Abstract
The rostral cranial fossa (RCF) consists of the sphenoid and ethmoid bones, which accommodate the olfactory bulbs and nerves along the recesses of the cribriform plate. Neoplasms located in the vicinities of the RCF can compress and/or invade the cribriform plate. Here we describe the clinical and pathologic findings of neoplasms involving the cribriform plate in 32 dogs and 17 cats autopsied over a 13-y period. The average ages of affected dogs and cats were 9.2 y and 9.7 y, respectively. No sex or breed predisposition was evident in dogs, but 13 of 18 cats were spayed females and 14 of 18 were domestic shorthair cats. The main clinical signs were seizures (10 cases) and epistaxis (5 cases) in dogs, and red-to-brown nasal discharge (5 cases) and seizures (4 cases) in cats. In dogs, the 22 sinonasal neoplasms included adenocarcinoma (14 cases), transitional carcinoma (4), squamous cell carcinoma (2), lymphoma (1), and histiocytic sarcoma (1); the 10 intracranial neoplasms consisted of high-grade gliomas (3 cases), psammomatous meningiomas (2), histiocytic sarcomas (2), olfactory neuroblastomas (2), and a meningeal granular cell tumor (1). In cats, the 14 sinonasal neoplasms included lymphoma (8 cases), adenocarcinoma (4), adenosquamous carcinoma (1), and squamous cell carcinoma (1); the 3 intracranial neoplasms consisted of oligodendroglioma (1), transitional meningioma (1), and olfactory neuroblastoma (1).
The rostral cranial fossa (RCF) is an area of the skull formed by the sphenoid and ethmoid bones. 18 It accommodates the olfactory bulbs and nerves, which are embedded within the recesses of the cribriform plate, a sieve-like portion of the ethmoid bone that physically separates the inner rostral skull from the caudal nasal mucosa. 18 In humans and other animal species, including sheep, rodents, and dogs, the cribriform plate is an important route of cerebrospinal fluid (CSF) drainage to the cervical lymph nodes.3,4 CSF percolates along the olfactory bulbs, traverses the subarachnoid space through the cribriform plate and olfactory nerves, and flows directly within lymphatics in the nasal mucosa. 3 Once in the nasal tissue, CSF is absorbed by cervical lymphatics or leaks into the nasal cavity (CSF rhinorrhea).3,4
Defective CSF drainage through the RCF is thought to contribute to the development of central nervous system disorders, including multiple sclerosis and Alzheimer disease in humans and congenital hydrocephalus in brachycephalic dogs.5,19–21 Infectious agents such as SARS-CoV-2 in humans, bovine herpesvirus in cattle, and Cuterebra in cats can also reach the CNS directly through the cribriform plate after infection of the nasal mucosa.7,12,17
Similarly, neoplasms arising from the soft tissues or bones in the vicinities of the RCF, particularly those affecting the brain and rostral inner skull base, nasal cavity, and paranasal sinuses, can compress or invade the cribriform plate and cause destruction of adjacent tissues.2,8–11,13,15,23 Although many of those neoplasms have been described in the veterinary literature, a search of PubMed, CAB Direct, Web of Science, Scopus, and Google revealed no investigations focusing on the cribriform plate and its association with different adjacent neoplasms.
It would be useful for clinicians, neurologists, radiologists, and pathologists to have a list of the most common neoplasms that affect the cribriform plate of dogs and cats so that the most likely differential diagnoses and potential prognosis or treatment protocol can be considered following a clinical diagnosis of a mass within or around the sinonasal area and RCF. Here we describe the clinical and pathologic findings of a set of sinonasal and RCF neoplasms involving the cribriform plate in dogs and cats autopsied at 4 veterinary diagnostic institutions during a 13-y period.
The electronic web-based archive system of the Athens Veterinary Diagnostic Laboratory (College of Veterinary Medicine, University of Georgia, Athens, GA, USA), Schwarzman Animal Medical Center (New York, NY, USA), New York State Animal Health Diagnostic Center, Section of Anatomic Pathology (College of Veterinary Medicine, Cornell University, Ithaca, NY), and Department of Pathobiology (School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA, USA) were searched for autopsy cases of dogs and cats with a diagnosis of a neoplasm involving the cribriform plate using the keywords “cribriform plate”, “ethmoid”, and “ethmoidal” between January 2010 and December 2023. Submission clinical forms and autopsy reports from retrieved cases were reviewed for patient signalment, clinical signs, and pathology findings. We selected and included cases in our study if there was compression or invasion of the cribriform plate by an adjacent neoplasm. Cases with no final diagnosis were not included. Based on autopsy reports and review of archived H&E-stained slides, neoplasms were assigned a specific anatomic location (sinonasal region rostral to the ethmoid bone or paranasal skull base region caudal to the ethmoid bone) and diagnosis according to published standards.13,23 Infiltration and/or compression of the cribriform plate or other surrounding tissues, as well as the presence of metastases associated with the neoplasms were also recorded.
