Abstract
There have been significant changes to Wisconsin agriculture since ~2010, one of which is the growth of commercial farming of white-tailed deer (WTD; Odocoileus virginianus). These high-density populations may lead to the emergence of previously unrecognized or under-recognized disease trends. I evaluated archived pathology records at the Wisconsin Veterinary Diagnostic Laboratory (WVDL) from 2009–2021 for captive WTD postmortem cases and included records from 277 WTD cases. Diagnoses were found in 81.9% of cases, with an average of 1.3 diagnoses per animal. Submissions with a clinical history of respiratory disease were 7.0 times more likely to have a diagnosis than the average case. Fawns were 1.9 times more likely to have a diagnosis. The most common diagnoses were bronchopneumonia and enteritis and/or enterocolitis (both 16.2% of total diagnoses). The most common isolates from bronchopneumonia cases were Pasteurella multocida, Bibersteinia trehalosi, and Trueperella pyogenes. The pathogens identified most often in enteritis and/or enterocolitis cases were rotavirus, Clostridium perfringens, and Escherichia coli. The most common non-infectious diagnoses were poor nutritional status or inanition, hepatic lipidosis, and selenium and/or vitamin E deficiency. Focusing on testing for pathogens in fawns and in cervid respiratory disease complex submissions may be recommended for laboratories expanding their WTD testing.
Commercial farming of white-tailed deer (WTD; Odocoileus virginianus) has expanded in the state of Wisconsin over recent years. In 2017, 7,943 animals were recorded in the Census of Agriculture, giving Wisconsin the fourth-largest inventory of captive WTD in the United States. 16 The primary health concern for these populations is chronic wasting disease (CWD), which is heavily monitored. As these deer populations have grown, an increased number of postmortem specimens from WTD have been received at the Wisconsin Veterinary Diagnostic Laboratory (WVDL; University of Wisconsin–Madison, Madison, WI, USA) for investigation of possible causes of morbidity and mortality other than CWD. Although ruminant species are generally afflicted by similar processes, there is less information about mortality affecting WTD in captivity and no population-level reports in captive WTD of Wisconsin. Information regarding case submissions may also be useful in identifying factors that influence whether a diagnosis is reached.
I searched for cervid postmortem cases in the archived report database recovered from the laboratory information management system at the WVDL from January 1, 2009, to December 31, 2021. Cases originating from outside the state of Wisconsin, species other than WTD, and free-ranging WTD were excluded from further analysis. In total, I included records of 277 WTD postmortem cases. Information recorded from each case, when available, included the type of submission (whole-body autopsy or fresh and fixed tissues from a field autopsy), signalment (sex, age), city of origin, date of sample submission to WVDL, pathology diagnosis(es) (as reported by the primary pathologist of each case), specific infectious agents when found, whether autolysis was noted in the autopsy report (Y/N), clinical signs, whether the animal was euthanized (Y/N), and CWD status. Some animals had > 1 diagnosis or > 1 infectious agent involved. Given that many of the reports indicated that animals had several causes leading to death or euthanasia, up to 3 diagnoses and 3 infectious agents were included for each animal. Cases were classified as “undiagnosed” if this term or similar terms (inconclusive, indeterminate, unknown, unsure) were used in the reported final diagnosis.
Submissions were divided into age categories (based on standard ages of weaning and previous publications9,15): abortion (“fetus”), neonate (1–7 d or indicated as “newborn”), fawn (1 wk–6 mo or indicated as “fawn”), juvenile (> 6 mo–2 y), and adult (>2 y or indicated as “adult”). Submission dates were divided into seasonal categories: spring (March–May), summer (June–August), autumn (September–November), and winter (December–February). Reports included gross and microscopic examinations of whole bodies or various tissues received from field autopsies. Microbiology tests, including bacterial and fungal cultures, fecal and ingesta parasitology procedures, PCR, and trace mineral analyses, were elected for each case by the primary pathologist at the time of examination. Percentages and averages were calculated in Excel (Microsoft). Odds ratios and CIs were determined by 2-sided Fisher exact tests and by multivariable simple logistic regression model using STATA/SE 17.0 (StataCorp).
