Abstract
A 3-month-old fawn from a group of 12 captive white-tailed deer (Odocoileus virginianus) displaying cutaneous lesions was presented to the Mississippi Veterinary Research & Diagnostic Laboratory for necropsy. Postmortem examination identified multiple discrete, round, alopecic, flat, proliferative dermal lesions scattered along the skin of the lips, muzzle, pinna, ventral thorax, medial limbs, and most notably the abdomen. Multiple ulcers were present on the commissures of the lips, dorsal surface of the tongue, and left caudal buccal surface of the oral cavity. The abdomen was filled with fibrinopurulent exudate and ruminal contents. Multiple to coalescing transmural ulcers were identified in the rumen. Histopathological evaluation of the skin revealed markedly thickened epidermis and focal areas of superficial dermal fibrosis, intracytoplasmic, eosinophilic inclusions in swollen keratinocytes and lymphocytic and plasmacytic perivascular dermatitis. The rumen ulcers were surrounded with necrotic cellular debris mixed with fibrin, bacteria, hemorrhages, and a collection of mixed inflammatory cells. Some swollen ruminal mucosal epithelia had eosinophilic intracytoplasmic inclusions. Poxvirus was isolated from the skin and rumen tissue specimens. Electron microscopy detected viral particles with poxvirus morphology. Polymerase chain reaction assays detected A21, a gene conserved within family Poxviridae, in the skin and rumen tissues. Phylogenic analysis of the A21 sequences indicated that the viral isolate (M10-9055) was closely related to known members of genus Cervidpoxvirus. In conclusion, findings indicate that Deerpox virus can produce extensive lesions in white-tailed deer.
The Poxviridae family, one of several viral families representing large DNA viruses, consists of 2 subfamilies, Entomopoxvirinae and Chordopoxvirinae. Based on morphological and biological characteristics, 3 genera plus 1 unassigned genus are placed in the Entomopoxvirinae subfamily, while 9 genera plus 1 unassigned genus are recognized within the Chordopoxvirinae subfamily (http://www.ictvonline.org/virusTaxonomy.asp).3,9 All members of Chordopoxvirinae are pathogens of vertebrate hosts with 8 genera infecting mammals and 1 genus, Avipoxvirus, infecting birds.3,9 Advances in the understanding of Deerpox virus (DPV) led to the proposition and ratification of the new genus Cervidpoxvirus, in subfamily Chordopoxvirinae, family Poxviridae, by the International Committee on Taxonomy of Viruses (ICTV) in 2006.
The composition of the poxvirus particle is complex. During the process of virus replication, poxvirus produces 2 types of virions, extracellular and intracellular.3,9 The double-stranded DNA genome of poxvirus is fairly large and encodes all the enzymes and structural proteins for its life cycle. The central region of the viral genome is essential, and it is conserved among most members in subfamily Chordopoxvirinae.3,4,9 Poxviruses replicate in the cytoplasm of infected cells resulting in the formation of cytoplasmic inclusion bodies. Like other poxviruses, DPV is enveloped and oval-shaped, 200 nm in diameter, and 230 nm in length. 8 When examined with an electron microscope (EM), the surface membrane of DPV displays tubular or filament structures, a typical appearance of all poxviruses. 8 Complete sequence determination of 2 DPVs (W-848-83 and W-1170-84) has revealed that the size of the DPV genome varies between 166 and 170 kilobases (kb), comprising 169 and 170 genes, respectively. 1 In addition to the genes common to all poxviruses, several unique genes potentially involved in the modulation of the host immune system have been identified in the genome of DPV. 1
Although the prevalence and pathogenicity of DPV are not fully understood, the virus has been isolated from mule deer (Odocoileus hemionus hemionus), reindeer (Rangifer tarandus), and black-tailed deer (Odocoileus hemionus columbianus).10,11 It has also been shown that experimental infection of black-tailed deer via intravenous or intracutaneous routes can induce clinical symptoms. 2 Phylogenic and morphologic characterizations of DPVs isolated from different host species located in various geographical regions indicate that these viruses are closely related. 8 In the current study, a report on the isolation of DPV from a white-tailed deer (Odocoileus virginianus) is documented.