We selected and included 49 cases (32 dogs, 17 cats) in our study (Tables 1, 2). Affected dogs were 1–17-y-old (x̄ = 9.2 y), and there was no evident sex (14 spayed females, 2 females, 13 castrated males, 3 males) or breed predisposition. Clinical signs were typically acute and consisted mainly of seizures (10 cases) and epistaxis (5 cases). Affected cats were 2–18-y-old (x̄ = 9.7 y). Most cats were spayed females (12 cases), with 5 castrated males. Fourteen cats were domestic shorthair cats. Clinical signs consisted mainly of red-to-brown nasal discharge (5 cases) and seizures (4 cases).
Anatomic location, diagnosis, tumor infiltration, and metastasis of neoplasms with cribriform plate involvement in 32 dogs.
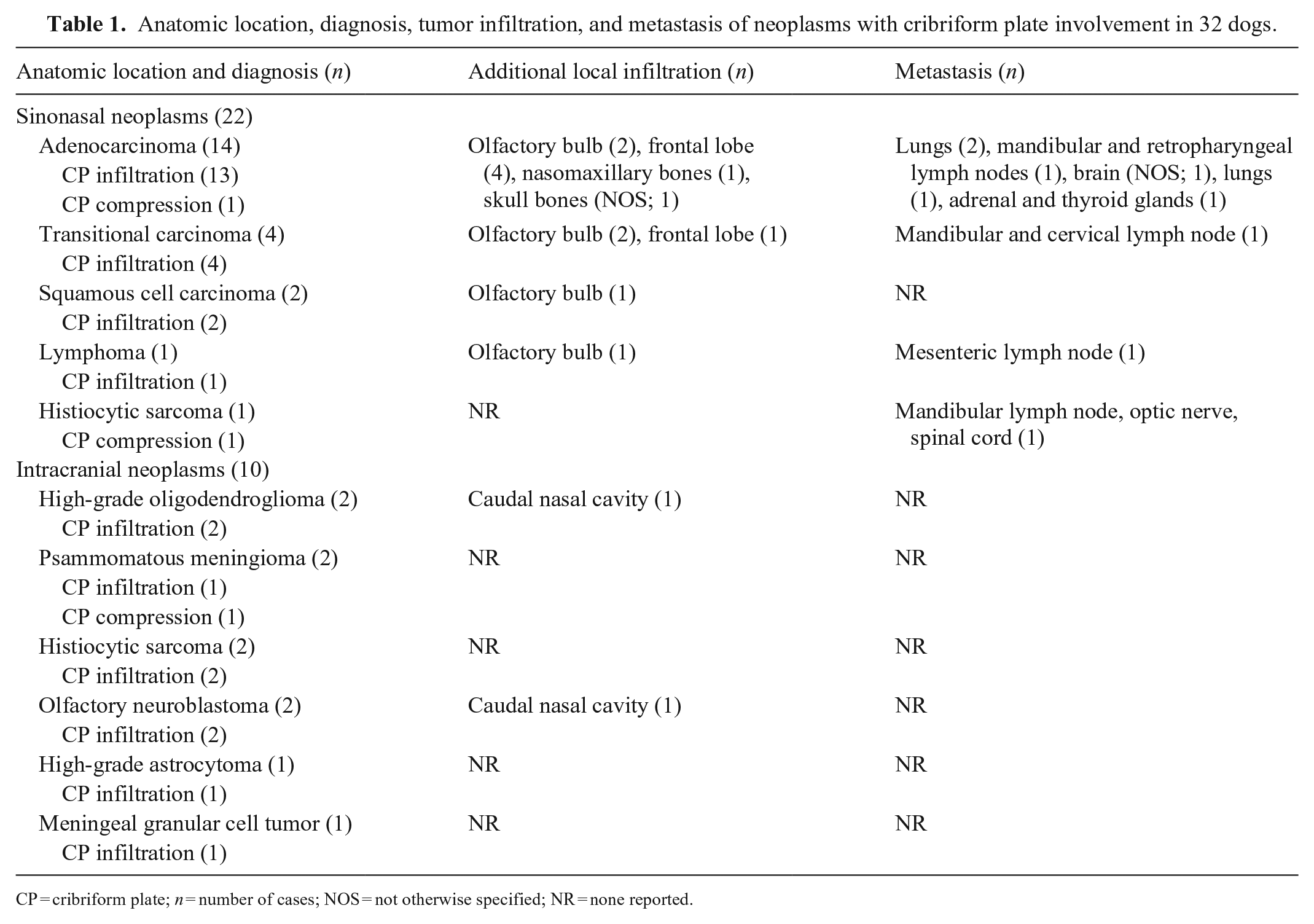
CP = cribriform plate; n = number of cases; NOS = not otherwise specified; NR = none reported.
