Abstract
Clinical signs in 4 cases of salmonellosis in cats included vomiting, diarrhea (2 cases each), fever, dystocia, icterus, and seizures (1 case each). Three cats died, and one was euthanized. Grossly, all cats were in poor body condition and had yellow-to-dark-red perianal feces (3 cases), oral and ocular pallor (2 cases) or icterus (1 case), fluid or pasty yellow intestinal contents (4 cases), white or dark-red-to-black depressed areas on the hepatic surface (2 cases), yellow abdominal fluid with swollen abdominal lymph nodes (1 case), and fibrin strands on the placental chorionic surface (1 case). Histologically, all cats had necrotizing enterocolitis and random hepatocellular necrosis. Other histologic findings included mesenteric (4 cases) or splenic (2 cases) lymphoid necrosis, and endometrial and chorioallantoic necrosis (1 case). Gram-negative bacilli were observed within neutrophils and macrophages in the intestinal lamina propria (4 cases), liver, spleen, lymph node, endometrium, and placenta (1 case each). Aerobic bacterial culture on frozen samples of small intestine, mesenteric lymph node, lung, and liver yielded Salmonella enterica subsp. enterica. Serotyping was consistent with S. Enteritidis (cases 1, 3) and S. Typhimurium (cases 2, 4).
Keywords
Salmonella spp. (family Enterobacteriaceae) are gram-negative facultative anaerobic bacilli responsible for life-threatening zoonotic infections in a wide variety of animals.7,13 Genus Salmonella consists of 2 species: S. enterica and S. bongori. Clinical disease in domesticated animals and humans is most commonly associated with 7 subspecies (enterica, arizonae, diarizonae, indica, houtenae, salamae, and an as-yet-unnamed species designated as S. enterica subspecies VII) and ~2,600 serovars of S. enterica. 13 Infection typically involves fecal-oral transmission or inhalation of organisms in contaminated feed, water, and environment. 7 Infected individuals may develop clinical disease associated with enterocolitis, septicemia, and abortion or become subclinical carriers and act as a source of infection to other susceptible hosts. 13 Systemic disease caused by Salmonella spp. in humans, horses, cattle, and pigs is typically associated with the host-adapted serovars S. Typhi, S. Typhimurium, S. Dublin, and S. Choleraesuis, respectively. 13
In companion animals, clinical disease caused by Salmonella spp. infection is infrequently reported and typically associated with hospitalization, antibiotic therapy, immunosuppression, and consumption of raw-food diets.7,12 Salmonella spp. are highly prevalent in feces from clinically healthy or diarrheic dogs and cats, 10 particularly in stray or sheltered individuals or those fed raw-food diets. 4
The main source of infection for indoor cats is the ingestion of raw-meat diets or other types of contaminated food.4,12 Infection of outdoor cats typically occurs via exposure to contaminated environments during the scavenging of, or hunting for, rodents, birds, or reptiles.4,5,8,12 Clinical signs may range from self-limited diarrhea to severe and often fatal clinical disease characterized by anorexia, abdominal pain, fever, and diarrhea resulting from enterocolitis and septicemia.2,4,7,12 Extraintestinal disease, including conjunctivitis, pneumonia, and abortion, has been described infrequently.1,2,9
Given that salmonellosis is infrequently reported in cats and most descriptions focus on the epidemiologic or clinical aspects of the infection,1,4,5,10,12 the pathologic changes associated with clinical disease are not well documented in the veterinary literature.12,14 Here we describe the clinical and pathologic findings of 4 cases of systemic salmonellosis in cats autopsied at the Athens Veterinary Diagnostic Laboratory (AVDL; University of Georgia, Athens, GA, USA) from 2015 to 2022.
Case 1 was an 8-wk-old male Bengal cat with acute vomiting and diarrhea that was autopsied in August 2015. The patient had seizures before dying during a physical examination. Case 2 was a 1-y-old female Siberian cat with acute fever and dystocia that was autopsied in August 2018. Euthanasia was elected because of the suspicion of feline infectious peritonitis virus infection. Case 3 was a 3-wk-old male domestic shorthaired cat found dead in the cage and autopsied in September 2019. Case 4 was a 7-wk-old male domestic shorthaired cat (autopsied in October 2022) that was part of a group of 8 kittens and a feral queen that were captured by animal control and held in a shelter environment. In a period of 2 d, 5 kittens died after acute onset icterus, vomiting, and diarrhea. Two cats (cases 1, 4) were fed commercial canned food; the type of food was unknown for cases 2 and 3. Cases 1 and 4 were positive for feline leukemia virus (FeLV) and negative for feline immunodeficiency virus (FIV); FeLV and FIV status was not reported for cases 2 and 3. Cases 1–3 were from Georgia; case 4 was from South Carolina.
