Abstract
Listeriosis is a disease of humans and domestic mammals (mainly ruminants) with variable manifestations, primarily encephalitis, septicemia, and abortion. Although Listeria monocytogenes readily causes illness in ruminants, the prevalence among domestic South American camelids (llamas and alpacas) is low and has not been documented in their wild counterparts, the vicuna and guanaco. We describe herein the clinical signs, autopsy findings, and histopathology of septicemia and suppurative meningoencephalitis caused by L. monocytogenes in 2 neonatal llamas (Llama glama) from the same herd. L. monocytogenes was isolated in pure culture and identified by real-time PCR on fresh and paraffin-embedded tissue samples of the brain from both crias. This presentation of septicemic listeriosis with meningoencephalitis in 2 animals from the same group is unusual, especially among llamas.
Listeria monocytogenes is a gram-positive, facultatively anaerobic, rod-shaped bacterium and the causative agent of listeriosis in mammals (including humans) and birds.2,3,6,8 The organism grows at pH 5–9 and a temperature of 4–45°C. It is often associated with spoiled silage (pH >5.5), and can act as a saprophyte or pathogen.2,3,7 The disease in humans and ruminants typically occurs in 3 main forms: septicemia, encephalomyelitis and/or meningoencephalitis, and abortion.2,3,6–8 Less common forms of listeriosis include mastitis, conjunctivitis, otitis, and gastroenteritis.3,7 Transmission typically occurs via contaminated feed (e.g., silage), bacteria shed in feces, or transplacentally from the dam.3,7 Although listeriosis is a fairly common disease in ruminants, it has a low prevalence among South American camelids (SACs); meningoencephalomyelitis, abortion, and septicemia caused by L. monocytogenes have been documented in a few case reports in domestic llamas and alpacas.2,4–8,12,14 Herein we describe the clinical signs, and gross, histopathologic, and molecular findings, as well as the supporting bacterial culture results, that confirm listeriosis in 2 neonatal llamas (Llama glama) from the same herd. This presentation of septicemic listeriosis with meningoencephalitis in 2 animals from the same group is unusual, especially among llamas.