Anatomic location, diagnosis, tumor infiltration, and metastasis of neoplasms with cribriform plate involvement in 17 cats.

CP = cribriform plate; n = number of cases; NOS = not otherwise specified; NR = none reported.
Diagnostic imaging findings data were limited and reported in 12 of 32 dogs and 9 of 17 cats. MRI or computed tomography (CT) scan findings were consistent with a sinonasal mass (9 of 12 canine cases; 6 of 9 feline cases) or intracranial mass (3 of 12 canine cases; 3 of 9 feline cases) with infiltration of adjacent tissues, including the cribriform plate (4 of 12 canine cases; 3 of 9 feline cases). The clinical outcome for the canine patients was euthanasia (27 cases), spontaneous death (3), or unknown (2). All cats were euthanized.
The most common neoplasms of dogs and cats were sinonasal tumors (22 canine cases; 14 feline cases), followed by intracranial neoplasms affecting the RCF (11 canine cases; 3 canine cases). The 22 canine sinonasal neoplasms included adenocarcinoma (14 cases), transitional carcinoma (4), squamous cell carcinoma (2), lymphoma (1), and histiocytic sarcoma (1); the 10 intracranial neoplasms consisted of high-grade glioma (3 cases), psammomatous meningioma (2), histiocytic sarcoma (2), olfactory neuroblastoma (2), and a meningeal granular cell tumor (1). In cats, the 14 sinonasal neoplasms consisted of lymphoma (8 cases), adenocarcinoma (4), adenosquamous carcinoma (1), and squamous cell carcinoma (1); the 3 intracranial neoplasms consisted of oligodendroglioma (1 case), transitional meningioma (1), and olfactory neuroblastoma (1).
Grossly (Figs. 1–4), most neoplasms expanding the sinonasal region were poorly demarcated, white to pale-yellow, soft-to-firm homogeneous masses that often effaced the cribriform plate. In the rostral paranasal region, gliomas were relatively well-demarcated masses that expanded the neuroparenchyma; the oligodendrogliomas were white-to-red, soft, and gelatinous; and the astrocytoma was pale-yellow and soft. Meningiomas were white, granular, and firm. The histiocytic sarcomas, olfactory neuroblastomas, and granular cell tumor were irregular, pale-yellow, soft-to-firm masses. There was a variable amount of green-to-brown mucus within the frontal sinuses (5 cases) or nasal cavity (3 cases).