Grossly, all cats were in poor body condition, with scant subcutaneous and visceral adipose tissue stores. Autopsy findings consisted of fluid or pasty yellow intestinal contents (cases 1–4), yellow-to-dark-red perianal feces (cases 1, 3, 4), oral and ocular pallor (cases 1, 3), 1–15-mm diameter, irregular, white (case 2) or dark-red-to-black (case 4) areas of necrosis and/or hemorrhage on the capsular and cut surface of the liver (Figs. 1, 2), oral and ocular icterus (case 4), and yellow fluid in the abdomen with swollen mesenteric and ileocecal lymph nodes (case 2). The gravid uterus (case 2) had 3 full-term fetuses with yellow fibrin strands on the chorionic surfaces of the placenta.
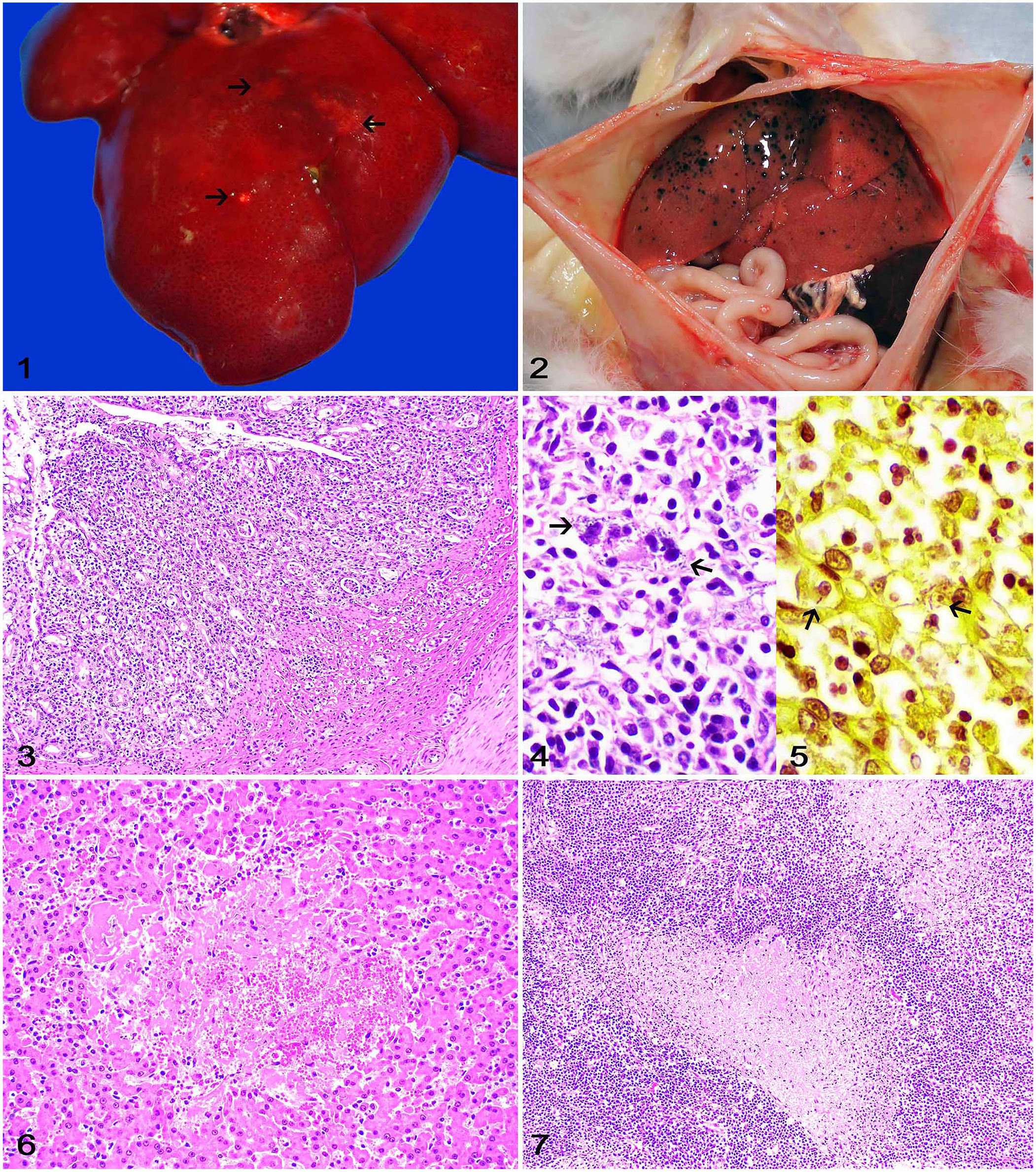
Systemic salmonellosis in cats.