The animals (1 female and 1 male) were 2-wk-old and originated from the same herd. Over a 4-d period in May 2015, both animals exhibited extreme lethargy, anorexia, and pyrexia. The female cria also exhibited signs of abdominal pain with abnormal vocalizations. Both animals were treated with florfenicol and flunixin meglumine, yet died within 1 wk of the onset of clinical signs. The animals were submitted to the University of Georgia, Tifton Veterinary Diagnostic and Investigation Laboratory, for autopsy.
On gross examination, the brain of the male cria was markedly congested and discolored red to dark-red. The meningeal surfaces over the sulci were cloudy and mottled white to light beige (Fig. 1A). The umbilicus of the female cria was wet and exuded small amounts of caseous exudate. The thoracic cavity, abdominal cavity, and pericardial space contained small amounts of fibrin. The meninges over the brainstem were thickened and mottled yellow to white. Other gross findings in the female included hepatic congestion, pulmonary congestion, and serous atrophy of pericardial fat.
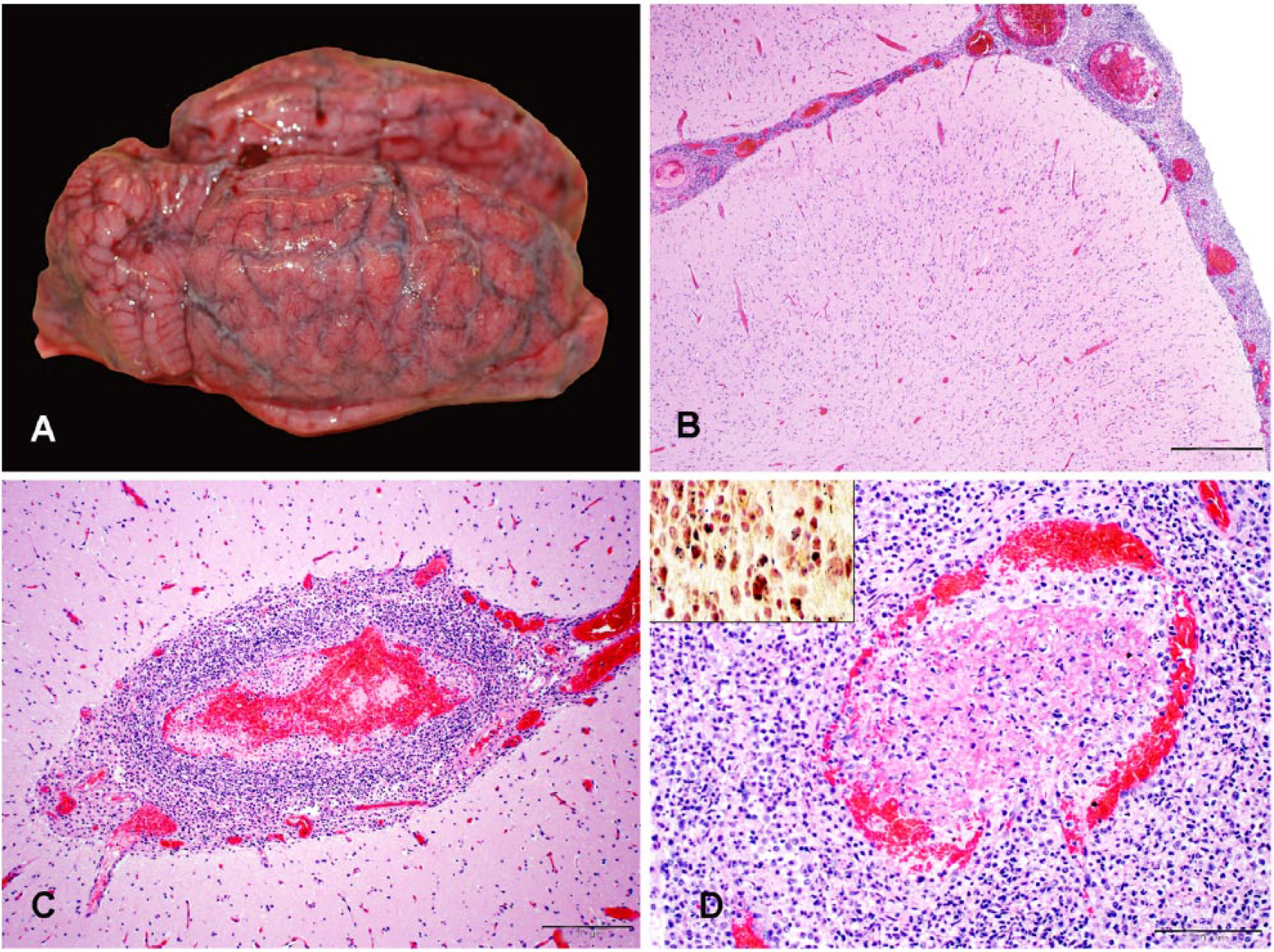
Meningoencephalitis in a neonatal llama.
Samples of brain, lung, spleen, liver, heart, kidney, tongue, thyroid gland, lymph node, small intestine, large intestine, third stomach compartment, trachea, skin, and urinary bladder from both animals were fixed in 10% buffered formalin solution. Following routine processing, 4-µm sections of paraffin-embedded tissue were stained with hematoxylin and eosin. Gram stains (Brown and Brenn) were performed on paraffin-embedded sections of brain and liver.
In both animals, the most prominent microscopic changes were in the brain and consisted of marked, multifocal-to-coalescing meningoencephalitis. Meningeal vessels were surrounded by dense aggregates of neutrophils with fewer macrophages (Fig. 1B). In the cortical gray matter, Virchow–Robin spaces were filled with similar perivascular cuffs of leukocytes. In vasculitic vessels, the tunica intima and media were effaced by fibrinoid necrosis with karyorrhectic debris and degenerate neutrophils present (Fig. 1C). Occasional vessels were partially to fully occluded by thrombi (Fig. 1D); numerous intralesional, gram-positive, short rod-shaped bacteria were present within neutrophils and macrophages in brain lesions (Fig. 1D, inset). Microabscesses in gray matter neuropil were identified predominantly in brainstem sections (Fig. 2). Perivascular cuffs and microabscesses contained moderate numbers of intracellular, short rod-shaped bacteria.
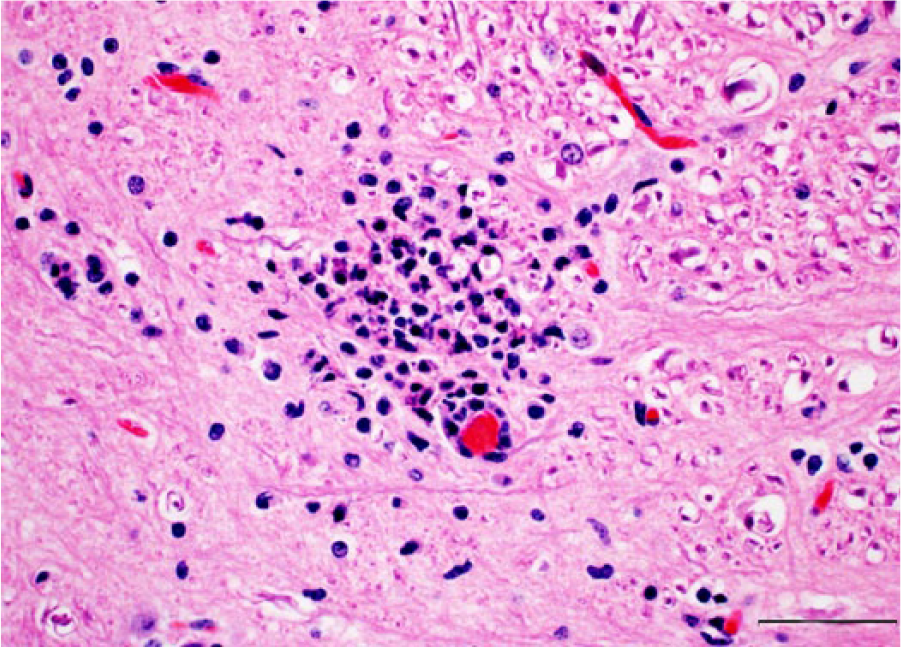
Microabscess in the brainstem. H&E. Bar = 50 µm.
