Abstract
Equine ingesta-associated choledocholithiasis is a rare cause of morbidity and mortality. We describe here the clinical, gross, histologic, and microbiologic features of this condition in 2 horses and compare the features to 2 previous cases. Case 1 was a 4-y-old Thoroughbred mare with colic. Case 2 was an 18-y-old American Paint Horse mare with colic, chronic weight loss, and inappropriate mentation. Both had elevated biochemical markers of hepatocellular injury and cholestasis and were euthanized given a poor prognosis. Case 1 had a well-formed 5-cm choledocholith surrounding a piece of hay, and had chronic neutrophilic cholangiohepatitis, bridging fibrosis, and extrahepatic obstruction. Case 2 had an ill-formed choledocholith with occasional hay fragments, wood stick, and twigs, and had regionally extensive hepatocellular necrosis with mild neutrophilic cholangiohepatitis and bridging fibrosis.
Formation of biliary tract stones (cholelithiasis), including gallbladder stones (choleliths) and common bile duct stones (choledocholiths), are relatively uncommon phenomena in animals.16,17,20,29 Specifically, horses, which lack a gallbladder, are reported to develop choledocholiths with an estimated prevalence of 0.08%.
21
Equine choledocholithiasis can be clinically silent and only detected at autopsy; alternatively, choledocholiths can lead to cholangitis, extrahepatic biliary obstruction, hepatic fibrosis, and cholangiohepatitis, ultimately resulting in significant hepatobiliary injury and clinical signs and biochemical indices of hepatobiliary dysfunction.15,19,28 The pathogenesis of equine choledocholithiasis is unknown, but bacteria, mostly enteric species such as
Case 1 was a 4-y-old Thoroughbred mare that was presented to the North Carolina State University College of Veterinary Medicine (NCSU-CVM; Raleigh, NC, USA) with a 3-d history of intermittent and progressive colic. Rectal palpation by the referring veterinarian (rDVM) identified a suspected large colon displacement. Clinical signs initially resolved with administration of flunixin meglumine, but recurred 2 d later. Pain was refractory to medical intervention, peripheral blood lactate was 5.7 mmol/L (RI: < 2 mmol/L), and referral to the NCSU-CVM was elected. Upon arrival to NCSU-CVM, physical examination revealed mildly decreased borborygmi in the left quadrants and right dorsal displacement of the large colon on abdominal palpation per rectum, which resolved on repeat examination 8 h later. Thoracic and abdominal ultrasound were unremarkable. A CBC revealed moderate neutrophilic leukocytosis and hypofibrinogenemia (Table 1). An initial limited after-hours serum biochemistry panel at NCSU-CVM revealed markedly increased activity in aspartate aminotransferase (AST; 11.4 µkat/L, RI: 2.9–5.7 µkat/L) and gamma-glutamyl transferase (GGT; 8.58 µkat/L, RI: 0.08–0.40 µkat/L), and moderate hyperbilirubinemia (103 µmol/L; RI: 9–39 µmol/L). A repeat serum biochemistry panel specifically evaluating hepatic enzymes and biomarkers was obtained 8 h after initial presentation to the NCSU-CVM. This panel confirmed the initial aberrations in AST, GGT, and bilirubin, and further identified markedly elevated bile acids and moderately increased activity of succinate dehydrogenase (SDH; Table 1). Serum ammonia and lactate were within RIs. Given these findings, severe hepatopathy was suspected. Clinical signs persisted despite medical management. The mare was ultimately euthanized and submitted for postmortem examination.
Relevant CBC and serum biochemistry findings for horses with ingesta-associated choledocholithiasis. Values (RIs) are those seen on presentation at the referral hospitals.
