Abstract
Distinct patterns of local infiltration are a common feature of canine oligodendroglioma and astrocytoma, and typically involve the surrounding neuroparenchyma, ventricles, or leptomeninges. Infiltration of adjacent extraneural sites is rare and has not been well documented in veterinary medicine. Here we describe 6 canine gliomas with cribriform plate involvement (compression or infiltration) and caudal nasal invasion confirmed by neuroimaging, autopsy, and/or histology. All affected dogs were adults (9–12-y-old), and 3 were brachycephalic. Clinical signs were associated with the brain tumor, with no respiratory signs reported. Magnetic resonance imaging in 2 patients revealed a rostral intraparenchymal telencephalic mass with extension into the cribriform plate. All dogs were euthanized. Gross changes consisted of poorly demarcated, white or pale-yellow, soft, and, in oligodendrogliomas, gelatinous, intraparenchymal masses that expanded the rostral portions of the telencephalon and adhered firmly to the ethmoid bone and cribriform plate. Gliomas were classified as high-grade oligodendrogliomas (4 cases) and high-grade astrocytomas (2 cases) based on histology and immunohistochemistry for OLIG2 and GFAP. In all cases, there was evidence of cribriform plate invasion and, in one case, additional invasion of the caudal nasal cavity.
Keywords
Oligodendrogliomas and astrocytomas, the most common canine gliomas, are 2 of the CNS neoplasms that are diagnosed most frequently in dogs, accounting for ~35% of all canine primary brain tumors.4,13 Most oligodendrogliomas and astrocytomas occur in adult-to-older dogs and affect mainly brachycephalic breeds, including Boston Terriers, Bulldogs, and Boxers. 2 Tumors are typically intraparenchymal and occur mainly in the frontal, parietal, and temporal telencephalic lobes, and less often in the brainstem and spinal cord.2,4,9
Local tumor infiltration is a common feature of canine oligodendroglioma and astrocytoma, with distinct distribution patterns that vary in frequency according to the tumor type.1,2 Although tumor infiltration into the surrounding neuroparenchyma is commonly observed in oligodendrogliomas and astrocytomas alike, 2 intraventricular7,10 and leptomeningeal spread2,3,6 are more frequently reported in oligodendrogliomas than astrocytomas. Tumor infiltration into adjacent extraneural soft tissues or bones is exceedingly rare and not well documented in the veterinary medical literature.1,8 Here we describe 6 canine gliomas with cribriform plate involvement (compression or invasion) or caudal nasal cavity invasion diagnosed at 3 academic institutions over a period of approximately 20 y.
We searched for cases of canine glioma with cribriform plate involvement in the autopsy archives of the Athens Veterinary Diagnostic Laboratory (AVDL; College of Veterinary Medicine, University of Georgia, Athens, GA, USA) and the Department of Pathobiology (School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA, USA) between 2000 and 2022, and the Department of Veterinary Pathology (Federal University of Rio Grande do Sul, Brazil) between 2012 and 2022 using the keywords: canine, glioma, oligodendroglioma, astrocytoma, bone, ethmoid, cribriform plate. Submission forms and autopsy reports were reviewed for case selection. Archived slides and H&E-stained tissue sections from selected cases were retrieved and reviewed to characterize the morphology and anatomic distribution of the neoplasms. Immunohistochemistry (IHC) for oligodendrocyte transcription factor 2 (OLIG2) and glial fibrillary acidic protein (GFAP) was performed for diagnostic confirmation (Suppl. Tables 1, 2).
Six cases met the criteria for inclusion in our study (Table 1). Cribriform plate compression and/or infiltration was confirmed using neuroimaging (cases 1, 6), autopsy findings (cases 1–6), and/or histology (cases 1, 2, 6). All dogs were adults (9–12-y-old), and 3 were brachycephalic (2 Boxers, 1 Cane Corso). Neurologic clinical signs varied according to the neurolocalization of the neoplasms, and no respiratory signs were reported. Magnetic resonance imaging (MRI) in 2 patients (cases 1, 6) revealed a rostral telencephalic mass with extension into the cribriform plate (Fig. 1). All dogs were euthanized because of the poor prognosis associated with the progressive nature of the clinical signs or a clinical diagnosis of a CNS neoplasm. Gross changes in the brain consisted of poorly demarcated, white or pale-yellow, soft, and, in oligodendrogliomas, gelatinous masses that expanded the rostral portions of the telencephalon, paranasal region, and rostral cranial fossa, and adhered firmly to the ethmoid bone and cribriform plate (Fig. 2).
Signalment, clinical signs, neuroimaging, gross neuropathology, and diagnosis of glioma with cribriform plate and nasal infiltration in 5 dogs.

CM = castrated male; E = euthanasia; HGA = high-grade astrocytoma; HGO = high-grade oligodendroglioma; MRI = magnetic resonance imaging; NOS = not otherwise specified; NR = none reported; SF = spayed female.