The livers of both animals contained multiple, randomly distributed areas of acute hepatocellular necrosis admixed with low-to-moderate numbers of degenerate neutrophils. Occasionally, nearby sinusoids contained irregular thrombi that disrupted the hepatic cords. Low numbers of intralesional, gram-positive, short rod-shaped bacteria were present intracellularly in neutrophils and macrophages.
The renal pelvis and medulla of both animals contained multifocal, moderate to high density, predominantly perivascular, aggregates of degenerate and intact neutrophils admixed with macrophages. A few glomeruli were partially distorted by aggregates of degenerate neutrophils and karyorrhectic debris, and occasional glomerular capillaries were occluded by microthrombi.
The lungs of both animals were severely congested and edematous. Other histologic changes included marked neutrophilic and necrotizing omphalophlebitis in the male cria and mild splenic lymphoid depletion in the female cria.
Samples of lung, kidney, liver, spleen, brain, and meningeal, pericardial, thoracic, and peritoneal swabs were cultured on modified Oxford agar. Cultures were incubated at 35°C and, within 24 h, all samples yielded a few colonies to heavy growth of L. monocytogenes. In both animals, the liver, brain, and meningeal swabs yielded heavy growth of L. monocytogenes.
Real-time PCR (rtPCR) was performed on scrolls of paraffin-embedded sections of brain from both animals, along with fresh frozen samples of brain from the female cria. DNA was extracted using the QIAamp DNA mini kit (Qiagen, Valencia, CA) according to the manufacturer’s protocol, and rtPCR was performed using the MicroSEQ L. monocytogenes detection kit (Applied Biosystems, ThermoFisher Scientific, Grand Island, NY) according to the manufacturer’s protocol. Real-time PCR on all tissue samples was positive for Listeria monocytogenes with threshold cycle (Ct) values of 25.9 and 25.3 on paraffin-embedded tissue sections from the female and male cria, respectively. The Ct value on the fresh brain tissue from the female cria was 29.4.
Fresh samples of brain and lung were negative for bluetongue virus (BTV), equid herpesvirus 1, bovine viral diarrhea virus (BVDV), and bovine herpesvirus 1 by fluorescent antibody testing. Immunohistochemistry for BVDV was performed on 4-µm sections of paraffin-embedded skin samples from both llamas, and the sections were negative. Fresh frozen brain samples from both crias were negative for epizootic hemorrhagic disease virus (EHDV) by rtPCR, and no viruses were isolated at the University of Georgia, Southeastern Cooperative Wildlife Disease Study (Athens, GA).
The gross changes in both animals were indicative of suppurative meningoencephalitis that was confirmed by histologic examination of the brain. The pattern of severe neutrophilic encephalitis with brainstem involvement and microabscess formation was strongly suggestive of L. monocytogenes infection. This suspicion was confirmed by the presence of high numbers of intralesional, intracellular gram-positive, short rod-shaped bacteria within macrophages and neutrophils of the brain, heavy growth of L. monocytogenes cultured from the brain, and detection of L. monocytogenes DNA from the affected tissue. The histologic changes, culture results, and PCR results confirmed meningoencephalitis caused by L. monocytogenes.