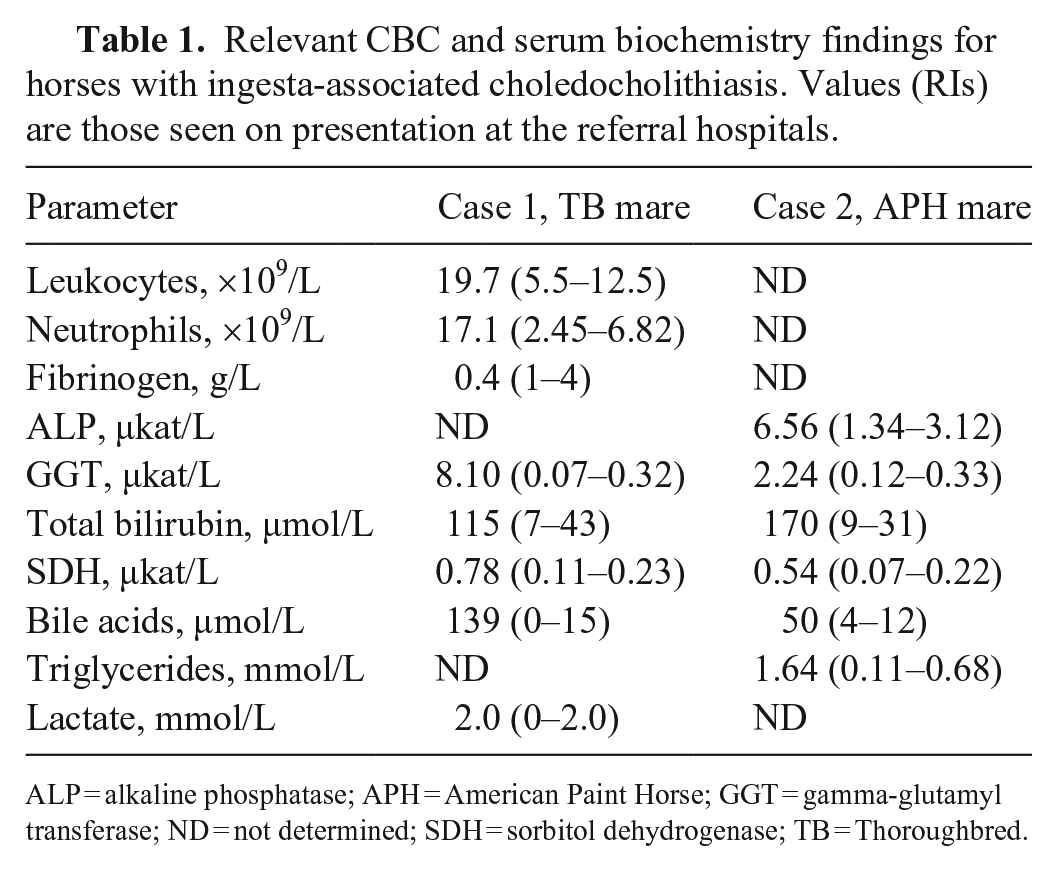
ALP = alkaline phosphatase; APH = American Paint Horse; GGT = gamma-glutamyl transferase; ND = not determined; SDH = sorbitol dehydrogenase; TB = Thoroughbred.
On gross postmortem examination, the liver was firm, resistant to crushing on palpation, and mildly enlarged. At the hilus of the liver, filling and emerging from the common bile duct and extending into the intrahepatic biliary tree, was a firm-to-hard, orange-brown, 3.5 × 3.5 × 5.5-cm ovoid choledocholith with a granular surface (Fig. 1). A few (< 10) similar, 2–5-mm, more friable choleliths were present in the adjacent intrahepatic biliary tree. Regional hepatic lymph nodes were moderately enlarged, measuring up to 4.5 × 2.5 × 0.9 cm (Fig. 1). On cut surface of the liver, bile ducts were prominent with fibrous tissue surrounding portal areas; these changes were most pronounced in parenchyma at the hilus adjacent to the choledocholith. The duct mucosa associated with the choledocholith was red-to-purple and eroded (Fig. 1).
