Abstract
Tumoral macrocysts (grossly observable cysts) are common in human pilocytic astrocytomas but are rarely reported in canine astrocytomas. Here we describe 7 canine astrocytomas with macrocysts. The median age of affected patients was 9.6 y, and 2 dogs were brachycephalic. Clinical signs reported in 6 cases included head tilt (4 cases), falling (4 cases), and ataxia (3 cases). Gross changes consisted of well (5 cases) or poorly demarcated (2 cases), tan-to-pale-yellow masses with distinct, 2–10-mm fluid-filled cysts that expanded the cerebellum (4 cases) or telencephalon (3 cases). Histology and immunohistochemistry revealed low-grade astrocytoma in 6 cases and high-grade astrocytoma in 1 case. Neoplastic cells were elongate, formed bundles embedded in an eosinophilic fibrillary stroma, and had oval-to-elongate nuclei. Gemistocytic differentiation was common (4 cases). Cystic areas were lined by neoplastic astrocytes and were either apparently empty or contained scant, eosinophilic, proteinaceous material and a few neoplastic astrocytes, foamy macrophages, and occasionally other leukocytes. The mitotic activity was absent or low across cases. Cytoplasmic immunolabeling for glial fibrillary acidic protein was detected in 6 cases. Our findings indicate that canine cystic astrocytomas are rare (our cases span several decades) and are located mainly, but not exclusively, in the cerebellum.
Intratumoral or peritumoral cysts (sometimes dubbed macrocysts if detectable with diagnostic imaging or gross examination) are a common feature of human pilocytic astrocytomas (PAs), but are less common in other gliomas and rare in meningiomas.7,9,23 In veterinary medicine, intratumoral macrocysts are documented in MRI investigations of subsets of canine glioma, particularly in high-grade astrocytomas and oligodendrogliomas, 24 and are rarely reported in meningiomas.3,4 However, these intratumoral macrocysts are rarely described as a gross feature of canine glioma, with most reports focusing on astrocytomas with pilocytic morphology similar to human PAs. 22
In humans, PAs account for ~17% of all primary brain neoplasms of childhood, and PA is the most common glioma in patients ≤19-y-old; PAs rarely affect adults.9,23 Macroscopic cysts are a hallmark feature of >80% of these pilocytic astrocytomas, in which cysts may efface much of the mass, leaving only a mural tumor nodule. 6 Most tumors are classified as WHO grade 1 astrocytomas and occur in the cerebellum (particularly in children), optic nerve and chiasm, and hypothalamus.8,9,14 Distinct from their human counterpart, PAs are rarely reported in dogs of many ages.20,22 Tumors are reportedly more common in the thalamus. 22 MRI findings in humans and dogs consist of well-demarcated, T1W-hypointense and T2W-hyperintense cystic areas that contrast with a brightly enhancing solid tumor mass.8,9,22
Grossly, most human and canine PAs are well-demarcated, soft gray masses with intratumoral cysts. 22 Histologically, these neoplasms have low cellularity and consist of elongate bipolar neoplastic cells arranged in fascicles admixed with stellate cells with branching cytoplasmic processes that enclose the macrocysts and numerous microcysts.14,20 Because macrocysts are rarely reported grossly and are not used as part of the canine glioma classification and grading system, 13 their potential association with a particular glioma subtype or grade remains undetermined. Here we describe the occurrence of intratumoral cysts in 7 canine astrocytomas.
