Abstract
Herein we describe a rare case of systemic Listeria monocytogenes infection with concurrent pleural mesothelioma in a stray cat that was found dead and submitted for autopsy. Gross pathology changes consisted of thoracic clear yellow fluid admixed with suspended fibrin strands; clear-to-tan, variably sized, <3 mm diameter pulmonary nodules; and enlargement of the submandibular, retropharyngeal, and prescapular lymph nodes. Histologic changes consisted of extensive areas of suppurative inflammation and necrosis with mineralization that partially effaced the pulmonary parenchyma and lymph nodes. Random, distinct necrotic foci were present throughout the hepatic parenchyma. Extending from the pleura, within perinecrotic alveolar spaces, and infiltrating the submandibular, retropharyngeal, and prescapular lymph nodes were dense sheets of neoplastic epithelioid cells with moderate pleomorphism and occasional karyomegaly and multinucleation. Neoplastic cells exhibited immunolabeling for pancytokeratin AE1/AE3 and vimentin, consistent with pleural mesothelioma. Aerobic bacterial culture of lung yielded heavy growth of L. monocytogenes. Immunohistochemistry (IHC) for L. monocytogenes revealed clusters of bacteria in the lung, lymph node, and liver. Pathologic changes were consistent with systemic listeriosis, confirmed by bacterial culture and IHC, and concurrent pleural mesothelioma.
Listeriosis is a zoonotic bacterial infection that affects a wide range of animals. Organisms are found worldwide and are ubiquitous in the environment, with most infections acquired from ingestion of contaminated soil and foodstuffs, as well as feces, urine, and milk of normal or diseased animals. 20 Although Listeria monocytogenes is the main cause of listeriosis in domestic animals, L. ivanovii can also cause disease, primarily in ruminants.16,20 In addition to domestic animals, there have been multiple reports of listeriosis in wildlife and domestic species, including dogs,1,3,15 camelids,8,10 and horses.13,19 L. monocytogenes is the third most common cause of death from food poisoning in humans, and most commonly affects pregnant women, newborns, the elderly, and immunocompromised individuals. 16
Listeriosis causes 3 distinct clinical manifestations, with neurologic, septicemic, or reproductive problems.6,7,20 Neurologic disease, seen most commonly in cattle, sheep, and goats, is characterized by encephalitis or meningoencephalitis.6,7,20 Septicemia occurs mainly in aborted or newborn ruminants, foals, piglets, rabbits, and birds.6,7,20 Reproductive disease includes metritis, placentitis, or fetal infection with subsequent abortion or stillbirth.6,7
Listeriosis in cats is exceedingly rare, and cases are mainly associated with systemic infection, 5 cutaneous wounds, 11 encephalomyelitis, 18 and lymphadenitis. 9 Herein we describe a case of systemic L. monocytogenes infection in a cat with concurrent pleural mesothelioma.
A stray young adult spayed female domestic medium-hair cat was found dead with no previous clinical signs and submitted for autopsy. The carcass was in poor body condition with scarce subcutaneous and visceral adipose tissue stores. The main gross pathology findings were restricted to the lungs and submandibular, retropharyngeal, and prescapular lymph nodes. The thoracic cavity contained 33 mL of clear yellow fluid admixed with suspended fibrin strands. The pulmonary and costal pleura, as well as the pericardium, were covered with fine strands of fibrin (Fig. 1). The pericardium was white, opaque, and firmly, circumferentially adhered to the thoracic body walls. The cranioventral pulmonary pleura was adhered to the left thoracic wall by fibrin. The lungs were red and collapsed and had numerous pale white-to-tan, ~3 cm diameter, well-demarcated parenchymal nodules scattered randomly throughout all lobes (Fig. 2). The submandibular, retropharyngeal, and prescapular lymph nodes were markedly swollen and had a pale-yellow and firm cut surface intercalated with extensive accumulations of yellow suppurative exudate. No other significant changes were present.