Tumors were classified as high-grade oligodendrogliomas (cases 1, 3, 5, 6) and high-grade astrocytomas (cases 2, 4) based on histology and IHC. 2 The 4 high-grade oligodendrogliomas consisted of closely packed sheets of round cells supported by a fine network of branching capillaries and a faintly basophilic mucinous stroma (Fig. 3). Neoplastic cells had low pleomorphism, with indistinct cytoplasmic borders and a moderate amount of eosinophilic cytoplasm with a prominent perinuclear clear halo. Nuclei were round and had finely stippled to dense chromatin and indistinct nucleoli (case 1) or finely stippled chromatin with 1–2 nucleoli (cases 3, 5). The mitotic count was 2 (case 1) and 3 (cases 3, 5, 6) in 2.37 mm2 (10 FN22/40× fields). Areas of geographic necrosis and microvascular proliferation were present in all cases. Neoplastic cells had multifocal-to-widespread, moderate-to-robust nuclear immunolabeling for OLIG2 and no cytoplasmic immunolabeling for GFAP (Fig. 4). High-grade astrocytomas (cases 2, 4) consisted of closely packet sheets of polygonal to slightly elongate cells supported by a faintly eosinophilic fibrillar stroma (Fig. 5). Neoplastic cells had moderate-to-high pleomorphism, with distinct cytoplasmic borders and abundant eosinophilic cytoplasm. Nuclei were round-to-elongate and had finely stippled chromatin with 1–2 nucleoli. The mitotic count was 3 (case 2) and 12 (case 4) in 2.37 mm2. There were areas of geographic necrosis and microvascular proliferation throughout. One astrocytoma (case 2) had an area of leptomeningeal spread within the ventral portion of the frontal lobe. Case 2 had widespread, moderate nuclear immunolabeling for OLIG2; case 4 had no OLIG2 immunolabeling. Widespread, robust cytoplasmic GFAP immunolabeling (Fig. 6) was observed in both cases.
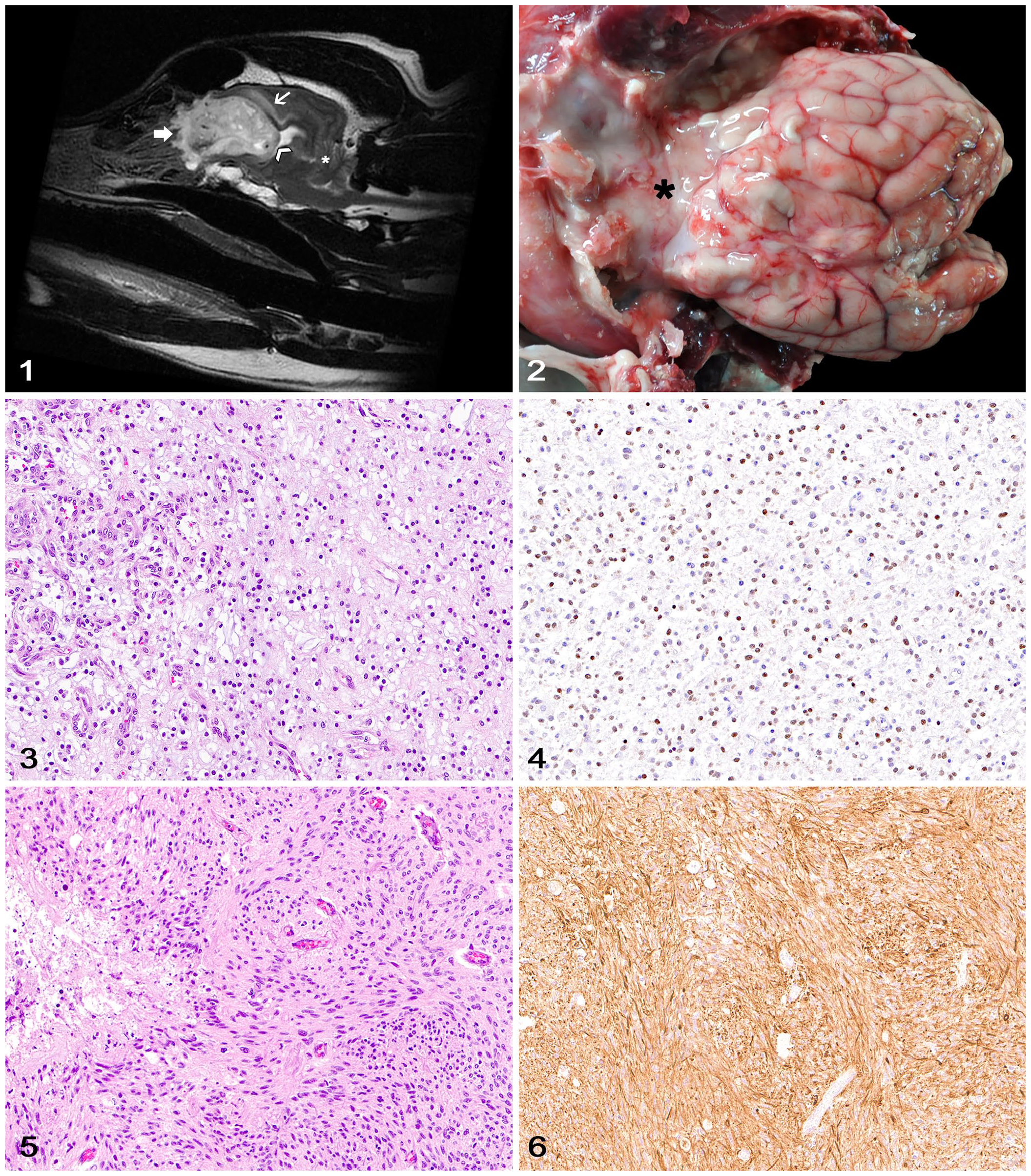
Canine glioma with cribriform plate involvement.