We searched for cases of canine cystic astrocytoma with macrocysts in the autopsy archives of the Athens Veterinary Diagnostic Laboratory (AVDL; College of Veterinary Medicine, University of Georgia, Athens, GA, USA) and the Department of Population Medicine and Diagnostic Sciences (Section of Anatomic Pathology, College of Veterinary Medicine, Cornell University, Ithaca, NY, USA) between 2010 and 2024 using the keywords “canine”, “glioma”, “oligodendroglioma”, “astrocytoma”, “macrocystic”, “cystic”, “macrocysts”, and “cysts”. Cases were also searched in the private archives of one author (Brian A. Summers) in a period spanning 45 y (1978–2023). Clinical forms and autopsy reports were reviewed, and cases in which tumoral macrocysts were described were selected. Archived H&E-stained tissue sections and immunohistochemistry (IHC) slides (when available) were reviewed to characterize the morphology and IHC features of the neoplasms. H&E and IHC were performed at the time of diagnosis based on historically validated methods according to laboratory SOPs at the Cornell University (case 1–6) and AVDL (case 7). 13
We retrieved 7 cases for inclusion in our study (Table 1). The mean age of affected dogs was 9.6 y, with no apparent sex or breed predisposition. Clinical signs were reported in 6 of 7 cases and reflected mainly caudal fossa involvement, including vestibular signs such as head tilt (4 of 6 cases), falling (4 of 6), and ataxia (3 of 6). Because of the progressive nature of the clinical signs, euthanasia was elected in 6 of 7 cases (1–3, 5–7); the clinical outcome was not known in 1 case (4).