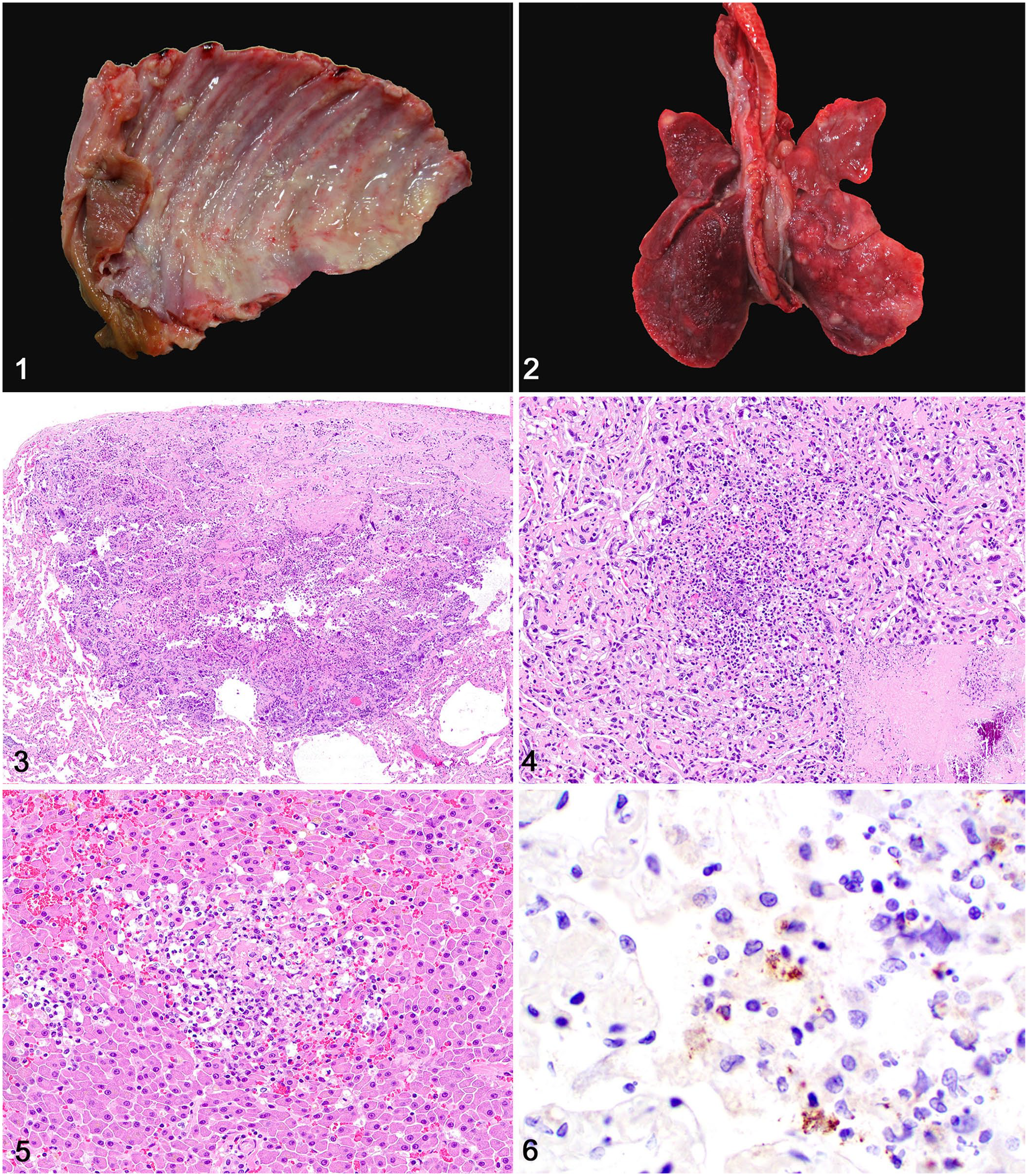
Systemic listeriosis in a cat.
