Abstract
Most canine gliomas occur in adult and aged dogs, and reports in puppies < 12-mo-old are exceedingly rare. Here we describe the occurrence of gliomas in 5 dogs ≤ 12-mo-old. The affected patients (4 males, 1 female) were 3–12-mo-old (x̄ = 6.6-mo-old). None of the dogs were brachycephalic. Clinical signs consisted of dullness (2 cases), seizures (2 cases), vestibular signs, and deafness (1 case each). All patients were euthanized. Grossly, neoplasms were pale-tan or red, soft masses in the telencephalon (4 cases) or gelatinous leptomeningeal thickening in the brain and spinal cord (1 case). Neoplasms were classified as astrocytomas (3 cases) and oligodendrogliomas (2 cases) based on histology or histology and IHC. Our findings confirm that, while exceptionally rare, canine gliomas occur in the first year of life, and are clinically, morphologically, and immunohistochemically similar to gliomas in adult and aged dogs.
Oligodendrogliomas and astrocytomas account for nearly 35% of all primary canine brain tumors.3,5,11 These gliomas occur mainly in adult and aged dogs (5–13-y-old), particularly those of brachycephalic breeds, such as Boxers, Bulldogs, and Boston Terriers.5,9,12,17 A male sex predisposition has been suggested. 5 Gliomas in young adult dogs are uncommonly reported,3,5,6,8,11,13 and those in dogs < 12-mo-old are exceedingly rare, with only sporadic single case reports in the veterinary literature.4,7,12,16 Clinicians, neurologists, and pathologists need to be aware that although unlikely, a glioma should still be suspected in young puppies with neuroimaging evidence of an intracranial mass. Here we describe the occurrence of gliomas in 5 dogs ≤ 12-mo-old.
We searched for cases of canine glioma in the autopsy archives of the Athens Veterinary Diagnostic Laboratory (AVDL; College of Veterinary Medicine, University of Georgia, Athens, GA, USA) between 2010 and 2023 using the keywords: canine, glioma, oligodendroglioma, and astrocytoma. Cases were also searched from the private archives of one author (BA Summers) in a period spanning 45 y (1978–2023). Clinical forms and autopsy reports were reviewed, and cases diagnosed in patients ≤ 12-mo-old were selected. Archived H&E-stained tissue sections and immunohistochemistry (IHC) slides were retrieved and reviewed to characterize the morphology and IHC features of the neoplasms. H&E and IHC were performed at the time of diagnosis based on historically validated methods according to laboratory SOPs at the AVDL (cases 3, 5) and Cornell University (cases 1, 2, 4).
Over the studied period (2010–2023), 89 canine gliomas were diagnosed at the AVDL, with only 2 cases (cases 3, 5) in dogs ≤ 12-mo-old. Similarly, only 4 cases were retrieved from the personal collection of one author (BA Summers) in 45 y; 3 of these 4 cases were included here (cases 1, 2, 4); 1 undefined glioma (originally published as oligoastrocytoma) in a 9-mo-old male Cavalier King Charles Spaniel was retrieved from the same collection, but it was not included in our study because it has been published previously as a case report. 16 A search in the archives of 5 other veterinary institutions (The Schwarzman Animal Medical Center, New York, NY, USA; Department of Pathobiology, School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA, USA; Department of Pathobiology, College of Veterinary Medicine, Auburn University, Auburn, AL, USA; Department of Population Medicine and Diagnostic Sciences, Section of Anatomic Pathology, College of Veterinary Medicine, Cornell University, Ithaca, NY, USA; Department of Veterinary Pathobiology, School of Veterinary Medicine & Biomedical Sciences, Texas A&M University, College Station, TX, USA; Department of Veterinary Pathology, Federal University of Rio Grande do Sul, Rio Grande do Sul, Brazil) between 2010 and 2023 retrieved no cases of glioma in dogs < 12-mo-old. Our findings confirm that gliomas occur in young dogs but are exceptionally rare in patients ≤ 12-mo-old.