Of the 277 WTD cases identified, most were field autopsies (58.5%; Table 1); 43.3% were male and 39.7% were female (remaining cases did not designate sex). Most cases were received in summer (171; 61.7%) followed by autumn (62; 22.4%), winter (27; 9.8%), and spring (17; 6.1%). Autolysis was noted in 37.9% of reports. Twenty-five (9.0%) animals were euthanized: 11 by gunshot, 6 by intravenous barbiturate injection, 3 by captive bolt, 1 by bowshot, 1 by exsanguination, and 3 by an unknown method; 187 (67.5%) animals died (remaining cases did not indicate the manner of death). Two animals (0.7%) tested positive for CWD by rectal biopsy (1) and brainstem (1) immunohistochemistry. Twenty (7.2%) additional animals were tested for CWD, which was not detected (CWD status of remaining cases is unknown). Clinical histories were summarized and categorized as acute death (68; 24.5%), diarrhea (44; 15.9%), lethargic (34; 12.3%), respiratory disease (33; 11.9%), poor doer (24; 8.7%), neurologic or ataxic (8; 2.9%), other (8; 2.9%), and abortion (4; 1.4%). Remaining cases (54, 19.5%) had no history provided.
Captive white-tailed deer cases received at WVDL, 2009–2021, including type of submission (fresh and fixed tissues from a field exam or whole-body postmortem examination), sex, season submission was received, age category, whether autolysis was noted in the autopsy report (Y/N), whether the animal was euthanized (Y/N), CWD status, and clinical presentation category. Diagnostic rate, odds of finding a diagnosis(es) compared to average, and CIs as calculated by a Fisher exact test are listed.
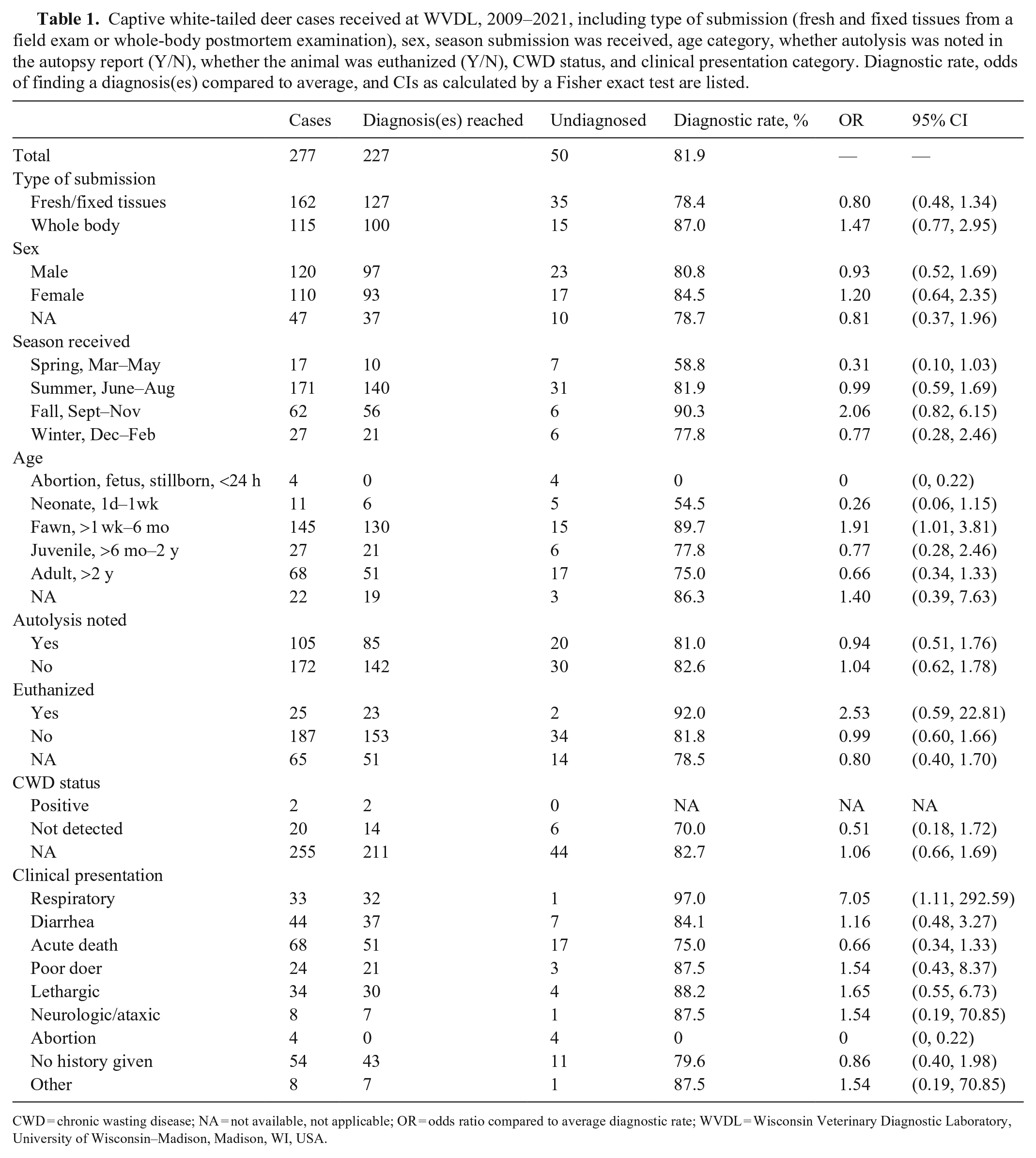
CWD = chronic wasting disease; NA = not available, not applicable; OR = odds ratio compared to average diagnostic rate; WVDL = Wisconsin Veterinary Diagnostic Laboratory, University of Wisconsin–Madison, Madison, WI, USA.