Samples of multiple tissues were fixed in 10% neutral-buffered formalin, processed routinely for histology, and stained with hematoxylin and eosin. Fresh samples of lungs were submitted for a fluorescent antibody test (FAT) for feline coronavirus and for aerobic bacterial culture. Histologic examination of lungs and lymph nodes revealed extensive areas of necrosis admixed with a neoplastic cell population. In the lungs, extensive areas of necrosis partially effaced the parenchyma (Fig. 3). These necrotic foci consisted of dense infiltrates of degenerate neutrophils admixed with eosinophilic amorphous material, pyknotic and karyorrhectic debris, and mineralization (Fig. 4). Subpleural spaces were expanded with fibrin, hemorrhage, and cell debris admixed with neutrophils and macrophages. Necrotic areas intercalated with neoplastic cells similar to those in the lungs effaced the cortical and medullary tissue architecture of affected lymph nodes (Suppl. Fig. 1). The pericardium was expanded with fibrin, edema fluid, and hemorrhage admixed with a small number of neutrophils and macrophages.
The pulmonary nodules noted grossly corresponded histologically to areas of necrosis, as described above, and additionally to a neoplastic cell population. Extending from the pleura and into the alveolar spaces surrounding necrotic areas (Suppl. Fig. 2) were clusters of epithelioid neoplastic cells with a moderate amount of eosinophilic, homogeneous cytoplasm with distinct cell borders (Suppl. Fig. 3). Nuclei were round-to-oval and had finely to coarsely stippled chromatin with 1–3 prominent nucleoli. Anisocytosis and anisokaryosis were marked, with occasional karyomegaly and multinucleate cells. There were 5 mitoses per 2.37 mm2 (equivalent to 10 FN22/400× fields). Random, distinct necrotic foci with a small number of neutrophils and macrophages were present throughout the hepatic parenchyma (Fig. 5).
Aerobic bacterial culture of the lung yielded heavy growth of L. monocytogenes. FAT for feline coronavirus was negative. Immunohistochemistry (IHC) for L. monocytogenes (rabbit polyclonal antibody, 1 in 100 dilution for 45 min; United States Biological) performed on tissue sections of lung, lymph node, and liver revealed clusters of bacteria throughout the necrotic areas (Fig. 6). Additional IHC was performed on sections of lung: CD3 (rabbit polyclonal antibody, 1 in 1,000 dilution for 60 min; Dako), CD79a (mouse monoclonal antibody, 1 in 50 dilution for 60 min; Biocare Medical), multiple myeloma oncogene 1/interferon regulatory factor 4 (MUM1; rabbit monoclonal antibody, 1 in 50 dilution for 60 min; Biocare Medical), ionized calcium-binding adapter molecule 1 (Iba1; rabbit polyclonal antibody, 1 in 8,000 dilution for 60 min; Wako), pancytokeratin (AE1/AE3, mouse monoclonal, 1 in 100 dilution for 90 min; Biocare Medical), e-cadherin (mouse monoclonal, 1 in 500 dilution for 60 min; BD Biosciences), thyroid transcription factor-1 (TTF-1; mouse monoclonal antibody, ready-to-use dilution for 60 min; Cell Marque), and vimentin (mouse monoclonal, 1:3,000 dilution for 60 min, BioGenex). CD3, CD79a, and MUM-1 IHC was performed additionally on lymph node. Neoplastic cells exhibited robust immunolabeling for pancytokeratin AE1/AE3 (Suppl. Fig. 4) and vimentin (Suppl. Fig. 5). All other IHC results were negative.
