Abstract
Our study describes a case of systemic Trichosporon loubieri infection in a cat with acute dyspnea, anorexia, and aggressiveness. Physical examination revealed multiple ulcerative cutaneous lesions on the abdomen, neck, and thorax. Thoracic radiographs and ultrasound showed multiple mediastinal nodules and marked pleural effusion, respectively. A cutaneous biopsy from the ulcerated wounds revealed necrogranulomatous dermatitis and panniculitis with numerous intralesional fungal hyphae. Fungal culture on fresh swab samples from the cutaneous lesions yielded growth of a fungal organism that was further identified as Trichosporon loubieri by PCR and DNA sequencing. The cat was subsequently euthanized and submitted to autopsy. Gross pathology changes consisted of multifocal to coalescing white nodules ranging from 5 to 10 mm in diameter that expanded the mediastinal fat, intrathoracic lymph nodes, lungs, and costal pleura. These lesions consisted of areas of necrogranulomatous inflammation with numerous intralesional fungal hyphae morphologically similar to those observed in the cutaneous biopsy sample. Gross and histologic changes were consistent with a systemic fungal infection, and the etiologic diagnosis was supported by fungal culture. Fungal identity was confirmed by DNA sequencing of D1-D2 and TS1 regions.
Trichosporon is a genus of yeast-like anamorphic fungi with distinct morphological features of true mycelium that forms arthroconidia. 15 These organisms are not considered primary pathogens and are widely distributed in the environment, normally inhabiting the cutaneous, oral, gastrointestinal, respiratory, and reproductive tract of humans and other animals.7,19 To date, there are 50 reported species of Trichosporon classified within different clades; only 15 species are of potential medical interest. 7 Infection and clinical disease in humans is typically superficial and mild in immunocompetent individuals, but systemic and fatal in immunocompromised patients. 7 In addition, allergic pneumonia has been attributed to inhalation of Trichosporon arthroconidia from the environment during the summer in Japan. 2 In veterinary medicine, multiple species of Trichosporon have been associated with clinical disease, including dermatitis, rhinitis, and cystitis in cats,9,12,17 meningoencephalitis in dogs, 5 mastitis in cattle,8,11 and dermatitis in horses and nonhuman primates. 14
Our study describes a case of systemic T. loubieri infection in an 8-year-old Domestic Longhair cat that was presented to a private clinic because of acute dyspnea, anorexia, and aggressiveness. No history of Feline immunodeficiency virus and Feline leukemia virus infection was available, but the cat had never been vaccinated. Thoracic radiographs and ultrasound revealed a possible diaphragmatic hernia and diffuse pleural effusion, respectively. The cat was placed under general anesthesia and subjected to a brief exploratory laparotomy. No diaphragmatic hernia was detected, but 2 ulcerative lesions were noted in the abdominal skin during surgery. Thoracic fluid was removed by thoracocentesis, and additional radiographs revealed multiple thoracic nodules, suggesting a possible lymphoma or granulomatous disease. The cat was discharged after recovery from anesthesia and prescribed cefovecin, buprenorphine, and furosemide, but re-presented a week later with severe dyspnea. Cytological examination of impression smears obtained from the thoracic effusion after a new thoracocentesis revealed scattered neutrophils and macrophages. Multiple other ulcerative lesions were also observed over the left aspect of the neck, right and left thorax, and abdomen (Fig. 1). Dyspnea persisted for an additional week, and other cutaneous lesions were found. Multiple punch biopsies and a swab sample were taken from the cutaneous lesions for histologic examination and culture, respectively. Biopsy samples were received immersed in 10% neutral buffered formalin, and were routinely processed for histology and stained with hematoxylin and eosin (HE). Histologic changes consisted of necrogranulomatous dermatitis and panniculitis with numerous intralesional fungal hyphae (Fig. 2). The dermis and subcutaneous tissue were multifocally effaced by extensive areas of necrosis containing fibrin and cell debris or clusters of foamy and epithelioid macrophages admixed with neutrophils and numerous free and intrahistiocytic organisms morphologically consistent with fungal hyphae. The inflammation occasionally extended into the underlying skeletal muscle. Adjacent periadnexal areas had mild lymphoplasmacytic inflammation, and the overlying epidermis had areas of ulceration and suppurative inflammation or hyperplasia with orthokeratotic hyperkeratosis. Replicate sections stained with Grocott methenamine silver special stain (GMS; Fig. 3) and periodic acid–Schiff (PAS; Fig. 4) reaction revealed myriads of 4–6 um wide, septate fungal hyphae with non-parallel walls, nondichotomous branching, and terminal bulbous dilations. Swab culture yielded heavy growth of a nonpigmented fungus with arthroconidia on all media, but identification based on morphology was not possible. PCR and DNA sequencing of the D1-D2 and TS1 regions revealed a 100% similarity of the organism to Trichosporon loubieri (GenBank accession JN939448.1).