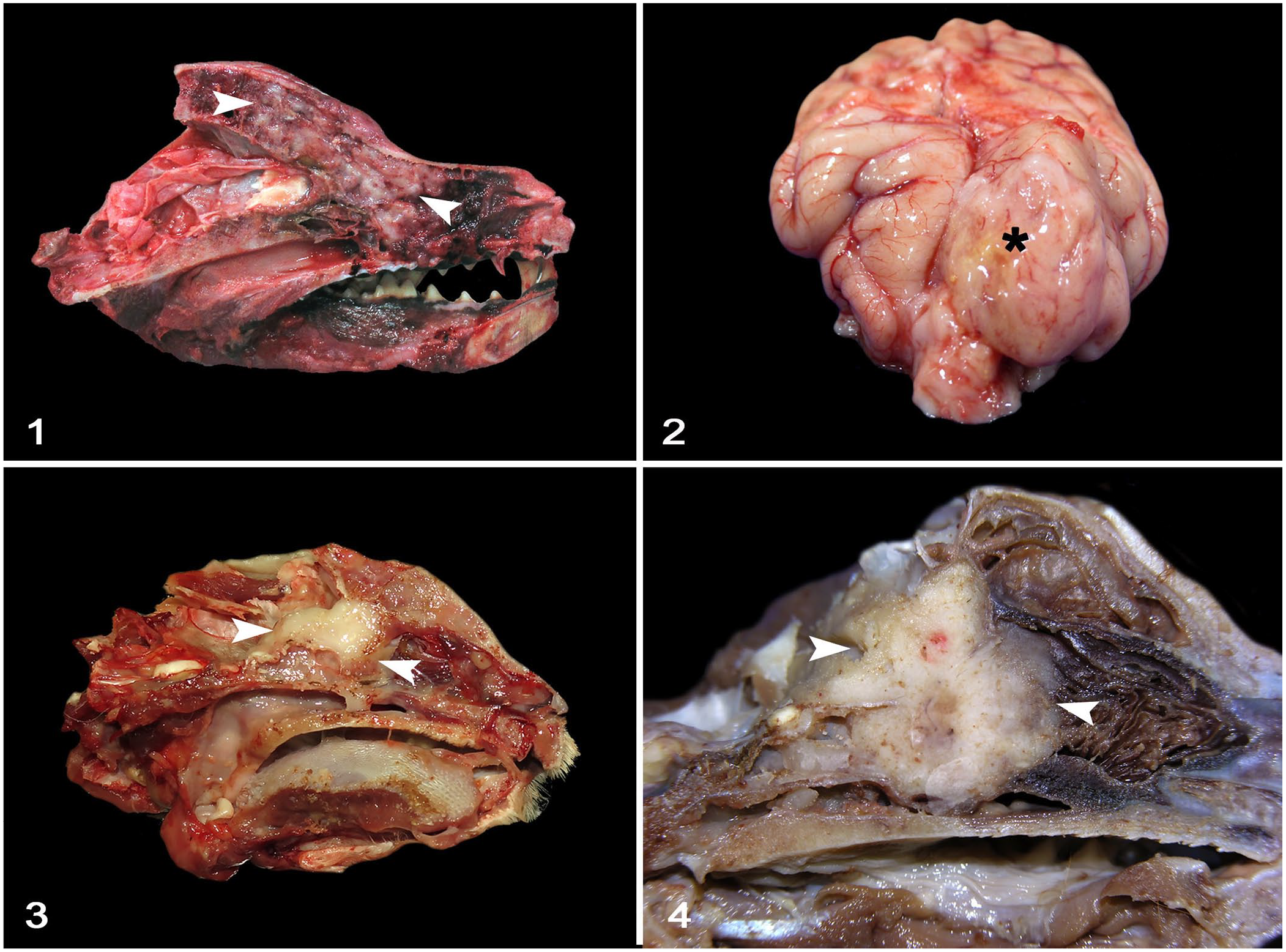
Rostral cranial fossa and sinonasal neoplasms with cribriform plate involvement in dogs and cats.
Histologically, canine and feline sinonasal carcinomas were locally invasive tumors (Fig. 5). Adenocarcinomas consisted of polygonal neoplastic cells forming acini and tubules with secretory material and were supported by a fine fibrovascular stroma (Fig. 6). Solid adenocarcinomas (5 of 13 adenocarcinomas) had rare acini and consisted of extensive solid groups of polygonal neoplastic cells supported by a scant fibrovascular stroma (Fig. 7). Neoplastic cells in transitional carcinomas were columnar and formed stratified, palisading layers distributed perpendicularly to the basement membrane, and were supported by a fine fibrovascular stroma (Fig. 8). Squamous cell carcinomas were composed of polygonal neoplastic cells with squamous differentiation supported by a fibrovascular-to-collagenous stroma (Fig. 9). One adenosquamous carcinoma had numerous acini and tubules admixed with areas of squamous differentiation. Sinonasal lymphomas were characterized by sheets of round neoplastic lymphocytes supported by a scant fibrovascular stroma (Fig. 10). Sinonasal and intracranial histiocytic sarcomas consisted of sheets of highly pleomorphic and atypical neoplastic round cells with scattered multinucleation supported by a fine fibrovascular stroma (Fig. 11). Oligodendrogliomas consisted of sheets of round neoplastic cells with clear perinuclear halos supported by a mucinous and scant