An approximately 3-month-old, female white-tailed deer fawn was found dead and presented to Mississippi Veterinary Research & Diagnostic Laboratory for necropsy in August 2010. The animal had a history of conjunctivitis, skin lesions, oral ulcers, and abdominal distention. Skin lesions were frequently alopecic, circumscribed, proliferative, and eroded. The fawn was 1 of 12 captive bred white-tailed deer fawns in an exotic and domestic hoof stock enclosure in Mississippi showing similar clinical signs. The affected animals were bred and housed in a 900-acre exotic and domestic hoof stock enclosure. Additional animal species in the enclosure included red deer (Cervus elaphus), fallow deer (Dama dama), axis deer (Axis axis), African blackbuck antelope (Antilope cervicapra), llama (Lama glama), barbados blackbelly sheep (Ovis aries), mouflon sheep (Ovis aries orientalis), feral hogs (Sus scrofa), horses, and emu (Dromaius novaehollandiae). Among all animals kept on the premises, white-tailed deer was the only host species affected by the disease. The 12 clinically affected deer fawns were treated with procaine penicillin G (20,000 IU/kg via subcutaneous route once a day) to control secondary bacterial infections. Skin lesions in 11 fawns resolved completely within 3 weeks of the onset of clinical signs.
On necropsy, the fawn had multiple, widely scattered circumscribed, 0.5–1.5 cm in diameter, alopecic, flat, thickened cutaneous lesions on the lips, muzzle, pinna, thorax, and, most notably, the abdomen (Fig. 1A, 1B). Additionally, the fawn had unilateral corneal ulceration, bilateral conjunctivitis, and periocular alopecia and erosions. The lip commissures, dorsal surface of the tongue, and left caudal buccal surfaces of the oral cavity had multiple to coalescing variable sized ulcers. The abdomen was filled with malodorous, opaque liquid, mixed with flocculent material, fibrin strands, and ruminal contents. The pale rumen contained multiple to coalescing, 0.5–1 cm in diameter, transmural ulcers (Fig. 1C). The liver, kidney, adrenal glands, and remaining intestinal tract had no significant findings.
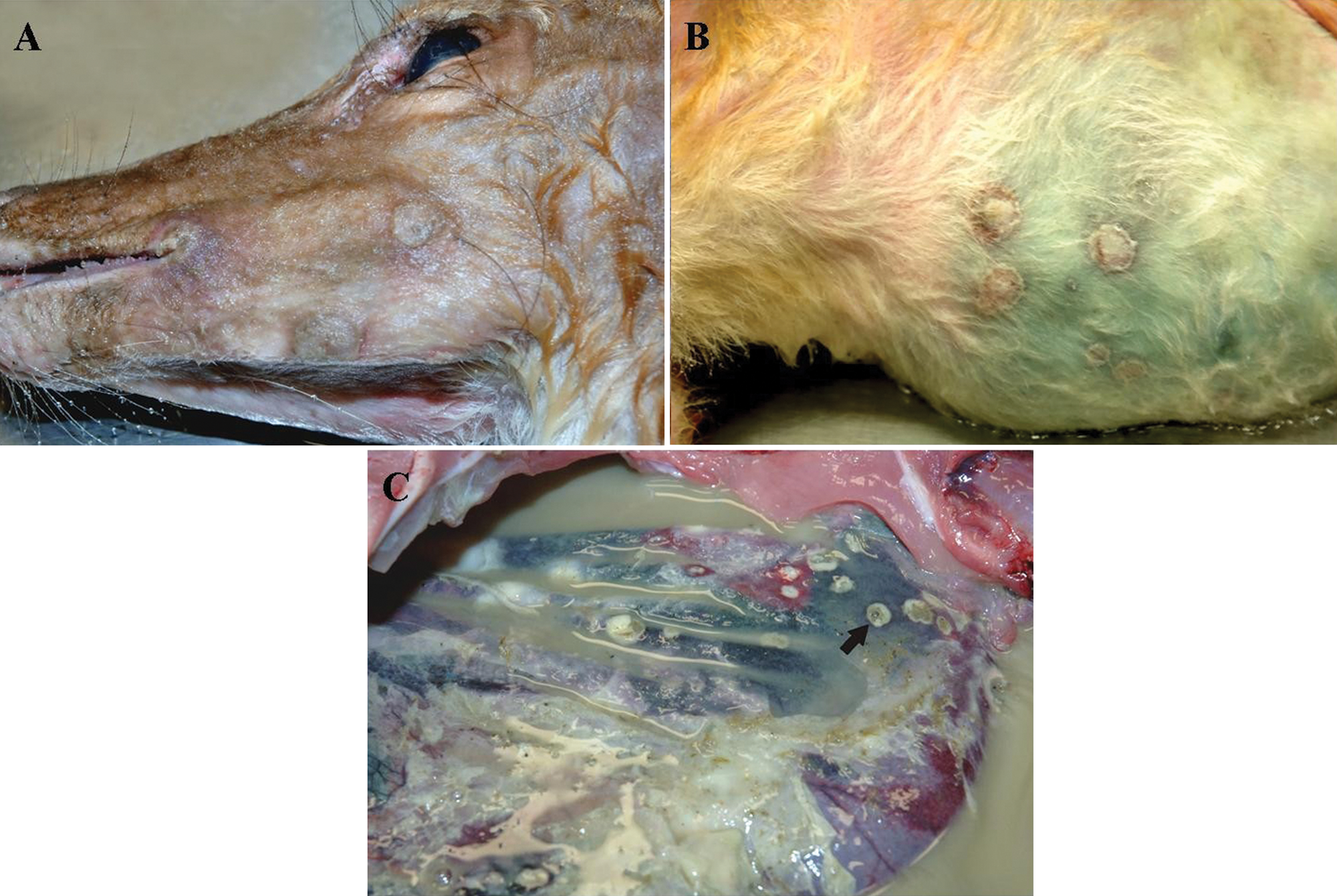
White-tailed deer (Odocoileus virginianus).
Representative tissue samples of skin, oral mucosa, rumen, and spleen were fixed in neutral buffered, 10% formalin solution, trimmed, routinely processed, embedded in paraffin, sectioned at approximately 5 μm, and stained with hematoxylin and eosin. Microscopically, the skin tissue sections had multiple raised epidermal plaques and nodules, predominantly composed of markedly thickened epidermis over areas of granulomatous dermatitis, with fibrosis, and perivascular collections of lymphocytes and plasma cells. Keratinocytes of the stratum spinosum and follicular infundibular root sheath had marked cytoplasmic vacuolation and swelling, or ballooning degeneration, with scattered, distinct, round to oval, eosinophilic inclusion bodies surrounded by a clear, unstained zone. Cells within the expanded stratum corneum above often retained the nuclei (parakeratotic hyperkeratosis).