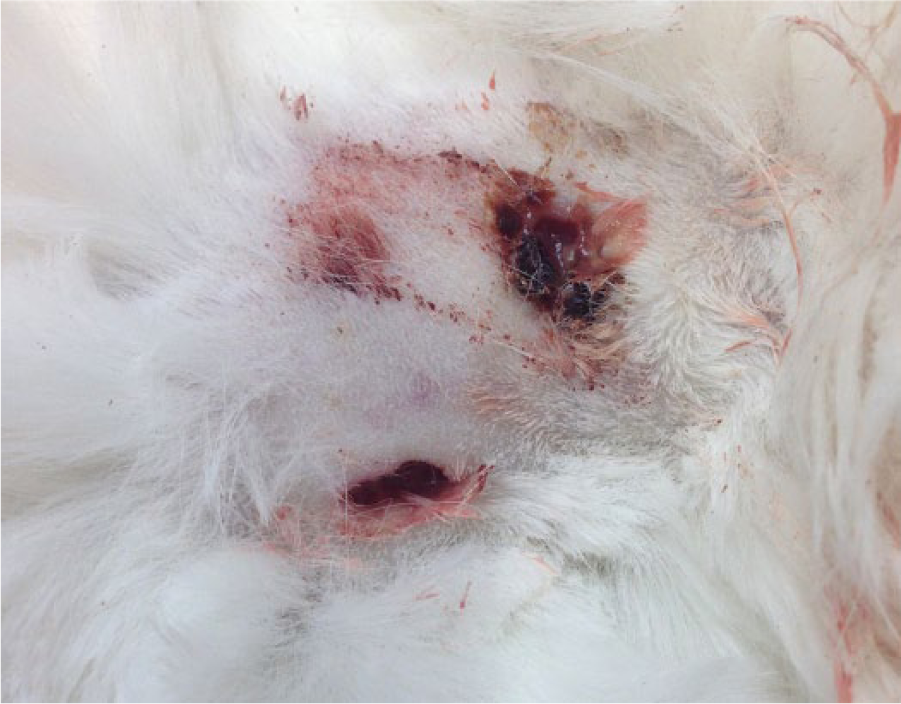
Three ulcerative lesions with hemorrhage in the ventral abdomen.
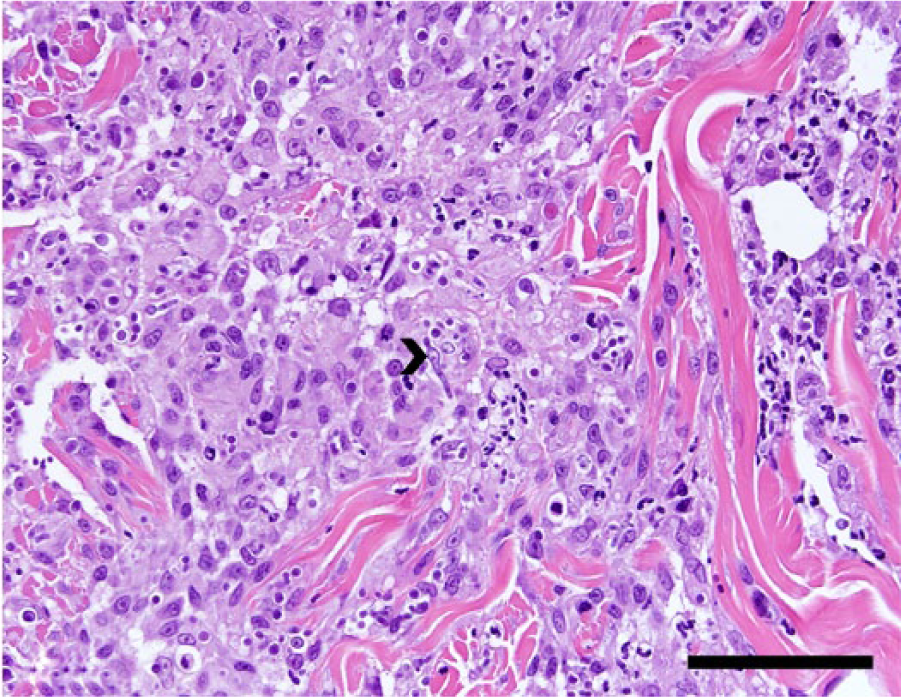
Punch biopsy from the cutaneous lesions reveals dense pyogranulomatous inflammation in the dermis with intralesional fungal hyphae (arrowhead). Hematoxylin and eosin. Bar = 50 µm.