Diagnoses were achieved in 227 of 277 (81.9%) cases. Of these 227 cases, 110 (48.5%) reports indicated that the cause of death or euthanasia was multifactorial with an average of 1.3 diagnoses/animal. I classified diagnoses according to the affected body system (Table 2). Respiratory disease, gastrointestinal disease, and systemic disease (including septicemia, parasitism, suboptimal nutritional status or inanition, and peritonitis) were the most common categories. Suboptimal nutritional status or inanition was defined as poor body condition, serous atrophy of fat, or as otherwise indicated by the primary pathologist at the time of evaluation. Bronchopneumonia and enteritis and/or enterocolitis were the most common diagnoses (both 16.2% of total diagnoses). The most common non-infectious diagnoses were suboptimal nutritional status or inanition (7.0%), hepatic lipidosis (2.0%), and vitamin E and/or selenium deficiency (1.9%).
Diagnoses and infectious agents found in captive white-tailed deer postmortem examination reports at WVDL, 2009–2021, listed by organ system. Up to 3 diagnoses and 3 infectious agents were included for each animal.

NA = not available, not applicable; WVDL = Wisconsin Veterinary Diagnostic Laboratory, University of Wisconsin–Madison, Madison, WI, USA.
Of 370 total diagnoses found in 277 cases.
Several factors were associated with diagnostic rates less than the average, including field autopsy cases, male animals and those with undesignated sex, cases received in spring and winter, abortion, neonatal, juvenile, and adult submissions, cases with autolysis noted in the report, cases without a method of death indicated, and cases with no clinical history provided or a history of acute death. Spring submissions were 0.31 times as likely to have a diagnosis as the average WTD case (p = 0.028) when compared by a 2-sided Fisher exact test. No abortion or stillborn cases were diagnosed (p = 0.001). Neonatal cases were 0.26 times as likely to have a diagnosis as the average case (p = 0.039).
The fawn age category and cases with a clinical history of respiratory disease both had diagnostic rates statistically significantly higher than average. Fawns were 1.9 times more likely to have a diagnosis compared to the average WTD case (p = 0.046). Cases with a clinical history of respiratory disease were 7.0 times more likely to have a diagnosis as the average case (p = 0.025). Of those cases submitted with a clinical history of respiratory disease, 84.8% had a diagnosis of respiratory disease. Of the 219 diagnoses in the fawn age category, 27.4% involved the respiratory system. Fawns were more likely to be diagnosed with enteritis and/or enterocolitis and septicemia compared to juveniles and adults. Enteritis and/or enterocolitis was also more likely diagnosed in summer than in other seasons.
A multivariable logistic regression model was used to evaluate the association between factors and each of the top 2 diagnoses (bronchopneumonia, enteritis and/or enterocolitis). Explanatory variables (Table 1) included season (4 categories), age (6 categories), sex (3 categories), autolysis (2 categories), euthanasia (3 categories), and CWD status (3 categories). Association between each explanatory variable and each outcome was examined using univariable logistic regression models. A likelihood ratio test p ≤ 0.25 from the univariable analysis was used to screen explanatory variables for inclusion in the multivariable logistic regression model. Season was the only factor influencing the odds of a diagnosis of bronchopneumonia (OR = 1.60, 95% CI = 1.08–2.37; p = 0.019); age was the only factor influencing the odds of a diagnosis of enteritis and/or enterocolitis (OR = 0.75, 95% CI = 0.57–1.00; p = 0.047). Secondary Fisher exact tests indicated that a diagnosis of bronchopneumonia was 0.47 times more likely in the summer than other seasons (95% CI = 0.26–0.85; p = 0.005) and a diagnosis of enteritis/enterocolitis was 5.27 times more likely in fawns than in other age groups (95% CI = 2.43–12.6; p < 0.00001).