The multifocally ulcerated ruminal mucosal surfaces were outlined by an accumulation of amorphous necrotic cellular debris, bacteria, and mixed inflammatory cells embedded in a layer of fibrin (Fig. 2). Occasionally, the intact ruminal mucosa had discrete, circumscribed, and thickened foci of ballooning degeneration, with intracytoplasmic inclusion bodies. Similar findings were observed on the tongue. Moderate lymphocytes depletion in splenic lymphoid follicles was observed. No other tissues were evaluated. Based on pathological findings, a primary poxvirus infection, with secondary bacterial infection was suspected.
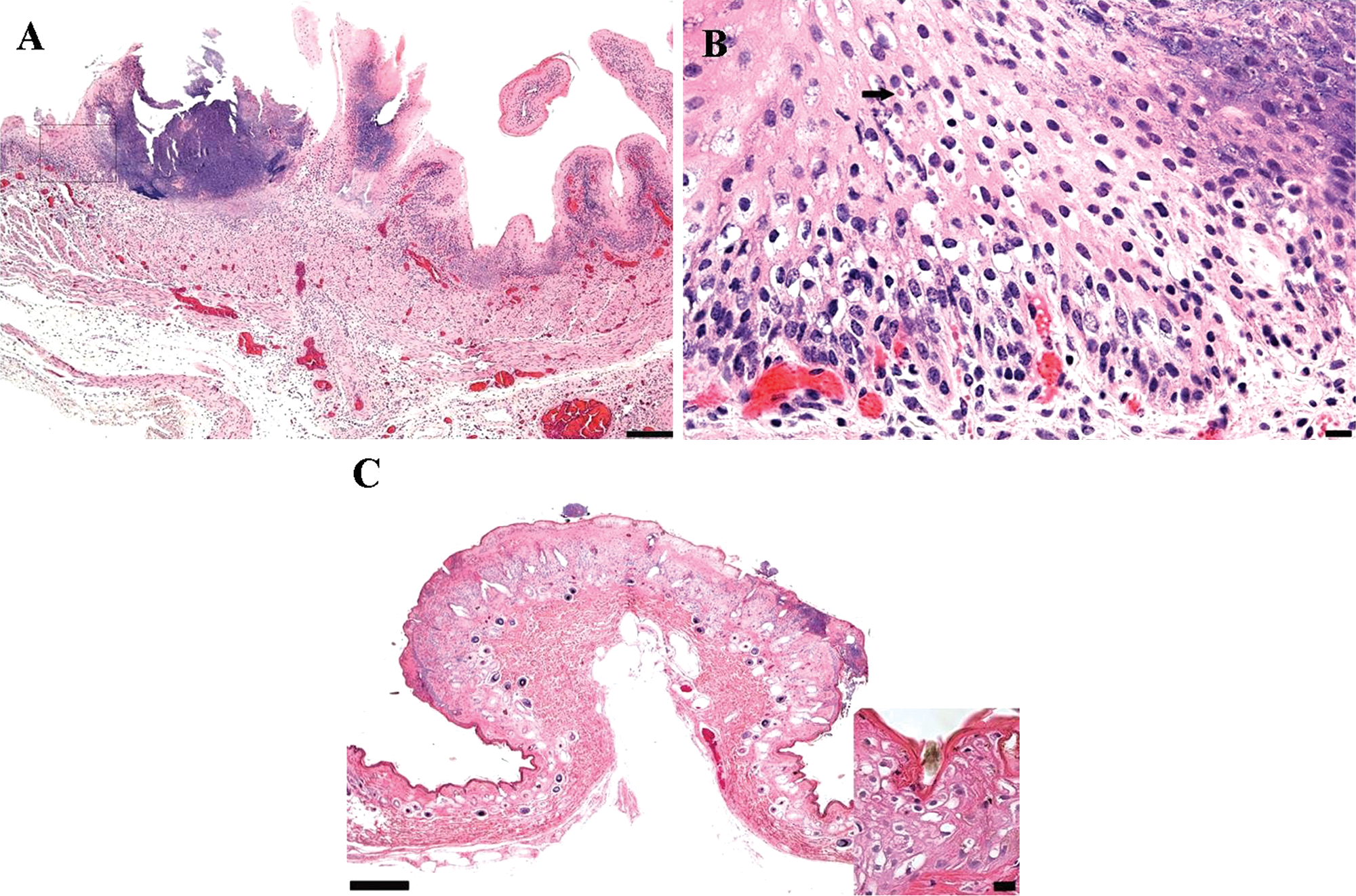
White-tailed deer (Odocoileus virginianus).
For virus isolation, tissue specimens, including skin, oral mucosa, and rumen, were processed as described previously.2,8 In brief, tissue homogenates were inoculated onto primary testicular cells of black-tailed deer maintained in minimal essential medium a supplemented with 10% fetal bovine serum. b The inoculated cultures were examined daily for up to 7 days for cytopathic effect characterized by enlarged cytoplasm containing many inclusion bodies and cell detachment. Poxvirus was isolated from skin and ruminal lesions, and the virus identity was confirmed by electron microscopy (oval-shaped virions of 200 nm × 230 nm; Fig. 3). Poxvirus-like viral particles were also observed ultrastructurally in tissue sections prepared from skin, oral mucosa, and rumen (data not shown).