In addition, the random necrotizing hepatitis with intralesional, gram-positive, bacterial rods and evidence of early embolic nephritis in both llamas was indicative of septicemia. Heavy growth of L. monocytogenes from liver confirmed septicemic listeriosis in these animals. Given the evidence of omphalophlebitis in both animals and fibrinous peritonitis in the female cria, the presumed entrance site for the bacteria was the umbilicus with subsequent hematogenous spread to liver, kidney, and brain. Although the encephalitic form typically results from spread along the cranial nerves subsequent to oral trauma, 3 with the young age of our animals, acute time frame, and evidence of embolic disease, it is more likely that the encephalitis developed hematogenously. Other potential sources of infection include fecal–oral transmission or from bacteria shed in the milk, including organisms shed from healthy carrier animals,2,3,13 as well as from contaminated bedding and water sources. 9
Listeriosis has been documented in SACs, but is considered an uncommon and not well-characterized disease in such species.7,14 The presentation of septicemia with meningoencephalitis as observed in the llamas in our report is unusual, even among more commonly affected domestic species (e.g., ruminants). A few case reports (mainly single case reports) have documented Listeria infection in the llama, with the 3 major disease forms represented (septicemia, abortion, encephalitis).2–8,12,13 L. monocytogenes septicemia and meningoencephalitis has been reported in a cria; however, this animal also had concurrent systemic Escherichia coli infection. 5 Previous reports of meningoencephalitis in llamas were observed in neonatal to adult animals. In many of the previous reports and in our case report, the neurologic condition was meningoencephalitis with extensive involvement of the meninges,5–7,13 which may be a more common feature of L. monocytogenes infections in SACs. The presence of microabscesses in the brainstem of both crias did raise concerns for a concurrent encephalitic form of listeriosis, which was carefully considered. However, the signalment of these animals, decreased likelihood of oral cavity trauma, lack of evidence of cranial nerve involvement, concomitant sepsis, and time frame of infection make the encephalitic form less likely for these cases. Furthermore, aside from lethargy, the 2 llamas did not exhibit the classic neurologic signs associated with the encephalitic form of listeriosis (i.e., head tilt, circling, ataxia, cranial nerve deficits).3,14 Based on our report and previous reports, the neurologic lesions of L. monocytogenes in llamas are predominantly observed as meningoencephalitis, potentially recognizable on gross examination.5,6,13
Two animals from the same herd succumbing to L. monocytogenes infection in such a short time frame raised concerns for a single, underlying predisposing factor. Indeed the mild lymphoid depletion of the spleen in the female would suggest some degree of immunosuppression; however, an underlying predisposing condition or cause was not identified. Outbreaks of listeriosis can occur among domestic ruminants, but these typically involve adult animals exhibiting signs of neurologic disease and are associated with the feeding of silage. 3 Given the time of year (i.e., late spring in south Georgia), lack of history of recent silage usage on the premises, and age of the animals, it is less likely that spoiled silage played a role in this particular case. L. monocytogenes is naturally present in the soil, therefore it is possible that certain environmental factors (e.g., changes in soil pH, soil moisture levels, stocking density3,7,9) may have led to an overgrowth of the bacteria in the animals’ environment.2,3,7 Alternatively, it is possible that these animals may have been exposed to the organism shed from the dam (e.g., feces or milk).2,3,7,13 The rapid disease progression, young age of the llamas, and prominent vasculitis observed, especially in the meninges, did raise concerns for an underlying orbiviral infection. Orbiviruses have been associated with mortality in SACs.1,10,11 However, in our case, both animals were negative for BTV and EHDV. Also, all other tests for viruses were negative, thereby making an underlying viral infection less likely in these animals.
Following the deaths of these animals, no other mortalities or animals developing similar neurologic disease were reported on the premises. The dams of both crias remained healthy and asymptomatic for listeriosis.
Information in the literature indicates that the prevalence of listeriosis among SACs is low.7,14 However, the reasons are not known and, to our knowledge, no studies focusing on the prevalence of L. monocytogenes in SACs have been reported. This apparent low prevalence in SACs compared to domestic ruminants may in part be the result of population differences across species. For instance, according to the National Cattlemen’s Beef Association, the total number of cattle in the United States as of 2016 was 92 million (https://goo.gl/wE7nSW) versus 76,086 llamas and 140,601 alpacas, according to census data from the U.S. Department of Agriculture in 2012 (https://goo.gl/ms5vMh). Another contributing factor could result from differences in management practices; among cattle, L. monocytogenes is more common in dairy herds and has historically been attributed to silage usage. Also, the stress associated with more intensive management systems could lead to increased shedding by animals. 9 Additionally, SACs may have a degree of natural resistance to L. monocytogenes, supported by the fact that listeriosis has not been reported in their wild counterparts: the vicuna (Vicugna vicugna) and the guanaco (Lama guanicoe). 7 Finally, listeriosis in SACs may be underreported, contributing to a perceived low prevalence in these species. L. monocytogenes should be included as a possible etiology for meningoencephalitis in neonatal SACs with gross changes suggestive of bacterial meningitis as observed in these 2 animals
Footnotes
Acknowledgements
We thank Dr. Leanna Wilder at the Macon County Veterinary Hospital for the case submission, history, and pertinent clinical information. We thank Dr. David E. Stallknecht and Clara Kienzle at the University of Georgia, Southeastern Cooperative Wildlife Disease Study for conducting Epizootic hemorrhagic disease virus testing. We also thank Dr. Murray E. Hines II for critically revising the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