Infectious pathogens identified included bacterial, viral, fungal, and parasitic agents (Table 2). Bronchopneumonia, interstitial pneumonia, and pleuropneumonia and/or pleuritis were associated with single and multiple bacterial agents, predominantly Trueperella pyogenes, Pasteurella multocida, and Bibersteinia trehalosi. T. pyogenes was also the most frequently isolated agent that was associated with abscesses in various organ systems (lung [4 of 5], intracranial [1 of 3], retrobulbar [1 of 1], and osteomyelitis [1 of 4]). Fusobacterium necrophorum was isolated from cases of necroulcerative laryngitis (2 of 4) and necrotizing stomatitis (4 of 5) in combination with other bacteria (T. pyogenes or B. trehalosi). The most frequently identified enteritis and/or enterocolitis-associated infectious agents included rotavirus, Clostridium perfringens, and Escherichia coli. C. perfringens was also associated with abomasitis (4 of 9), rumenitis (1 of 15), pleuritis (1 of 12), epicarditis (1 of 5), and myocarditis (1 of 4). Genotyping was attempted on 5 isolates from anaerobic cultures with corresponding lesions, including C. perfringens type A associated with enteritis (2), enterocolitis (1), and abomasitis (1), and C. perfringens type E associated with enterocolitis (1). The most commonly identified gastrointestinal parasites were strongyles, coccidia, and Strongyloides sp. Malignant catarrhal fever was identified in 3 cases, confirmed by PCR assay for ovine herpesvirus 2 (Macavirus ovinegamma2).
Although statistical methods identified few significant correlates between diagnostic outcomes and factors, sample size may have been a limitation for statistical significance. Descriptively, fawns were the largest age category of submissions and were predominantly received in summer, which is consistent with the WTD fawning season of late May to early June. Compared to other age groups, the fawn age category also had the highest number and percentage of diagnoses of bronchopneumonia, enteritis and/or enterocolitis, septicemia, poor nutritional status or inanition, and parasitism, which may indicate increased susceptibility of fawns to these diseases. Management data were not collected because of poor reporting of histories on submission forms. Fawn management on deer farms can vary dramatically, from animals being raised in a field alongside their does to being housed in barns or pens and being bottle-fed. Diet can also vary dramatically given that some facilities will feed specific formulas to promote antler growth in anticipation of these animals being used on hunting ranches; others provide only forage and browse. Weaning is another significant stress factor for fawns and, similar to traditional domestic ruminants housed in high-density populations, likely contributes to the occurrence of opportunistic gastrointestinal and respiratory disease. Additional information about management practices would likely contribute to a better understanding of the associations described in this population and the probability of definitive diagnoses.
There are few large-scale surveys of disease and pathology in captive cervid herds, and even fewer that include WTD. Those reviewed did find similar causes of death and associated infectious agents to my study in Wisconsin. In a 3-y review of mortality of captive WTD in Pennsylvania (2000–2003), the most common causes of death were bronchopneumonia, enterocolitis, malnutrition, and trauma. 9 Other studies, including survey data from deer farmers in addition to postmortem records, indicate that mortality is higher in fawns < 1-y-old.6,7,9 The most common infectious agents identified included Trueperella (formerly Arcanobacterium, Actinomyces, and Corynebacterium) pyogenes, F. necrophorum, E. coli, and Mannheimia haemolytica. 9 In free-ranging WTD, bronchopneumonia was frequently identified in those animals in which lung lesions could be assessed. 5 When pathogens were identified, infections were frequently mixed, including T. pyogenes, B. trehalosi, and P. multocida. C. perfringens was frequently cultured in association with enterocolitis 9 ; however, it should be noted that isolation of C. perfringens from postmortem tissue does not necessarily indicate that C. perfringens was a primary cause of disease. Similar to other ruminant species, proliferation of intestinal C. perfringens following rapid introduction of high-carbohydrate rations causing acute enterotoxemia has been proposed in WTD, 9 but this association has not been demonstrated in controlled conditions.