Signalment, clinical signs, neuropathology, and diagnosis of cystic astrocytomas in 7 dogs.
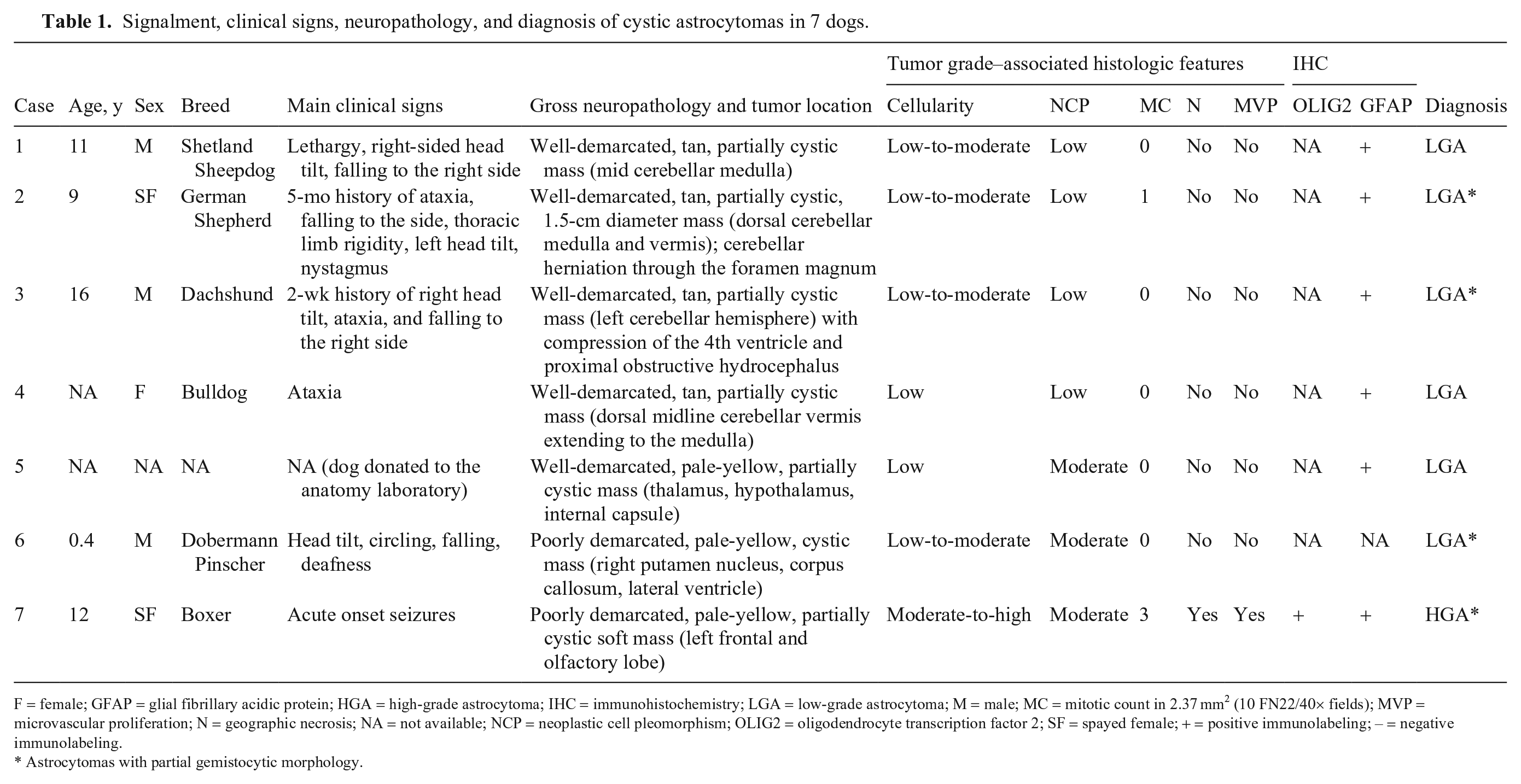
F = female; GFAP = glial fibrillary acidic protein; HGA = high-grade astrocytoma; IHC = immunohistochemistry; LGA = low-grade astrocytoma; M = male; MC = mitotic count in 2.37 mm2 (10 FN22/40× fields); MVP = microvascular proliferation; N = geographic necrosis; NA = not available; NCP = neoplastic cell pleomorphism; OLIG2 = oligodendrocyte transcription factor 2; SF = spayed female; + = positive immunolabeling; – = negative immunolabeling.
Astrocytomas with partial gemistocytic morphology.
Grossly, neoplasms were well-demarcated (5 of 7 cases) or poorly demarcated (2 of 7), tan-to-pale-yellow masses that expanded and partially effaced the cerebellum (4 of 7), thalamus, hypothalamus, and internal capsule (1 of 7), right putamen nucleus, corpus callosum, and lateral ventricle (1 of 7), and left frontal and olfactory lobe (1 of 7). The 4 cerebellar astrocytomas were either midline masses involving both the cerebellar cortex and medulla or lateralized tumors involving one cerebellar hemisphere. In all cases, 2–10-mm intratumoral cysts filled with variable amounts of clear fluid were grossly evident (Figs. 1, 2). Secondary pathologic changes in the brain were occasionally reported and included caudal cerebellar herniation through the foramen magnum, compression of the fourth ventricle, and proximal obstructive hydrocephalus (1 of 7 each).
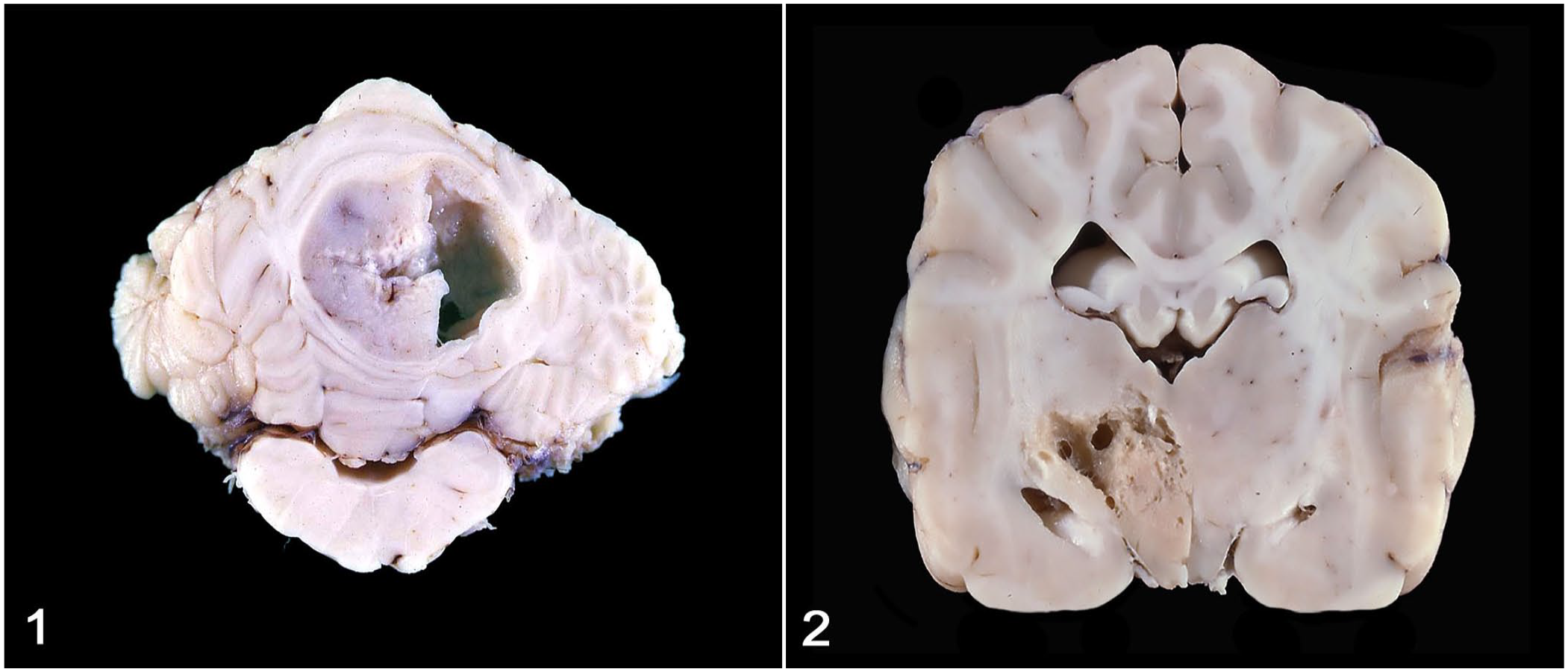
Canine cystic astrocytomas; formalin-fixed brains.