fibrovascular stroma (Fig. 12). Meningiomas were composed of bundles of polygonal-to-elongate meningothelial neoplastic cells with concentric whorls surrounding basophilic mineral concretions (psammomatous meningiomas; Fig. 13) or concentric whorls admixed with spindle cells (transitional meningiomas). One astrocytoma was composed of sheets of neoplastic cells with elongate nuclei and no evident cytoplasm, embedded in an eosinophilic, neuroparenchyma-like stroma (Fig. 14). Olfactory neuroblastomas were composed of round-to-oval neoplastic cells arranged in lobules supported by a fibrovascular stroma; neuroblastic rosettes and pseudorosettes were common throughout the neoplasms (Fig. 15). One meningeal granular cell tumor consisted of sheets of round neoplastic cells with granular cytoplasm and small, often eccentric, nuclei (Fig. 16). Immunohistochemistry (IHC) was performed at the time of diagnosis according to historically validated protocols in each participating institution but was not applied in all cases. Canine oligodendrogliomas had nuclear immunolabeling for OLIG2 and no cytoplasmic immunolabeling for GFAP; the canine astrocytoma had nuclear immunolabeling for OLIG2 and cytoplasmic immunolabeling for GFAP. For the feline tumors, IHC for CD3 and CD79 was performed on 4 of 9 lymphomas; neoplastic cells had membranous immunolabeling with CD79, consistent with B-cell lymphoma.
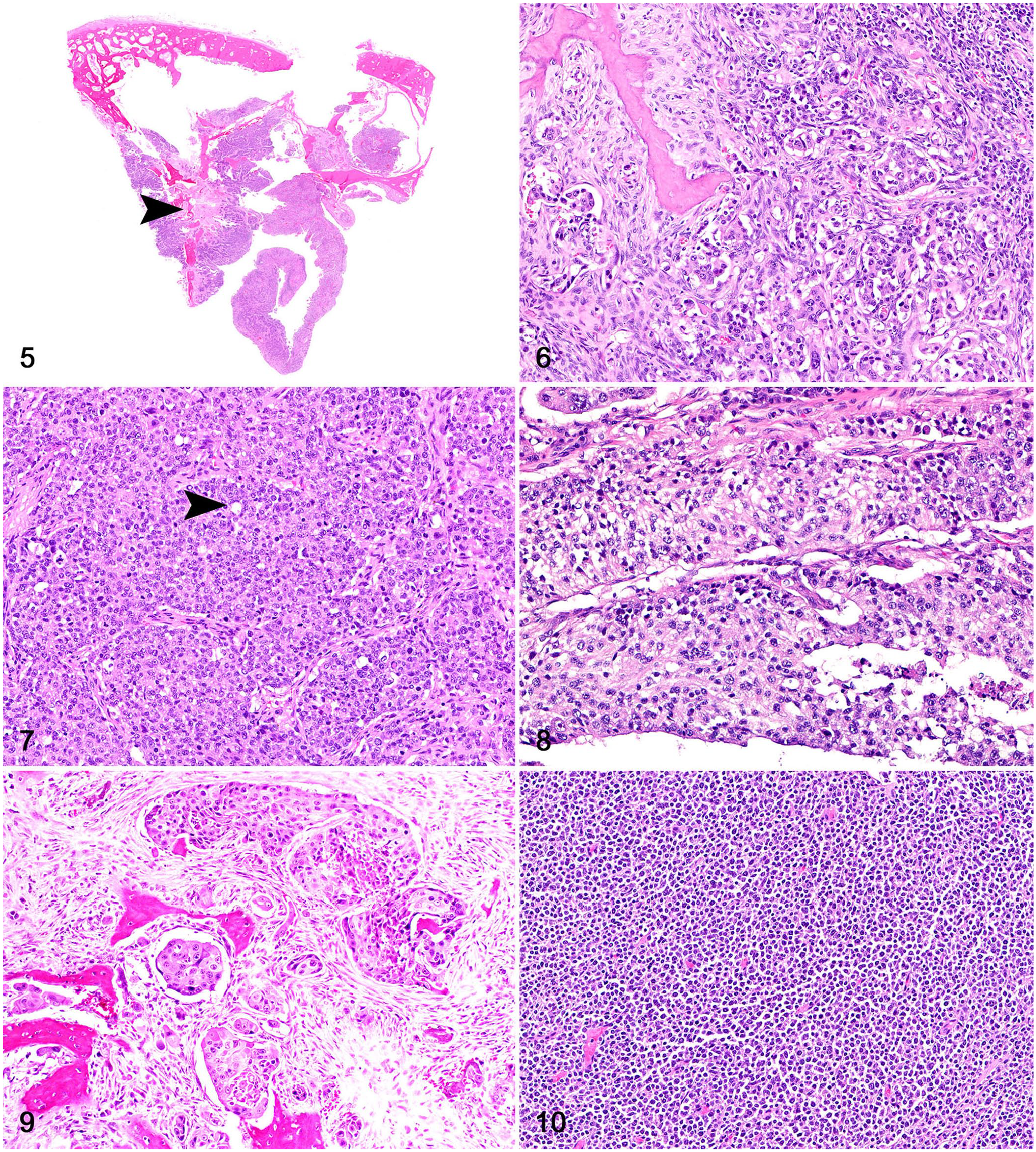
Rostral cranial fossa and sinonasal neoplasms with cribriform plate involvement in dogs and cats.