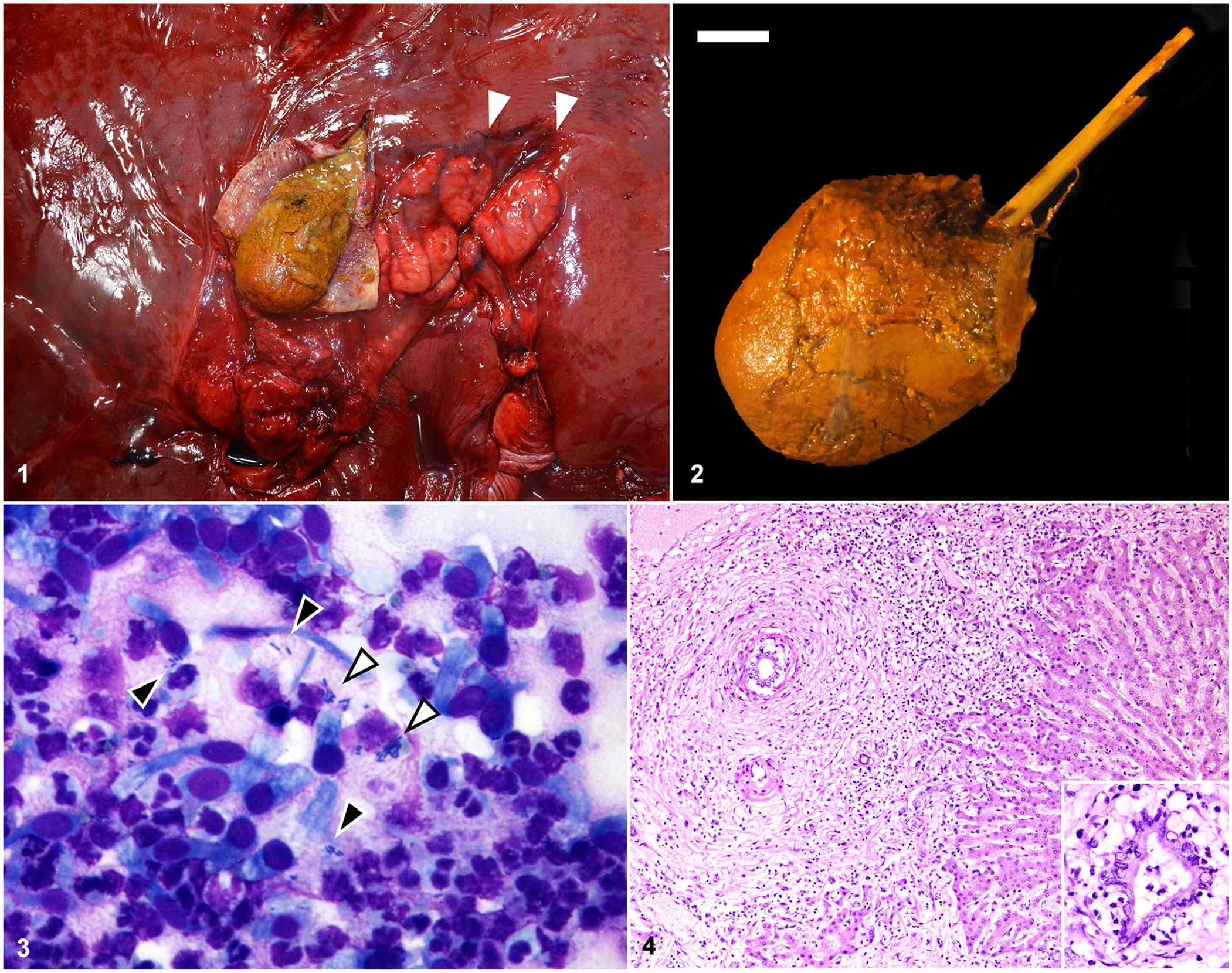
Gross, cytologic, and histologic features of ingesta-associated choledocholithiasis in case 1, a Thoroughbred mare.