Samples of multiple tissues were fixed in 10% neutral-buffered formalin, processed routinely for histology, and stained with H&E. Histologically, all cats had various degrees of enteritis (case 1) or colitis (cases 2–4) characterized by areas of mucosal necrosis admixed with a moderate number of neutrophils, macrophages, and fewer lymphocytes and plasma cells that occasionally extended into the submucosa or to the crypt lumina (Fig. 3). Intracellular gram-negative bacilli were observed within neutrophils and macrophages in the lamina propria in all cases (Figs. 4, 5). Other findings included well-demarcated areas of random hepatocellular necrosis (cases 1–4) that were replaced with fibrin, hemorrhage, and scattered neutrophils (Fig. 6), and extensive areas of lymphoid necrosis admixed with fibrin, macrophages, and scattered neutrophils that effaced most of the mesenteric lymph node (cases 1–4; Fig. 7) and/or spleen (cases 2, 4). Sections of liver, spleen, mesenteric lymph node, and intestine were subjected to modified Brown–Hopps Gram stain. Gram-negative bacilli were observed within necrotic areas in the liver (case 4), spleen, and lymph node (case 3). In addition, case 2 had extensive endometrial and chorioallantoic necrosis with fibrin and numerous neutrophils, macrophages, lymphocytes, plasma cells, and gram-negative bacilli.
Based on histologic findings and the suspicion of bacterial septicemia, frozen tissue samples were subjected to bacterial culture for diagnostic confirmation, at the discretion of each case pathologist. Aerobic bacterial culture on frozen samples of small intestine (case 1), mesenteric lymph node (case 2), lung (case 3), and liver (case 4) yielded moderate-to-heavy growth of S. enterica subsp. enterica in all cases. Samples were serotyped at the USDA National Veterinary Services Laboratories (Ames, IA, USA) as S. Enteritidis (cases 1, 3) and S. Typhimurium (cases 2, 4).
Approximately 1,600 cats were autopsied at the AVDL between August 2015 and October 2022, with only 4 confirmed cases of salmonellosis (a prevalence of 0.25%). The clinical signs in our cases were similar to those reported for feline salmonellosis elsewhere.4,5,12 However, given that these clinical signs are associated with enterocolitis and septicemia and are not specific for Salmonella spp. infection, a definitive clinical diagnosis requires bacterial culture or PCR testing performed on rectal swabs or fecal samples from affected patients.5,7 Serotyping in our cases revealed infection by S. Enteritidis and S. Typhimurium. Although S. Enteritidis is associated mainly with poultry infections, 11 S. Typhimurium has frequently been associated with feline salmonellosis.5,8
The pathologic changes associated with salmonellosis in cats have rarely been documented in detail.12,14 Reported gross changes include poor body condition, mucosal pallor, gray necrotic foci in the liver, and reddened intestines,12,14 mirroring many of our gross findings. Histologically, hepatocellular necrosis, neutrophilic and lymphoplasmacytic enteritis, and interstitial or suppurative pneumonia have been described, 12 many of which lesions were also observed in our cases. Similar to the clinical signs, most of these pathologic findings reflect bacterial septicemia. A final diagnosis of salmonellosis in our cases was achieved based on bacterial culture of frozen tissue samples.
Infection in cats occurs mainly through the fecal-oral route or ingestion of contaminated food or water.4,12 Feeding homemade or commercial raw diets to cats and dogs has become an increasingly popular practice.3,6 However, these diets have no scientifically proven benefit for pets and are based on the appeal to nature or antiquity, anti-corporate sentiment, and pseudoscience. 15 Moreover, because these products are not subjected to any heat processing or sterilization, they are not recommended by the U.S. Food and Drug Administration given the high risk of contamination with many types of pathogens, including Salmonella spp.3,6 Not surprisingly, raw-meat diets have been associated with cases of salmonellosis in cats.4,12
Other important sources of Salmonella spp. infection for cats include a contaminated environment or close contact with infected animals such as poultry, other wild or pet birds, rodents, and reptiles.4,5,8,12 The source of infection or the role of potential predisposing factors could not be determined in our cases; the specific type of canned food fed to 2 of our cases and the type of food available for the 2 other patients was not specified. However, S. Enteritidis is one of the most common serotypes associated with human and poultry infections, 11 which could suggest a potential human or avian source of infection in cases 2 and 3. Cases 1 and 4 were positive for FeLV, and the patients may have been suffering from immunosuppression, but that was not confirmed clinically. In addition, pregnancy (case 2) and a shelter environment (case 4) could have predisposed to infection through immunosuppression and close contact among individuals, respectively. 10
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