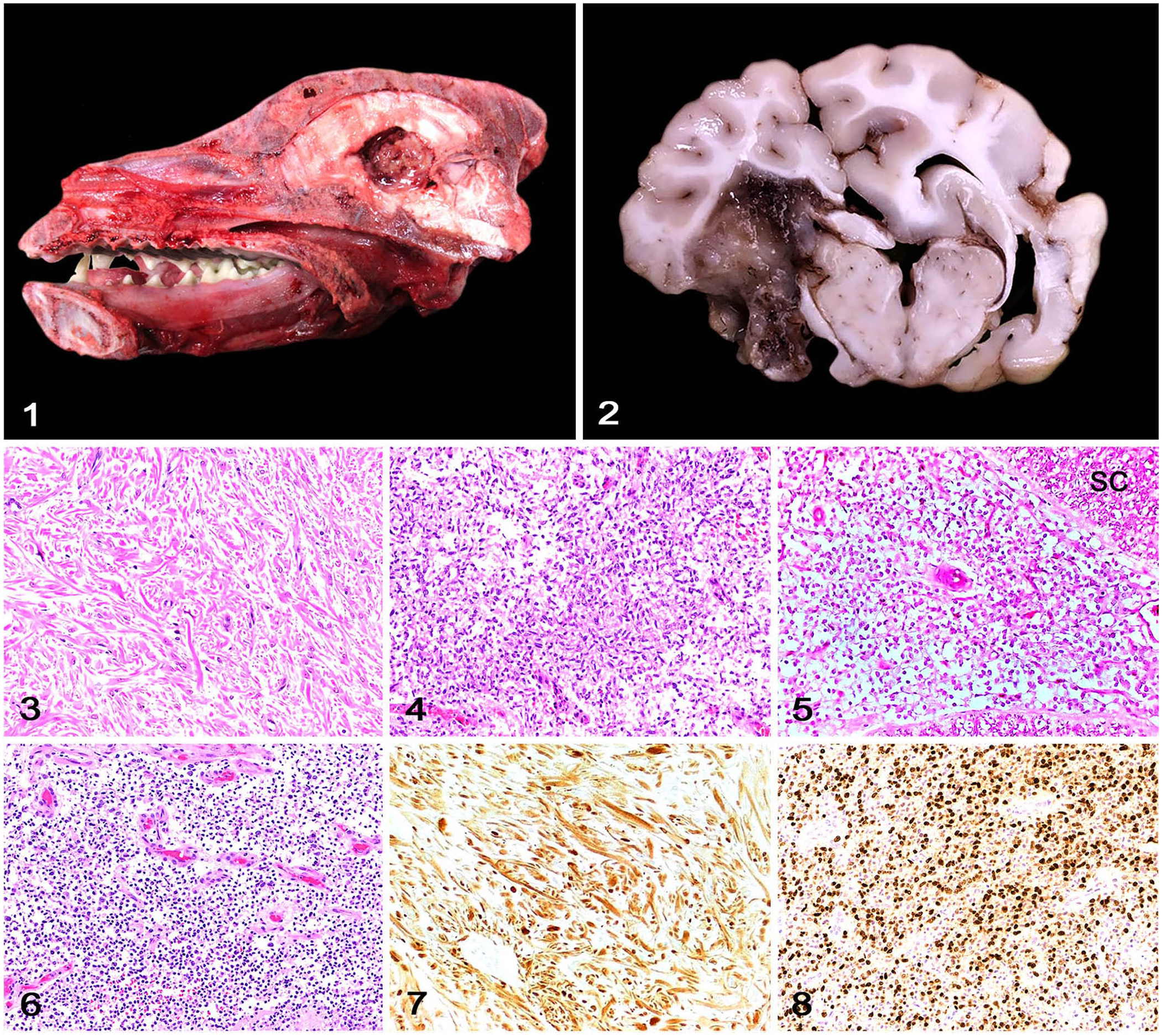
Five cases met the criteria for inclusion in our study (Table 1). The affected dogs (4 males, 1 female) were 3–12-mo-old (x̄ = 6.6 mo). None of the dogs were brachycephalic. Clinical signs consisted of dullness (2 cases), seizures (2 cases), vestibular signs (1 case), and deafness (1 case). All patients were euthanized because of the poor prognosis. Grossly, neoplasms were characterized as pale-tan or red, soft masses (Figs. 1, 2) in the telencephalon (cases 1–3, 5) or as gelatinous leptomeningeal thickening affecting the brain and spinal cord (case 4). Neoplasms were classified as astrocytomas (3 cases) and oligodendrogliomas (2 cases) based on histology or histology and IHC (Figs. 3–8). 5 Briefly, astrocytomas (cases 1–3) consisted of close bundles of oval-to-elongate cells with low-to-moderate pleomorphism and abundant eosinophilic cytoplasm supported by an eosinophilic fibrillar stroma. Nuclei were oval and had finely stippled chromatin with 1–2 nucleoli. In cases 1 and 2 (low-grade astrocytomas), no mitoses were observed in 2.37 mm2 (10 FN22/40× fields). In case 3 (high-grade astrocytoma), 3 mitoses were observed in 2.37 mm2, with areas of geographic coagulative necrosis throughout the neoplasm. Oligodendrogliomas (cases 4, 5) consisted of sheets of round cells with low-to-moderate pleomorphism supported by branching capillaries and a basophilic mucinous stroma. The cytoplasm was clear and formed the typical perinuclear clear halo. Nuclei were round and had dense chromatin and indistinct nucleoli. In case 5 (high-grade oligodendroglioma), 5 mitoses were observed in 2.37 mm2, with areas of geographic coagulative necrosis that were surrounded by palisaded neoplastic cells and microvascular proliferation.
Signalment, clinical signs, gross neuropathology, and diagnosis of glioma in 5 young dogs.
F = female; GFAP = glial fibrillary acidic protein; HGA = high-grade astrocytoma; HGO = high-grade oligodendroglioma; IHC = immunohistochemistry; LGA = low-grade astrocytoma; LGO = low-grade oligodendroglioma; M = male; MC = mitotic count in 2.37 mm2 (10 FN22/40× fields); MVP = microvascular proliferation; N = geographic necrosis with neoplastic cell palisading; NA = not available; NCP = neoplastic cell pleomorphism; NOS = not otherwise specified; OLIG2 = oligodendrocyte transcription factor 2; PLG = primary leptomeningeal gliomatosis; + = positive immunolabeling; – = negative immunolabeling.