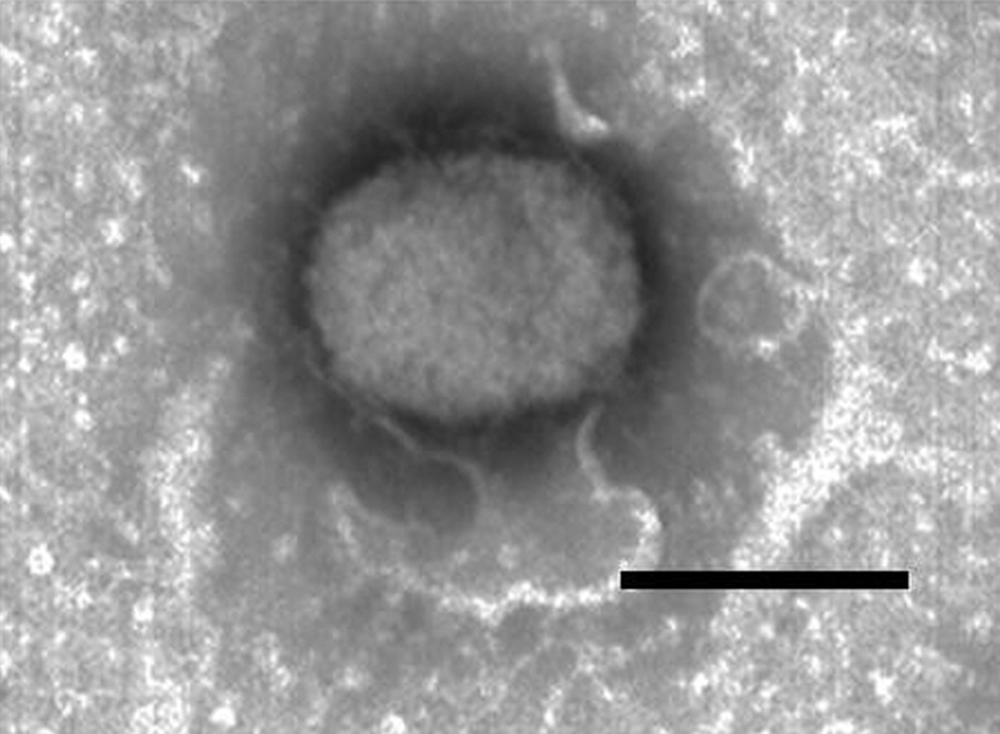
Electron micrographs of Deerpox virus (DPV) virions prepared by negative staining DPV particles harvested from infected Vero (African green monkey kidney epithelial) cell culture. Bar = 250 nm.
To rule out infection with Bluetongue virus (BTV), Bovine viral diarrhea virus (BVDV), Epizootic hemorrhagic disease virus (EHDV), and Malignant catarrhal fever virus (MCFV), polymerase chain reaction (PCR) tests were performed on available fresh tissues according to the in-house protocols. The PCR results were negative for BTV, BVDV, EHDV, and MCFV.
For identification of bacterial etiologic agent(s), fresh tissue samples were cultured on blood agar, MacConkey agar, and anaerobic phenylethyl alcohol blood agar plates c and incubated at 35°C for up to 72 hr. Anaerobic environment was created using a sealed anaerobic jar containing an anaerobic gas-generating sachet. d Rumen cultures showed heavy growth of Escherichia coli, Fusobacterium spp., Pasteurella multocida, and Pseudomonas aeruginosa. Peritoneal fluid cultures yielded heavy growth of P. multocida whereas cultures from skin lesions showed moderate growth of Staphylococcus xylosus. Identification of the bacterial species was based on colonial morphology, growth characteristics, and biochemical tests using a commercial identification system. e
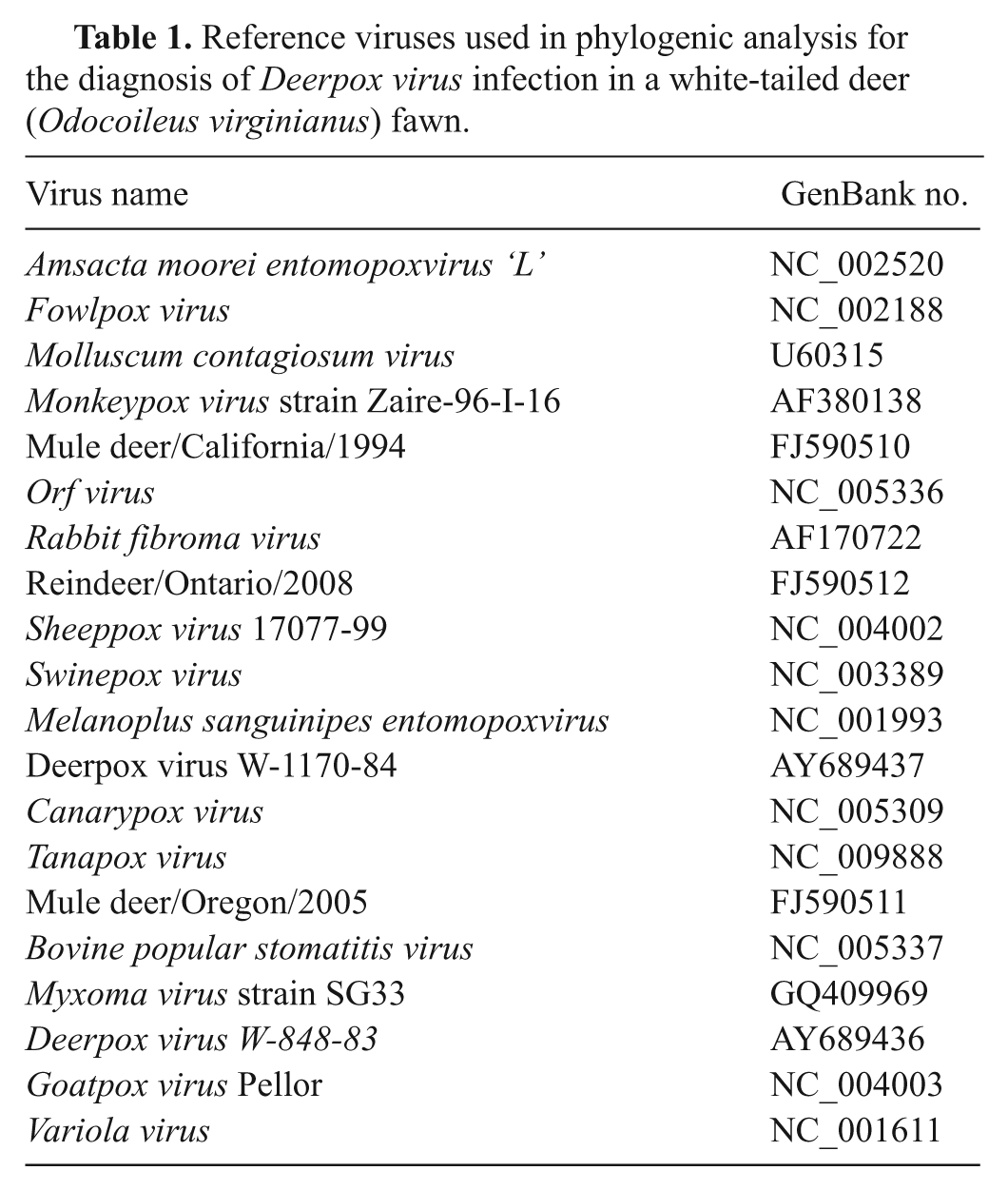
For a definitive characterization of the poxvirus isolate, DNA was extracted from samples of skin, oral mucosa, rumen epithelium, and infected cell cultures using a DNA extraction kit, f according to the manufacturer’s instructions. The PCR assay targeting genes, which encoded a conserved poxvirus virion membrane protein (A21), were conducted as previously described. 