Studies of other farmed cervid species have identified a similar array of diseases. A survey of Canadian and American farmed elk (Cervus elaphus) producers and a review of autopsy records identified the highest mortality rates in neonates, with the most common diagnosis being unknown or emaciation; however, there was a high rate of undiagnosed cases and unknown etiologies in every age group. 17 A review of farmed elk mortality in Pennsylvania identified gastrointestinal parasitism, pneumonia, enterocolitis, malnutrition, and rumenitis and/or acidosis as the most common causes of death. 10 T. pyogenes was the most frequently isolated etiologic agent in both elk studies. CWD remains a looming concern for farmed cervids in North America although not represented in these retrospective analyses, likely because CWD suspects are not typically submitted for routine autopsy.
T. pyogenes has frequently been associated with various diseases in WTD and other ruminants while also being a commensal organism of the skin, gastrointestinal, urogenital, and upper respiratory tracts in apparently healthy animals. 14 Reports of T. pyogenes–associated severe necrotizing pleuropneumonia, 13 similar to many descriptions in my study, attribute these infections to stress and presumed associated immunosuppression. Questions still surround the overall virulence of T. pyogenes and factors that may influence its transition from commensal to pathogen. 14 T. pyogenes is frequently associated with abscesses, particularly cranial and intracranial abscesses, in WTD. Geospatial clustering of cranial abscesses and isolation of similar T. pyogenes strains has been observed in free-ranging WTD, suggesting that both intrinsic and environmental factors contribute to the occurrence of disease. 1 For captive WTD, this raises questions regarding husbandry practices, stocking density, and appropriate preventive health measures.
In traditional domestic ruminants, infectious agents involved in pneumonia in young animals are frequently referred to as a “respiratory disease complex” to reflect contributing factors other than cultured bacteria, many of which are also commensals. Additional factors, such as viral infections, may predispose animals to severe bacterial disease with few to no recognizable histologic findings. 18 Viral infections targeting the respiratory tract have been documented in WTD and can be associated with immunosuppression and mild clinical disease, which may predispose infected animals to opportunistic bacterial agents.8,12 Seropositivity for bovine herpesvirus 1 (Varicellovirus bovinealpha1) and bovine parainfluenza virus 3 (Respirovirus bovis) has been documented in free-ranging WTD in Wisconsin; however, the clinical significance of these viruses for WTD is unclear. 3 Mycoplasmal respiratory infections, including Mycoplasma bovis 4 and M. ovipneumoniae, 11 with associated pulmonary lesions, have been described in WTD, which also may allow for opportunistic bacterial colonization. In my study, both M. bovis and M. ovipneumoniae were identified in one case of pneumonia each by PCR. PCR testing for bovine herpesvirus 1, bovine viral diarrhea virus 1 and 2 (Pestivirus bovis, Pestivirus tauri), bovine respiratory syncytial virus (Orthopneumovirus bovis), and bovine parainfluenza virus 3 was attempted sporadically, but no positive cases were identified. Coinfections of bacterial agents with underlying, undetected or under-detected, immunosuppressive viral or mycoplasmal infections may produce a synergistic effect and more severe disease. Further characterization of the respiratory disease complex in WTD may further elucidate these interactions.
Stress can play an even more pronounced role in disease in semi-wild species than in more traditional domestic animals. There is commonality, however, in the sources of stress, including capture, transport, weaning, changes in social structure, adverse climate, and poor nutrition. 13 Capture, which is an extremely stressful event in both free-ranging and captive WTD, can influence the probability of mortality. 2 The handling of deer presents difficulties in acquiring antemortem samples, administering preventive treatments, and increasing risk for pathogen spread. 12
Limitations of my study include its retrospective format, relatively small sample size, variations in tissue submission, and variation in sampling and testing of submissions by the primary pathologists at the time of postmortem examination. Accurate dates of death were not provided with all submissions, allowing for variation in postmortem intervals and seasonal classification of received samples. Autolysis was not found to be a significant factor affecting diagnosis, although this factor was associated with diagnostic rates less than the average. Additionally, cases received at the WVDL were likely a biased subset of the total Wisconsin captive WTD population, potentially excluding cases in which the cost of a postmortem examination was prohibitive, cases that were too autolyzed for submission, and cases that were diagnosed clinically or by gross postmortem examination only.
Footnotes
Acknowledgements
My thanks to WVDL pathologists and staff past and present who contributed autopsy reports to the archives.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
I received no financial support for the research, authorship, and/or publication of this article.