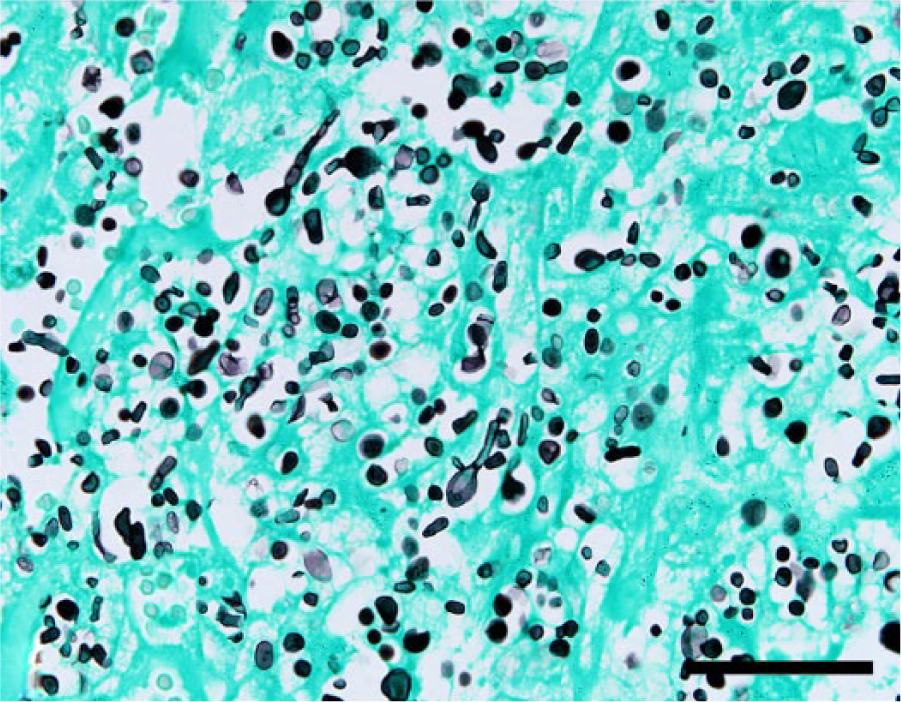
Fungal organisms are highlighted by Grocott methenamine silver special stain. Hyphae are 4–6 um wide; septate fungal hyphae with nonparallel walls, nondichotomous branching, and terminal bulbous dilations. Grocott methenamine silver stain. Bar = 50 µm.
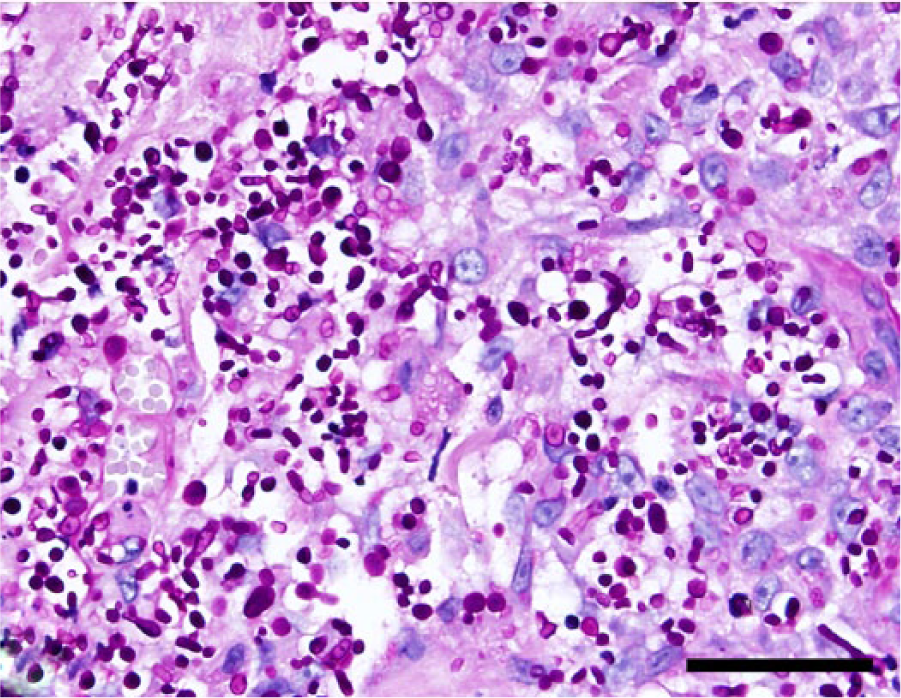
Same section shown in Figure 3 stained with periodic acid–Schiff reaction. Bar = 50 µm.