8 All primers were commercially synthesized. g The PCR amplicons were separated on 1% agarose gel and visualized under a gel documentation system. h Amplicons with the expected sizes (391 base pairs [bp]) were detected in all tissue and cell culture samples examined, suggesting that the viral isolate was a member of the Poxviridae family. Subsequently, PCR amplicons were sequenced from both directions by a commercial sequencing facility. g The sequences were compared with GenBank entries using the Basic Local Alignment Search Tool (BLAST), which indicated 100% identity among the A21 genes of the Mississippi isolate (MS10-9055) from white-tailed deer, Wyoming isolates (W83 and W84) from mule deer, and Ontario isolate (2008) from reindeer. To characterize the isolate, the sequences of the amplicons and A21 sequences of selected poxviruses were aligned using ClustalX version 2.0.12. 5 Maximum parsimony and neighbor-joining analyses were performed using PAUP* 4.0 Beta as described previously. 7 Maximum likelihood tree estimation was evaluated using GARLI version 0.951. i Tree topologies were confirmed between each of three methods. Bootstrapping support for tree topologies was performed using the neighbor-joining method implemented in PAUP* 4.0 Beta with 1,000 replicates. 7 Phylogenetic analysis of A21 sequences indicated that the isolate (M10-9055) and known DPVs formed a cluster that was closely related to the Capripoxvirus genus (Fig. 4). All sequences used in the study are listed in Table 1. The identity of M10-9055 was further confirmed by PCR amplification of Cervidpoxvirus-specific gene encoding a C-type lectin-like receptor (CTLR) homologue as previously described. 8 Comparison of CTLR sequences revealed genetic heterogeneity within the Cervidpoxvirus genus, with M10-9055 being most closely related to DPV W-1170-84, a mule DPV (data not shown).