An ~7 × 0.3 × 0.3-cm linear piece of plant material (hay) was identified at the core of the choledocholith (Fig. 2). The choledocholith was surrounded by 5–10 mL of turbid, yellow-green, opaque fluid that extended into the immediately adjacent intrahepatic bile ducts. Cytologic evaluation of an impression smear of the fluid revealed sloughed columnar biliary epithelial cells admixed with degenerate neutrophils, with scattered extracellular and intracytoplasmic short rods (Fig. 3). Bacterial culture of bile duct fluid yielded heavy growth of
Tissues were collected in 10% neutral-buffered formalin and processed routinely for histologic evaluation with H&E staining. Evaluation of the common bile duct where the choledocholith was lodged revealed moderate mucosal hyperplasia with expansion of the lamina propria by neutrophils, lymphocytes, plasma cells, and macrophages. Inflammation extended into deeper fibrous connective tissue separating the common bile duct from the adjacent pancreas but did not extend into the pancreatic parenchyma, which was histologically within normal limits. Evaluation of the liver revealed moderate-to-marked expansion of portal tracts by neutrophils, with fewer lymphocytes, plasma cells, and macrophages. Inflammatory cells tightly cuffed and infiltrated the biliary epithelium, with multifocal neutrophilic exocytosis, luminal accumulation of degenerate-to-intact neutrophils and necrotic cellular debris, and coagulative necrosis of epithelium, consistent with neutrophilic cholangitis (Fig. 4). In addition, within the portal regions, there was fibrosis, edema, proliferation of a few small caliber bile ducts (ductular reaction), and robust periductular proliferation of concentric spindloid cells (myofibroblasts), consistent with extrahepatic obstruction (Fig. 4). Fibrosis often bridged between portal tracts, and inflammatory cells multifocally extended beyond the limiting plate, with rare individual hepatocellular necrosis in portal areas, consistent with cholangiohepatitis (Fig. 4, inset). Low numbers of canalicular bile plugs were present adjacent to portal tracts.
Collectively, these findings informed a diagnosis in case 1 of segmental choledocholithiasis with plant material with secondary moderate-to-severe, chronic, neutrophilic choledochitis and cholangiohepatitis with bridging fibrosis, extrahepatic obstruction, and ductular reaction. In addition to the hepatic and biliary changes, the associated regional lymph nodes had marked, follicular hyperplasia with formation of prominent germinal centers. The medullary sinuses were moderately expanded by a mixture of macrophages, neutrophils, lymphocytes, and plasma cells. The brain was not evaluated histologically.
Case 2 was an 18-y-old American Paint Horse mare that was referred to The Ohio State University Veterinary Medical Center (OSU-VMC; Columbus, OH, USA) because of colic, chronic weight loss, inappetence, and acute inappropriate mentation. Blood work from the rDVM identified increases in activity of AST, GGT, and alkaline phosphatase (ALP), and hyperbilirubinemia (Suppl. Table 1). The mare was treated initially with intravenous fluids and flunixin meglumine by the rDVM, and referral to OSU-VMC was elected after persistence of clinical signs despite medical management.
On physical examination at the OSU-VMC, the mare was icteric, moderately tachycardic (70 bpm) with a 4 of 6 left holosystolic murmur and bilaterally decreased (ventral) to absent (dorsal) borborygmi. The mare was thin (body condition score: 2.5 of 9) with marked symmetric atrophy of epaxial and gluteal muscles and generalized weakness. Abdominal ultrasound revealed copious amounts of free heteroechoic and flocculated abdominal fluid suggestive of fibrin exudation. The liver was heterogeneous with large hypoechoic foci scattered randomly throughout the parenchyma. Abdominocentesis yielded dark-brown-to-orange turbid fluid with total protein of 38 g/L (RI: < 20 g/L), lactate of 1,750 mmol/L (RI: < 222 mmol/L), and total leukocyte count of 5.34 × 109 cells/L (RI: < 5 × 109 cells/L), with 20% neutrophils and 72% large mononuclear cells. Cytologic evaluation of the abdominal fluid revealed marked neutrophilic and histiocytic inflammation with a mixed population of extracellular bacteria including diplococci, short rods, and elongate rods, collectively suggesting a mixed septic peritonitis. Serum biochemistry performed at the OSU-VMC revealed numerous abnormalities (Table 1; Suppl. Table 2), the most significant of which were moderate-to-marked increases in activity of AST, ALP, GGT, and SDH, hyperbilirubinemia, and increased bile acids. Given the poor prognosis and concern for severe hepatopathy, the mare was euthanized and submitted immediately for postmortem examination.