Histologically, sections of the paranasal region and rostral cranial fossa (cases 1, 2, 6) had evidence of cribriform plate compression and infiltration by neoplastic cells morphologically and immunohistochemically similar to the corresponding adjacent gliomas (Figs. 7–12). Neoplastic cells compressed the thin and low-density cribriform plate bone and infiltrated through the olfactory foramina along branches of the olfactory nerves. No overt evidence of bone lysis was observed. Occasional infiltration of small clusters of neoplastic cells in the tissues adjacent to the caudal nasal mucosa was present in case 2.
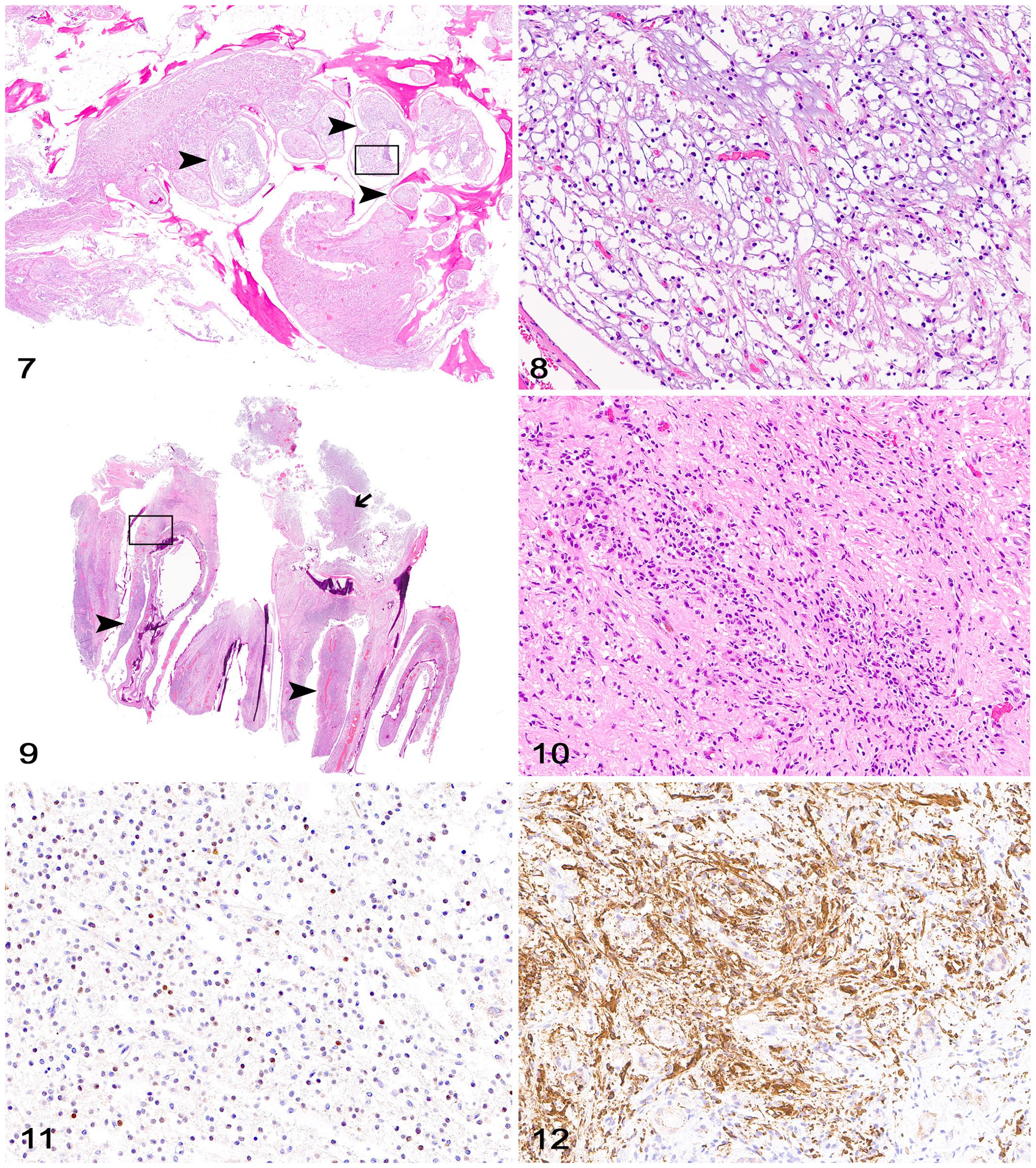
Canine gliomas with cribriform plate involvement.