Because of the likelihood of a systemic fungal infection and poor prognosis, the cat was subjected to euthanasia and autopsy. Gross pathology changes consisted of multifocal to coalescing white nodules ranging from 5 to 10 mm in diameter that expanded the mediastinal fat, intrathoracic lymph nodes, lungs, and costal pleura (Fig. 5). No other gross anatomic changes were observed. Fragments of multiple tissues were fixed in 10% neutral buffered formalin, routinely processed for histology, and stained with HE. Histologic examination of multiple samples revealed disseminated, fairly nodular areas of necrogranulomatous inflammation with numerous intralesional fungal hyphae throughout the costal and visceral pleura, lungs, mediastinal fat, and thoracic lymph nodes (Fig. 6).
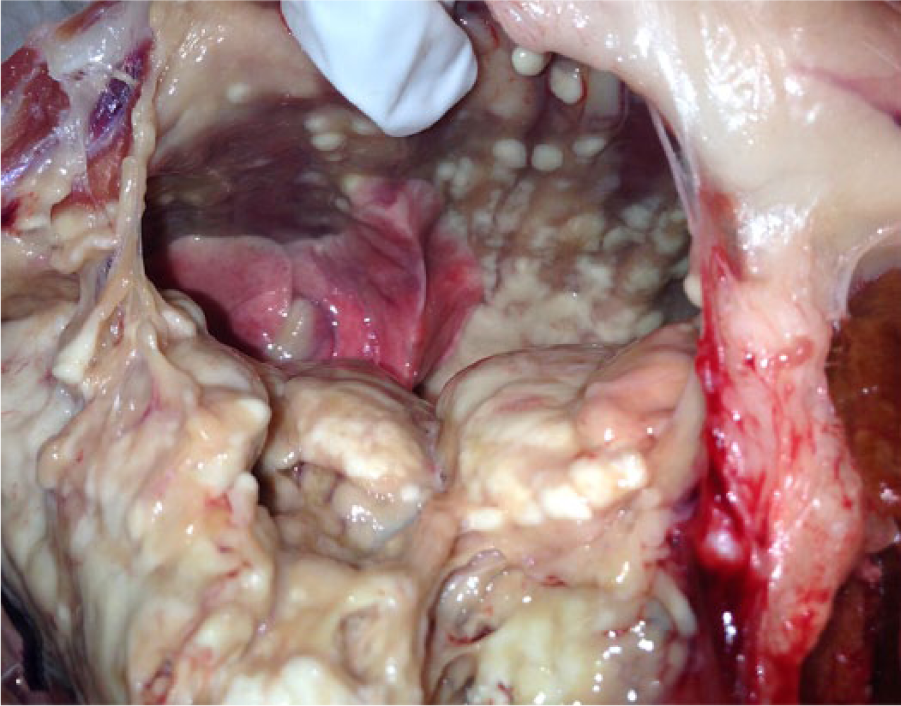
Multifocal to coalescing white nodules expand the mediastinal fat, intrathoracic lymph nodes, lungs, and costal pleura.
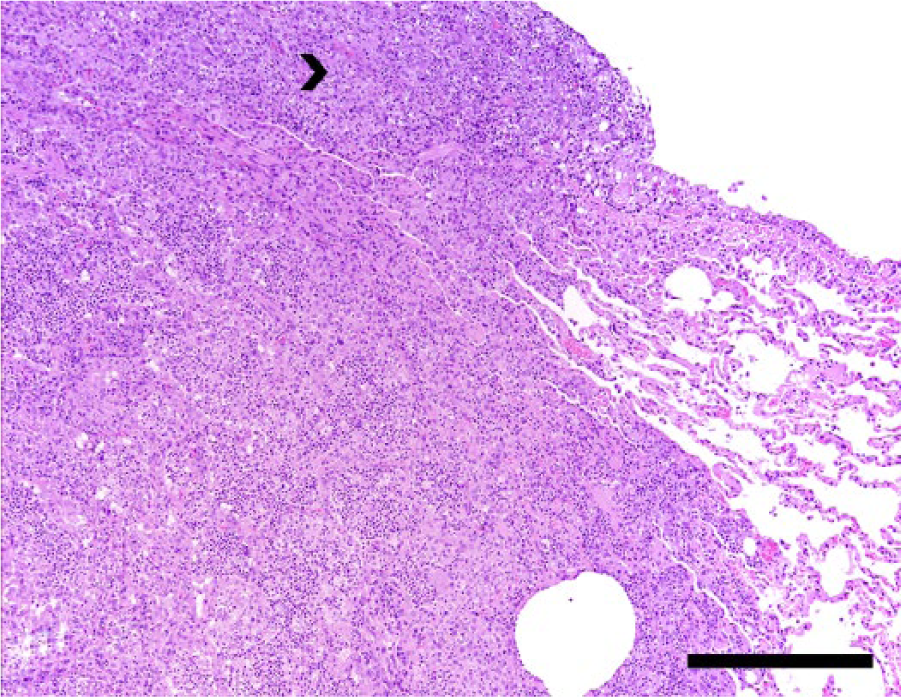
A pulmonary granuloma expanding and compressing the parenchyma (right) and forming pleural nodules (arrowhead). Hematoxylin and eosin. Bar = 500 µm.