On gross postmortem examination, there was severe diffuse icterus of the mucous membranes, sclerae, and adipose tissue. The peritoneal cavity contained ~15 L of green-brown-to-red, opaque fluid with no observable organ rupture. Copious amounts of fibrin coated the omentum, spleen, liver, and ventral abdominal body wall; fibrin was most abundant overlying the left side of the liver. Affecting ~50% of the liver, and predominantly the left lobes, were 5–8, multifocal-to-coalescing, large (up to 30 cm diameter), well-demarcated, circular-to-irregularly shaped soft red-brown-to-tan foci of coagulative necrosis. Necrotic areas were emphysematous and, on cut section, were diffusely red-brown. Along the periphery of the left lobe, affecting ~10% of the liver, was a poorly demarcated green region of liquefactive necrosis.
Filling the entire common bile duct, from liver to duodenum, was a 4 × 4 × 12-cm green-yellow, granular concretion (choledocholith; Fig. 5). At the level of the major duodenal papilla, a brown-black 5 × 0.2 × 0.2-cm wood stick extended from the choledocholith toward the duodenum (Fig. 5). After fixation and sectioning of the bile duct, the choledocholith was comprised of bile concretions and pieces of plant material (hay and wood twigs; Fig. 6). Aerobic and anaerobic bacterial culture of the liver yielded heavy growth of
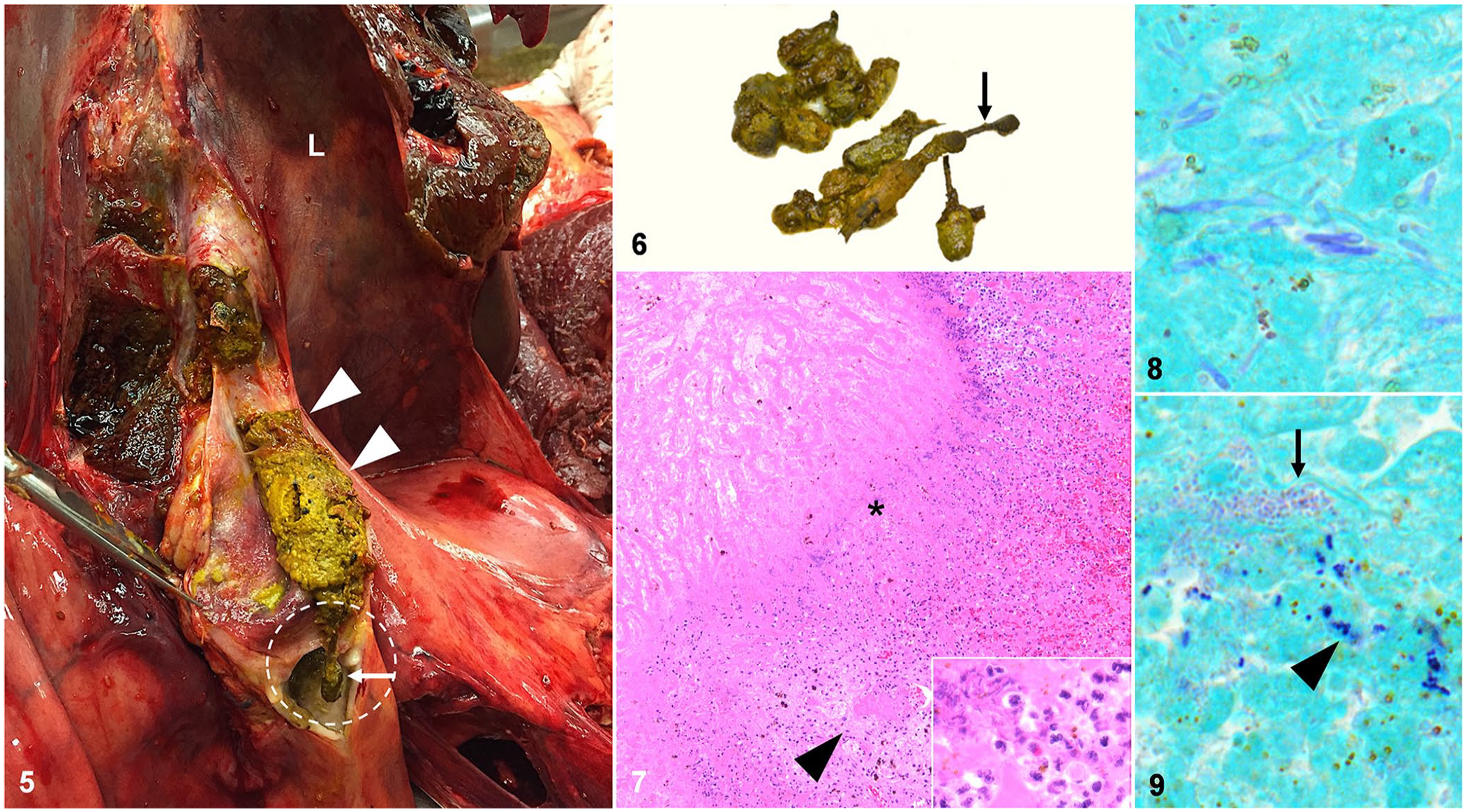
Gross and histologic features of ingesta-associated choledocholithiasis in case 2, an American Paint Horse mare.