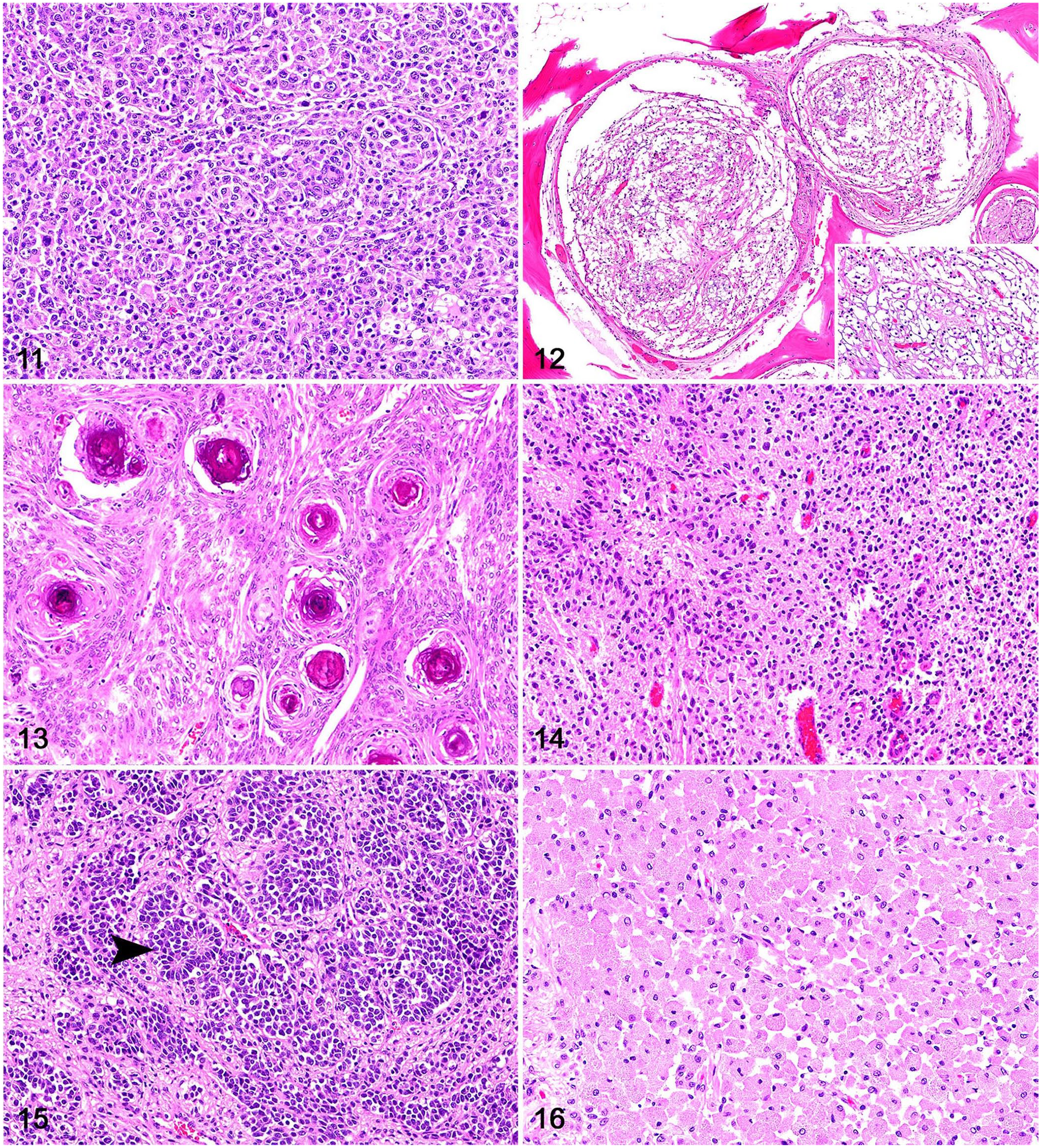
Rostral cranial fossa and sinonasal neoplasms with cribriform plate involvement in dogs and cats.