Canine glioma in the first year of life.
Pediatric primary CNS neoplasms account for nearly 20% of all cancer in children < 15-y-old 1 ; ~50% of these neoplasms are astrocytomas, followed by embryonal tumors (~24%) and rarely other neuroepithelial tumors (including oligodendroglioma), meningiomas, and nerve sheath tumors.1,2 Primary CNS tumors in children < 1-y-old are rare and consist mainly of medulloblastomas and other embryonal tumors, astrocytomas, ependymomas, and choroid plexus tumors. 10 In dogs, the prevalence of primary intracranial neoplasms increases with age, with a median age of 11 y for meningiomas 11 and 8 y for gliomas.3,5,11,14,15 There are rare detailed reports of gliomas in puppies.7,16
Although MRI was not performed in our cases, limited MRI data from previously published cases suggest that neuroimaging findings in these young dogs are similar to those found in other age groups with brain gliomas.7,16 The clinical signs observed included seizures and altered mentation, as are encountered with canine brain tumors in adults. 3 Based on our findings and previous cases, early-life canine gliomas are also morphologically and immunohistochemically similar to gliomas in adult and aged dogs.7,16 Our 5 cases included both low- and high-grade gliomas, the latter having the conventional histologic hallmarks of malignancy. 5
Veterinary clinicians and neurologists should consider glioma as a highly unlikely but possible differential diagnosis in young dogs with MRI evidence of an intracranial mass.7,16 Three of our 5 dogs were only 3–4-mo-old, and thus it is possible that some tumors may have developed in utero and were present at birth.
Footnotes
Acknowledgements
We thank Drs. Taryn A. Donovan (The Schwarzman Animal Medical Center), Molly E. Church (University of Pennsylvania), Jennifer W. Koehler (Auburn University), Andrew D. Miller (Cornell University), Brian F. Porter (Texas A&M University), and Saulo P. Pavarini (Federal University of Rio Grande do Sul) for their willingness to search for cases in their respective institutions. Dr. A. de Lahunta kindly shared case 1 with Brian A. Summers.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