Most canine gliomas are intraparenchymal and occur predominantly in the telencephalic hemispheres, particularly frontal, temporal, and parietal lobes.1,2,4 Tumor infiltration into the surrounding neuroparenchyma (common in both oligodendrogliomas and astrocytomas) or ventricles and leptomeninges (more common in oligodendrogliomas) are relatively common and well-documented features of canine glioma.1,2,10 Infiltration of the surrounding neuroparenchyma can be focal (nodular gliomas with one of more areas of infiltration) or diffuse (nodular gliomas with poorly defined margins and circumferential infiltration), and can be accompanied by secondary structures of glioma such as clusters of neoplastic cells surrounding neurons (perineuronal satellitosis), blood vessels (perivascular satellitosis), ependymal lining, or leptomeninges. 2
Glioma infiltration into the adjacent extraneural tissues such as skull bones, pituitary gland, cribriform plate, ethmoturbinates, and nasal mucosa is exceedingly rare and not well documented in the veterinary literature.1,8 A large population study of 74 canine gliomas highlighted MRI evidence of tumor infiltration in the adjacent skull bones in 12% of cases (18% of astrocytomas and 12% of oligodendrogliomas), with infiltration of the pituitary gland in 8% of cases. 8 Infiltration of the cribriform plate, ethmoturbinates, and nasal mucosa was recorded in 6% of cases (3 oligodendrogliomas) in which that information was available. 8 The cribriform plate is a portion of the ethmoid bone that physically separates the intracranial cavity from the nasal mucosa and allows for the passage of olfactory nerves from the nasal mucosa to the brain. 12 Most canine gliomas occur in the frontal, temporal, and parietal telencephalic lobes,1,2,4 as evidenced by our cases. Given the rostral neuroanatomic distribution of our gliomas, it is not surprising that the lesions associated with extradural spread were more common in the cribriform plate. 1 However, it is also possible that our gliomas may have arisen from the olfactory system and secondarily infiltrated the rostral portions of the brain, as reported in subsets of human gliomas. 15
Extradural infiltration of primary brain tumors in humans has been reported only rarely in the literature and is typically associated with dural disruption as a result of neurosurgical intervention or radiation therapy. 14 Although one of our dogs (case 1) underwent surgical resection of a right frontal lobe glioma with subsequent chemotherapy 27 mo before euthanasia, too few cases have been documented in dogs to draw any conclusions between associations of neurosurgery with extraneural glioma infiltration. Another proposed mechanism of extradural tumor spread in humans is neoplastic cell infiltration along dural exit points associated with blood vessels and cranial nerves.5,11 The presence of neoplastic cells infiltrating along perineural spaces in our cases lends support for this mechanism.
Diagnostic confirmation of extraneural glioma infiltration in dogs relies on neuroimaging, 8 and special procedures, such as midline section of the skull and evaluation of cross-sections of the cribriform plate, nasal cavity, and paranasal sinuses, are needed for confirmation during autopsy. For these reasons, it remains undetermined whether extraneural infiltration of canine glioma is intrinsically rare or may be missed during a standard autopsy, particularly in patients that did not undergo a full neurologic examination and neuroimaging for neurolocalization of lesions before death or euthanasia.
Most patients in our study had typical neurologic signs that were likely associated with intracranial disease, which was subsequently confirmed by neurologic examination and neuroimaging. However, it is possible that extensive cribriform involvement on MRI may be a confounding factor in the accurate clinical diagnosis of a primary intracranial tumor, given that extension of sinonasal tumors into the brain is a more common finding in dogs.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231195291 – Supplemental material for Glioma with cribriform plate involvement in 6 dogs
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231195291 for Glioma with cribriform plate involvement in 6 dogs by Vicente A. A. Reyes, Elizabeth W. Howerth, Uriel Blas-Machado, Simon R. Platt, Saulo P. Pavarini, Lucas T. Castro, Molly E. Church and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