Tissue samples were collected in 10% neutral-buffered formalin and processed routinely for histologic evaluation with H&E staining. The common bile duct had severe mucosal ulceration with accumulation of abundant intact and degenerate neutrophils, fibrin, and luminal lakes of bile and plant material. Well-demarcated regions of acute coagulative-to-liquefactive necrosis of the hepatic parenchyma affected over half of the liver section randomly (Fig. 7). At the interface of necrotic foci and intact hepatic parenchyma, there was infiltration by large numbers of neutrophils, abundant fibrin, and myriad mixed bacteria, including mostly long rods with fewer short rods and cocci (Fig. 7, inset). Occasionally, the short rods and cocci were present centrally within the necrotic regions. Gram staining revealed 3 populations of bacteria: numerous long gram-positive rods, fewer large gram-positive cocci, and small gram-negative short rods, consistent with
Collectively, these findings informed diagnoses in case 2 of focally extensive, severe, subacute ulcerative and fibrinosuppurative choledochitis with a grossly identified choledocholith with intra-choledocholith plant material; severe, multifocal-to-coalescing, acute hepatocellular necrosis with intralesional mixed bacteria; and mild, chronic, multifocal neutrophilic cholangitis and cholangiohepatitis with bridging fibrosis and ductular reaction.
Our 2 cases demonstrate a unique cause of equine colic in the form of choledocholithiasis secondary to ingested plant material that had ascended into the common bile duct. We identified 2 additional cases of ingesta-associated choledocholithiasis by searching for “horse cholelithiasis”, “equine cholelithiasis”, and “horse cholelithiasis foreign body” in PubMed, NCSU, and OSU Library databases; one of the cases had a 7-cm wooden stick, and the other case had grass awns, within the core of a choledocholith7,8 (Table 2). Meaningful evaluation of trends in this cohort is precluded given the low number of cases; however, a few observations can be made.
Features of reported cases of foreign body–associated equine choledocholithiasis.