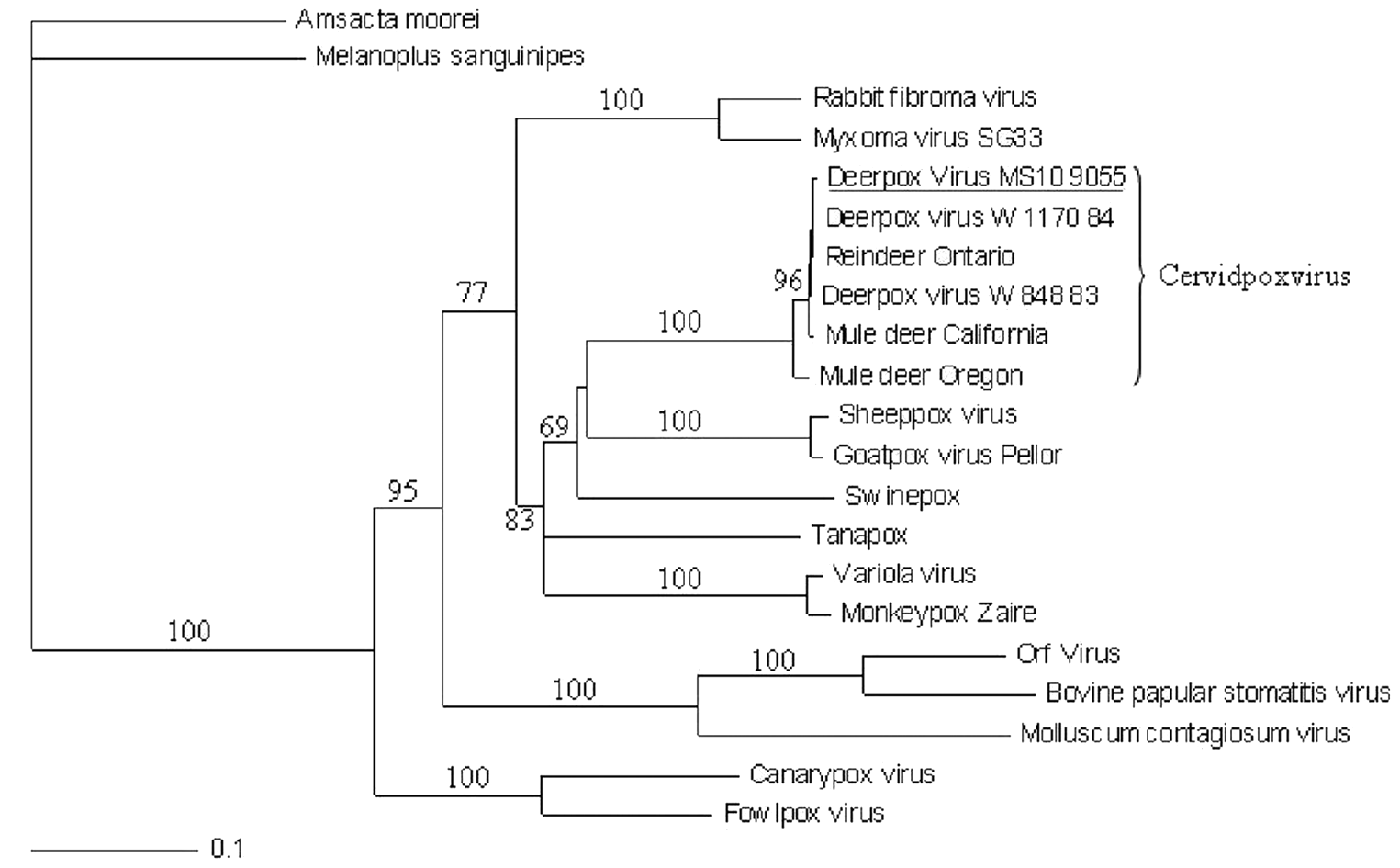
Phylogenic analysis of the Deerpox virus (DPV) isolated from a white-tailed deer (Odocoileus virginianus) fawn (M10-9055) and representative poxviruses. Nucleotide sequences of A21 gene were aligned as described in the text. Maximum parsimony and neighbor-joining analyses were performed using PAUP* 4.0 Beta, and the tree is shown in a rooted format. The bootstrap values from 1,000 replica samplings and the divergence scale are indicated.
