used readily by veterinarians to screen both healthy and clinically ill dogs for infection with TBPs. A potentially “active” TBP infection can be inferred when the presence of certain clinical signs, such as fever and thrombocytopenia, among others, are combined with seropositive POC immunoassay results. It is likely that, for some of these clinically ill dogs, different diagnostic and treatment decisions would have been made with earlier Anaplasma seroreactivity results, especially for the 4 dogs that were not initially treated with doxycycline. Based on these limited cases, use of the 2-Gen could have reduced the number of tests performed and time in the hospital for those dogs that were admitted.
As of 2022 June, all SNAP 4Dx Plus tests contain the new Anaplasma and Ehrlichia peptides referred to in our study. Although improved serologic detection of TBPs will likely lead to improved management of TBDs in dogs, it will also likely lead to an increase in the number of seropositive, overtly healthy dogs needing additional testing, including CBC, SBP, urinalysis, and/or PCR to confirm an active infection. Furthermore, it will be important for veterinarians to recognize that potential increases in regional Anaplasma and Ehrlichia seroprevalence after this product was changed in 2022 could reflect the improved immunoassay sensitivity and not necessarily an increase in TBD in their region.
There were several limitations to our study. First, for most of our sensitivity calculations, we used PCR positivity as the gold standard. It is likely that some of the PCR+ dogs were acutely infected and had not yet generated antibodies, therefore some of the sensitivities we report are lower than those reported by other studies using seroreactivity as the gold standard.1,28 Second, we used samples from dogs naturally infected with Anaplasma or Ehrlichia, so we did not determine specificity or compare temporal differences in antibody detection between the immunoassays because dogs can have antibodies from concurrent or previous infections. Specificity and detection time differences were addressed previously for the 2-Gen using experimentally infected dogs. 1 Third, there was an insufficient amount of archived serum or blood to test all of the dogs retrospectively, head-to-head with both the 2-Gen and 1-Gen; thus, 1-Gen-historical data was used for 15 dogs. However, differences between the 1-Gen-historical and 1-Gen-retrospective results were not statistically significant. These findings support the notion that archived antibodies in serum, stored at cold temperatures, are stable for use in future serologic studies. Another limitation was a lack of medical data for the dogs in our study. Only 15.4% of the veterinary institutions we contacted responded to our request, limiting our ability to evaluate clinicopathologic data from dogs with discordant immunoassay results.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231172723 – Supplemental material for A second-generation, point-of-care immunoassay provided improved detection of Anaplasma and Ehrlichia antibodies in PCR-positive dogs naturally infected with Anaplasma or Ehrlichia species
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231172723 for A second-generation, point-of-care immunoassay provided improved detection of Anaplasma and Ehrlichia antibodies in PCR-positive dogs naturally infected with Anaplasma or Ehrlichia species by Safari S. Richardson, Celine A. Mainville, Andrea Arguello-Marin, Darcy Whalley, Wade Burton, Edward B. Breitschwerdt and Barbara A. Qurollo in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the veterinarians and clients whose dogs were included in our study. We also thank the technologists in the North Carolina State University, Vector-borne Disease Diagnostic Laboratory for their help with contacting veterinarians and sample acquisition, and we thank statistician James Robertson for his help with statistical analyses.
Declaration of conflicting interests
Safari S. Richardson has no conflicting interests. Celine A. Mainville, Andrea Arguello-Marin, Darcy Whalley, and Wade Burton are all employees of Idexx Laboratories. Barbara A. Qurollo receives support for a portion of her salary from Idexx Laboratories as co-director of the Vector Borne Disease Diagnostic Laboratory within the Department of Clinical Sciences, College of Veterinary Medicine, North Carolina State University. Edward B. Breitschwerdt is a paid consultant for Idexx Laboratories. E. B. Breitschwerdt has received compensation from Idexx Laboratories for providing continuing education to veterinarians.
Funding
Funding for student support at North Carolina State University, College of Veterinary Medicine to evaluate a database, acquire clinicopathologic data, retrieve diagnostic samples, and write the manuscript was provided by Idexx Laboratories and the North Carolina State University, Vector-borne Disease Diagnostic Laboratory. Funding for retrospective immunoassay testing was provided by Idexx Laboratories.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