Our report documents a case of systemic infection by T. loubieri in a cat, a fungal organism that has been associated with rhinitis but no systemic infection in this species to date. 18 Infection was characterized by ulcerative and pyogranulomatous dermatitis and panniculitis, pleuritis, pneumonia, and thoracic lymphadenitis. Intralesional GMS- and PAS-positive fungal organisms were abundant in all examined tissues and were morphologically consistent with what has been described for this fungal organism.5,9,12 The etiologic diagnosis was supported by fungal culture on swab samples from the cutaneous lesions, and fungal identity was achieved by PCR with subsequent DNA sequence analysis. These tests are typically required for a definitive diagnosis because identification of these fungal organisms by conventional methods, such as biochemical profile and morphology, is often difficult and frequently inconclusive.3,6,18
Systemic fungal infections in cats are typically divided into those caused by opportunistic agents, such as Cryptococcus neoformans, and those where the organisms are able to infect an immunocompetent host. The latter usually takes place via inhalation of conidia and include Blastomyces dermatitidis, Coccidioides immitis, and Histoplasma capsulatum infection. 13 Trichosporon is an ubiquitous organism and is not considered a primary pathogen. 6 Organisms are widely distributed in nature, and although most species are associated with superficial cutaneous infection in immunocompetent humans, opportunistic infections causing systemic disease may affect immunocompromised and/or hospitalized patients worldwide. 7 Although superficial infection (referred to as white piedra) is mild and restricted to the hair shafts in the scalp, face, axilla, and genitalia, 7 systemic disease is frequently fatal and is characterized by arthritis, endocarditis, peritonitis, esophagitis, meningitis, cerebral, splenic and hepatic abscesses, and urinary tract infection. 7 Infection by Trichosporon spp. has been rarely documented in the veterinary medical literature. Most cases have been reported in cats and consist of nasal granulomas associated with T. pullulans and T. loubieri infection,12,18 suppurative and hemorrhagic cystitis as a result of T. beigelii and T. domesticum infection,9,17 and ulcerative dermatitis caused by infection by an unclassified Trichosporon sp. 9 Although systemic infection in cats may also be associated with chronic debilitating disease, such as lymphoma or diabetes mellitus, no clinical or pathological evidence of any other underlying disease was detected in the cat of the current report. 9 Cutaneous lesions in cats infected with B. dermatitidis, C. immitis, and H. capsulatum typically reflect an underlying systemic infection that occurs primarily by inhalation of the organisms. 13 Therefore, based on the pathological changes in the present case, it is plausible that the respiratory infection may have preceded the cutaneous lesions. Other animal species have also been reported as susceptible to infection by Trichosporon. T. montevideense has been associated with granulomatous meningoencephalitis in dogs, 5 and T. cutaneum has been implicated as the cause of otitis externa or dermatitis and nasal granulomas in dogs.4,10 In addition, T. beigelii has been associated with outbreaks of mastitis in dairy cattle,8,11 and T. montevideense was isolated from a case of onychomycosis in a Japanese monkey. 1 Although the zoonotic potential of Trichosporon has not been confirmed, transmission from affected animals to humans should be considered in case infection is suspected. Treatment appears to be ineffective in the majority of Trichosporon infections in humans and other animals, mainly because these organisms show species-related resistance to a variety of antifungal drugs. 3 Therefore, early diagnosis of fungal infection and speciation of clinical samples is recommended to distinguish highly pathogenic from less pathogenic strains and to promptly determine a susceptibility drug profile.3,16,18
Footnotes
Acknowledgements
We thank Amy S. McKinney, Ashely E. Phillips, and Ingrid Fernandez for their technical assistance with fungal culture and PCR.
Authors’ contributions
DR Rissi contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. KD Kirby contributed to design of the study, and contributed to acquisition of data. S Sanchez contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