Histologically, most neoplasms were consistent with low-grade astrocytomas (cases 1–6), with one high-grade astrocytoma (case 7). 13 Tumors were sparsely to moderately cellular (cases 1–6) and well-demarcated (cases 1–5), with occasional areas of invasion into the surrounding neuroparenchyma (cases 2, 3, 7). A small area of enlarged neoplastic cells with nuclear atypia (case 1) and an infiltrative pattern (case 5) were rarely observed.
Neoplastic cells were arranged in bundles embedded in eosinophilic fibrillary stroma (Fig. 3). Most neoplastic cells had indistinct cell boundaries with elongate cytoplasm, oval-to-elongate nuclei with dense to finely stippled chromatin, and rare distinct nucleoli. Areas with gemistocytic morphology were common among tumors (cases 2, 3, 6, 7) and consisted of dense sheets of neoplastic cells with abundant, round-to-polygonal, eosinophilic, glassy cytoplasm with distinct cell margins and eccentric, oval-to-elongate nuclei with finely stippled chromatin (Fig. 4). The variably sized, solitary-to-coalescing cystic areas were located within the mass of neoplastic astrocytes and their cytoplasmic processes (Fig. 5). In addition, many microcysts measuring ~200–800 µm were present throughout. Cystic spaces were either apparently empty or contained scant, acellular, flocculent eosinophilic material admixed with a few isolated neoplastic astrocytes, foamy macrophages, and occasionally other leukocytes (Fig. 6). Mitotic activity was absent or low across cases. Geographic coagulative necrosis and microvascular proliferation were observed in the high-grade astrocytoma (case 7). Perivascular accumulations of lymphocytes (cases 2) or scattered areas of mineralization (case 7) were occasionally observed.