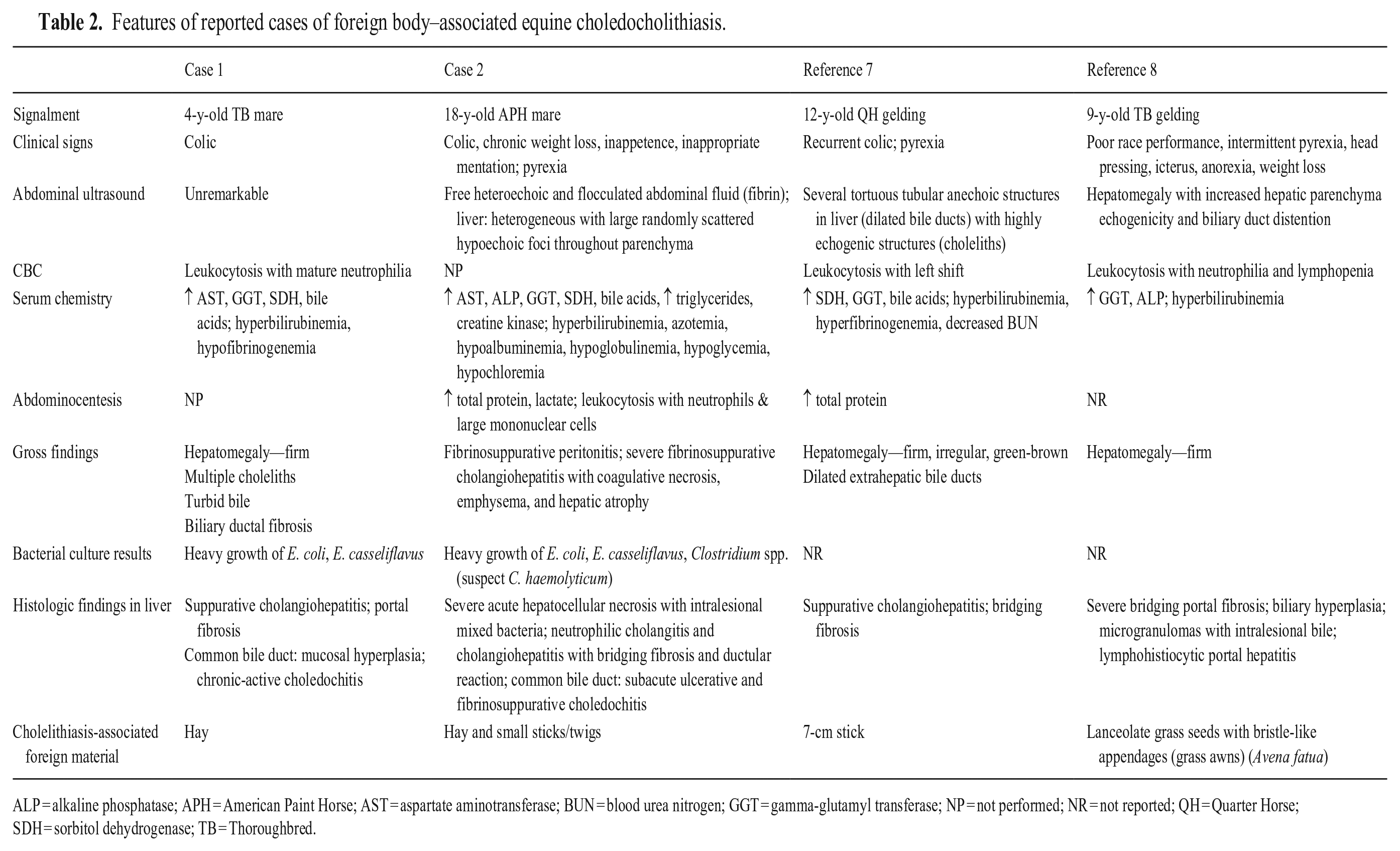
ALP = alkaline phosphatase; APH = American Paint Horse; AST = aspartate aminotransferase; BUN = blood urea nitrogen; GGT = gamma-glutamyl transferase; NP = not performed; NR = not reported; QH = Quarter Horse; SDH = sorbitol dehydrogenase; TB = Thoroughbred.
Ingesta-associated choledocholiths in horses have been reported in a wide age range (4–18 y); 2 of 4 horses were Thoroughbreds.7,8 Clinically, these horses had signs similar to horses with non–ingesta-associated choledocholiths, 15 including colic (3 of 4), pyrexia (3 of 4), and/or neurologic signs (2 of 4; Table 2).2,7,8 Only 3 of the 4 horses had CBC data, and for all 3, leukocytosis with neutrophilia was present. Hepatobiliary serum biochemical derangements were common, with elevations in hepatocellular enzyme activity including AST (2 of 4) and SDH (3 of 4), and increased cholestatic enzyme activity (GGT and ALP [in 2 of 4]). All 4 horses had hyperbilirubinemia, and 3 cases had elevated bile acid concentrations.
Three horses, including our case 1, had gross hepatomegaly with firm consistency along with either dilation or fibrosis of the bile ducts.7,8 Histologically, these cases had lesions consistent with extrahepatic obstruction including myofibroblast proliferation within portal regions, bridging fibrosis, and portal hepatitis and/or neutrophilic cholangiohepatitis.