A diagnosis of systemic L. monocytogenes infection was made based on the pathologic changes and aerobic bacterial culture with further confirmation by IHC. The rare reported cases of listerial infection in cats have been associated mainly with systemic infection, 5 a cutaneous wound caused by a tree weta (Hemideina crassidens) bite, 11 encephalomyelitis, 18 and lymphadenitis. 9 The low occurrence of listerial infection in cats is corroborated by data from the Athens Veterinary Diagnostic Laboratory (Athens, GA). From 2000 to 2020, only 2 cats with confirmed listerial infection were submitted to our diagnostic service. One autopsy case was diagnosed with suppurative rhombencephalitis and meningitis, and diagnostic confirmation was made by FAT and bacterial culture. In the second case, a diagnosis of necrotizing cervical lymphadenitis confirmed by bacterial culture was made after histologic examination of autopsy samples submitted by a veterinarian.
Listeriosis is a food-borne disease and thus the gastrointestinal tract is considered the major site of entry for Listeria spp. 20 The main sources of infection are the environment, food and water, and other infected individuals.3,12,20 The increasing popularity of commercial pet food products containing raw livestock or wildlife meat in recent years has increased the risk of food-borne bacterial infections in dogs and cats, including listeriosis.3,9,12 Studies have shown L. monocytogenes in up to 16% of tested commercial raw pet foods, 17 corroborating the potentially harmful effects associated with this pseudoscientific approach to pet nutrition. 3 Given that the animal in our case was a stray cat, the source of infection remains unknown.
Abortion in adult ruminants and septicemia in many newborn animals occur after Listeria spp. gain access to the bloodstream through the gastrointestinal tract. 20 In contrast, neurologic disease in adult ruminants typically follows centripetal axonal migration of organisms to the central nervous system via the trigeminal nerve and other cranial or spinal nerves.6,7 A similar mechanism has been proposed for a case of neurologic listeriosis in a cat. 18 Our case shares morphologic features that are consistent with septicemic listeriosis, with multiple areas of necrosis in the lungs, cervical and thoracic lymph nodes, and liver.5,9,20 Interestingly, the more severe and extensive lesion in our case was necrotizing pneumonia. The random distribution of pulmonary necrosis suggests that the primary site of bacterial entry was hematogenous, with the gastrointestinal tract serving as the possible portal of entry. Given the lack of airway-associated infection and necrosis, it is less likely that the primary site of bacterial entry was the lung, as is reported rarely in immunosuppressed humans. 14 Human immunodeficiency virus infection is a major risk factor for the development of listeriosis in human patients. 20 Given that listerial infection in cats is exceedingly rare, a possible role of immunosuppression in the development of clinical disease has not been established in feline species, 9 to our knowledge. However, immune deregulation with abnormal levels of interleukin-10 has been shown to be associated with increased risk of clinical listeriosis in cats infected with feline immunodeficiency virus (FIV). 4 Although the pleural mesothelioma with metastasis to the submandibular, retropharyngeal, and prescapular lymph nodes in our case could have caused immunosuppression, the lack of a complete blood examination and knowledge of FIV status did not allow for the evaluation of the immunocompetence in our case.
Neoplastic cells in our case were morphologically consistent with an epithelioid mesothelioma, an uncommon malignant neoplasm that arises from mesothelial cells. Mesotheliomas are rarely reported in cats, in which tumors affect mainly the pleura and peritoneum. 2 Diagnostic confirmation can be achieved based on anatomic location, morphology, and immunolabeling for pancytokeratin AE1/AE3 and vimentin. 2 Differential diagnoses in our case included a poorly differentiated round cell tumor and a pulmonary carcinoma, which were ruled out by the negative IHC for CD3, CD79a, and Iba1, as well as for TTF-1, respectively.
Supplemental Material
Supplemental_material – Supplemental material for Systemic Listeria monocytogenes infection and concurrent pleural mesothelioma in a cat
Supplemental material, Supplemental_material for Systemic Listeria monocytogenes infection and concurrent pleural mesothelioma in a cat by Jessica A. Elbert and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Ashley Alden for her technical assistance with bacterial culture.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