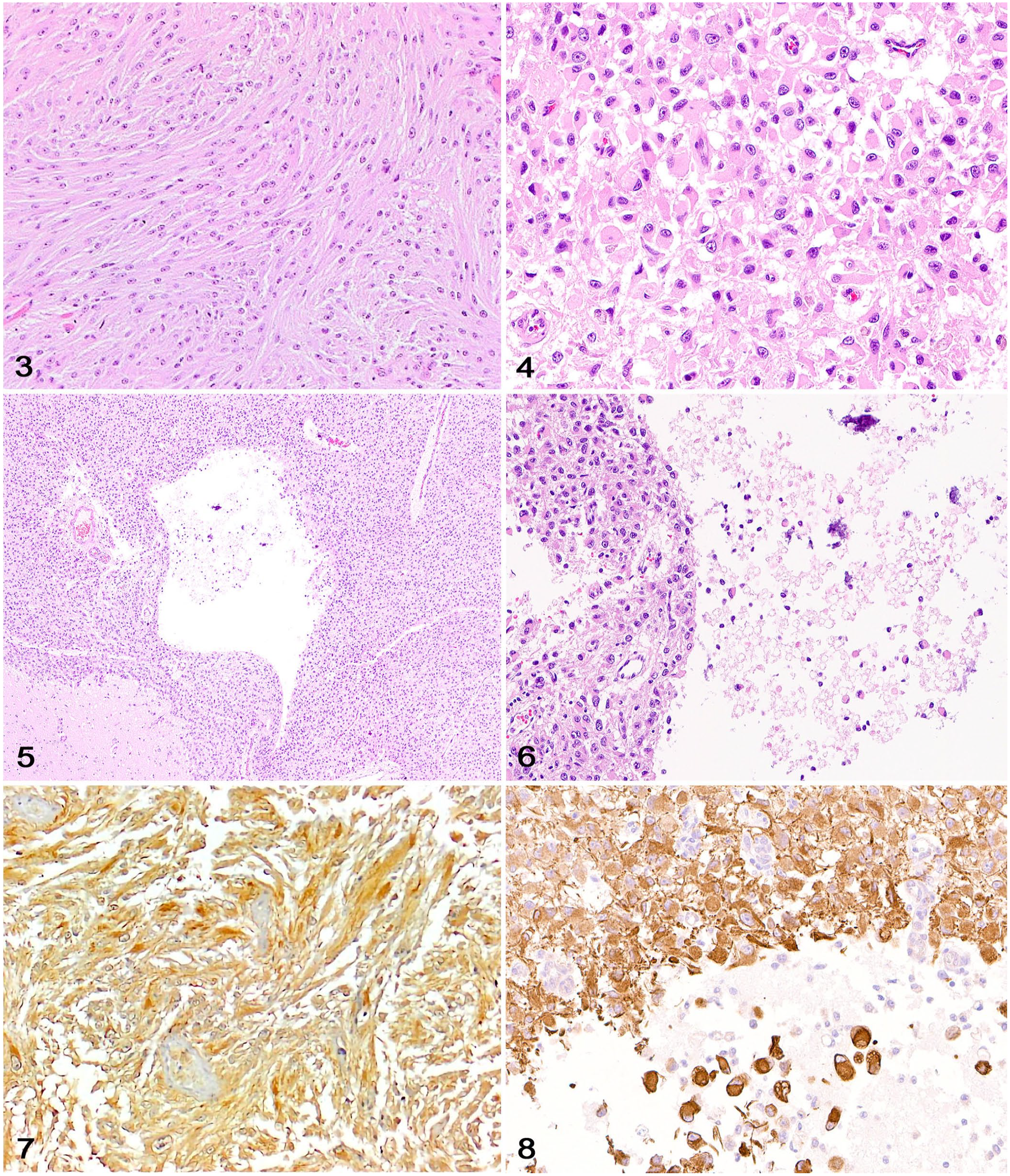
Canine astrocytomas with macrocysts.
Approximately 50–80% of neoplastic cells in 6 of 7 cases (case 6 not available for IHC) had moderate-to-robust cytoplasmic immunolabeling for glial fibrillary acidic protein (GFAP), particularly within areas with gemistocytic differentiation (Figs. 7, 8). GFAP immunolabeling highlighted neoplastic astrocytes surrounding or within the cystic spaces. In contrast to reactive astrocytes, which had uniform immunolabeling with GFAP, neoplastic astrocytes often had mild heterogeneity in the strength of GFAP immunolabeling. Nuclear immunolabeling for oligodendrocyte transcription factor 2 (OLIG2), which was reported in case 7, is variably encountered in astrocytomas, 17 including human pilocytic astrocytomas. 6
Intratumoral macrocysts are occasionally reported as an MRI finding in subsets of canine glioma and may be better known to veterinary radiologists than pathologists.5,12,18,24 Except for sporadic accounts of macrocysts in canine PA,20,22 macrocysts are rarely reported grossly in dogs. Our findings support the assertion that canine cystic astrocytomas are exceedingly rare, as 6 of our cases were collected over several decades by one author (B.A. Summers) and only one was retrieved from the AVDL autopsy archives over a 14-y period in which 89 canine gliomas were diagnosed. Tumor macrocysts were originally reported in 6 of our 7 cases. Although no macrocysts were described in the original autopsy report from case 7, cysts were distinctly observed when reviewing autopsy images of the case, suggesting that tumoral macrocysts may be rare but may also be overlooked or not reported during autopsy or tissue sectioning.