Reference viruses used in phylogenic analysis for the diagnosis of Deerpox virus infection in a white-tailed deer (Odocoileus virginianus) fawn.
The present study described a DPV infection in a white-tailed deer and possibly an outbreak of DPV in an exotic and domestic hoof stock enclosure. Deerpox virus infections have previously been reported in mule deer, reindeer, and black-tailed deer.10,11 Clinical presentations of DPV infections in these host species are usually keratoconjunctivitis and proliferative-ulcerative skin lesions on the face.10,11 Although bronchopneumonia and ulceration of the esophagus, with accompanying fibrinopurulent inflammation were observed in the black-tailed deer naturally infected with DPV, the observed pathological changes were not solely attributed to DPV infection because adenovirus infection was also detected in the affected animal. 10 Experimental infection of black-tailed deer with DPV isolated from mule deer induced multifocal cutaneous lesions without significant involvement of internal organs. 2 Compared to the pathological changes found in black-tailed deer and mule deer, the wide distribution of the cutaneous lesions, from head to abdomen, and the extensive involvement of digestive tract of the white-tailed deer fawn were characteristic. Although E. coli, Fusobacterium spp., and possibly P. multocida might have contributed to the development of ulcerative and necrotizing rumenitis followed by fibrinopurulent peritonitis, the pathological changes, such as parakeratosis and eosinophilic intracytoplasmic inclusion bodies, confirmed DPV as the primary etiology. The extensive involvement of the digestive tract could be a result of compromised host defense, as supported by the extremely poor physical condition of the affected animal. However, the only histological evidence was moderate lymphocyte depletion in splenic follicles. The thymus and lymph nodes were not examined histologically because no appreciable gross lesions were observed. It must be pointed out that 12 animals initially displayed similar clinical symptoms, such as keratoconjunctivitis and ulcerative cutaneous lesions. Thus, it is reasonable to speculate that all other 11 white-tailed deer were also infected with the same viral agent, namely DPV. However, no specimens from these animals were received for laboratory testing. It is also noteworthy that many animals of the closely related species, including red deer, fallow deer, axis deer, and African black buck antelope, were comingled with the affected white-tailed deer, but did not show any clinical symptoms. This phenomenon raises the possibility that the virus was only able to infect a limited range of hosts or induce clinical symptoms in certain species. Although the source of the infection was not determined, certain management practices, such as open access and comingling of species, may have been risk factors for exposure and transmission.
Diagnosis of poxvirus infections is usually achieved by observation of rather specific clinical signs, histopathological examination of suspected lesions, and identification of pathogens3,9 Because of the large size and unique morphology, presumptive diagnoses could be made by EM examination of negative stained samples of purified virus or ultra-thin section of tissue specimens. Molecular tests, such as the guanine–cytosine content-based pan-pox universal PCR and the A21-gene PCR have been developed for the screening and diagnosis of poxvirus infections.6,8 To identify a poxvirus to the genus level, PCR assays targeting specific genes or phylogenic analysis of conserved genes become the tests of choice. In the current study, both conserved and genus-specific genes were amplified and sequenced. Phylogenetic analysis of the A21 gene made it possible to place the viral isolate to the genus Cervidpoxvirus. Amplification of CTLR further proves that the new isolate belongs to the Deerpox virus species.
Footnotes
Acknowledgements
The authors would like to acknowledge Latoya Sly, Tina Hay, Jay Kay Thornton, Corey Williams, and Candy Zhang for their technical assistance.
a.
MEM medium, Invitrogen Corp., Carlsbad, CA.
b.
Fetal bovine serum, Sigma-Aldrich, St. Louis, MO.
c.
Prepared culture media, Remel Inc., Norcross, GA.
d.
AnaeroGen™, Remel Inc., Norcross, GA.
e.
Sensititre ARIS® 2X, TREK Diagnostic Systems Inc., Cleveland, OH.
f.
DNeasy® Blood & Tissue Kit, Qiagen Inc., Valencia, CA.
g.
Custom primers, Eurofins MWG Operon, Huntsville, AL.
h.
AlphaImager™ Gel Documentation, Cell Biosciences Inc., Santa Clara, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported in part by Mississippi Veterinary Research and Diagnostic Laboratory.