Sinonasal neoplasms are uncommon in dogs and cats and account for ~2% of all canine neoplasms and 8% of all feline neoplasms.2,9 In humans, neoplasms involving the cribriform plate originate most frequently from the nasal cavity and paranasal sinuses, including sinonasal squamous cell carcinomas, adenocarcinomas, and lymphomas, among others. 6 Similarly, canine and feline neoplasms involving the cribriform plate in our study also originated most commonly from the sinonasal area. The tumor types in our cases were also similar to the human tumor types, consisting of adenocarcinomas and transitional carcinomas in dogs, and lymphomas and adenocarcinomas in cats.
Regardless of their primary anatomic site in the nasal cavity or sinuses, most canine sinonasal neoplasms are epithelial, locally invasive, and malignant tumors,10,15 which was the case for ~90% of our canine sinonasal tumors. In addition to cribriform plate involvement, canine sinonasal carcinomas occasionally infiltrated other adjacent structures, such as the olfactory bulbs (5 cases), frontal telencephalic lobes (5), and skull or nasomaxillary bones (2). Metastases from sinonasal carcinomas are uncommon to rare and occur mainly in regional lymph nodes, brain, lungs, and eyes, 8 as evidenced by a few cases in our study. Although most of the canine nasal adenocarcinomas formed papillary or acinar patterns, neoplastic cells in 5 cases were arranged in solid groups with rare acini or tubules, which was consistent with solid adenocarcinoma.13,23 This distinction is important from a diagnostic standpoint, as solid carcinomas with no evidence of glandular differentiation should be classified as undifferentiated carcinomas. 23 It is currently unknown whether those differences may have any clinical implications for the patient.
The classification of neoplasms that contain patterns resembling the transitional epithelium (between the squamous and respiratory nasal epithelium) has been controversial in human and veterinary medicine. 13 The term transitional carcinoma has been abandoned from the World Health Organization classification of sinonasal tumors and replaced with non-keratinizing squamous cell carcinoma, 22 a step that has also been suggested in veterinary medicine. 13
Lymphoma was the most common sinonasal neoplasm in our feline cases (8 of 14 cases). IHC for CD3 and CD79 was performed in 4 of 9 cases, and all 4 cases were consistent with a B-cell lymphoma. In cats, sinonasal tumors are also mostly malignant, with carcinomas (particularly adenocarcinomas and squamous cell carcinomas) and lymphomas predominating over other neoplasms. 11
Clinical signs in dogs and cats were acute and variable, typically reflecting the sinonasal or intracranial nature of the lesions. In humans, diagnostic imaging can provide useful information associated with the extent and degree of infiltration of lesions affecting the sinonasal area, rostral cranial fossa, and adjacent tissues. However, it is typically not possible to determine the tumor type based on diagnostic imaging, and biopsy is often required for a definitive diagnosis. 6 Diagnostic imaging findings were not available for most of our cases, but a clinical diagnosis of a sinonasal or intracranial mass with distinct degrees of infiltration into the surrounding tissues was achieved in almost 50% of our cases, confirming that MRI or CT scans are reliable tools for the clinical diagnosis and verification of the degree of invasiveness of sinonasal or intracranial masses.9,14
Intracranial neoplasms consisted mainly of gliomas, meningiomas, and olfactory neuroblastomas. Although meningiomas and olfactory neuroblastomas are known to occur in the vicinities of the paranasal region, which likely contributes to compression and/or infiltration of the cribriform plate,1,16 glioma infiltration of adjacent extraneural tissues (including cribriform plate or nasal cavity) is exceedingly rare. 14 A detailed description of the 2 high-grade oligodendrogliomas and 1 high-grade astrocytoma with cribriform plate and nasal infiltration in our study was published elsewhere. 14
Footnotes
Acknowledgements
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declare that they received no financial support for their research and/or authorship of this article.