Most of the canine astrocytomas in our report were low-grade tumors with a mixed neoplastic cell morphology. Human PAs typically occur in the cerebellum, and the sizeable tumoral cysts provide a helpful diagnostic imaging or gross anatomic feature for the diagnosis. 9 Interestingly, 4 of our 7 astrocytomas occurred in the cerebellum, and 4 of 7 tumors had areas of gemistocytic differentiation, but none of the neoplasms had pilocytic morphology. Diagnostic imaging data was not available for any of our patients and thus it remains undetermined whether the macrocysts could have been detected clinically, as reported elsewhere.5,12,18,24
The pathogenesis of cyst formation within gliomas is poorly understood and may involve more than one mechanism. 1 Intratumoral cysts may be associated with vasogenic edema secondary to disruption of the peritumoral blood-brain barrier, with subsequent hypoxia, ischemia, and liquefaction of the edematous tissue. 1 Cysts may also develop because of longstanding edema and defective fluid resorption within the tumor, which leads to entrapment of extravasated fluid and subsequent formation of macrocysts from confluent microcysts (those that only can be visualized histologically). 1 Intratumoral microcysts containing proteinaceous material or mucin (which were present in our cases) are a known feature of human pilocytic astrocytoma and subsets of canine oligodendroglioma and astrocytoma. 13 It is thought that in some human astrocytomas, these microcysts could eventually coalesce and become macrocysts. 6 More clinical and pathologic data would be necessary to address this possibility in dogs. Finally, macrocysts could be the result of chronic tumor necrosis,1,15 which would have important implications for tumor grading. 13 However, the high concentration of plasma proteins within the cysts supports a blood-brain barrier disruption and not tumor necrosis as a mechanism for their development.1,15
As observed in our cases, tumoral cysts in human gliomas are lined by neoplastic cells, 1 contrasting with cysts that may occur as a consequence of surgery and chemotherapy, which are typically lined by neuroparenchyma or dura mater and only occasionally by neoplastic cells.1,16 With most studies focusing on the clinical aspects of chemotherapy and radiotherapy on canine glioma, information on the morphologic effects of these treatments on the tumor tissue are lacking.11,21 We have rarely autopsied dogs with glioma that were treated with chemotherapy or radiotherapy. In these few cases, the tumor site is almost completely cystic, collapsed, and replaced by semi-translucent, brown, gelatinous tissue. Histologically, lesions consisted of extensive cystic spaces containing necrotic debris with hyalinized blood vessels associated with numerous hemosiderin-laden macrophages, astrocytosis, and residual glioma (Daniel R. Rissi, Andrew D. Miller, pers. comm., 2024 Jul 3). These findings are consistent with the radiation-treatment effects described in human gliomas and are distinct from those observed in our macrocystic astrocytomas. 19
As evidenced by 4 of 7 cases in our report, cystic astrocytomas may occur more commonly in the cerebellum of dogs, as reported in humans. 9 Further, as described here, low-grade canine astrocytomas seem most prone to undergo cystic change. However, more canine cases are needed before a link between cystic change and predisposed neuroanatomic location or tumor grade can be drawn. In our cases, there was some variation in neoplastic cell morphology that did not appear to be a factor in cystic change. Glial cell heterogeneity has been proposed and increasingly recognized in humans, and it is possible that a distinct subset of astrocytes may account for the development of gliomas with cystic change.2,10
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declare that they received no financial support for their research and/or authorship of this article.
*
Deceased.