In contrast, our case 2 had no evidence of extrahepatic obstruction, but instead had large regions of grossly apparent and histologically confirmed hepatic coagulative and liquefactive necrosis with a much less prominent component of cholangiohepatitis and bridging fibrosis. Of note, this is also the only horse of the 4 that had evidence of massive hepatocellular dysfunction (i.e., hypoalbuminemia, hypoglycemia). These distinguishing features from the other 3 reported cases are attributed to the presence of
There are a few potential pathogeneses for the hepatocellular injury in case 2. First, biliary rupture secondary to ascending bacterial cholangitis may have resulted in release of bile into the parenchyma, resulting in regional hepatocellular necrosis (so-called “bile infarcts”) and creation of an anerobic environment. We consider this the most likely pathogenesis. Second, significant extrahepatic obstruction and increased retrograde biliary pressure could similarly result in bile infarcts, but is considered less likely given the lack of histologic evidence of extrahepatic obstruction. Third, intrahepatic bile canalicular cholestasis could have impacted sinusoidal blood flow, leading to hypoxic conditions and hepatocellular necrosis. However, this is also considered less likely given the lack of significant canalicular cholestasis in case 2. In all of these scenarios, local necrosis and an anaerobic environment within the liver could have allowed the
Additionally, in the bile or liver of our 2 cases, there was growth of
Choledocholiths and choleliths associated with foreign material are not reported in other domestic species, although foreign material, such as sand and seeds, has been identified in the gallbladders of sheep and pigs, respectively. 5 Anatomic features specific to the horse extrahepatic biliary tree, including the short length and wide caliber of the common bile duct as well as lack of a gallbladder, may contribute to the occurrence of this phenomenon in horses. Given the lack of a gallbladder, it is possible that there is more frequent opening of the major duodenal papilla for bile excretion, allowing a higher likelihood of ingesta ascending the common bile duct and predisposing to cholangitis and stone formation.
Foreign body–associated choleliths and choledocholiths in humans are most commonly associated with a history of biliary tract surgery with surgical sutures and Ligaclips (Ethicon) found within choleliths and choledocholiths. 1 Although uncommon, ingesta-associated choleliths and choledocholiths have been reported in humans in association with fish bones, toothpicks, tomato skin, and celery stalks.3,11,12,27,30 In these cases, biopsy and culture were not reported; however, these patients often had epigastric pain or icterus, and had evidence of common bile duct dilation with or without concurrent hepatic chemistry abnormalities.11,12,27,30 In humans, although bacterial culture is not always performed, there is evidence that bacteria within the biliary tract have a role in stone formation, specifically given their ability to deconjugate bilirubin.25,26 This reaction leads to the formation and aggregation of calcium bilirubinate “pigment” stones. In people, the risk of recurrent choledocholithiasis is associated with biliary tract infections and advanced age, both of which have also been reported in horses.6,18
Although rare, we recommend assessment of the bile duct for the presence of choledocholiths in horses with persistent colic, pyrexia, and elevated hepatocellular and cholestatic biomarkers. Any retrieved choledocholiths should be evaluated for potential ingested foreign material that may serve as a nidus for choledocholith formation and ascending bacterial infection.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231177251 – Supplemental material for Ingesta-associated choledocholithiasis in horses: 2 cases and literature review
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231177251 for Ingesta-associated choledocholithiasis in horses: 2 cases and literature review by Sarah C. Linn-Peirano, Kate Hepworth-Warren, Hannah Kinsella, Dubraska Diaz-Campos, Bonnie M. Brenseke, Rachel E. Cianciolo, Eric Schroeder and Megan E. Schreeg in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank and acknowledge the histology laboratory staff of North Carolina State University College of Veterinary Medicine as well as the Comparative Pathology and Digital Imaging Shared Resource of The Ohio State University College of Veterinary Medicine. In addition, we thank the North Carolina State University Clinical Microbiology and Molecular Diagnostics and The Ohio State University Diagnostic and Clinical Microbiology Service for their assistance with bacterial culture.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
